Abstract
Background:
Depression and anxiety are common in synucleinopathies and often present during prodromal stages, including idiopathic/isolated REM sleep behavior disorder (iRBD). However, the specific profiles of depression/anxiety and their predictive values for phenoconversion remain unclear.
Objective:
To assess the predominant manifestations, predictive value, and changes over time in depressive and anxiety symptoms in iRBD.
Methods:
Patients with polysomnography-confirmed iRBD (n = 114) and healthy controls (n = 44) were recruited. The Beck Depression Inventory and Beck Anxiety Inventory were administered at baseline, which was repeated prospectively over follow-up. Factor solutions were generated to delineate symptom clusters within the scales, and to help disentangle primary mood symptoms from other neurodegenerative confounds. Total scores, individual scale items, and factors were evaluated to 1) compare patients and controls, 2) assess progression of symptoms over time, and 3) assess predictive value for phenoconversion.
Results:
At baseline, iRBD patients had more severe depressive (9.0 = 6.7 vs 5.8 = 4.8) and anxiety (7.0 = 7.9 vs 4.5 = 6.0) symptoms than controls. Increased scores were seen in numerous individual scale items and most scales’ factors. For depressive symptoms, there was no progression of total scores or factors over time. However, anxiety scores worsened slightly over prospective follow-up (annual slope = 0.58 points, p < 0.05). Over an average 2.4 = 3.1-year follow-up, 37 patients phenoconverted and 72 remained disease-free. Neither baseline depressive nor anxiety symptoms predicted phenoconversion to clinical neurodegenerative disease.
Conclusions:
Depressive and anxiety symptoms are common in iRBD. However, they do not predict phenoconversion and show only modest progression over time, solely for anxiety.
INTRODUCTION
Idiopathic or isolated REM sleep behavior disorder (iRBD) is a parasomnia characterized by loss of muscle atonia during REM sleep [1]. It is part of the prodromal stage of neurodegenerative diseases such as Parkinson’s disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA). As many as 80%of patients may phenoconvert to these diseases [2]. Depression and anxiety are common prodromal features of PD, DLB, and iRBD, with a clinical diagnosis of major disorder reported in about 30%of PD [3], 25%of DLB, and 20–30%percent of iRBD patients [4]. Of note, RBD itself can be triggered or augmented by antidepressant medications; prior studies have suggested that patients with antidepressant-triggered RBD have early signs of prodromal synucleinopathy but may be at an earlier stage of neurodegeneration and at lower phenoconversion risk [5]. Outside of antidepressant-triggered RBD, it has remained unclear whether depression and anxiety in iRBD predict speed of phenoconversion to dementia or parkinsonism; most studies have found no clear predictive value [5–7]. It is also unclear to what degree these symptoms change over time as patients phenoconvert to defined neurodegenerative diseases.
The severity of depressive and anxiety symptoms are typically evaluated clinically through the use of self-reported questionnaires, such as the Beck Depression Inventory, Version II (BDI) [8] and Beck Anxiety Inventory (BAI) [9]. However, these symptoms can have variable presentations in early neurodegeneration. Moreover, these questionnaires include items that may be confounded by other symptoms of neurodegeneration; for example, the BDI and BAI contain items on fatigue, sleep disturbances, sexual changes, tremor, cognition, and appetite. Therefore, analyzing scales as a whole may therefore mix true mood disturbances with non-mood symptoms of synucleinopathy.
One way to obtain a more precise picture of depression and anxiety profiles is to use factor solutions. For example, in non-RBD samples, the 21 items of the BAI have been split variably into two factors (cognitive and somatic), four factors (cognitive, autonomic, neuromotor, and panic), or six factors (panic, autonomic hyperactivity, somatic, nervousness, motor tension, and fear) [10]; similarly, the 21 items of the BDI have been split into multiple two-factor solutions (cognitive and somatic-affective; cognitive affective and somatic; somatic-affective and cognitive), a three factor solution (cognitive, affective, and somatic), and a four factor solution (somatic, cognitive, self-criticalness, and anhedonia) [11]. These factor solutions may not apply well to patients with prodromal synucleinopathy, given the high prevalence of direct confounds in our patients.
