Abstract
Despite clinical evidence of poor oral health and hygiene in Parkinson’s disease (PD) patients, the mouth is often overlooked by both patients and the medical community, who generally focus on motor or psychiatric disorders considered more burdensome. Yet, oral health is in a two-way relationship with overall health—a weakened status triggering a decline in the quality of life. Here, we aim at giving a comprehensive overview of oral health disorders in PD, while identifying their etiologies and consequences. The physical (abnormal posture, muscle tone, tremor, and dyskinesia), behavioral (cognitive and neuropsychiatric disorders), and iatrogenic patterns associated with PD have an overall detrimental effect on patients’ oral health, putting them at risk for other disorders (infections, aspiration, pain, malnutrition), reducing their quality of life and increasing their isolation (anxiety, depression, communication issues). Interdisciplinary cooperation for prevention, management and follow-up strategies need to be implemented at an early stage to maintain and improve patients’ overall comfort and condition. Recommendations for practice, including (non-)pharmacological management strategies are discussed, with an emphasis on the neurologists’ role. Of interest, the oral cavity may become a valuable tool for diagnosis and prognosis in the near future (biomarkers). This overlooked but critical issue requires further attention and interdisciplinary research.
Keywords
INTRODUCTION
“As far as the fields of dentistry and neurology are concerned, they should be especially close [ . . . ] I am convinced that every neurologist should know more dentistry, and that the average dentist would profit in many ways if he had more information about neurology.”
Roland P. Mackay (1937) Points of contact between dentistry and neurology. The Journal of the American Dental Association and The Dental Cosmos
To this day, oral health is not prioritized in the management of neurodegenerative diseases. Patients, caregivers, and the medical community generally focus on other high-priority problems, like dementia or movement disorders [1, 2]. Parkinson’s disease (PD) is no exception [3–5]. Yet, oral health is not isolated from general health: it is a critical component of overall health, well-being and quality of life [6–10].
PD is now recognized as a complex multisystem neurodegenerative disorder [11–13], with growing awareness and treatment of nonmotor symptoms [14, 15]. Patients’ oral and dental health has, however, been under-documented, with a lack of high-quality studies 1 and conflicting findings [17]. To our knowledge, no overview of the possible relationships between oral health, PD clinical picture and the ramification of their intricacy on general health has been published. Recent efforts have been undertaken to guide the management of PD patients within dental offices [18] and the inclusion of dentists into a new outpatient integrated 2 “ PD care model [19–21]. This narrative review therefore aims at summarizing published literature regarding oral health in PD, exploring the interplay between oral and general health in PD and highlighting unmet needs in clinical care and research.
A literature search for original studies, case reports, case series and review articles published up to January 31, 2021 (language: English) was performed using the MEDLINE, PubMed, and Google Scholar databases, with the following inclusion criteria as search terms: “Parkinson disease”, “Oral Health”, “Stomatognathic Diseases”, “Dentistry” (MeSH terms), “oral disorders”, “dental”, and “buccal” (non-MeSH terms). Reference lists of relevant articles were additionally screened to supplement electronic searching and subsequently reduce the risk of missing relevant information. Data were combined and analyzed through a multi-expertise approach involving a PD specialist (MV), neuroscientists (MA, MV), a pharmacist (MA), dentists (VM, EB, MBM), and microbiologists (VM, EB, MBM) to carry out a comprehensive review and draw new hypotheses.
We firstly report the complex picture of oral health in PD. Orofacial manifestations, weakened oral health, and reduced oral care in both home-based and professional settings are described and explained. Secondly, we summarize what is at stake for PD patients and how oral health disorders may have adeleterious impact on the quality of life and PD progression. Thirdly, we highlight the central role of the neurologist and the need for early and close collaboration with the dental team. Such integrated care would be empowering and potentially be both short- and long-term life-changing for patients. Finally, we address unmet needs by paving the way towards better awareness, assessment, and care of oral health in PD. We propose future research avenues to unravel the mechanisms of this two-way relationship and the potential diagnostic and predictive values of the oral ecosystem.
ORAL HEALTH IN PARKINSON’S DISEASE: A COMPLEX PICTURE
Oral health is multidimensional (physical, psychological, emotional, and social), subjective, and dynamic. It is essential for daily functions like eating, swallowing, speaking, and socializing [10, 22–24]. These functions involve the action of the teeth, lips, cheeks, tongue, and oro-facial-pharyngeal muscles. The imbalance caused by motor (voluntary and involuntary muscle control), nonmotor, and sensory deficits in PD leads to a variety of orofacial manifestations [25, 26]. To date, there is no consensus regarding the most prevalent issues in PD patients, and results vary across studies [3, 28]. Several factors may account for these discrepancies [1, 30], as many papers failed to adjust for clinical, environmental, and pharmaceutical variables. Differences are identified within study populations: gender, age, comorbidities, and subjects randomly selected or actively seeking dental treatment. Moreover, specific differences are also found within PD groups: idiopathic PD versus parkinsonism, PD stage, subtype, severity, and duration, antiparkinsonian medications, living at home versus institutionalized. The quality, accessibility and affordability of the healthcare systems also account for discrepancies. Other contributing factors are methodological: subjective and objective assessments, questionnaires, and ambiguous definitions. However, all studies but two [29, 30] point out towards a weakened or disturbed oral status with objective and subjective differences between PD patients and control groups.
