Abstract
Background:
People with Parkinson’s disease and freezing of gait (FOG; freezers) suffer from pronounced postural instability. However, the relationship between these phenomena remains unclear and has mostly been tested in paradigms requiring step generation.
Objective:
To determine if freezing-related dynamic balance deficits are present during a task without stepping and determine the influence of dopaminergic medication on dynamic balance control.
Methods:
Twenty-two freezers, 16 non-freezers, and 20 healthy age-matched controls performed mediolateral weight-shifts at increasing frequencies when following a visual target projected on a screen (MELBA task). The amplitude and phase shift differences between center of mass and target motion were measured. Balance scores (Mini-BESTest), 360° turning speed and the freezing ratio were also measured. Subjects with Parkinson’s disease were tested ON and partial OFF (overnight withdrawal) dopaminergic medication.
Results:
Freezers had comparable turning speed and balance scores to non-freezers and took more levodopa. Freezers produced hypokinetic weight-shift amplitudes throughout the MELBA task compared to non-freezers (p = 0.002), which were already present at task onset (p < 0.001). Freezers also displayed an earlier weight-shift breakdown than controls when OFF-medication (p = 0.008). Medication improved mediolateral weight-shifting in freezers and non-freezers. Freezers decreased their freezing ratio in response to medication.
Conclusion:
Hypokinetic weight-shifting proved a marked postural control deficit in freezers, while balance scores and turning speed were similar to non-freezers. Both weight-shift amplitudes and the freezing ratio were responsive to medication in freezers, suggesting axial motor vigor is levodopa-responsive. Future work needs to test whether weight-shifting and freezing severity can be further ameliorated through training.
INTRODUCTION
People with Parkinson’s disease (PD) have poordynamic postural control associated with an incre-ased risk of falling [1], and this is more prevalent in people with PD who suffer from freezing of gait (FOG; freezers) [2]. FOG can be defined as a brief, episodic absence or marked reduction of forward progression of the feet despite the intention to walk [3]. FOG often occurs when steps are made with high frequency (cadence) and small amplitude (step length) [4]. Dynamic postural control is defined as the ability to control the center of mass (CoM) during continuo-usly changing conditions, including the transfer ofbody weight between the legs (weight-shifting) during walking. Effective treatment of axial symptoms, such as dynamic postural instability and FOG, req-uires insight into how these symptoms interfere with each other.
Tasks that require mediolateral weight-shifting, such as turning, stepping in place and gait initiation,have demonstrated greater postural instability in freezers than in non-freezers [2]. Turning is also the most consistent trigger of FOG, especially when full turns (360°) are performed at fast speed [5]. The close link between FOG and turning is also testified by the fact that the ratio between power in the freezing band (3–8 Hertz (Hz)) and gait band (0.5–3 Hz) during repeated 360° turning was shown to be a valid measure of freezing severity [6]. FOG-episodes during turning were preceded by a less medial position of the CoM and a reduction of step width [7]. Furthermore, during daily-life turning, freezers exhibited a greater mediolateral jerkiness than non-freezers, especially during large-angle turns [8]. In addition, small and inefficient mediolateral weight-shifts have been sh-own to be related to freezing during a stepping in place task, even at a self-selected speed [9]. Med-iolateral weight-shifting is also an important component in more automated tasks, such as gait initiation, in which an initial posterior weight-shift (the im-balance phase) is followed by a weight-shift in the mediolateral direction towards the stance leg, so that the swing leg can be lifted of the ground (unloading phase). Generally, these anticipatory postural adjustments (APAs) were demonstrated to be hypometric in people with PD [10, 11]. The mediolateral weight-shift of the unloading phase was found to be smaller in freezers compared to non-freezers during self-ini-tiated gait without FOG, while co-contraction of thetensor fasciae latae was higher in freezers [12, 13]. This co-contraction might be a compensatory strategyto protect against postural instability in the mediolateral direction. Of note, other studies found no fre-ezing-related APA deficits for self-initiated uncued gait [14–16], but the first step was wider in freezers[14], which may also point to mediolateral imbalance. As postural control and stepping are tightly coupled during turning, stepping in place and gaitinitiation, it is not possible to distinguish if impaired weight-shifting is due to deficits in postural control or step generation. Hence, studying a dynamic balance task from which step generation is removed, may further unravel the common-end mechanisms between FOG and dynamic balance disturbances.
Multimodal therapy is needed to treat axial symptoms in people with PD [17]. Therefore, it is critical to map which axial symptoms are responsive and which are unresponsive to dopaminergic medication. Levodopa has been shown to reduce the number andduration of FOG episodes in most people with PD, but did not ameliorate FOG altogether [18, 19]. Also, turning improved with levodopa in both freezers and non-freezers, [20], but its effect on turning was found to be smaller than on straight-line gait [21]. As for APAs, the imbalance phase showed to be levodopa-responsive while the unloading phase was unresponsive [11]. By and large, the above-men-tioned axial problems involving stepping proved only partially responsive to dopaminergic medication [21, 22] and showed a diverse responsiveness across various postural or gait domains. Therefore, determining the effect of dopaminergic medication on a dynamic balance task without a stepping component in freezers and non-freezers may clarify the inconsistent results found so far on the dopaminergic involvement in freezing-related axial problems.
The aim of this study was to compare mediolateral weight-shifting in freezers, non-freezers and healthy controls subjects. For this purpose, we used the mediolateral balance assessment task (MELBA) in which repetitive weight-shifting is required at an increasing frequency to follow a visual target withthe CoM [23]. This task has been developed and validated in older people, and appears more sensitive to dynamic postural control decline than traditionalparadigms [23]. As freezers show mediolateral dyn-amic balance impairments and repetitive movements at increasing frequencies can elicit freezing [24], we hypothesize that MELBA outcomes will be more affected in freezers than in non-freezers and may even elicit FOG when OFF medication. Second, we tested the impact of dopaminergic medication on mediolat-eral weight-shifting, expecting that non-freezers will improve their performance more than freezers. Finally, an exploratory correlation analysis was carried out between the MELBA outcomes and the clinical characteristics.