In this study, we compared total BDI and BAI scores as well as individual BDI and BAI item scores between RBD patients and controls at baseline. Additionally, we measured changes in the scales prospectively over time and at time of phenoconversion. We then developed factor solutions for the BDI and BAI using SPSS in order to compare the profiles of depression and anxiety in RBD patients and controls. We also investigated how the factors changed over time and whether phenoconversion to PD or DLB resulted in any changes to the factors.
METHODS
Participants
Patients were recruited from the Center for Advanced Research in Sleep Medicine at the Centre Intégré Universitaire de Santé et de Services Sociaux du Nord-de-l’Île-de-Montréal –Hôpital du Sacré-Coeur de Montréal (CIUSS-NÎM), Canada from 2004 to 2019 as previously described [12]. All patients had polysomnographic-proven RBD and met criteria for idiopathic RBD as defined by the standard International Classification of Sleep Disorders-III criteria [13]. Patients currently taking antidepressants were excluded from analysis because of potential confounding by antidepressant-triggered RBD [5]. Controls were selected from the general population and underwent polysomnographic testing to prove the absence of RBD. Ethics approval was obtained from the ethics review board of l’Hôpital du Sacré-Coeur de Montréal and all patients gave informed consent prior to participating. We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Procedures
All patients received an extensive neurological and neuropsychological examination, as has been described in detail in previous publications [7, 12]. This study primarily used two self-reported questionnaires which assess the severity of typical symptoms of anxiety and depression: the Beck Anxiety Inventory (BAI) [9] and the Beck Depression Inventory, second edition (BDI-II) [10]. These questionnaires were administered at the baseline visit and were repeated annually since 2013. Because repeat annual administration of these questionnaires only began in 2013, most patients only had 3 or 4 years of follow-up visits. While data exists past 4 years for some patients, the number of patients was too low to allow for reliable assessment, and these questionnaires were not included.
Factor analysis
Factoring of each questionnaire was performed using the dimension reduction factor analysis feature of SPSS. An unrotated principle component analysis (PCA) was run first to establish a baseline for comparison. Combinations of other factoring techniques (principle axis factoring [PAF], maximum likelihood, unweighted least squares, and generalized least squares) and rotations (quartimax, equamax, direct oblimin, varimax, and promax) were then executed. The factoring techniques aim to explain the variance in the questions in order to correlate like questions and reduce the total number of variables. Rotations further correlate variables to make for a simpler factor solution; an unrotated factor solution may have some heavily loaded factors, but by rotating the solution we were able to explore more correlations so that each factor may be loaded less heavily. A factoring solution was chosen when each individual component had a higher correlation coefficient than the unrotated PCA solution. Components were then sorted into factors based on highest correlation coefficient.
Statistical analysis
All statistical analyses were performed in Statistical Package for the Social Sciences version 24 statistical software (SPSS, Chicago, IL, USA). Baseline comparisons between RBD patients and controls on individual items of the BDI and BAI were performed using independent sample t-tests. The progression of individual questions over time as well as factors was also assessed using linear regression in which baseline responses were compared to responses in subsequent years. An additional exploratory analysis used independent sample t-tests to compare scores of patients who converted to parkinsonism or dementia in the year they converted versus their own baseline scores and the scores of the entire cohort. For all t-tests, a p-value of 0.05 was considered significant. Because of the exploratory nature of the study, no correction for multiple comparisons was performed; therefore, all results should be considered exploratory in nature.
RESULTS
A total of 114 patients and 44 controls were recruited. The average age was 69.0 = 9.1 years for RBD patients and 66.3 = 9.8 for controls and 88 RBD patients (77%) and 31 controls (70%) were male. Average RBD duration for these patients was 7.9±7.0 years. 113 had baseline BDI and 111 had BAI. Of this, 73 had at least one follow-up for BDI (n = 44 at 2 years, 33 at 3 years, and 19 at 4 years) 70 had at least one follow-up for BAI (n = 50 at 2 years, 34 at 3 years, and 17 at 4 years. Over the course of this study, nine patients were lost to follow up and one patient died.