Orofacial manifestations of PD
PD is characterized by a wide range of motor and nonmotor symptoms, stemming from both dopaminergic and non-dopaminergic alterations [14, 31]. These symptoms include a variety of orofacial manifestations, underpinned by sensory and motor dysfunction, occurring in premotor, early, and later stages [25]. Hyposmia is a well-known prodromal sign [31, 32]. However, other orofacial manifestations can also precede the onset of motor symptoms and be presenting features: hyposialorrhea [33], tongue tremor [34–36], upper-body axial symptoms [37], or even dysphagia on rare occasions [38]. Speech disorders, dysphagia, and drooling are the most frequently described orofacial disorders in PD throughout the disease course [14, 40]. At least one of these symptoms was found in more than half (65%) of a cohort of 419 French PD patients, whilst a combination of symptoms was present in one third of them [41]. Regardless of disease duration, drooling, appetite/weight, speech, loss of smell/taste and swallowing disorders all appear in the list of the “24 most bothersome PD-related symptoms/conditions” as evaluated by patients themselves [42]. Notably, the loss of smell/taste is one of the 5 most prevalent complains in early PD (<6 years), while drooling is one of the 5 most prevalent complaints in late PD (≥6 years) [42]. As these disorders have been extensively described and studied in the literature, this article will not focus on them (for recent reviews, please refer to the following references: dysphagia [43, 44], speech disorders [45, 46], and sialorrhea [47]). Functional changes of the anatomic system comprising teeth, jaws and associated soft tissues are reported in PD. They include masticatory and temporomandibular joint disorders (muscle rigidity and incoordination, reduced muscle tone) [5, 48–54], hypomimia, tremor, dyskinesia [55], dystonia [56], oral festination [57], and orofacial pain [49, 59]. Dysgeusia [60–64], bruxism [54, 66], and burning mouth syndrome [66–70] are also inconsistently reported.
Oral health is poorer in PD
Few international studies have been published on oral health in PD patients (Table 1; for detailed results, please refer to Supplementary Table 1). Differences in methodologies, populations, and countries (underpinning differences in health care accessibility and affordability) are notable, which complicates both the interpretation and comparison of results. However, while many of these studies do not represent large samples of patients (ranging from single case reports to hundreds of subjects), they highlight a concerning oral health deficit, with symptoms coexisting in different combinations. Despite being often underreported, these oral symptoms increase the overall disease burden as PD progresses [3, 72]. Compared to controls, PD patients appear to have a significantly weakened oral status [5, 72], with periodontal diseases. 3 and dental caries (tooth decay), leading to more mobile and missing teeth [3, 72–76]. Patients report more discomfort (pain, dry mouth, denture issues) as well as chewing difficulties (related to either teeth, denture, or masticatory function) and dysphagia. They have a worse perception of their oral health [3, 77], and a decreased oral health-related quality of life [4, 79]. Of interest, changes in the oral microbiota are also reported, though the significance and extent of such alterations, as well as their link with PD remain to be determined (causal/correlative/consecutive) [74, 80–82].
Summary of the main findings found in international literature regarding oral health disorders in patients with Parkinson’s disease (orofacial symptoms, oral health, oral hygiene). For detailed results, please refer to Supplementary Table 1
LEDD, levodopa equivalent daily dose; PD, Parkinson’s disease; NA, not assessed. *Since the present study only deals with patients with mild symptoms, it should be noted that the results shown here may not be applicable to advanced PD patients. **Authors’ hypotheses: “general conscientiousness in patients and relatives”. # Periodontal disease account for gingivitis, periodontitis.
This significant oral health inequality is associated with difficulties in coping with dental treatment at the dental office and a decreased ability to maintain adequate levels of daily home oral care, arising from both motor and nonmotor disorders [5, 83].
PD: An impediment to oral and dental care
Despite being aware of the importance of good oral care and displaying normal to high toothbrushing habits and yearly dental visits [3, 75], PD patients do not seem to perform and receive sufficient quality of care. This could be related to the fact that PD has direct and indirect impact on oral health and hygiene. As highlighted in Fig. 1, the physical, psychological, and iatrogenic (see Table 2) patterns associated with PD are predisposing factors of diminished oral health [5, 84–98]. PD severity and duration are both associated with an increased severity of orofacial and dental problems [5, 72]. Motor difficulties, cognitive deficiencies and behavioral changes (food and hygiene habits) compromise daily activities, reducing the quality and quantity of daily oral care and dental care utilization [1, 72]. Additionally, PD patients are more prone to falls, owing to several conditions, such as gait disorders, postural instability and orthostatic hypotension [99, 100]. Falls can in turn lead to maxillofacial trauma and traumatic dental injuries [101].
