Abstract
The increasing prevalence of neurodegenerative conditions such as Parkinson’s disease (PD) and related mobility issues places a serious burden on healthcare systems. The COVID-19 pandemic has reinforced the urgent need for better tools to manage chronic conditions remotely, as regular access to clinics may be problematic. Digital health technology in the form of remote monitoring with body-worn sensors offers significant opportunities for transforming research and revolutionizing the clinical management of PD. Significant efforts are being invested in the development and validation of digital outcomes to support diagnosis and track motor and mobility impairments “off-line”. Imagine being able to remotely assess your patient, understand how well they are functioning, evaluate the impact of any recent medication/intervention, and identify the need for urgent follow-up before overt, irreparable change takes place? This could offer new pragmatic solutions for personalized care and clinical research. So the question remains: how close are we to achieving this? Here, we describe the state-of-the-art based on representative papers published between 2017 and 2020. We focus on remote (i.e., real-world, daily-living) monitoring of PD using body-worn sensors (e.g., accelerometers, inertial measurement units) for assessing motor symptoms and their complications. Despite the tremendous potential, existing challenges exist (e.g., validity, regulatory) that are preventing the widespread clinical adoption of body-worn sensors as a digital outcome. We propose a roadmap with clear recommendations for addressing these challenges and future directions to bring us closer to the implementation and widespread adoption of this important way of improving the clinical care, evaluation, and monitoring of PD.
INTRODUCTION: THE “VISION”—ARE WE THERE YET?
At the 2013 World Congress of the International Society of Posture and Gait Research, a keynote speaker predicted that digital health technology such as body-worn sensors (BWS) would soon become a routine, widely used tool to augment the clinical examination of patients with Parkinson’s disease (PD) and, more importantly, enhance patients’ quality of life [1]. Accumulating evidence at that time demonstrated that low-cost, easy-to-use BWS could be used in the clinic to provide new information and add needed objectivity to the assessment of PD motor symptoms, gait, and mobility. Use of BWS could, therefore, help patients by enhancing their therapeutic management, function and quality of life, and offer them a personalized approach to their symptoms. Moreover, an emerging, exciting set of studies demonstrated that continuous, “24/7” remote monitoring empowered by BWS had the potential to measure, characterize, and quantify both subtle and large changes in mobility and other critical motor symptoms of a patient with PD, providing a robust comprehensive map of the patient’s function and its changes over time. In this vision, a patient would receive a small package in the mail containing a BWS several weeks before a routine clinical exam. After following simple instructions and wearing the device continuously for one week, the information collected would be uploaded to a cloud, automatically analyzed, summarized, sent to the clinician for review and provide feedback to the patient. At the clinical exam, the healthcare professional would read an objective, detailed report on the patient’s motor function that could be compared and contrasted to the results of previous years, even before the patient took a single step into the clinic. In this way, the clinical visit would be transformed into a more productive and informed meeting, enhancing clinical care in a truly personalized manner.
This optimistic vision has not yet been realized [2, 3]. Nonetheless, at a time when COVID-19 is wreaking havoc throughout the globe, the need has become even greater. In this short review, we provide an overview of the current use of BWS (accelerometers, inertial measurement units (IMUs)) for the remote monitoring of PD motor symptoms, summarize the challenges that must be overcome to achieve that potential, and outline steps that should and are being taken, in the long road ahead [2] to address this important opportunity for improving the evaluation and monitoring of PD motor symptoms.
STATE OF THE ART: WHERE ARE WE AT?
