Abstract
Background:
Several small-scale studies have shown that motor performance in Parkinson’s disease (PD) fluctuates throughout the day. Studies specifically focusing on de novo patients are, however, lacking.
Objective:
To evaluate the effect of clock time on motor performance in de novo drug-naïve patients with PD.
Methods:
We retrieved MDS-UPDRS III scores for 421 de novo PD patients from the PPMI cohort and stratified them into three groups based on time of assessment: group 1) 7:00–10:00; group 2) 10:00–13:00, and group 3) 13:00–18:00. Groups were compared using Kruskal-Wallis test and results corrected for multiple testing. In addition, we obtained 27 wearable sensor reports, objectively capturing bradykinesia scores in a home setting over a 6-day continuous period, in 12 drug-naïve patients from the Parkinson’s Kinetigraph Registry held at King’s College Hospital London. Time spent in severe bradykinesia scores were broken down into five daytime (06:00–21:00) three-hourly epochs and scores compared using the Friedman test.
Results:
There were no group differences in demographic or other clinical variables for the cross-sectional analysis. MDS-UPDRS III total scores worsened significantly during the course of the day (median 18 (group 1); 20 (group 2); and 23 (group 3); p = 0.001). In the longitudinal wearable sensor cohort, diurnal variations were present in percentage of time spent in severe bradykinesia (p < 0.001) with the lowest percentage during the 09:00–12:00 epoch (69.56±16.68%), when most patients are awake and start daily activity, and the highest percentage during the 18:00–21:00 epoch (73.58±16.35%).
Conclusion:
This exploratory study shows the existence of a diurnal pattern of motor function in patients with de novo PD. The results obtained were corroborated by objective measurements in a small longitudinal cohort confirming a similar diurnal motor score variation.
INTRODUCTION
The biological clock, located in the hypothalamic suprachiasmatic nuclei (SCN), regulates many of the 24 h patterns observed in several physiological processes in humans [1]. In healthy subjects, but also in patients with Parkinson’s disease (PD), several smaller-scale studies have shown that non-motor and motor performance fluctuates throughout the day and across seasons, with a rise in activity after awakening, stable patterns during the morning and early afternoon, and worsening later during the day [2–7]. An example in PD is the study by van Hilten and colleagues who observed that in mildly to moderately affected patients diurnal patterns of motor activity were present, with greatest motor activity in the morning [5]. Also, other PD features appear to fluctuate according to time of the day, including gait patterns [6] and some non-motor symptoms [8, 9].
The recognition of physiological diurnal patterns holds importance for the use of dopaminergic medication in PD, as dopaminergic neurotransmission directly influences the biological master clock. In rodent studies it has been shown that the administration of Haloperidol, a dopamine receptor antagonist, increases the expression of mPer1, one the crucial clock genes in the SCN [10]. This effect might account for some of the observations that have been made in PD patients with regards to the disappearance of a diurnal pattern in activity counts in the advanced stage of the disease, whereas PD patients in Hoehn and Yahr stages 1 and 2 display a diurnal activity pattern similar to healthy controls [5]. Similar daytime patterns in tapping speed were also observed by Bonuccelli and colleagues, although this pattern was only present in patients with stable and advanced disease stages, but not in de novo patients [11]. In the latter study, the authors showed that the diurnal pattern might be explained by changes in levodopa pharmacodynamics despite stable repeated medication administration [11]. This is also supported by findings in advanced PD patients, where the clinical effects of intrajejunal levodopa infusion diminish in the afternoon despite stable continuous infusion rates of Levodopa [12].
In order to exclude the potentially confounding effect of dopaminergic medication, we studied diurnal patterns of motor performance in a large cross-sectional cohort of de novo PD patients. In addition, we aimed to corroborate these findings in a longitudinal setting using objective wearable sensor data.