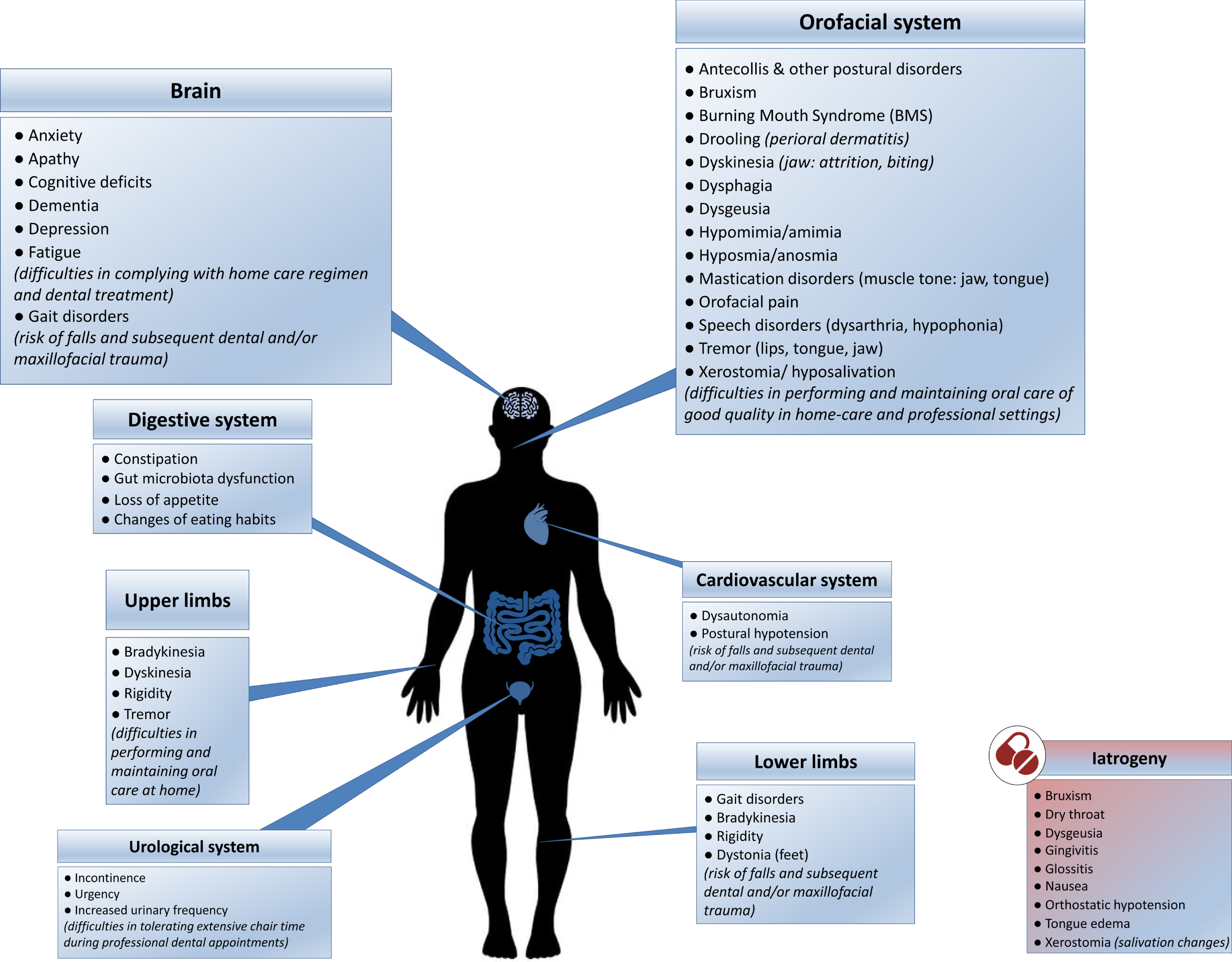
The direct and indirect impact of Parkinson’s disease on oral health and hygiene: contributing physical, psychological (blue boxes) and iatrogenic (red box) factors. (See online version for colour figure.)
Considering the fact that orofacial symptoms and a weakened oral health status are present even at early stages, additional factors, different from motor and cognitive impairments, may also be involved [25, 81]. (Non)dopaminergic imbalance and autonomic dysfunction likely play a role in the emergence and aggravation of these orofacial and oral health disorders [41].
Problems faced in maintaining good oral care at home
Brushing, flossing, and interproximal brushing are critical components of oral self-care [3, 79]. These hygiene maneuvers require muscle-eye coordination, digital dexterity, and tongue-cheek-lip control. Such actions become more difficult to achieve as PD progresses, due to tremor of hands, lips and tongue, bradykinesia, dyskinesia of the hands and jaws, fluctuations, muscle stiffness, and postural deformities [3, 103]. Decline in the quality of the toothbrushing (not necessarily in the frequency [74]) and lack of interdental cleaning reduce the efficacy of plaque control, promoting gingivitis, and initiating or aggravating periodontitis and dental caries. Moreover, such fine movement impairments induce more difficulties in the frequency and quality of prosthesis hygiene, leading to biofilm accumulation [78]. Discomfort with mouthwashes, possibly due to dysphagia and fear of choking, has also been reported [68]. These ineffective oral hygiene measures add to the pre-existing disturbed self-cleaning mechanisms of the mouth (dysphagia, chewing disorders, oral dyskinesia and saliva changes) [4, 80].
Finally, when present, neuropsychiatric disorders also contribute to patients’ difficulties: apathy, depression, fatigue, anosognosia, cognitive decline and dementia may all lead to negligence in daily care, making compliance with a home care regimen difficult [1, 68].
Dental appointments: a challenge to both patients and dentists
The use of dental care services mainly depends on their accessibility (distance, transportation, reduced mobility access) and affordability (healthcare system, dental insurance, socioeconomic status) [10, 105]. While several barriers have been identified at the individual, organizational and policy levels [104] for elders and vulnerable people, PD patients have to face even more challenges in accessing and undergoing dental treatment.
Cognitive decline and dementia may prevent patients from reporting oral and dental symptomatology and complying with dental treatment [1, 106]. In addition, cognitive disorders make them more likely to miss dental appointments or react with care-resistant behavior [107] when receiving assistance [1, 106].
For dental clinicians, PD patients are a challenge, as PD symptoms complicate treatment plans, execution, and outcome [1, 109]. Akinesia, hypokinesia, bradykinesia, gait disorders, orthostatic hypotension, postural deformities [103, 110], and muscle related problems (spasms, rigidity, tremors, and dyskinesia) cause difficulties in getting in and out of the dental chair and staying still during dental treatment [18, 86]. Because of motor and nonmotor fluctuations, patients’ behavior can change from minute to minute and may require stopping dental procedures [18, 83], while anxiety and stress worsen motor symptoms [111]. Muscle rigidity, poor muscle control (lips, jaw, tongue), oral festination [57], facial dystonia [56], orolingual tremor [34, 112–114], dyskinesia (flycatcher tongue), impaired neck and head posture, or dysphagia (saliva accumulation) lead to difficulties in dental examination and oral rehabilitation [30, 109], and can even compel the use of general anesthesia [102]. The supine position during dental procedures may increase the risk of aspiration and ingestion of dental instruments, adding to the already existing risk factors related to PD [26, 115]. Fatigue, urinary [116] and bowel issues may require treatment cessation, preventing extensive treatment plans which require extended chair time [18, 108].
Finally, dentists may have limited knowledge and experience regarding the specific needs and management of PD patients [17, 102]. The patient-dentist relationship is hampered by several PD-related verbal and non-verbal communication impairments. Hypomimia and amimia [117] are linked with reduced facial expressiveness, fixed gaze, disruption of facial emotion recognition [118], and alterations in the facial expression of pain [119]. Speech and language disorders [45, 120] (dysarthria, hypophonia) directly impair verbal communication, while cognitive impairment as well as micrography and other handwriting abnormalities [121] complicate the picture.