The rapidly expanding field of remote monitoring has been the subject of several recent systematic reviews [4–7]. Here, we provide an overview of recent representative work (2017–2020) on remote monitoring of PD motor symptoms using BWS (Table 1). We report studies using BWS for remote monitoring (real-world, at-home and in the community) in both unsupervised or scripted conditions, in order to quantify digital outcomes, focusing on papers related to motor symptoms and motor complications: tremor, bradykinesia, dyskinesia, postural instability, gait disturbances and turning, falls risk, freezing of gait (FoG) and physical activity. Using a previously proposed framework [8], in Table 1 we report:
Representative studies examining remote monitoring of Parkinson’s disease (PD) motor symptoms using body worn sensors (BWS), between 2017 and 2020. Validation of digital outcomes has been classified ‘yes’ for each of the following criteria: 1. criterion validity: if digital outcome has been validated against a gold standard reference in the study cited, or in previous studies; 2. construct validity: if digital outcome has been validated (e.g., correlated) against clinical scales (convergent validity) and/or it has shown significant differences between groups (discriminant validity) in the study cited, or in previous studies. Digital outcome regulated/qualified has been classified ‘yes’ if BWS and/or digital outcome has received FDA (510K1) or EMA2 positive decision/qualification
*Night excluded. ABC, Activity specific Balance Confidence scale; ADLs, Activities of Daily Living; CL, Controls; DL, Deep Learning; ECG, Electrocardiogram; F, Fallers; FES, Falls Efficacy Scale; H&Y, Hoehn and Yahr; MET, Metabolic Equivalent; ML, Machine Learning; MS, People with Multiple Sclerosis; NF, Non-fallers; NFOG-Q, New freezing of gait questionnaire; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s disease Rating Scale; OA, Older Adults; PAL, Physical Activity Level; PD, People with Parkinson’s disease; PKG, Parkinson’s KinetiGraph; SVM, Super Vector Machine; TUG, Timed Up and Go; UPDRS-III, Unified Parkinson’s disease Rating Scale, Part III. 1https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm. 2https://www.ema.europa.eu/en/human-regulatory/research-development/scientific-advice-protocol-assistance/novel-methodologies-biomarkers/opinions-letters-support-qualification-novel-methodologies-medicine-development
the clinical concept of interest; quantified digital outcomes; validity: criterion validity: digital outcome validated against a reference system, construct validity: digital outcome validated against clinical scales (convergent validity) and/or showed known groups differences (discriminant validity)); digital outcome regulatory/qualification status assessed by regulatory bodies (e.g., EMA, FDA) [9, 10].
Tremor, bradykinesia, dyskinesia, motor fluctuations
BWS have been used to automatically detect and evaluate tremor, bradykinesia, dyskinesia and on/off medication state [11–17]. The main techniques for identification of these symptoms are based on machine learning (ML) models (e.g., support vector machines (SVM)). Models are usually fed with digital outcomes that are signal-based features (e.g., frequency domain) extracted from the BWS.
In terms of criterion validity, studies tend to validate the digital outcome against a reference system (e.g., videos, self-report), using mainly ML techniques, showing good accuracy (>90%) [13, 17]. Construct validity is generally tested utilizing clinical scales (e.g., Unified Parkinson’s disease Rating Scale (UPDRS)), but is less explored.
Although preliminary results are promising for some digital outcomes, excluding Farzanehfar et al. [12], these studies include a limited number of subjects (≤25) and, therefore, the generalizability of ML models and related validity for clinical adoption is problematic. We note using BWS to study rigidity, one of the cardinal symptoms of PD, is especially challenging [18].
Postural instability, gait disturbances, and turning
The importance of postural instability, gait, and turning as diagnostic, prognostic, and progression markers in PD is well recognized [19–21]. Nonetheless, static balance tests are usually confined to laboratory environments. Difficulty in identifying and discriminating periods of static, “quiet” standing balance from sedentary behavior during everyday activities (especially using single BWS on the trunk) and achieving a “totally unsupervised” postural instability assessment is challenging.
Evaluations of digital outcomes are based on: first identification in the BWS signal of the clinical concept of interest (e.g., gait, turning) using either ML methods or previously validated signal-based methods (e.g., methods developed from lab-based validation against gold standards); and second on the quantification of digital outcomes in the identified segments of the signals. Digital outcomes often include signal-based features (e.g., extracted from the BWS signal – frequency domain) or clinically relevant and “translatable” features (e.g., walking speed).
Recent studies on the construct validity of turning and gait corroborated that real-world gait and turning performances of PD were impaired (e.g., slower, more variable, and with lower cadence), compared with older adults [22–24] and reported moderate correlations with clinical scales (e.g., UPDRS) [25, 26]. Only a few studies have reported criterion validity for digital outcomes, and this was limited to in laboratory or home-like environments [25, 28]. Real-world validation remains challenging and relies mainly on videos as a reference.
Falls risk, freezing of gait
BWS can help advance our understanding of fall risk. For these concepts of interest, the main techniques are again use of ML models to identify relevant segments of the BWS signals, and then quantification of digital outcomes by using signal-based features or validated clinically relevant digital outcomes (e.g., walking speed, variability).