METHODS
PPMI cohort
We included 421 de novo PD patients from the Parkinson’s Progression Markers Initiative (PPMI) database. The PPMI is an ongoing prospective, observational, international, multicentre study aimed at identifying clinical biomarkers of PD in a large cohort of participants with early PD at enrolment [13]. The aims and methodology of the study have been extensively described elsewhere and are available at www.ppmi-info.org/study-design. Inclusion criteria were age 30 years or older, diagnosis of PD (based on one of the following: presence of 1) asymmetrical resting tremor or 2) asymmetrical bradykinesia or 3) at least two of either of resting tremor, bradykinesia, and rigidity), and a disease duration of 1 to 24 months, Hoehn and Yahr (H&Y) stage of 1 to 2, and presence of striatal dopamine transporter deficit on 123I-ioflupane SPECT imaging (DaTSCAN). In this cross-sectional study, the selected patients had both recorded MDS-UPDRS III scores and exact time of this assessment. Patients were stratified into three groups: 1) Patients with an assessment performed between 7:00 and 10:00 (Group 1), 2) Patients with an assessment between 10:00 and 13:00 (Group 2), and 3) Patients with an assessment after 13:00 (Group 3). No assessment was performed after 18:00. The study was approved by the institutional review board of all participating sites. Written informed consent was obtained from all participants before inclusion in the study. We obtained data from the PPMI database on December 2, 2019 in compliance with the PPMI Data Use Agreement.
Data extracted from the PPMI database included demographics, age at onset, disease duration, HY staging, Movement Disorder Society-Unified Parkin-son’s Disease Rating Scale (MDS-UPDRS) (including part I-Non-Motor Aspects of Experiences of Daily Living, part II-Motor Aspects of Experiences of Daily Living, and part III-Motor Examination) [14], SCOPA-Autonomic (SCOPA-AUT) [15], Montreal Cognitive Assessment (MoCA) [16], 123I-FP-CIT striatal binding ratios. The following motor subscores were additionally calculated from the original MDS-UPDRS-III: rigidity score (item 3), bradykinesia score (sum of items 2, 4–9 and 14), tremor score (sum of items 15–18), and axial score (sum of items 1 and 9–13).
Wearable sensor cohort
For this cohort we included 27 wearable sensor readouts from 12 different de novo PD patients from the Parkinson’s Kinetigraph™ Registry (PKG Registry) study at the Parkinson’s Foundation Centre of Excellence at King’s College Hospital London, UK. This registry obtained ethical approval from the London –Riverside Research Ethics committee (REC reference: 17/LO/1010, IRAS ID: 215965). All patients gave written informed consent, and the study was conducted in line with the Declaration of Helsinki.
For this analysis we included patients who: 1) had a diagnosis of idiopathic PD according to the UK Brain Bank criteria; 2) had an available PKG recording within the frame of the PKG Registry; 3) were under the care of the Parkinson’s Foundation Centre of Excellence at King’s College Hospital in London; and 4) had never been on dopaminergic treatment for PD.
The PKG watch which was used is a wrist-worn device that provides continuous monitoring of motor symptoms in PD. The watch is worn for a period of six consecutive days, after which data is downloaded and analysed using a proprietary algorithm to calculate the scores for bradykinesia (BKS) and dyskinesia [17]. The latter scores were not used in the current study, as de novo patients do not exhibit dyskinesia. The BKS score is the median value of bradykinesia over a specific period during the day. These values have been shown to correlate to the Unified Parkinson’s Disease Rating Scale part III scores, and with the modified Abnormal Involuntary Movement Score assessed at the time of PKG [18, 19]. The BKS scores used in the current study were broken down into five three-hourly epochs for the daytime (06:00–21:00), as follows: epoch 1) 06:00–09:00; epoch 2) 09:00–12:00; epoch 3) 12:00–15:00; epoch 4) 15:00–18:00; and epoch 5) 18:00–21:00; due to the way these data are provided by the manufacturer of the PKG watch, Global Kinetics, it was not possible to have hourly breakdowns for this data. For each of these epochs, the percentage of time spent in severe bradykinesia, defined as BKS values over the 75th percentile (BKS≥III), was used for analysis as they are of functional significance [17]. Other data used in the current study included baseline demographics, such as gender, disease duration, Scales for Outcomes in Parkinson’s disease (SCOPA) [20] scores, Hoehn and Yahr staging [21], and Non-Motor Symptoms Scale (NMSS) [22] total scores.