Although not surprising to the neurologist, these issues can become very difficult to overcome and lead to frustrating situations for both patient and dental practitioner if the professional is not aware of them and is therefore unable to adjust. Such challenges make patients and dental team reluctant to embark on complex dental procedures [17, 86]. As a result, orofacial symptoms in PD are rarely assessed and detected, making them unaddressed for too long [106].
ORAL HEALTH DISORDERS: WHAT IS AT STAKE FOR PD PATIENTS? A REVIEW AND NEW HYPOTHESES
Oral diseases are associated with a variety of local and general disorders [6, 122–126] and can severely impair quality of life [7, 22]. Being chronic and progressive, they are major functional, psychosocial and economic burdens [10], like PD. Epidemiological data, and studies on microbiological and inflammatory processes have highlighted a link between dysregulated immune and inflammatory response in periodontitis and the initiation and progression of systemic diseases, such as diabetes, cardiovascular disease (atherosclerosis, stroke), cancer, Alzheimer’s disease, or respiratory tract infections [6, 127–130]. The inattention to oral health is therefore concerning in PD patients, considering its direct (pain, orofunctional problems, psychosocial wellbeing) and indirect (increased risk of infections, inflammation and systemic diseases) impacts (Figs. 23). Table 3 summarizes the possible etiologies and contributing factors to oral health disorders in PD patients, their consequences on general health and available prevention/management strategies.
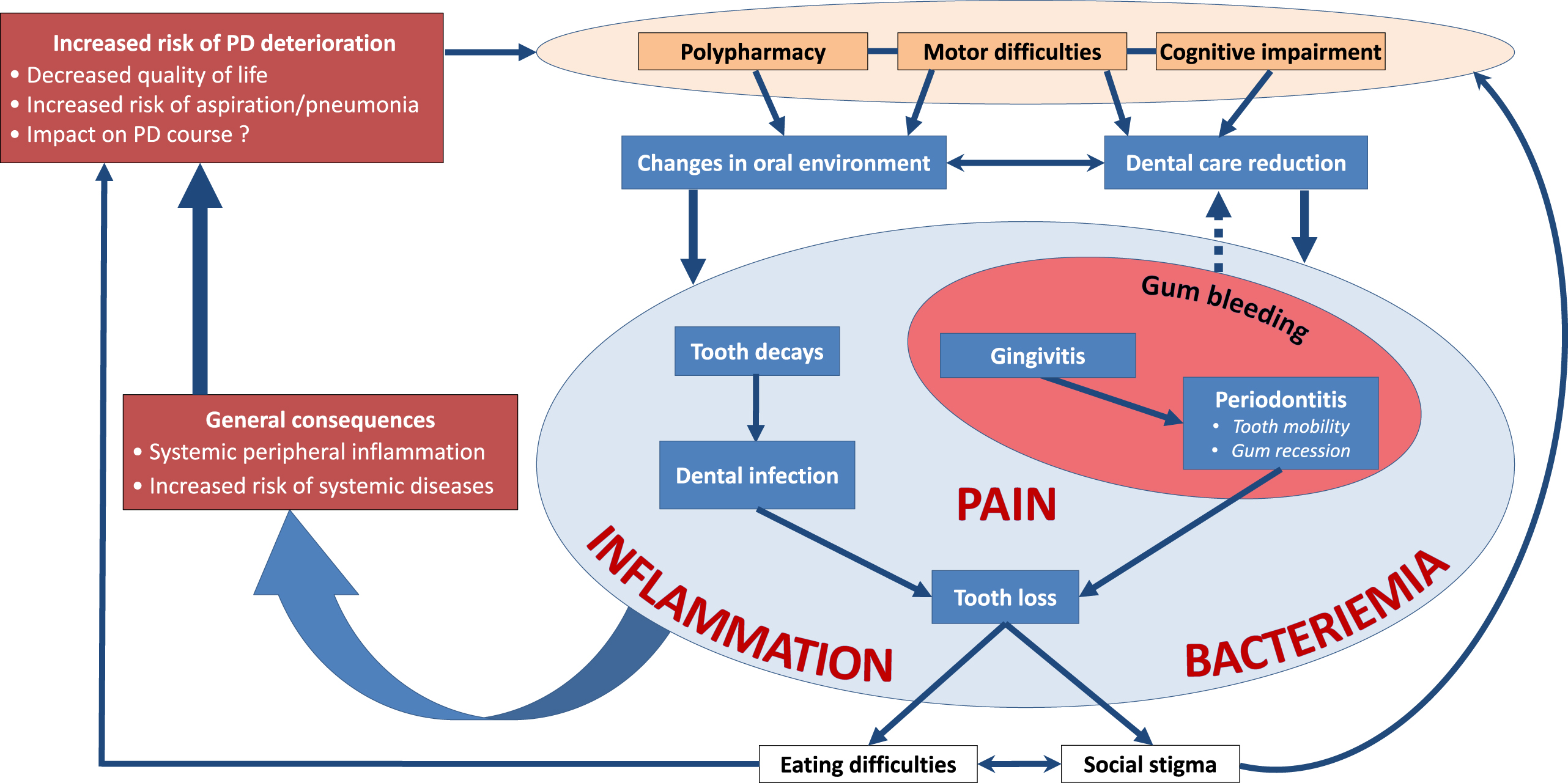
More than meets the eye: Consequences of oral health disorders in Parkinson’s disease.