METHODS
Participants
Twenty-two freezers, 16 non-freezers, and 20 healthy control subjects participated in this study. Observation of FOG during testing or self-reported freezing on item 1 of the new freezing of gait questionnaire (NFOG-Q) was used to determined FOG status in the PD cohort. The NFOG-Q item 1 involves indicating whether FOG has been experienced in the past month after showing a video of different types of freezing episodes, including very mild ones [25]. The inclusion criteria for PD were: 1) Diagnosis of idiopathic PD based on the UK PD Brain Bank criteria; 2) Stable medication regime for the past month; 3) Mini-Mental State Examination Score > 23/30; 4) Ability to stand unassisted for ≥20 minutes. Control subjects were screened according to inclusion criteria 3-4. Participants were excluded if other diseases were present that might interfere with the experiment. The Medical Ethics Committee of UZ/KU Leuven Research (B322201628931) approved the study and written informed consent was obtained from all participants prior to enrolment.
Study design
The present study had a cross-sectional between-group design. People with PD visited the lab twice,once ON and once OFF their dopaminergic medications (after overnight withdrawal) with approximately four weeks in-between visits. As symptomatic benefits of levodopa may persist for up to two weeks, the term “OFF state” actually refers to a partial OFF-state, which for sake of brevity is termed here as “OFF”. The order of ON and OFF tests was counterbalanced per PD sub-group. A counterbalanced design was chosen to avoid the influence of order effects. Controls were assessed once and compared to the PD sub-groups OFF medication to gain a better insight in the impact of PD on dynamic postural control.
Measurements
MELBA Task
Participants stood with the arms crossed on the chest in the middle of a fixed platform in a computer-assisted rehabilitation environment and were wearing a safety harness (CAREN, Motek Medical BV, Amsterdam, The Netherlands). The MELBA task, originally developed by Cofre Lizama et al. [26], requires participants to track a mediolaterally moving visual target at increasing frequencies with their CoM by shifting their weight mediolaterally from one leg to the other [23]. The target and the CoM were displayed as a white and red sphere, respectively, in real-time on a large screen in front of the participants (see Fig. 1 for experimental setup, see Supplementary Material 1 for CoM calculation). The feet position was personalized by setting the between-heels distance to 11% of body height and a 14 degrees between-feet angle. Participants were instructed to stand straight and were told not to lock their knees. Participants were instructed to track the target by moving their whole body sideways as an inverted pendulum (without moving their feet) for as long as possible, even if they could no longer follow the target precisely. The maximum frequency of the MELBA target was set to 1.3 Hz as opposed to 2 Hz previously used in older adults [23] to keep the task manageable for people with PD. The target signal consisted of 13 frequency blocks, starting at 0.1 Hz and increased with steps of 0.1 Hz per block. The first block lasted 20 seconds, blocks two until eight lasted 10 seconds and blocks nine until thirteen lasted 5 seconds in order to gradually increase the difficulty of the test to help patients feel comfortable and engage with the task. The amplitude of the target signal was normalized for each subject at 50% of stance width. One practice trial was performed, followed by three test trials. The averages of the three test trials were used to determine the MELBA outcome measures.
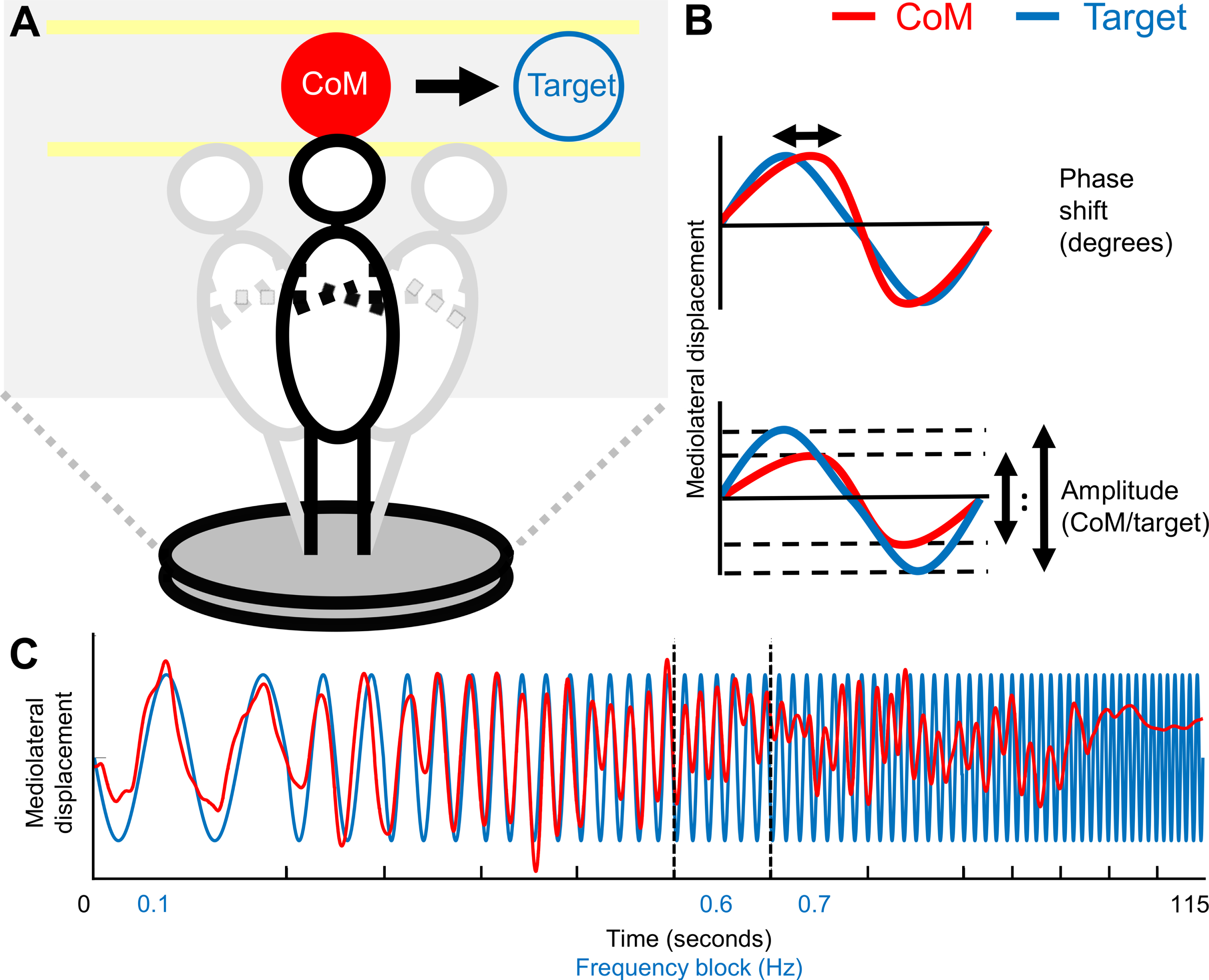
Visual representation of the mediolateral balance assessment task. A) Experimental setup. The participant is moving the center of mass (CoM), visualized in real time by the red sphere, in the mediolateral direction to follow the target sphere by shifting weight from one leg to the other. B) Visualization of the phase shift and amplitude outcome measures. The phase shift represents the delay of the CoM with regards to the target in degrees and the amplitude represents the ratio between the mediolateral displacements of the CoM and the target. C) Visualization of the mediolateral target and CoM displacement over the 115 seconds of a trial. The frequency blocks are separated by the ticks on the x-axis. The CoM of a randomly chosen freezer is also displayed. Dashed vertical lines represent breakdown of weight-shift amplitude (0.6 Hz) and phase shift (0.7 Hz). CoM, center of mass.