Statistical analysis
The primary outcome for the PPMI cohort was to assess motor differences across time of assessment. Since the variables were non-normally distributed (normality of variables was tested with Shapiro-Wilk tests), continuous variables were compared using the Kruskal-Wallis H test with additional post-hoc pairwise comparisons. Categorical variables were compared using Fisher’s exact test. Additionally, we explored the association between MDS-UPDRS III score and time of assessment using a Multiple Linear Regression analysis with the MDS-UPDRS III score as the dependent variable, and age, time of assessment, and striatal binding ratios (123I-FP-CIT) as independent variables.
The primary outcome for the wearable sensor cohort was to address differences in the severity of BKS between the five studied epochs. Also here, data were not normally distributed (Shapiro-Wilk tests), and therefore, for analysis of the outcome, we used the Friedman test with additional post-hoc pairwise analyses for comparing the scores between individual epochs.
A p-value≤0.05 was considered statistically significant, with a correction for multiple testing (Benjamini-Hochberg procedure) where relevant. Data are presented as mean±standard deviation, median (interquartile range) or number (percentage), unless otherwise specified. All data were analysed using SPSS Version 26 (IBM SPSS Statistics for Windows, Version 26.0. Armonk, NY: IBM Corp.).
RESULTS
PPMI cohort
Of the 421 included patients, 174 patients with de novo PD received a motor assessment between 7.00 and 10.00, 181 between 10.00 and 13.00 and 63 between 13.00 and 18.00. No demographic differences were found between the groups (Table 1).
Demographic data and motor assessment scores at different timepoints in de novo Parkinson’s disease patients enrolled in the Parkinson’s Progression Markers Initiative.
Data presented as median (25th-75th percentiles) or percentage; p-values corrected for multiple testing using the Benjamini-Hochberg procedure. aBased on MDS-UPDRS part-III items. MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment; SCOPA-AUT, SCales for Outcomes in PArkinson’s disease Autonomic
Group comparison showed that motor functions, measured by MDS-UPDRS III total score, were different among the groups (median: 18 (group 1), 20 (group 2), and 23 (group 3), p = 0.001, Fig. 1 and Table 1). Post-hoc analysis revealed worsening over the course of the day in patients with de novo PD (Groups 1-2: p = 0.052; Groups 2-3: p = 0.011; Groups 1–3: p = 0.001). Detailed motor characterisation using MDS-UPDRS III subscores revealed that specifically bradykinesia (p < 0.001) caused the group differences in MDS-UPDRS III total scores. There were no other group differences, except for a trend toward statistical significance in HY scores (p = 0.054). No differences were found in Striatal Binding Ratio (DaTSCAN) (p = 0.447).