Results show that quality (micro) and quantity (macro) digital outcomes describing gait and turning are associated not only with falls status (fallers vs. non-fallers) but also with PD specific characteristics (e.g., PD fallers showing higher variability than older adult fallers) [27, 29]. Real-world digital outcomes show promising results to quantify novel composite indexes (e.g., combining information on falls rate with walking activity) sensitive to change in fall risk in intervention studies [30–32]. Despite the availability of real-world falls repositories [33], methodologies for real-world automatic fall detection remain challenging, prone to the detection of false positives [34], and not thoroughly addressed in PD [35].
FoG is also notoriously difficult to fully replicate and detect, increasing the potential value of remote monitoring. To elicit FoG episodes, studies have tested participants in both ON and OFF conditions during scripted tests in the lab and home and used ML models [36–38]. The sensitivity and specificity for FoG detection both increase (88.09% and 88.01% respectively) when personalized (BWS data labeled by the participant, so user-dependent) rather than generic (automatic, user-independent) models are used [36, 37]. Comparisons between freezers and non-freezers indicate that the “quality of turning” digital outcome (e.g., turning angle smaller), rather than quantity of mobility, was impaired in PD freezers [25].
While construct validity is often reported (in terms of moderate relationship with clinical scales and discriminant validity) for fall risk and FoG, criterion validity is often limited to testing in the laboratory, rather than real-world environments for falls risk. Videos and ML techniques are mainly devoted to FoG.
Physical activity
Daily-living physical activity is one of the more mature applications of BWS. Outcomes such as the intensity of movement (e.g., energy expenditure, METs, step count) and temporal periods (bouts) of physical activity can be quantified [39]. Quantification of physical activity DMOs are based on features that describe the “magnitude” of BWS signals (e.g., counts, METs) or walking related features (based on identification of events—e.g., steps in the BWS signal for step count).
Commercial devices are widely utilized in PD for quantifying physical activity [40–42]. Although people with PD have lower levels of physical activity compared to older adults (discriminant validity), construct validity provides contrasting results with either no [42], moderate [43] or strong [44] relationships with clinical scales (e.g., UPDRS, Hoehn and Yahr staging) [40]. Criterion validity was again limited to laboratory-based tests (e.g., MET) rather than real-world environments [44], where the use of self-report diaries limit validity assessment due to subjectivity and recall issues [42].
Current limitations
Across the clinical concepts of interests presented, the vast majority of studies use a single or combination of BWS for data logging and off-line analysis with developed analytics. Only a few examples developed connected systems (e.g., multiple sensors systems, smartphones) and online/m-health (“cloud”) platforms to achieve true remote monitoring in real-time (e.g., REMPARK, PD_Monitoring) [14, 45]. These m-health platforms have been used in small studies [45], focusing on selective aspects (e.g., ON-OFF state). Although good usability and user satisfaction results were reported, feasibility aspects (e.g., limited sensor battery time of 20 hours) for clinical adoption were not thoroughly investigated.
Generally, studies using BWS are cross-sectional, with only a few examples assessing the ability to detect change and responsiveness in longitudinal or interventional studies [30, 46]. This aspect needs to be better explored. Only when digital outcomes derived from BWS demonstrate robust criterion and construct validity and equal or superior clinimetric properties (e.g., sensitivity to change, prediction of outcomes) compared to conventional clinical outcomes will their application become widespread.
Importantly, except for the Parkinson’s Kinetigraph for motor symptoms (tremor, bradykinesia and dyskinesia) [12] and Fitbit for electrocardiogram App [40], another common characteristic across clinical concepts of interest and BWS is the lack of qualification reports accepted for a PD context of use by regulatory bodies (e.g., EMA, FDA). This absence precludes the widespread clinical adoption of BWS and their related digital outcomes [2]. Therefore, despite the promise of BWS for remote monitoring, technical, validity, and regulatory limitations remain significant barriers to their uptake.
FUTURE PERSPECTIVES: HOW CAN WE MOVE FORWARD?