Data analysis was performed with Matlab version 2018B (The Mathworks, Natick, MA, USA). MELBA performance was characterized per frequency block by calculations of the difference in phase shift and amplitude (i.e., mediolateral displacement) between the visual target and CoM, both in the frequency domain [23] (see Fig. 1B). Phase shift and amplitude were determined for the target frequency present in each block. The phase shift characterizes how much the CoM lags behind the target (in degrees), with other words it captures the delay of the CoM with regards to the target. When the target is perfectly tracked, there is no delay and the phase shift would be 0. The absolute value of the phase shift was used for the analysis, so that a positive or negative phase shift was interpreted similarly. The amplitude outcome represents the ratio between the mediolateral displacement amplitudes of the CoM and the target. When the CoM amplitude is exactly the same as the target amplitude, the amplitude is 1. When the participant cannot follow the target any longer, the CoM will lag behind and/or show a smaller mediolateral displacement and thus a smaller amplitude.
Three outcomes for phase shift and amplitude were determined. The phase-shift outcomes were: 1) Phase shift breakdown: the frequency at which the phase shift increased over 90 degrees; 2) Mean phase shift: the mean phase-shift over the frequency blocks from the start frequency (0.1 Hz) until phase shift breakdown and 3) Start phase shift: the phase-shift at the start frequency. Similarly, amplitude outcomes were: 1) Amplitude breakdown: the frequency at which the amplitude fell below 0.5 of the target amplitude, 2) Mean amplitude: the mean amplitude from the start frequency until amplitude breakdown and 3) Start amplitude: the amplitude at the start frequency. For the breakdown outcomes, recovery of phase shift and amplitude in the first three blocks was allowed. Start phase shift and start amplitude were calculated in order to determine if possible differences in the mean amplitude or mean phase shift were already present at onset. The phase shift and amplitude were calculated with a linear constant coefficient transfer function between the CoM and target, using a Welch algorithm over windows of 0.25 times the length of the target and with 90% overlap between windows [23]. A coherence analysis between the CoM and target was used to determine the validity of phase shift and amplitude outcomes (see Supplementary Material 1 for more details).
Secondary outcomes
Participants performed alternating 360° right and left turns on the spot for one minute at self-preferred speed while wearing wireless synchronized inertial sensors (Opals by APDM, Inc) on the shins and sacrum [6]. Average and peak angular velocity within each turn were calculated and averaged over turns to give the mean and peak turning speed. Turning fluidity was characterized through turn jerkiness, calculated as the integral of the squared time derivative of the accelerometer signal in the mediolateral direction [6]. The FOG-ratio during turning was calculated from the accelerometer signals from the shins in the anteroposterior direction as per prior work [6]. Overall, the FOG ratio has been shown to match well with clinical assessment of FOG, but needs further validation work on capturing all the complex features of FOG.
Descriptive outcomes
Three trials of hip abduction strength were performed with a handheld dynamometer. Hip abduction strength was found to be a determining factor of MELBA outcomes in older adults [27]. The Mini-BESTest, a clinical test for global balance capacity, was also administered [28]. A cognitive test battery was carried out including the Benton’s Jugdement of Line Orientation test (JLO) for visuospatial function [29]. The Flanker test [30] was used to test executive function, more specifically inhibition. The Montreal Cognitive Assessment (MoCA) was executed as a measure of global cognitive capacity [31]. In the PD sub-groups alone, disease duration, the NFOG-Q and disease severity, assessed by the Movement Disorder Society Unified Parkinson’s Disease Rating Scale part III (MDS-UPDRS-III), were administered. The Levodopa Equivalent Daily Dose (LEDD) was calculated from a medication anamnesis [32]. Fall rate in the past 6 months and the Falls Efficacy Scale-International (FES-I) were collected to test overall balance confidence [33].
Statistical analysis
Data were analyzed with IBM SPSS statistics for Windows version 26 (IBM corp., Armonk, NY, USA). Alpha was set at 0.05, unless otherwise indicated. To determine the effect of group when people with PD were OFF medication, one-way ANOVAs with Tukey post hoc tests were performed (group: freezers, non-freezers, controls). If the data distribution was abnormal, a log-transformation was used. If the data remained abnormal, we used the Kruskal-Wallis test, with Mann-Whitney U tests as post hoc tests. We applied a Bonferroni correction for multiple comparisons with a critical value of 0.017 for these post hoc tests (three comparisons). As previous research has shown that MELBA outcomes were related to hip abduction strength [27], we performed an additional analysis of covariance (ANCOVA) including hip abduction strength when group effects were present (Supplementary Material 2). Also, to ensure that differences in disease characteristics did not explain differences between freezers and non-freezers in our primary and secondary outcomes, extra ANCOVAs were performed using MDS-UPDRS-III and disease duration as a covariate when MELBA or secondary outcomes were different between freezers and non-freezers (Supplementary Material 5).