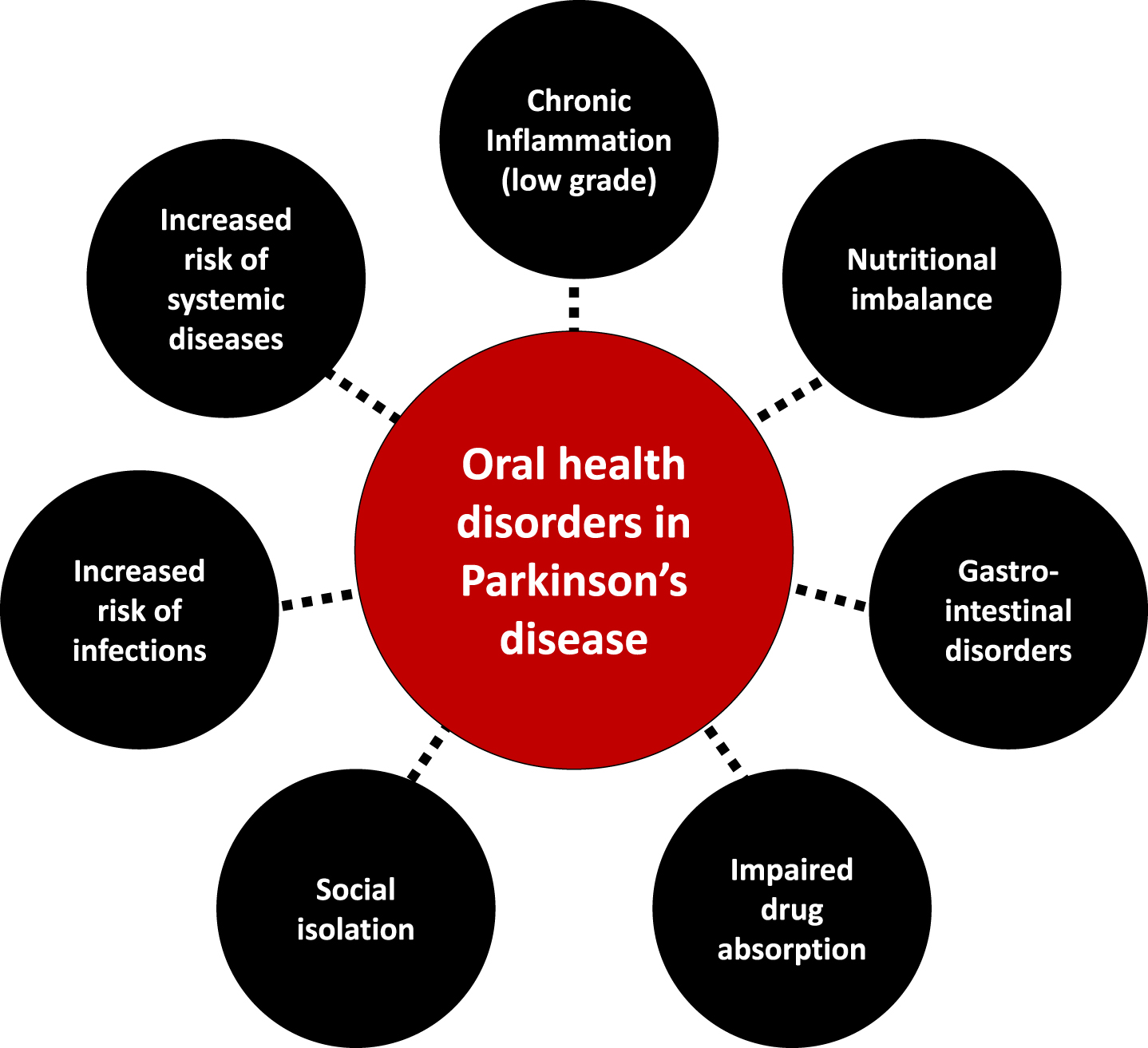
The interplay between oral health disorders and Parkinson’s disease.
Oral health disorders in parkinsonian patients: symptoms, etiologies, consequences (on local & general health) and available prevention and management strategies
AB, awake bruxism; BMS, burning mouth syndrome; BTX, botulinum toxin; CNS, central nervous system; DA, dopaminergic; L-dopa, levodopa; MAO-B, monoamine oxidase-B; PD, Parkinson’s disease; SB, sleep bruxism.
Polypharmacy, motor disorders, cognitive impairment, and the subsequent decline in dental care utilization put patients at risk for dental caries and periodontal diseases (Table 1), paving the way to tooth mobility and, finally, tooth loss [131]. Speech disorders, drooling, halitosis and tooth loss impair appearance and phonetics, reducing social life and increasing isolation, thereby triggering a decline in quality of life [4, 132–134].
A compromised oral health and impaired orofacial function can alter basic function like eating [132, 135], negatively impacting food intake [136, 137]. In PD, taste disturbances, hyposmia/anosmia [64, 138], orofacial pain, masticatory disorders and dysphagia [52, 139] induce prolonged mealtimes and ingestion of insufficiently masticated food, putting the patient at risk of a decline in the quality and quantity of food intake, up to malnutrition. Gastrointestinal disorders may also arise and hamper drug absorption [140, 141]. Conversely, changes in eating habits [142] may favor cariogenic environments, inducing oral diseases [74, 143]. Such modifications include taste preferences [61, 144] and craving for sweets [80], need for softer food [73] or high-energy food supplements, and use of syrup-based medications [145].
Poorly cleaned dentures [71, 78] facilitate biofilm accumulation, making them a reservoir for respiratory pathogens and encouraging opportunistic oral infections [146]. The oral cavity may also act as an important reservoir of β-lactam resistance genes facilitating infections with multiple drug resistant bacteria [147]. Oromuscular dysfunction contributes to changes in the oral flora: aerobic Gram negative bacilli have been isolated in the oropharynx of a small sample of PD patients, potentially shedding light on contributing factors to aspiration pneumonia [148], aspiration of oropharyngeal bacteria being linked to pneumonia [6, 149], which is the main cause of death in PD [150]. Enhanced oral hygiene interventions [151–153] may therefore be of interest in PD to mitigate the risk for aspiration and pneumonia, though evidence is still lacking at the time of our writing. Bacteriemia and infections stemming from the oral cavity are also a concern regarding the motor deterioration that may follow systemic infections and inflammation in PD [154].
Along with the increased risk of systemic diseases, periodontal diseases (gingivitis, periodontitis) induce a systemic peripheral inflammation that may contribute to oxidative stress, neurodegeneration and influence PD progression [28, 156]. However, the extent of it needs to be prospectively explored [81].