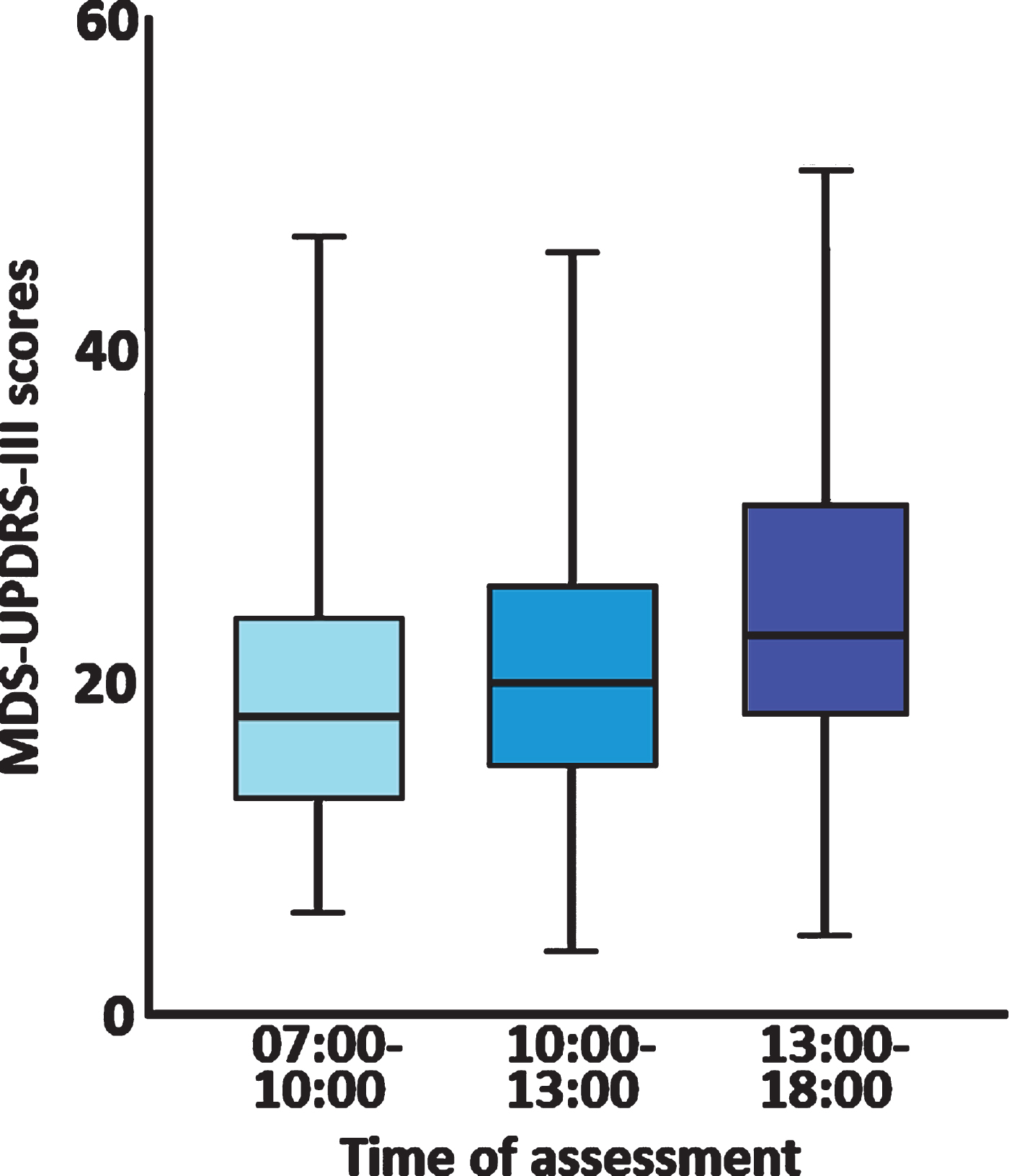
Cross-sectional motor assessment scores (Movement Disorder Society –Unified Parkinson’s Disease Rating Scale III total scores (MDS-UPDRS III)) during three different time epochs in 421 de novo Parkinson’s disease patients enrolled in the Parkinson’s Progression Markers Initiative study showing significant worsening of daytime MDS-UPDRS III scores (p < 0.001) as the day progressed. Error bars represent 95% confidence intervals.
After identification of significant associations in a univariate model, we included age and time of assessment as independent variables in a multiple linear regression model, with MDS-UPDRS III as dependent variable. In the latter model with a poor model fit (R2 = 0.109, p < 0.001), time of assessment was the most important predictor of MDS-UPDRS-III scores (β= 0.180, p < 0.001) after striatal binding ratios (β=–0.188, p < 0.001) (Table 2).