To test the effect of dopaminergic medication on MELBA and turning performance, repeated measures ANOVAs were performed with group (freezers, non-freezers) as between-subjects factor and medication state (OFF, ON) as within-subjects factor. In case of a medication*group interaction effect, the simple effects were analyzed. Also, in case of a medi-cation effect or a medication*group interaction eff-ect, we performed a repeated measures ANCOVA with the mean-centered LEDD as a covariate [34] (Supplementary Material 5). In some cases, when dis-tributions were abnormal, a log-transformation was applied (mean and 95% confidence intervals were back-transformed). The effect of dopaminergic medication on the MDS-UPDRS-III, Mini-BESTest and hip abduction strength was also determined with repeated measures ANOVAs. In addition, we determined the responsiveness of MELBA outcomes to levodopa with the standardized response mean (SRM). The SRM was calculated by dividing the mean change between ON and OFF by the standard deviation of this change. We interpreted standardized response means > 0.2 as small, >0.5 as moderate and > 0.8 as large responsiveness to levodopa [21].
Finally, in an exploratory correlation analysis aimed to better understand our findings, we grouped freezers and non-freezers to represent the full spectrum of the PD sample. We performed Spearman’s correlations between the MELBA mean amplitude and the FOG-ratio, the turning outcomes, the Mini-BESTest scores and disease severity measures (UPDRS-III and LEDD) collected when OFF-medication. Correlations performed were two-tailed and the Benjamini-Hochberg procedure was used to control for the false discovery rate (FDR) [35]. Last, the correlation between MELBA mean amplitude and the NFOG-Q was determined for freezers.
RESULTS
Participant characteristics
Participant demographics and clinical characteristics are presented in Table 1. Groups showed sim-ilar age, gender, length, weight, global cognition andvisuospatial functioning. However, the Flanker score was different among groups. Specifically, non-fre-ezers showed lower Flanker scores than controls (U = 85.00, p = 0.016). Compared to controls, both freezers and non-freezers experienced more falls (U = 80, p < 0.001 and U = 101, p = 0.013) and had a higher FES-I score (U = 59, p < 0.001 and U = 55, p = 0.001), though no differences were found between PD sub-groups (falls: U = 118, p = 0.074; FES-I: U = 125, p = 0.131). No differences were found between fre-ezers and non-freezers on the Mini-BESTest (p = 0.124), while controls had higher Mini-BESTest scores than both freezers and non-freezers. Similarly, hip abduction strength was also greater in controls than in freezers (p < 0.001) and non-freezers (p = 0.020), while no differences between freezers and non-freezers were present (p = 0.466). Freezers had higher LEDDs and longer disease durations than non-freezers, while the UPDRS-III was not significantly higher in freezers in OFF (p = 0.091, Table 1).
Participant characteristics (for PD, values OFF medication are displayed)
Mean±SD are reported. For PD subgroups, outcomes OFF medication are reported. NF - C = significant difference between controls and non-freezers. F - C = significant difference between controls and freezers. F - NF = significant difference between non-freezers and freezers. *Non-parametric Kruskal-Wallis tests with Bonferonni corrected Mann-Whitney post-hoc tests were used (reported as median (Q1–Q3)). LEDD, levodopa equivalent daily dose; MDS-UPDRS-III, Movement Disorders Society Unified Parkinson’s Disease Rating Scale (part III); MMSE, Mini-Mental State Examination; MOCA, Montreal Cognitive Assessment; JLO, Judgement of Line Orientation, N, Newton.
Group comparisons: controls versus non-freezers versus freezers (OFF medication)
MELBA outcomes
Freezers had significantly smaller mean amplitudes of their mediolateral weight-shifts during MELBA performance than non-freezers (p = 0.002) and controls (p = 0.001), while controls and non-freezers did not differ (p = 0.999; Fig. 2B and Table 2). Similarly, start amplitude was also significantly reduced in freezers compared to the other groups (p < 0.001). Further, Fig. 2A shows that in all groups the amplitude increased (until about 0.3–0.4 Hz) and then decreased towards the point of amplitude breakdown. Freezers showed an earlier breakdown of amplitude than controls (p = 0.008), but not when compared to non-freezers (p = 0.336). Non-freezers did not differ from controls on amplitude breakdown (p = 0.306). No group effects were found for any of the phase shift descriptors. Interestingly, one freezer reported a freezing-like experience during MELBA, indicatingan inability to initiate weight-shift movements, des-pite the intention to do so.