Finally and yet importantly, delivery systems targeting the mucosa of the oral cavity [157–159] for administering PD drugs have been investigated (piribedil [160]), are under development (ropinirole [161], levodopa/carbidopa or selegiline [162]) or have recently reached the market (apomorphine [163]). As the integrity of the oral mucosa influence the extent of drug absorption [159], oral health disorders definitely need to be addressed.
THE NEUROLOGIST’S ROLE IN SUPPORTING SUCCESSFUL PREVENTION, MANAGEMENT, AND FOLLOW-UP ORAL HEALTH STRATEGIES
Oral and dental “problems caused by PD are accentuated by physicians’ lack of dental knowledge and dentists’ lack of awareness of many of the oral implications of PD” [88]. However, oral diseases are largely preventable (Table 3) even in vulnerable populations [22, 88]. The maintenance of the oral health of PD patients should be of prime importance in providing complete health care [21, 88]. Initial steps have been recently taken, with the publication of clinical practice guidelines [18], although evidence-based recommendations are still scarce [15]. As dentists and dental hygienists play an integral role in providing health care to PD patients, they must be involved as early and often as possible [18, 88].
As PD experts, neurologists andPD nurses have a major role in initiating integrated care cooperation (Fig. 4). They should coordinate appropriate, continuous, individualized, either pharmacological or non-pharmacological interventions and follow-up that target oral health. This should be done through regular patient referral to the other members of the interdisciplinary [19] team: dentists, dental hygienists, community pharmacists, nutritionists/dieticians and speech therapists [20, 164–166]. Due to the progressive nature of PD, early and preventive interventions are necessary. Dental input should be requested at a very early stage, ideally immediately after initial PD diagnosis, to favor prompt and comprehensive interventions, including restorative procedures if needed, and prevent further complications [18, 143]. Periodontally compromised teeth should be evaluated and endodontic treatment considered, as later infection may affect patients’ motor state and response to PD treatment [154]. A conservative approach may be more appropriate in later stages of PD, considering patients’ increased disability in tolerating longer appointments, performing oral hygiene maneuvers and making medical decisions as the disease progresses [18, 83]. Treatment plan should be realistic, collaborative, and continuous with routine prophylaxis and regular visits, ideally 3 to 4 times a year. Interventions should be tailored to the patient’s immediate and long-term dental needs. They should take into account concomitant medications (Table 2), medical prognosis, homecare abilities and settings, as well as psychosocial dimensions (anxiety, cosmetics needs), with input from the neurologist [18, 167]. Dental appointments should be as brief and as comfortable as possible, with an empathetic approach, and an understanding of PD specificities and limitations [18, 108]. Table 4 summarizes recommendations for building PD-suited dental appointments, which can be discussed between the dental and the movement disorders teams.
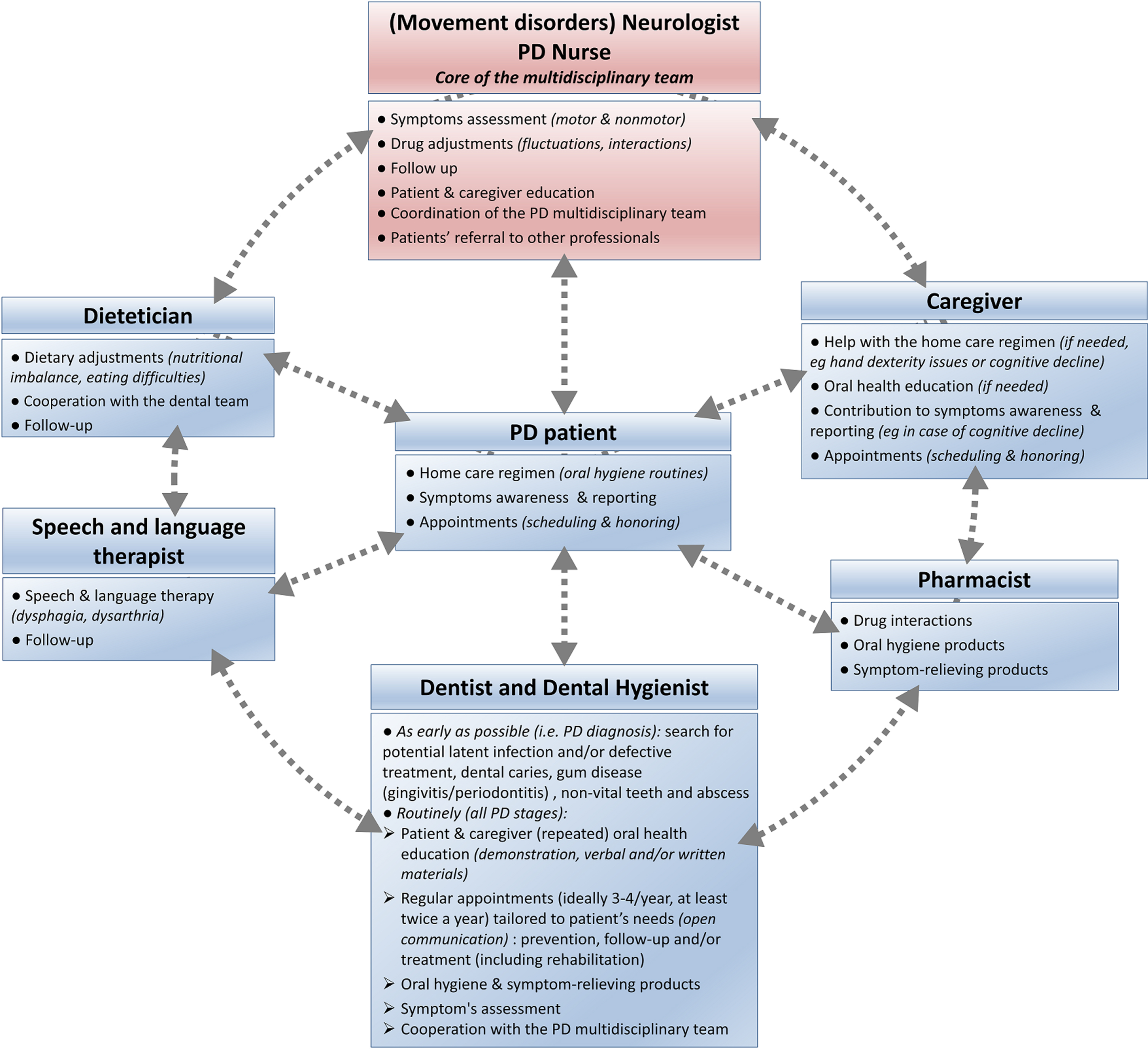
Addressing oral health disorders in Parkinson’s disease: An integrated care approach, coordinated by the movement disorders team (neurologist and PD nurse).