Multiple linear regression models with predictors of MDS-UPDRS part III scores in de novo Parkinson’s disease patients enrolled in the Parkinson’s Progression Markers Initiative. MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale
Wearable sensor cohort
Patient demographics for the 12 patients and 27 PKG readouts are shown in Table 3. We observed that a significant diurnal variation was present in percentage of time spent in severe bradykinesia between the five epochs studied (p < 0.001; Table 3; Fig. 2). Specifically, we observed that percen-tage of time spent with severe bradykinesia (defined as a bradykinesia score above the 75th percentile) was highest (78.97±15.00%) during epoch 1 (06:00–09:00), and lowest (69.59±16.68%) during epoch 2 (09:00–12:00) (Table 3; Fig. 2). Post-hoc analysis showed that significant differences were present between epoch 2 (09:00–12:00) and epoch 1 (06:00–09:00; p < 0.001), epoch 4 (15:00–18:00; p = 0.015), and epoch 5 (18:00–21:00; p < 0.001); and between epoch 3 (12:00–15:00) and epoch 1 (06:00–09:00; p < 0.001), and epoch 5 (18:00–21:00; p < 0.001) (Table 3).
Percentage of time spent with severe bradykinesia (grade III (75th percentile) and above) and demographics in 12 drug-naïve Parkinson’s disease patients, based on wearable sensor data, during five daytime epochs studied (n = 27 readouts).
PKG: Parkinson’s Kinetigraph™. Data represented as mean±standard deviation or median (25th-75th percentile); *Friedman’s test. SCOPA, Scales for Outcomes in Parkinson’s disease; NMSS, Non Motor Symptoms Scale
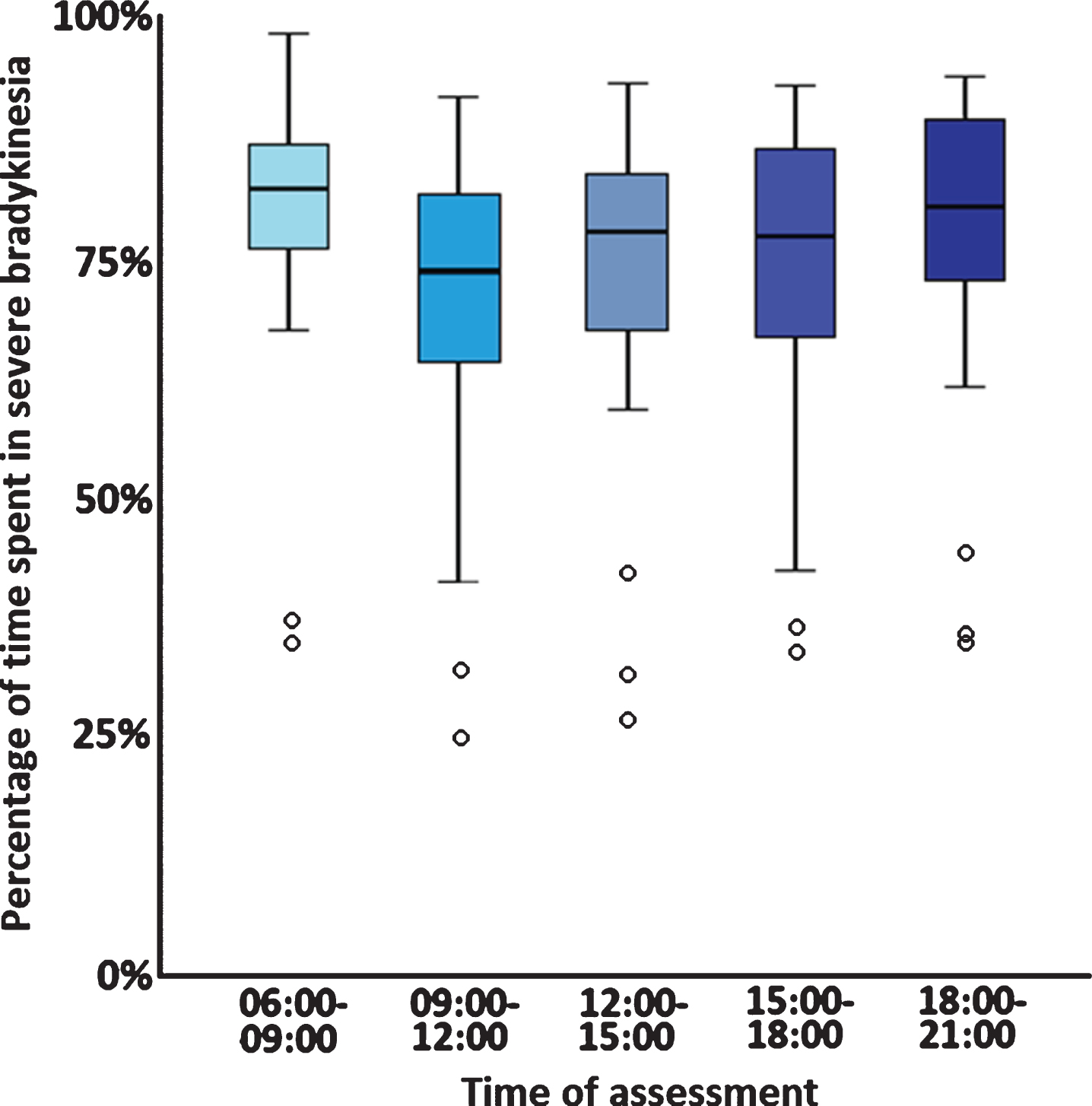
Motor assessment scores using wearable sensor data (Parkinson’s Kinetigraph™ PKG) in de novo Parkinson’s disease patients. Data is presented as the average percentage of time spent with severe bradykinesia (PKG bradykinesia score ≥III) during five different daytime epochs showing significant worsening of daytime bradykinesia as the day progressed (p < 0.001); error bars represent 95% confidence intervals and open circles represent outliers.
As some participants may have been asleep during part of the 06:00–09:00 epoch, we decided to repeat the previous analysis while omitting the first epoch (06:00–09:00). Nonetheless, the diurnal variation with increasing percentage of time spent with severe bradykinesia as the day progressed remained significant (p < 0.001). Post-hoc analysis revealed that significant differences were present between epoch 2 (09:00–12:00) and epoch 4 (15:00–18:00; p = 0.004), and epoch 5 (18:00–21:00; p < 0.001); and between epoch 3 (12:00–15:00) and epoch 5 (18:00–21:00; p < 0.001).