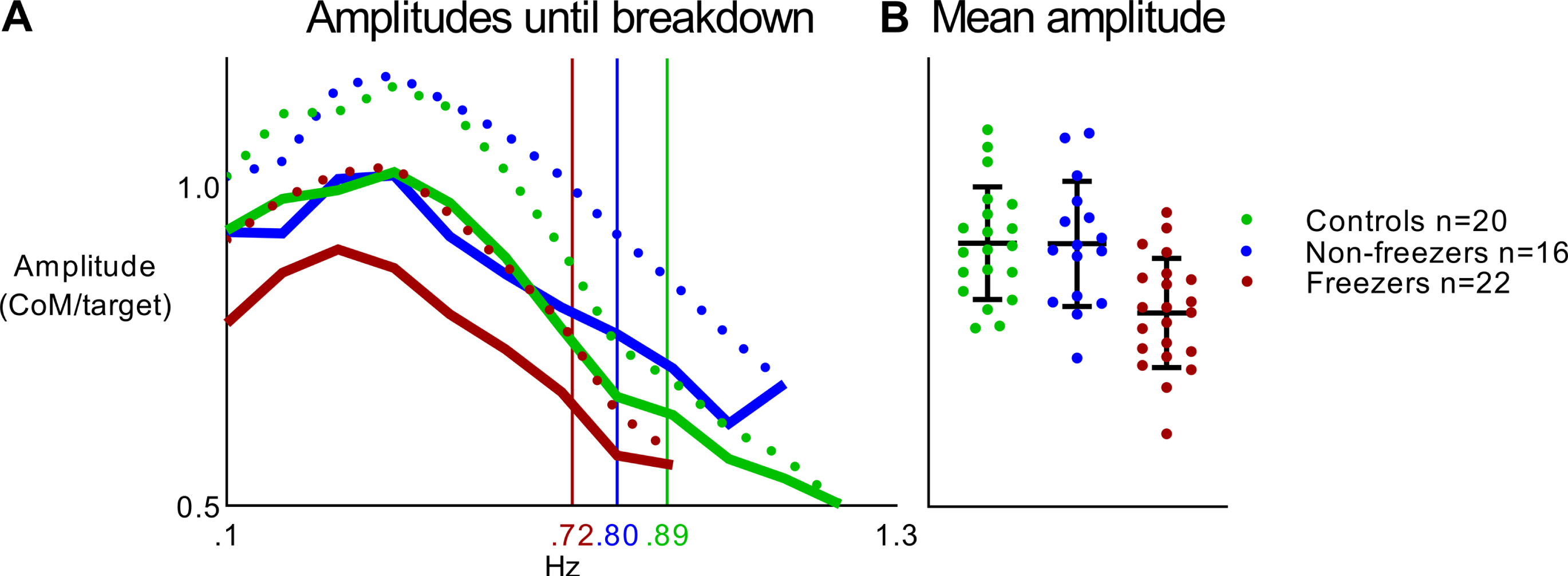
A) Visualization of the amplitudes until the weight-shift amplitude breakdown (freezers and non-freezers OFF medication and controls). Solid lines represent group mean and dashed lines represent standard deviation. Each participant contributed to the group mean until each individuals’ breakdown. Thus, not all participants contributed to the amplitudes in the higher frequencies. Vertical lines represent amplitude breakdown. Freezers showed a significantly earlier amplitude breakdown than controls. B) Mean amplitudes per group and per individual. Error bars represent the standard deviation. Freezers showed a significant lower mean amplitude than non-freezers and controls. Both figures have the same y-axis.
Group differences on the MELBA outcome measures (for PD sub-groups values OFF medications are displayed)
Values are displayed as mean±SD. F - C = significant difference between controls and freezers. F - NF = significant difference between non-freezers and freezers. *Data were log-transformed, back-transformed means (95% confidence intervals) are reported.
We added hip abduction strength as a covariatein ANCOVAs to check whether hip abduction str-ength influenced the significant group effects of the MELBA outcomes (amplitude breakdown, mean amplitude and start amplitude). The covariate hip abduction was not significant (Supplementary Material 2). Similarly, we checked the effects of disease duration and disease severity (MDS-UPDRS-III) when added as covariates in ANCOVAs on mean amplitude and start amplitude. The covariates disease duration and MDS-UPDRS-III were not significant (Supplementary Material 5).
Secondary outcomes
Group differences on the secondary outcomes aredisplayed in Supplementary Table 1 (Supplementary Material 3). One non-freezer did not perform the360° turning task due to fatigue at the end of theexperiment. The FOG ratio and turning jerkinessshowed higher values in freezers (FOG ratio = 1.69 (1.15–2.50) and jerkiness = 0.33 (0.28–0.40) m2/s5) compared to non-freezers (FOG ratio = 0.48 (0.30–0.78) and jerkiness = 0.24 (0.20–0.28); p < 0.001 and p = 0.026, respectively) and controls (FOG ratio = 0.37 (0.26–0.53) and jerkiness = 0.23 (0.20–0.28); p < 0.001 and p = 0.014), after log-transforma-tion. Controls showed a higher mean turning speed (101.13±26.41°/sec) and peak turning speed (167.76±44.16) than both freezers (mean speed = 59.44±15.42 and peak speed = 111.43±25.24; bothp < 0.001) and non-freezers (mean speed = 72.91±16.44 and peak speed = 122.90±26.40; p = 0.001 and p < 0.001, respectively), but no differences were apparent between PD sub-groups (p = 0.124 and p = 0.564).
We checked whether disease duration and MDS-UPDRS-III had a significant effect on the secondary outcomes that differed between freezers and non-freezers (FOG-ratio and jerkiness) by adding them as a covariate to ANCOVAs. None of the covariatesshowed a significant effect, except for disease duration on FOG-ratio (F (1, 34) = 6.043, p = 0.019, Supplementary Material 5).
Effect of medication on the MELBA
One non-freezer showed ON medication an abnormally high mean weight-shift amplitude (1.36), which was more than three standard deviations above the group mean. As perfect amplitude tracking gives an amplitude of 1, this participant showed severe overshooting of the target implicating abnormal task performance. As it caused abnormal distribution ofthe data, we excluded this participant from the rep-eated measures ANOVAs. Exclusion of this partici-pant did not alter the results. Figure 3A and Table 3 display that the mean amplitude of the mediolateral weight shifts significantly increased with medication (F (1, 35) = 7.925, p = 0.008) while no medication*group interaction was present (F (1, 35) = 3.201, p = 0.082). Figure 3B illustrates that the mean amplitude showed a moderate responsiveness to levodopa in freezers (SRM = 0.68) in contrast tothe small responsiveness to levodopa in non-freezers (SRM = 0.21). Start amplitude also improved with medication (F (1, 35 = 4.918, p = 0.033), while no me-dication*group interaction was present (F (1, 35) = 1.007, p = 0.323). Both freezers and non-freezers showed a small responsiveness to medication forstart amplitude (SRM freezers = 0.46; SMR non-freezers = 0.29). For amplitude breakdown a significant effect of medication was apparent in both groups (medication: F (1, 35) = 13.449, p = 0.001; medication*group interaction: F (1, 35) = 0.032, p = 0.859), with a moderate responsiveness (freezers: SRM = 0.64; non-freezers: SRM = 0.57). Overall, effects of medication were less strong on phase shift parameters, only phase shift breakdown showed a significant increase with medication (medication: F (1, 35) = 5.708, p = 0.022; medication*group interaction: F (1, 35) = 0.355; p = 0.555; freezers SRM = 0.47; non-freezers SRM = 0.34).