Prevention and management strategies at the dental office: advices from the neurologist to the dental team [3,18,21,26,68,78,79,86,102,106,108,115,136,245–248, 3,18,21,26,68,78,79,86,102,106,108,115,136,245–248]
DBS, deep brain surgery; PD, Parkinson’s disease.
To improve oral hygiene and comfort, appropriate oral healthcare tools (electric toothbrushes) and symptom-relieving products (gels, sprays) are available, and patients choice should be guided by inputs from his dental team and pharmacist [3, 165]. PD patients and their caregivers should be educated and motivated to maintain good oral hygiene routines at home, by both the dental and movement disorders teams [18, 88]. Live demonstrations, verbal and written repeated instructions, and positive reinforcement are effective in PD [78, 136]. By contributing to a better oral health, they decrease the risk of local and general complications—thus reducing the associated health care costs [6].
FUTURE DIRECTIONS AND AREAS FOR FURTHER STUDY
To provide holistic care, clinicians must understand the overlap between neurology, internal medicine [168] and dentistry in PD. As previously mentioned, oral health education and interdisciplinary cooperation are all initiatives that favor patients’ autonomy and would therefore be empowering. The under-reporting of oral health disorders may be partly due to anosognosia. However, the fact that clinicians (neurologist and dentist alike) do not focus on these disorders certainly contributes to such issue [3]. This could be due to a lack of awareness, diagnostic criteria, and available routine screening tools, which need to be properly addressed.
Towards better oral health awareness in PD
Promotion of oral health awareness must be put on the agenda of movement disorders teams. Dentistry should be integrated into PD course of care, alongside neurology and other medical consultations [21]. It is probable that patients would be more active in searching for access to oral care if they were aware of the link between oral and systemic health [169]. Awareness-raising actions involving movement disorders teams, dental teams and PD patients’ associations are to be carried out.
Regarding clinicians’ education, oral health modules developed in cooperation with dentistry schools and targeted to neurologists and PD nurses should be considered, as previous experiences of integrating such educational programs into the medical curriculum were positively received [170–172]. In order to enhance knowledge of PD oral and general specificities and limitations, dental students should join PD rounds in teaching hospitals whenever possible [21]. PD modules [173] targeted to educate dental students and update oral health professionals on caring for PD patients should also be created and routinely implemented. This would increase the cooperation between dentists and neurologists.
Towards better assessment and care
The oral health deserves more attention in PD than a sole assessment of dysphagia, sialorrhea and dysarthria. To overcome the limitations of the MDS-UPDRS scale as a measurement of PD orofacial symptoms [75, 174], there is a need for better tools and questionnaires assessing all the oral symptoms associated with PD that could be used routinely by the neurologist and dental teams [3, 173]. Fully addressing these disorders is a first step for offering optimal treatment [41, 66].
In general, patients and caregivers should be encouraged to be aware of oral symptoms and to report them during consultations. Specific PD patient-rated assessments of discomfort and oral health impact on quality of life should also be developed, to favor symptoms reporting during dental and neurology consults.
The recently published guidelines [18] and this paper are a frame for designing PD-suited dental appointments (Table 4). However, it is critical to keep in mind that dental care, follow-up, and treatment should be patient-specific: PD stage, (non)motor features, comorbidities, medications and psychosocial factors are all to be considered. Successful cases of tailored rehabilitation have been described in the literature [58, 175–179]; this should encourage clinicians to engage in individualized treatment plans, even in complex cases. Home training and oral hygiene programs can be easily implemented and were proved to be successful [136]. Such programs must be broadly offered and can include training of jaw mobility and masticatory function, lip and cheek muscles, as well as use of special toothbrush, denture brush, interdental brush and flossing. Finally, home visiting dental services [180–182] for dental check-up and education could help tackle PD patients’ difficulties in receiving those services at dental clinics. Domiciliary professional oral care [183], with regular professional toothbrushing [184, 185] could improve the quality of oral hygiene and may contribute to better nutritional status and quality of life, although this has yet to be demonstrated in cohorts of PD patients.
Towards better understanding of the mechanisms involved
The lack of methodologically sound studies in the field of neurology in relation to dentistry needs to be corrected [41]. Longitudinal studies with larger samples and standardized assessments are needed to further explore the relationship and pathophysiology of oral health disorders associated with PD [1, 186].
It has recently been demonstrated in vivo that periodontal inflammation exacerbates gut inflammation [123, 187]. The connection between the oral mucosa and the gut (“oral-gut axis”) therefore needs to be investigated in PD, particularly considering the growing concerns regarding the role of the gut-brain axis and gut microbiota in promoting (neuro)inflammation [188], alpha-synuclein pathology [189–191], motor impairments [190, 192] and impairing levodopa metabolism [193–196]. Similarly, Porphyromonas gingivalis a keystone pathogen in periodontitis, along with Treponema denticola, have recently been associated with Alzheimer’s disease and PD, suggesting a possible bacterial involvement in the development and course of neurodegenerative disorders [197, 198]. Poor oral health and periodontitis have indeed been associated with an increased risk of developing PD later in life in several recent studies [127, 199–201] whereas dental scaling was associated with a decreased risk [202], pointing towards a “multiple-hit” hypothesis [203]. Conversely, the impact of PD on oral microbiota [204], chronic inflammation, neurodegeneration and the gut brain axis need to be thoroughly explored ([81, 148], BUCCO-PARK study, NCT03965390 [4]). 4 Taken together, these inter-connections highlight the relevance of an “oral-gut-brain” axis that needs to be further explored.