Factor analysis
For the BDI, a promax rotation PCA proved to be the most successful and resulted in 7 factors. Factor 1 consisted of loss of pleasure, guilty feelings, self-dislike, self-criticalness, loss of interest, and indecisiveness. Factor 2 consisted of past failure, agitation, and worthlessness. Factor 3 consisted of sadness, pessimism, punishment feelings, and suicidal thoughts. Factor 4 consisted of tiredness and fatigue, and loss of interest in sex. Factor 5 included loss of energy, changes in sleep pattern, and irritability. Factor 6 included changes in appetite and concentration difficulty, and factor 7 consisted of crying.
For the BAI, a promax rotation PAF proved to be the most successful. However, unlike the BDI factor solution, this rotation did not result in higher correlation coefficients for all variables, although it was the best of all of the generated solutions. From this solution, 5 factors were produced: Factor 1 consisted of fear of worst happening, terrified/afraid, nervous, fear of losing control, difficulty in breathing, fear of dying, scared, and faint/lightheaded; Factor 2 included feeling hot, feeling of choking, indigestion, face flushed, and hot/cold sweats; Factor 3 was composed of dizzy/lightheaded, heart pounding/racing, and unsteady; Factor 4 included numbness/tingling, feeling hot, and wobbliness in legs; Factor 5 was composed of hands trembling and shaky/unsteady.
Baseline results
Total BDI and BAI scores
First, we compared baseline total scores for the BDI and BAI between iRBD patients and controls (Tables 12). Patients had more severe depressive and anxiety symptoms than controls, even when those with possible antidepressant-triggered RBD were excluded (BDI total scores: 9.1±6.7 vs 5.8±4.8, p < 0.001, BAI total scores: 7.8 = 8.9 vs 4.5 = 6.0, p < 0.01).
Question-by-question comparison of average baseline scores of iRBD patients and controls on the Beck Depression Inventory
*denotes statistical significance at threshold < 0.05. Effect size is Cohen’s D. BDI, Beck Depression Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
Question-by-question comparison of average baseline scores of iRBD patients and controls on the Beck Anxiety Inventory
*denotes statistical significance at threshold < 0.05. Effect size is Cohen’s D. BAI, Beck Anxiety Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
Subscales and factors: baseline depression
A baseline comparison of average individual question scores of RBD patients and controls was then performed to obtain a more detailed view of the presentation of depressive symptoms in patients (Table 1). Effect sizes were calculated to compare the difference between scores of RBD patients and controls. These data highlighted differences in some, but not all, items, indicating that differences in certain aspects of depressive symptoms were more pronounced in iRBD patients. Of note, the two features with the highest effect sizes (Cohen’s d = 0.67), fatigue and loss of interest in sex, are also potential symptoms of synucleinopathies. Agitation and crying, features that could possibly be related to RBD itself (i.e., agitation and crying out at night) had the next highest effect sizes (Cohen’s d = 0.53). Smaller statistically significant differences were noted for self-dislike, self-criticalness, indecisiveness, and irritability.
Next, we compared the various factors generated by the SPSS factor solution between iRBD patients and controls (Table 3). In this analysis, all factors were found to be significantly different between patients and controls with the exception of Factor 3, which consists of sadness, pessimism, punishment feelings, and suicidal thoughts, and Factor 5, which consists of loss of energy, changes in sleep pattern, and irritability.
Differences in SPSS-generated factors of the BDI between iRBD patients and controls at baseline
*denotes significance at threshold p < 0.05. BDI, Beck Depression Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
Baseline anxiety
On BAI analysis (Table 2), many single items demonstrated differences between patients and controls, with no clear pattern of confounding by prodromal physical/autonomic symptoms. Both core psychiatric anxiety symptoms (fear of worst happening, unable to relax, nervousness) and potentially-confounded symptoms (wobbliness in legs, trembling in hands, shakiness/unsteadiness) showed significant differences and had similar effect sizes. On factor analysis, all factors were significantly different between patients and controls except for Factor 2 (which includes many items related to autonomic hyperactivation) (Table 4). Factor 1, which includes many core cognitive features of anxiety, showed only equivocal differences between patients and controls at our threshold (p = 0.05).