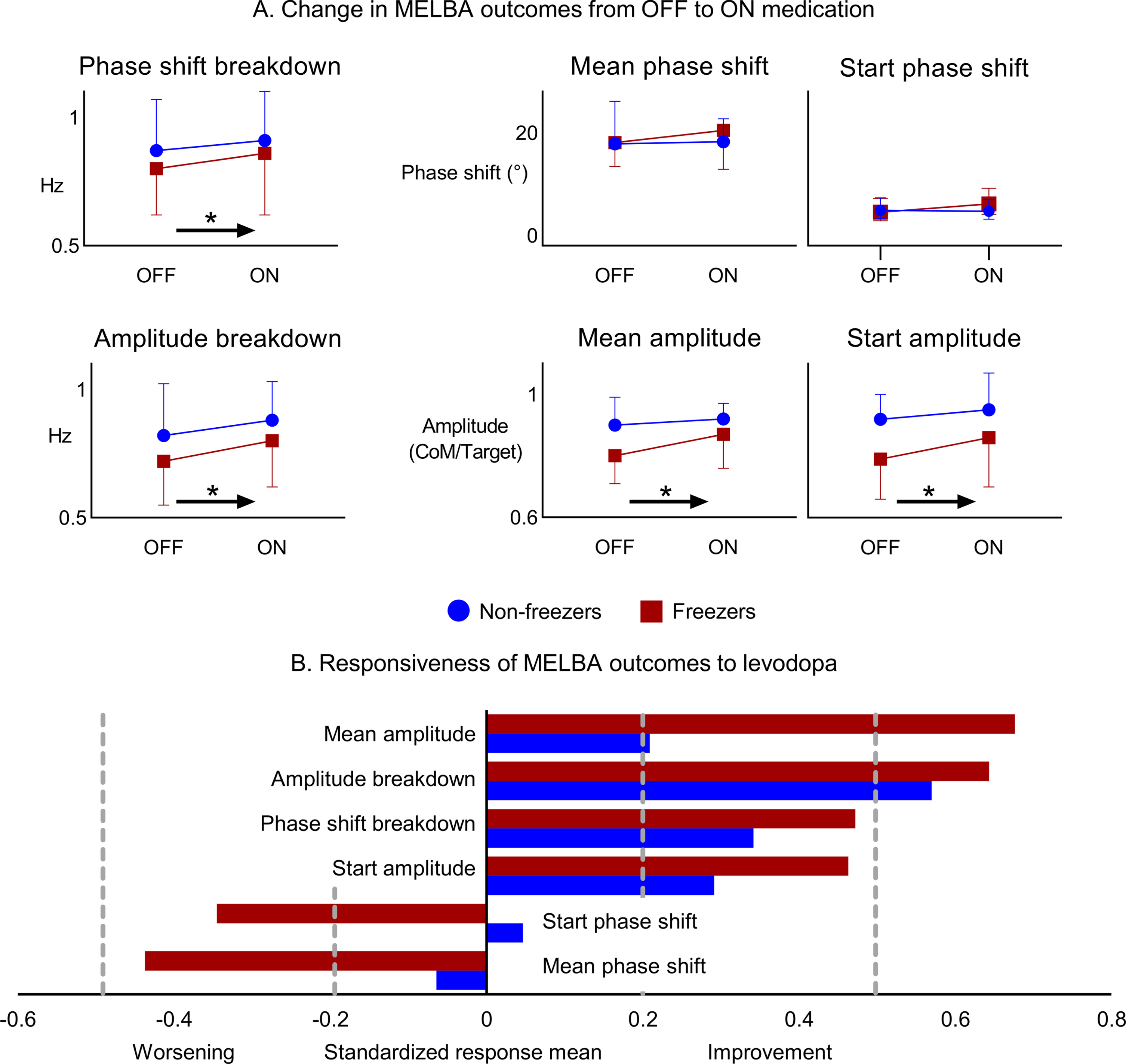
A) Change in MELBA outcomes from OFF to ON medication. Arrows with * represent a significant change from OFF to ON, irrespective of group. Mean and standard deviations are displayed, except for start phase shift. Start phase shift was log-transformed, the back-transformed means with 95% confidence intervals are displayed. B) Responsiveness of MELBA outcomes to levodopa as expressed with the standardized response mean. Vertical dotted lines represent a weak (0.2) and moderate (0.5) response to levodopa.
Using an ANCOVA with LEDD as a covariate, we checked if LEDD influenced the change from OFF to ON medication on the MELBA outcomes. We did not find any significant medication*LEDD interactions (Supplementary Material 5).
Effects of medication in freezers and non-freezers on the MELBA outcomes
Values are displayed as mean±SD. Significant values are displayed in bold. *Data were log-transformed, back-transformed means (95% confidence intervals) are reported.
Effect of medication on secondary outcomes
As for the turning task, data from 2 freezers and1 non-freezer were missing due to technical failureand fatigue. Therefore, 20 freezers and 15 non-free-zers were included in this analysis. The medication effects are summarized in Supplementary Table 2 (Supplementary Material 4). We found a significantgroup*medication effect for the FOG ratio (p = 0.001). Freezers improved (i.e., decreased) their FOG ratio with medication (OFF = 1.75 (1.20–2.55); ON = 0.96 (0.62–1.51); p = 0.001), while non-free-zers did not (OFF = 0.48 (0.30–0.78); ON = 0.62 (0.35–1.09); p = 0.168). The FOG ratio was higher in freezers than in non-freezers when OFF (p < 0.001) but not when ON (p = 0.201). We also foundthat mean turning speed increased in both non-freezers and freezers (freezers: OFF = 57.33 (49.91–65.87) and ON = 61.99 (50.13–76.67); non-free-zers: OFF = 71.40 (63.66–80.08 and ON = 80.85 (70.11–93.23); medication effect: p = 0.028; med-ication*group interaction effect: p = 0.602). Peak turning speed and turn jerkiness were not significantly affected by medication in both groups.
We added LEDD as a covariate to ANCOVAs on our secondary outcomes to determine if the difference in LEDD between groups could have influenced the medication effects (Supplementary Material 5). The medication*LEDD interaction was not significant for both the FOG ratio (p = 0.631) and mean turning speed (p = 0.137).