Microbiota on dentures or in weakened oral cavity needs to be evaluated as a potential reservoir for respiratory multiple drug resistant pathogens in PD patients [3, 146]. Additionally, the effect of antiparkinsonian drugs (drug class and levodopa equivalent daily dose, Table 2) on the oral ecosystem needs to be better characterized.
Towards new diagnostic and predictive tools: Landmark symptoms and biomarkers?
The chronology and combination of orofacial symptoms may give insights into disease progression [3, 48]. The Swedish “Jönköping Parkinson Registry” (N = 314 idiopathic PD patients, prospective study led between 1998 and 2014) has recently highlighted that orofacial symptoms were correlated with PD severity, symmetry of motor disturbances, and autonomic disorders. The more severe orofacial burden was at baseline, the faster patients progressed to more advanced stages [25]. As stated earlier, some orofacial symptoms occurs early in the disease course (hyposmia [31], hyposialorrhea [33], tongue tremor [34–36], upper-body axial symptoms [37]), whereas others are more apparent and frequent in advanced stages (dysphagia, drooling). Considering that only a few of these disorders have been thoroughly assessed in PD, more longitudinal studies with larger stratified samples are needed to further explore their predictive value. A recent Korean nationwide population-based cohort study has thus suggested that an increased number of missing teeth may be an important risk indicator of new-onset PD [205].
Identifying reliable biomarkers is of crucial importance in PD, particularly regarding the potential development of neuroprotective treatment strategies. Readily accessible, saliva allows non-invasive and convenient sampling for proteomic analysis in PD [206, 207]. Altered expression of α-synuclein, DJ-1 protein and microRNAs have been reported in the saliva of PD patients. They could become promising biomarkers for PD severity and differential diagnosis [206–212]. Correlating such peripheral biomarkers with the emergence and severity of clinical signs would be a further asset: a decrease in substance P saliva levels has been associated with early dysphagia [213]. Biopsies of the submandibular gland to detect pathological α-synuclein [214–216] may also hold promises, though such findings need to be supported by larger cohorts.
Finally, the oral microbiota may offer new insights into the pathophysiology of PD [81, 82], with a probable evolution of its composition throughout the course of the disease. A recent report suggests correlations between oral microbiota alterations and functional changes (cognition and balance) in early stage PD [81]. The currently undergoing BUCCO-PARK study (NCT03965390) is expected to provide longitudinal data of a cohort of 50 French PD patients in a prospective manner. Similarly to other diseases [217–220], future studies may even be able to identify microbiota signature associated with disease severity, clinical symptoms or PD medications in the future. Additionally, the possible interplay between the oral, nasal and gut microbiota demand further attention [82, 221–223].
CONCLUSION
Though closely located, the central nervous system and oral cavity are rarely assessed together. In PD, orofacial manifestations overlap neurology and dentistry, with direct and indirect impact on patients’ general well-being and quality of life, regardless of disease stage. There is still much to do to: 1) improve oral health awareness among medical professionals, patients and caregivers [169] and develop efficient interdisciplinary cooperation [166], 2) specifically assess and treat oral health disorders in PD patients, 3) understand the pathophysiological and chronological mechanisms underlying these disorders, and 4) determine whether the oral cavity may become an easy access valuable tool for diagnosis and prognosis. As a whole, neurologists should be aware of the clinical implications of oral health disorders in PD and should initiate and coordinate integrated care. On their own, dentists need to get more involved into the care of PD patients and adjust to a personalized approach for patient-specific needs. Familiarizing neurologists and dentists with oral health disorders in PD is critical for better recognition, diagnosis, and correct decision upon treatment. The consideration of the interplay between oral and general health is crucial to understand what is at stake for PD patients. Early, proactive, and preventive interventions are necessary to avoid reactive care stemming from oral and dental diseases accumulation. Of course, many of these statements apply to other neurological diseases involving cognitive or movement disorders, such as Alzheimer’s disease, multiple sclerosis, or Huntington’s disease. This paper therefore advocates to end the neglect of oral health in neurology, and to address the general paucity of strong literature. Considering the two-way relationship between oral and general health, and underlying multimorbidity, it is time to bridge the gap between neurology and dentistry, avoiding single provider tunnel vision and siloed care by working in concert towards patients’ overall health.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank Shao Bing Fong (PhD, The University of Melbourne) for proof-reading the manuscript. The authors also wish to thank France Parkinson for supporting the BUCCO-PARK project. Finally, MV and MA wish to thank France Développement Electronique (FDE) for its continued support and Homeperf for covering the expenses related to the presentation of this work at the 71st American Academy of Neurology meeting.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CONFLICT OF INTEREST
The authors declare that there are no conflicts of interest relevant to this work.
France Parkinson (a French Parkinson’s disease Association) financially supports the BUCCO-PARK clinical trial (Rennes, France -ClinicalTrials.gov Identifier: NCT03965390), an interdisciplinary research project based on some of the findings summarized in this paper.
PUBLICATION HISTORY
The intention and preliminary results of this review project have been presented as a poster at the 71st American Academy of Neurology (AAN) meeting in Philadelphia, USA on May 2019: Manon Auffret, Vincent Meuric, Emile Boyer, Martine Bonnaure-Mallet, Marc Verin. Oral cavity disorders in Parkinson’s disease: more than meets the eye (P5.8-038). Neurology Apr 2019, 92 (15 Supplement) P5.8-038. ![]()
Periodontal diseases are characterized by inflammation of tissues surrounding the teeth. They are of two types: gingivitis (non-destructive and reversible inflammation usually associated with bacteria accumulation) and periodontitis (chronic and dysregulated inflammation associated with multispecies bacterial community characterized by irreversible alveolar bone loss surrounding the teeth, leading to its mobility and loss)