This brief review of the state-of-the-art shows that, although promising, widespread adoption of BWS in clinical settings is yet to transpire, likely because of several factors. To date, there has been no comprehensive demonstration of criterion (“technical”) and construct (“clinical”) validity, with differences in BWS and measurement techniques accounting for differences in reporting of results of the same mobility variables [7]. As highlighted earlier, the majority of algorithms that have been developed have not been validated in real-world conditions, which may be due in part to a lack of gold-standard references against which to test. Establishing technical and clinical validity, in addition to the demonstration of feasibility and usability of BWS in patients is essential to obtain qualification approval by regulatory bodies, and, as a consequence, more widespread use of BWS by clinicians [3].
Moreover, to truly transform clinical and research conventions, there needs to be sufficient evidence to show that remote monitoring is clearly “better” in some way (e.g., cost, discriminative and predictive value, clinimetric properties, healthcare economics) than traditional scales and methods. Last year, the Movement Disorder Society (MDS) Task Force on technology published concrete steps to facilitate adoption of BWS in clinical practice [3]. Nonetheless, although there are many examples and attempts in the growing literature to address selective aspects of the Task Force recommendations (e.g., validity and utility), definitive prospective and comprehensive studies are lacking. To move this field forward significantly, previous suggestions included the need to improve cross-discipline communication and larger collaborative efforts. Recent Innovative Medicines Initiative projects (e.g., Mobilise-D (https://www.mobilise-d.eu/), IDEA-FAST (https://idea-fast.eu/)) are paving the way to achieve this.
To address this need, we tried to summarize previous recommendations [2, 48] by proposing a roadmap with clear milestones to guide the practical clinical adoption of BWS and digital outcomes (Fig. 1):
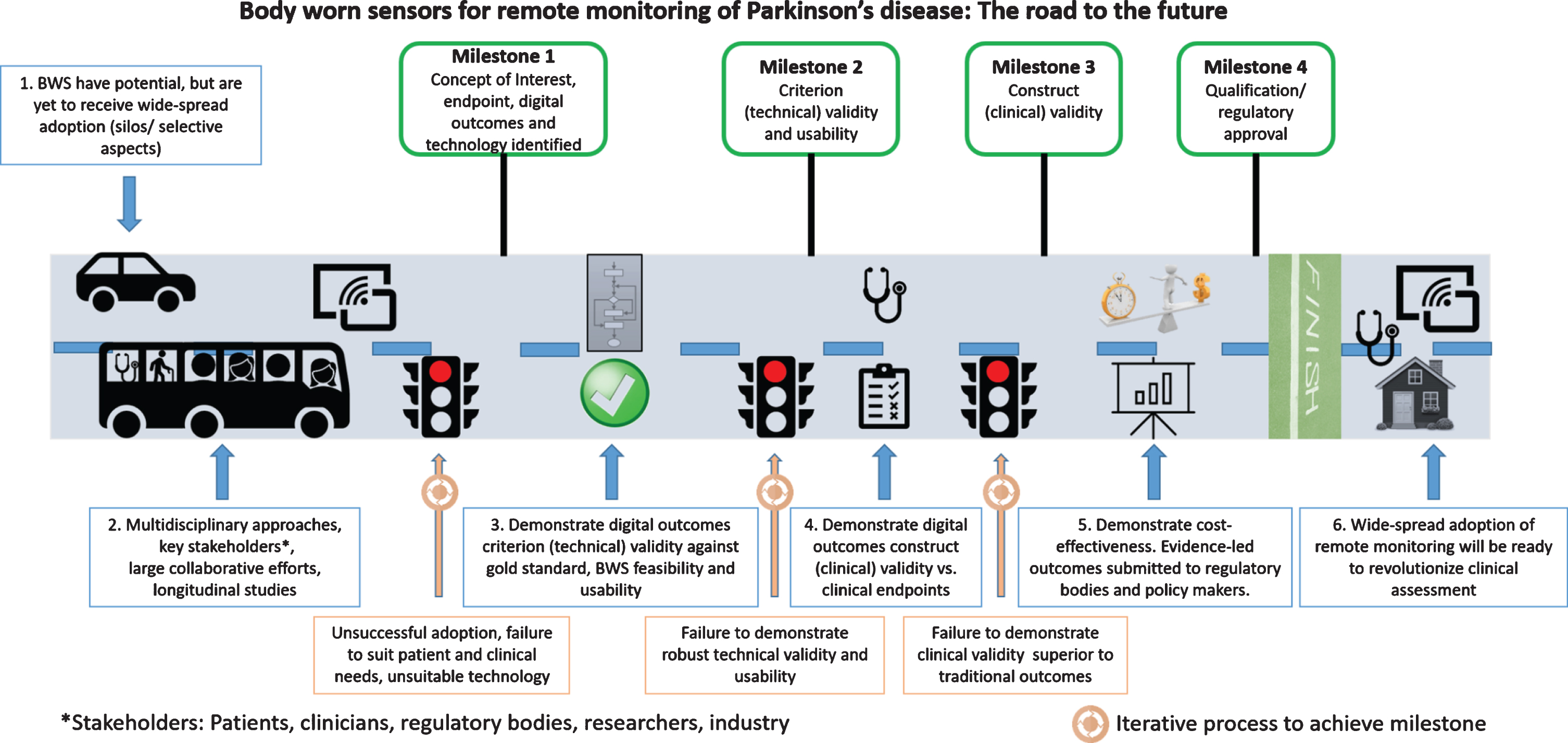
Roadmap for adoption of body worn sensors (BWS) and digital outcomes in clinical practice.