Differences in SPSS-generated BAI factors between iRBD patients and controls at baseline
*denotes significance at threshold p < 0.05. BAI, Beck Anxiety Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
Progression of depression and anxiety
We then examined how depressive and anxiety symptoms progressed over time in iRBD (Fig. 1). Over a four-year period, total depression scores showed little change, with total scores changing from 9.0 = 6.7 to 8.8 = 4.8. Anxiety, however, showed significant progression, changing from 7.1 = 7.9 to 9.3 =8.8 over a four-year period (slope = 0.58 per year, p < 0.05). Because confounding by non-mood symptoms could affect measures of progression, we then examined whether different patterns of progression could be seen in the individual scale factors. For depression, only Factor 6 (concentration difficulty/appetite, slope = 0.16, p < 0.05), showed progression over time (Fig. 2). For anxiety (Fig. 3), significant progression was seen in Factor 3 (slope = 0.16, p < 0.05), Factor 4 (slope = 0.25, p < 0.05), and Factor 5 (slope = 0.10, p < 0.05).
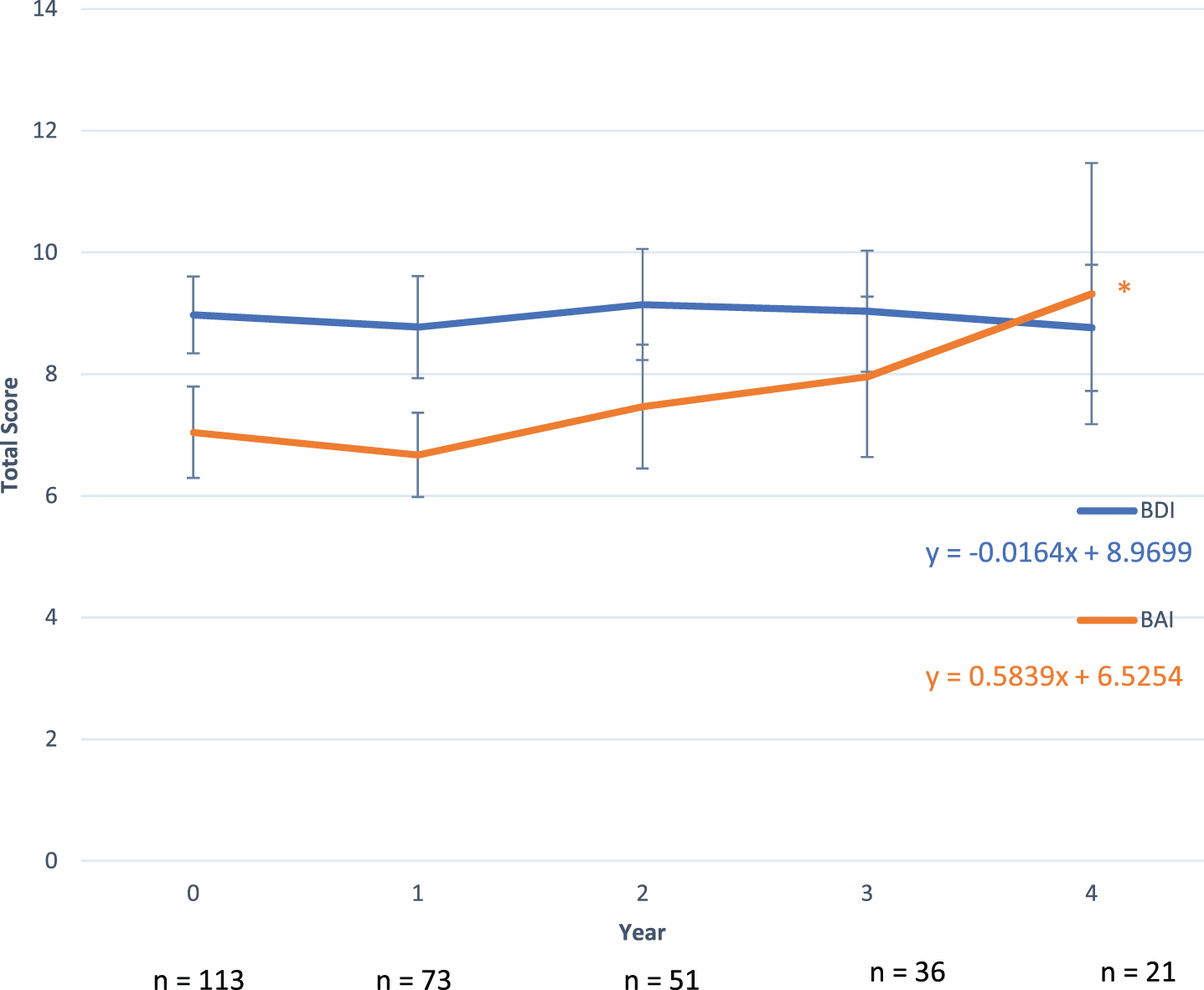
Change in total BDI and BAI scores over time in iRBD patients. * denotes significant progression at threshold p < 0.05. BAI, Beck Anxiety Inventory; BDA, Beck Depression Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
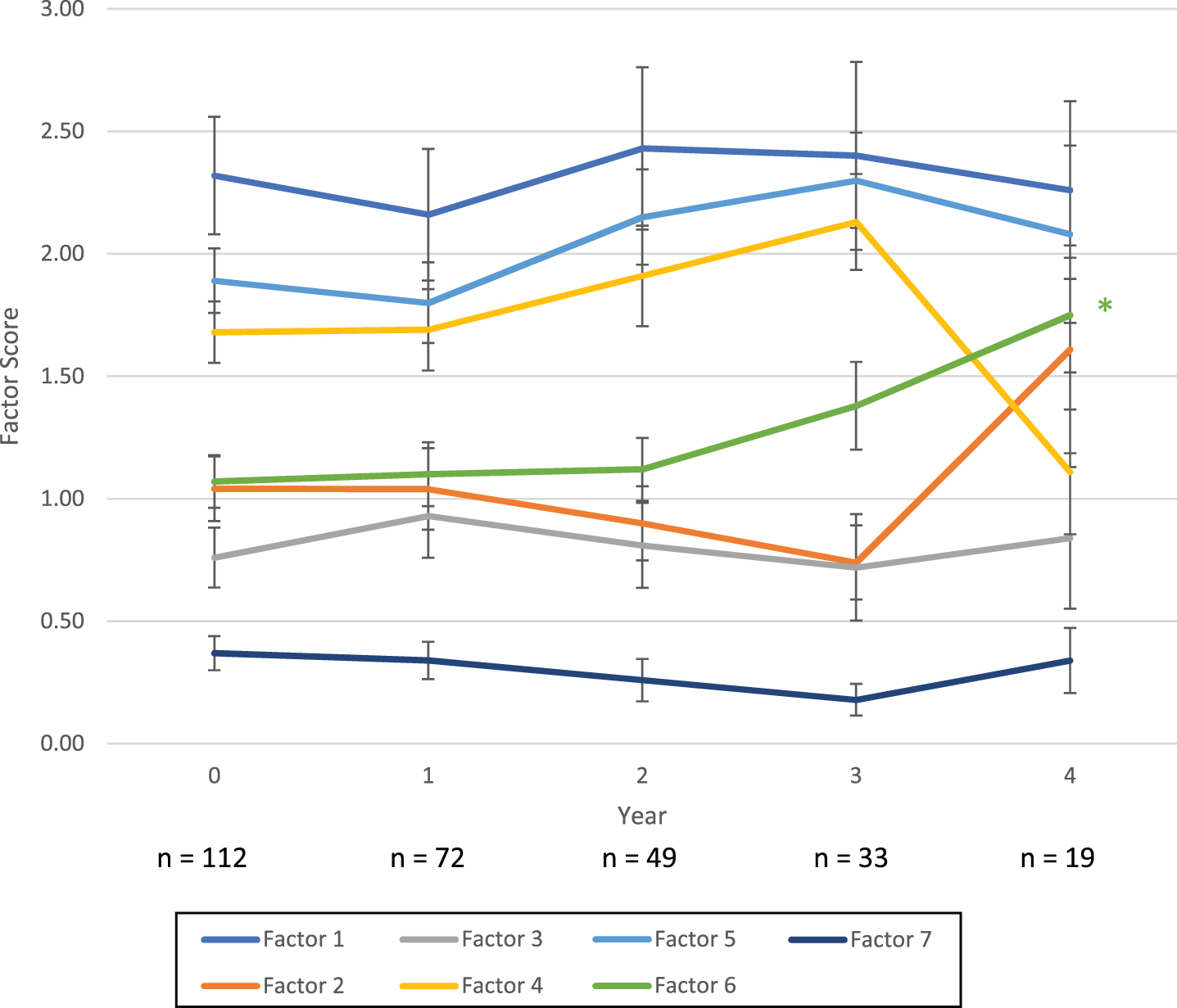
Progression of SPSS-derived BDI factors for iRBD patients. * denotes significant progression at threshold p < 0.05. BDI, Beck Depression Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
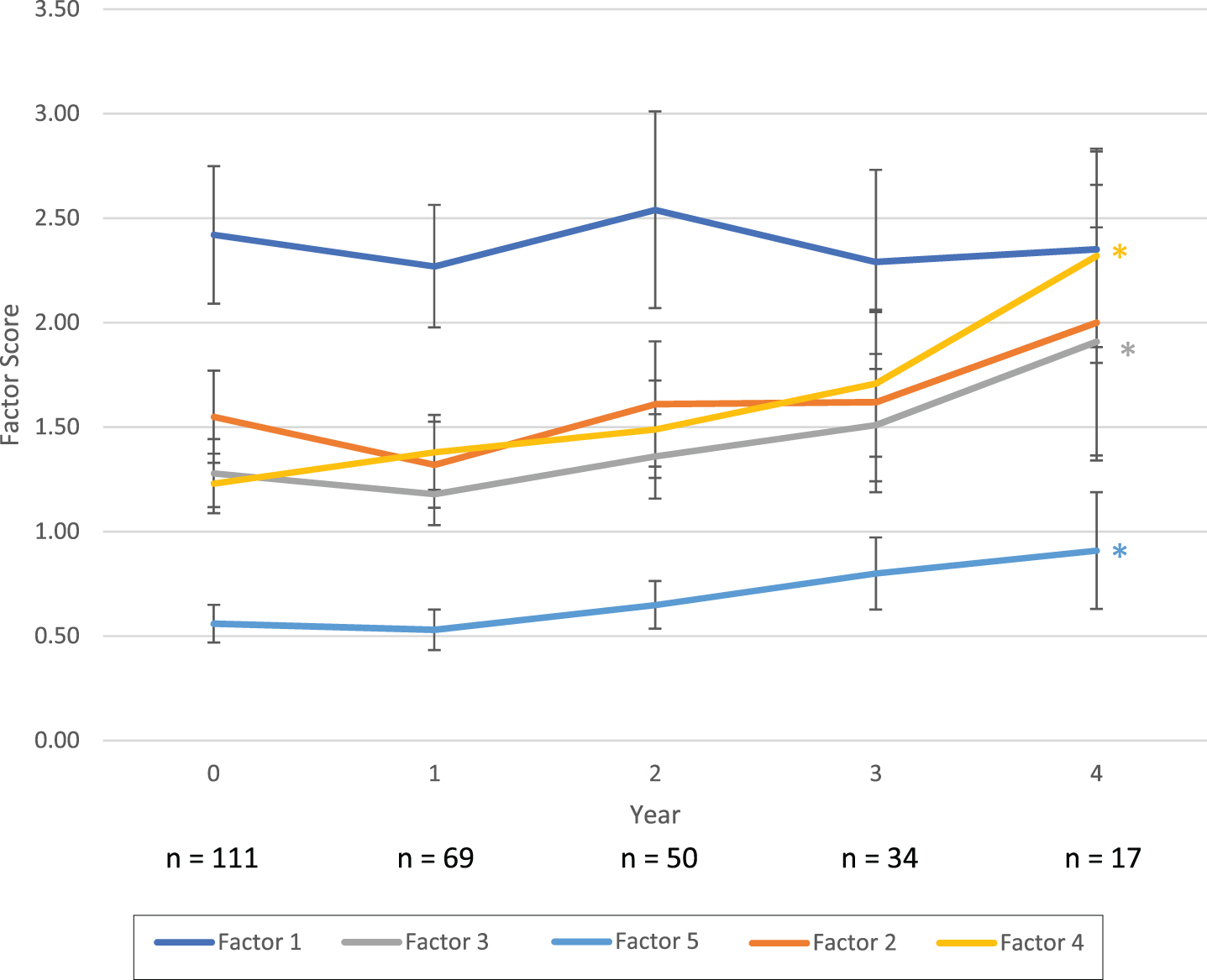
Progression of SPSS-derived BAI factors for iRBD patients. * denotes significant progression at threshold p < 0.05. BAI, Beck Anxiety Inventory; iRBD, Idiopathic/Isolated REM Sleep Behavior Disorder.
Depression and anxiety and phenoconversion