Effects of medication on clinical descriptors
Both freezers and non-freezers showed signifi-cantly higher UPDRS-III values during OFF ver-sus ON medication, meaning that both groups wereresponsive to medication (freezers: OFF = 36.23±10.46 and ON = 32.50±9.47; non-freezers: OFF = 31.19±5.69 and ON = 26.63±7.75; medication eff-ect: p < 0.001; medication*group interaction effect: p = 0.656). Details are reported in Supplementary Table 3 (Supplementary Material 4). Also, both gro-ups had significant benefits from medication ontheir Mini-BESTest scores (freezers: OFF = 21.50±3.22 and ON = 22.82±94.10; non-freezers: OFF = 23.44±3.74 and ON = 24.44±3.16; medication effect: p = 0.002; medication*group interaction eff-ect: p = 0.655) and abduction strength showed analmost barely significant increase in both PD sub-groups after medication intake (freezers: OFF = 95.5±31.4 and ON = 103.0±40.4; non-freezers: OFF = 110.3±37.9 and ON = 115.8±37.4; medication effect: p = 0.050; medication*group interaction effect: p = 0.770).
Exploratory correlation analysis
In our full PD sample, we found that the FOG ratio correlated negatively with MELBA mean amplitude (rs=–0.340, p = 0.039) OFF-medication, although this correlation did not survive FDR correction and was mainly driven by the non-freezers (see scatter plot and rho-values per subgroup in Supplementary Material 6). As for turning, MELBA mean amplitude had a moderately positive correlation with mean turning speed (rs = 0.430, p = 0.008) as well as peak turning speed (rs = 0.429, p = 0.008). Correlations with the MDS-UPDRS-III, LEDD, Mini-BESTest and turning jerkiness were not significant. The NFOG-Q did not correlate with mean amplitude in freezers.
DISCUSSION
The aim of this study was to determine whether freezers showed deficits during a dynamic balance task and to determine the influence of dopaminergic medication on dynamic balance. The most salient finding of this study was that freezers showed hypometric weight-shift amplitudes, compared to both non-freezers and healthy control subjects, during a visually driven accelerated weight-shifting task that did not involve stepping. Weight-shift amplitudes proved not related to disease severity measures, suggesting that this amplitude deficit may represent a contributing factor to the mechanism of FOG. In addition, breakdown of weight-shift amplitude occurred earlier in freezers compared to controls, whereas there was no difference between non-freezers and controls. Importantly, freezers were at least as responsive to dopaminergic medication as non-freezers for weight-shifting amplitude regulation.
Postural instability during dynamic balance control in freezers has been frequently reported during tasks involving step generation [2]. We now show that amplitude generation during accelerated mediolateral weight-shifting is impaired in freezers compared to both non-freezers and healthy control subjects, pointing towards impaired axial movement vigor (ability to put sufficient energy and power into movements) in freezers. The weight-shifting deficit in freezers affected the start amplitude at onset of the MELBA task and the mean amplitude throughout execution, but did not impinge on phase shift parameters. Previous work showed that the MELBA picked up both amplitude and phase shift deficits in older compared to young adults [23]. Thus, whilst freezers were capable of performing weight-shifts at a similar rhythm in relation to the target as non-freezers and controls, amplitude scaling was hypometric.
Isometric hip abduction strength has previously been shown to correlate with mean weight-shift amplitude during the MELBA task [27] and was in our sample lower in PD sub-groups than in controls. Hip abduction strength had no effect on both mean and start amplitudes when added as a covariate to our analysis. Therefore, we do not think that freezing-related deficits in weight-shift amplitudes were due to lower hip abduction strength. Earlier work also demonstrated a freezer-specific decline in step amplitude during turning, whereby freezers required on average five more steps to complete a 360° turn than non-freezers [36]. These smaller steps may have been underpinned by cautious behavior on the one hand and deficient amplitude generation on the other. We understand hypometric weight-shifting during the MELBA task as a basic failure in invigorating axial movement rather than a compensatory strategy to cope with postural instability, as subjects were supported by a safety harness throughout the task. Furthermore, we speculate that the precise modulation of axial motor vigor is crucial for dynamic postural control and will have repercussions for FOG and turning. Indeed, it has been shown that gait while wearing a weighted-belt, requiring greater motor vigor, also led to more FOG-episodes than normal gait [37]. We project that the uncovered mediolateral dynamic balance impairment in our study, found during a controlled experiment, will compromise the ability to cope with challenges to postural control in real life and exacerbate postural instability in freezers, particularly when OFF or wearing-OFF medication.
The eye-catching weight-shift amplitude deficits in freezers were apparent even in the presence of a visual cue. Previous research of upper limb tasks showed that freezing-related amplitude decline was normalized by auditory cues [38]. Cerebellar pathways are thought to be involved when visual cues induce a switch from internally driven motor behavior tovisually guided movements [39]. These compensatory cerebellar pathways could have been involved in the MELBA task, as the target served as a visual cue. Also, the predictive nature of the MELBA suggests a heavy reliance on feedforward control [23]. Feedforward control is one of the many complex functions of the cerebellum, within which several cerebellar circuits play a role in updating predictive models based on feedback error signals [39]. Inadequate compensatory cerebellar networks in freezers may explain why the weight-shift amplitude deficit was present even in the presence of a cue. In line, a recent systematic review of neuroimaging of FOG proposed that compensatory cerebellar circuits are impaired in FOG [40].
Freezer-specific deficits in amplitude generation during gait, turning and upper limb tasks have been shown to precipitate FOG episodes [38, 42]. Forexample, imposing a decreasing size of writing amp-litude, led to freezing of writing [42]. In the MELBA task, a decreasing amplitude was also observed, although not enforced. However, repetitive movements at increasing frequency were imposed by the MELBA, constituting another trigger for FOG [24]. One freezer reported a freezing-like feeling when performing the MELBA. This person was unable to re-initiate weight-shifting despite the intention to do so and this marked pattern repeated itself throughout the trials. To the best of our knowledge, this is the first report demonstrating freezing-like behavior in a dynamic balance task without ‘the motor intent’ of taking a step or by requiring automatic stepping adjustments during turning. This novel finding would suggest that FOG is not necessarily triggered by a coupling problem between postural control and step generation, but transpires as a result of a sequencing failure, in this case of axial movement repetitions. On a note of caution, this observation was only based on one patient’s self-report and needs further confirmation.