A joint effort between clinicians and end-users (e.g., patient groups [47]) should identify critical relevant clinical concepts of interest (e.g., gait), related digital outcomes (e.g., real-world walking speed), the context of use, and clinical endpoints (e.g., falls risk). Select or develop appropriate technology (BWS) with key stakeholders (industry, res-earchers, clinicians, end-users) for quantification of identified digital outcomes. If not already established, demonstrate criterion (technical, e.g., cross-sectional studies) validity of the digital outcome against a gold-standard reference, in real-world conditions, feasibility, and usability of the BWS for the end-users. This could be achieved, for example, by selecting a digital outcome (e.g., walking speed) and collecting data under controlled conditions with BWS and a gold standard that can quantify the same digital outcome and that can then be used also in real-world conditions. Demonstrate at least equivalent, but preferably superior construct (“clinical”, e.g., longitudinal studies) validity of the digital outcome with respect to traditional measures. This could be done, for example, by demonstrating, in a longitudinal study, that the selected digital outcome at baseline (e.g., real walking speed) has stronger correlation (or predictive power) with the clinical endpoint of interest (e.g., perspective number of falls) than clinical scales or questionnaires (e.g., UPDRS III). Describe the context of use and validation work for submission to qualification/regulatory bodies for approval (EMA, FDA). Demonstrate cost-effectiveness (e.g., saving time of the clinician, improving the quality of life of patients). The barrier for adoption of low-cost solutions is lower than that of solutions that require large monetary investments. This could be demonstrated by carrying out a cost-effectiveness analysis, for example, by quantifying the cost of the BWS (both in monetary and time/effort terms) versus that of a gold standard or clinical assessment and showing evidence of a lower healthcare expenditures and better outcomes achieved with BWS (e.g., as described in recommendation 3).
CONCLUSIONS
Although BWS and digital outcomes have shown potential for clinical management, they have not yet achieved widespread clinical adoption. We can imagine a future where true remote monitoring of digital outcomes is used to enhance PD diagnosis, monitor progression, and facilitate clinical management. We hope that the recommendations and practical roadmap that are outlined in Fig. 1 will help to move the field forward toward that vision and to better care and monitoring of people with PD.
CONFLICT OF INTEREST
JH reports having submitted a patent for assessment of mobility using wearable sensors in Parkinson’s disease. The intellectual property rights are held by the Tel Aviv Medical Center. All of the authors declare that they have no conflicts of interest.
Footnotes
ACKNOWLEDGMENTS
The work was supported by Innovative Medicines Initiative 2 Joint Undertaking (JU) under Grant agreement 820820 (Mobilise-D). The JU receives support from the European Union’s Horizon 2020 research and innovation programme and the European Federation of Pharmaceutical Industries and Associations (EFPIA). SDD, AJY and LR are also supported by the Newcastle Biomedical Research Centre (BRC) based at Newcastle upon Tyne and Newcastle University. The work was also supported by the NIHR/Wellcome Trust Clinical Research Facility (CRF) infrastructure at Newcastle upon Tyne Hospitals NHS Foundation Trust. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care or the funders.