Over an average 2.4 = 3.1 years of follow-up, 37 patients (29%) converted to defined neurodegenerative disease. Seventeen developed parkinsonism first (13 PD, 4 MSA) and 20 developed dementia first (all of whom, by definition, met criteria for probable DLB). Neither baseline BDI nor BAI were different in those who phenoconverted vs. those who did not (baseline score = 9.8 = 7.2 vs 8.4 = 6.4 for BDI and 7.2 = 7.9 vs 7.1 = 8.1 for BAI). An exploratory analysis of those who phenoconverted to different disease subtypes (i.e., PD-first, DLB-first, MSA) showed no significant differences at baseline nor at time of phenoconversion for BDI, (baseline in PD = 9.1 = 6.5, DLB = 10.5 = 7.4, and MSA = 9.0 = 3.6). For the BAI, baseline scores were similar among PD-first and DLB-first convertors (PD = 7.4 = 5.6, DLB = 8.3 = 9.9) but lower in the 4 MSA convertors (BAI = 1.3 = 0.5, p < 0.01 compared to combined Lewy-body group).
We hypothesized that phenoconversion may influence total BDI and BAI scores, and so we compared baseline scores of non-convertors, baseline scores of convertors, and scores of convertors in the year they converted (mean interval: 3.5 = 2.6 years) (Supplementary Table 1). We saw no significant differences between groups (9.8 = 7.2 vs 8.4 = 6.4 vs 10.2 = 7.2 for BDI and 7.2 = 7.9 vs 7.1 = 8.1 vs 6.7 = 5.4 for BAI). Upon examination of individual factors, only Factor 6 of the BDI (change in appetite, concentration difficulty) was higher in phenoconvertors than non-convertors (1.4 = 1.3 vs 0.88 = 1.0, p = 0.04).
We then explored potential progression of symptoms specifically in phenoconvertors by comparing baseline scores of phenoconvertors to the scores at their year of phenoconversion (note that power was lower for this analysis). There was no significant change in either total scores or scores on factors among phenoconvertors.