Both PD sub-groups responded well to dopaminergic medication as both the Mini-BESTest and UPDRS-III scores improved. FOG has been cast as a symptom with a notoriously complex response to levodopa [22]. Also, FOG has been associated with changes in the cholinergic system [43]. Therefore, we expected that levodopa would lead to a smaller improvement on MELBA outcomes in freezers than in non-freezers [44]. However, we found that both PD sub-groups showed a similar responsiveness to levodopa. Start and mean phase shift did not improve with medication, but these outcomes were not different between PD sub-groups OFF medication and controls. In contrast, all weight-shift amplitude outcomes improved with medication in both freezers and non-freezers. Freezers even showed a modera-te responsiveness to levodopa (SRM > 0.5) for meanweigh-shift amplitude whereas non-freezers onlyshowed a small levodopa responsiveness (SRM > 0.2). Of note, these results seemed independent from the higher LEDDs reported by freezers, but freezers did show lower weight-shifts than non-freezers and thus had more room for improvement. Therefore, we conclude that freezers were at least as responsive to dopaminergic medication for improving weight-shifting, especially weight-shifting amp-litude, as non-freezers. This is in line with previously reported levodopa-induced improvements in freezers in outcomes related to amplitude scaling during stepping in place (increase in step duration) and turning (decreased number of steps) [20, 45]. These findings together strengthen the notion that levodopa plays an important role in regulating motor vigor even in the axial dimension [46]. A recent review, including functional brain imaging and resting-state fMRI studies, indicated that dopamine-induced activations of M1, the supplementary motor area (SMA) and putamen were directly related to motor vigor improvements in PD [46]. Although this review per-tained to distal motor tasks [46], the strong correlation found recently between the dopamine reuptake transporter density of putamen and the spatial out-comes of gait initiation [11], suggests similar mecha-nisms in the axial domain. Indeed, in our study the FOG ratio and mean turning speed also improved with medication in freezers. A recent study of the long-term effects of levodopa on a drug-naïve cohort also showed that levodopa had positive effects onFOG, and postural instability and falling [44]. However, the smoothness of turning (jerkiness) andpeak turning speed remained unaffected by medi-cation in both freezers and non-freezers.
It is noteworthy that MELBA mean weight-shift amplitude in our total PD sample showed moderate correlations with mean and peak turning speed, but was not correlated to general disease severity orbalance measures. Interestingly, a higher FOG ratioseemed related to lower weight-shift amplitudes in PD, although this correlation did not surviveFDR correction and was mostly driven by non-fre-ezers. Hence, MELBA mean amplitudes appea-red to capture axial motor control important for freezing-related turning outcomes. Within freezers, the MELBA outcomes were not associated with the FOG questionnaire, which is in line with recent findings that self-reported freezing is highly variable within-patients, probably due to recall bias [41].
The finding that freezers suffer from a distinct weight-shifting impairment has clinical implications. A recent study illustrated that teaching people with PD to allocate conscious attention to weight-shifting, not only led to an amplitude increase of the unloading phase of APAs but also reduced unsuccessful attempts to initiate stepping in freezers [47]. In conjunction, personalized ‘rescue strategies’ have been shown to ameliorate FOG by upscaling mediolateral weight-shifts, e.g., through mimicking a skating movement [48]. Furthermore, it was recently shown that multimodal balance training improved postural instability in both freezers and non-freezers [49]. The present study enhances insight into why such behavioral training strategies are effective, and supports their use to complement levodopa in overcoming FOG.
The current results need to be interpreted against the limitation that we divided freezers and non-freezers in separate groups based on their subjective experience of FOG in the past month (i.e., item 1 on the NFOG-Q), which is the standard procedure in the field. We acknowledge that this procedure does not represent the reality that FOG is a gradient phenomenon, clinically heterogeneous and has a variable response to medication [22, 50]. Group allocation to freezer/non-freezer also led to imperfectly matched sub-groups in this study, which might mask the FOG-specificity of our findings. However, cognitive profiles were similar between freezers and non-freezers. In addition, disease characteristics were added as covariates to our analysis and were found not to have an effect on the MELBA outcomes. Similarly, LEDD was also higher in freezers compared to non-freezers, but did not seem to influence medication effects.
Characterizing FOG as a biological gradient within a wide spectrum of patients, who are more or less prone to FOG, is undoubtedly the most optimal future study approach. Currently, the FOG ratio probably serves as the best measure of this continuum [6]. However, the exact meaning of this metric still needs further validation, as it was recently shown not to correlate well with clinical ratings of very short FOG-episodes [51]. Mancini, et al. [51] suggested that decreased smoothness of gait could also be picked up as FOG by the algorithm. In line, in the present study we showed that freezers showed higher FOG ratios and turn jerkiness (worse turning smoothness) than non-freezers, even though FOG episodes were not frequently present. Future study needs to indicate if the FOG ratio can be used as a biological FOG gradient and if the FOG ratio can also identify non-freezers prone to FOG. Lastly, considering the cue dependency in freezers, the visual cues of the MELBA task may have concealed possible differences in phase shift or amplitude breakdown between PD sub-groups.
In conclusion, we demonstrated that freezers had a striking impairment in producing sufficient amplitudes during accelerated weight-shifting compared to non-freezers and controls. We believe that this impairment is a central mechanism, which explains one piece of the puzzle of why freezers fall more regularly and show impaired turning and dynamic postural control. While weight-shift amplitude regulation proved dopamine-responsive in freezers, training of weight-shifting may provide further benefits, especially as conscious weight-shifting is also strategy to overcome FOG. Future studies are needed to unravel the neural underpinnings of weight-shift amplitude deficits in PD and FOG.
Footnotes
ACKNOWLEDGMENTS
We like to thank all study volunteers who dedicated their time to participation in this study.
BWD was supported by the Research Foundation Flanders (FWO, grant number G.0867.15). MG received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 838576.
CONFLICT OF INTEREST
The authors declare no competing interests.