DISCUSSION
This study explored depressive and anxiety symptoms as potential prodromal markers in patients with iRBD using the BDI and BAI. We found clear differences in both total scores on the inventories and in individual questions between patients and controls. These included questions that targeted mood directly as well as questions potentially confounded by other neurodegenerative symptoms. Furthermore, when factored using an SPSS-generated solution, baseline differences exist between RBD patients and controls in most depression and anxiety factors. Depression scores did not progress over time, and analysis of factors that progressed suggested that this may have been driven by motor or autonomic symptoms. Anxiety scores did progress modestly; however, analysis of factors suggested that progression was mainly driven by factors potentially confounded by other neurodegenerative symptoms (e.g., wobbliness in legs, unsteadiness, hands trembling, etc.) and the core factor reflecting psychic/cognitive anxiety (Factor 1) did not progress. Moreover, neither total scores nor individual factors were higher among those who eventually phenoconverted to degenerative disease.
While studies have previously compared BDI and BAI total scores between RBD patients and controls and looked at the predictive value of these questionnaires [5, 12], no studies have assessed progression over time or used factor solutions to examine the profiles of depression and anxiety in RBD. Using factor analysis rather than relying on total questionnaire scores serves two useful purposes. First, factor solutions group similar questions together that tap into the same component, thereby allowing for more specific examination of thesis individual components. Of note, only 23 patients of this cohort (20.4%) had mild depression as defined by the BDI (total score ≥14) and 9 (7.9%) had moderate depression (total score ≥20). Using factor analysis, we can see that a larger proportion of participants exhibit some depressive symptoms and likely have mild subsyndromal depression is not captured by the BDI as a whole. Second, factoring can help draw out questions that are potentially confounded by other neurodegenerative symptoms (which is why factor solutions needed to be specifically created for this patient population, rather than relying on factor solutions generated in the general population). iRBD patients commonly have autonomic dysfunction, subtle cognitive loss, mild motor slowing, and primary sleep disturbances; these manifestations may have confounded previous attempts to document predictive value of depression or anxiety for phenoconversion or could have masked changes in depression and anxiety over time. In the end, however, analysis of factors showed few differences compared to analysis of scales overall. This may indicate that these factors are highly correlated (i.e., depression/anxiety, cognition, autonomic dysfunction all change similarly over time). Alternatively, patient insight may have played a role; patients may have understood the individual items as applying to the context of a depression/anxiety questionnaire, themselves discarding these same confounding factors (e.g., patients answered “hands trembling” or “shakiness/unsteadiness” questions as anxiety symptoms per se, rather than reporting primary tremor or balance symptoms unrelated to anxiety.
The primary limitation of this study is the relatively limited number of questionnaires for the progression analysis. This is because we began systematically tracking follow-up questionnaires starting in 2013 (9 years after the initiation of the cohort) Moreover, some patients were not followed up with every single year, were lost to follow up, or are newly-recruited patients with relatively few follow-up visits. This also affects the phenoconversion analysis as limited data is available at this time. This was an exploratory study without prior similar studies upon which to delineate primary outcomes; therefore, no adjustment for multiple comparisons was performed and results should be considered exploratory in nature [14]. However, our study is strengthened by our relatively large sample size at baseline. Furthermore, we used the BDI and BAI as the sole measures of depression and anxiety; future studies may want to consider using additional questionnaires or diagnostic measures to categorize depression and anxiety. Patients taking anti-depressants were excluded from analysis given the potential confound of anti-depressant triggered RBD (which is associated with lower prevalence of many neurodegenerative markers and phenoconversion/progression rate [5]; we observed similar results overall in sub-analysis including these patients (data not shown).
In conclusion, baseline differences exist in anxiety and depression scales between RBD patients and controls when assessed as total scores, individual items, and factor groupings. Total anxiety scores but not depression scores change modestly over time. Neither depression nor anxiety scores predict phenoconversion. Where progression is seen, it appears to be mainly driven by non-mood symptoms of neurodegeneration; however, these analyses should be repeated with larger sample sizes.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the Fonds de la Recherche - Sante Quebec (grant number POSRO0301 –28915) and the Canadian Institute of Health Research (Grant #286641).
CONFLICT OF INTEREST
The authors have no financial disclosures in relationship to this manuscript.