DISCUSSION
In this exploratory study, we showed in a large cohort of de novo PD patients that motor performance, and specifically bradykinesia, appears to be dependent on the time of assessment, supporting the notion that physiological diurnal patterns of motor performance are still present in early PD. These findings were corroborated by objective wearable sensor data in a longitudinal fashion in a small cohort of patients. Such diurnal variation holds importance for the interpretation of study results where motor assessments in PD patients are carried out, but could also be clinically relevant for the delivery of personalised medication regimes once these patients require dopaminergic medication.
Here we have illustrated that MDS-UPDRS III scores are lowest during the early morning hours (07:00–10:00) in de novo PD patients, and then seem to worsen over the course of the day, with highest scores in afternoon. In contrast, in the cohort with wearable sensor data we showed that bradykinesia scores were highest early in the morning (06:00–09:00), after which they improve, only to worsen again in the afternoon with a peak in the early evening (18:00–21:00). Since we cannot exclude that the high bradykinesia score in the early morning (06:00–09:00) could be due the fact that some patients were still sleeping or inactive, as this information was not available, we repeated the analysis excluding the first epoch (06:00–09:00). Nonetheless, the diurnal pattern with worsening bradykinesia in the afternoon remained and was very similar to the pattern identified in the cross-sectional cohort. The observed variations in motor symptom severity in both cohorts are also very similar to those that have been previously described [5, 23]. However, the main advance of our study is that we studied these patterns in de novo patients, ruling out the possible effect of dopaminergic medication. The latter is able to influence the SCN [10] and, as such, may interfere with the diurnal pattern in motor performance, which is likely to be imposed by the biological master clock [1, 2]. In addition, we had a large sample size in the cross-sectional cohort.
Not only motor symptoms appear to have a diurnal patterns, and in fact several studies have also reported that other symptoms, including non-motor symptoms, such as autonomic function [24–26] and sleep–wake cycles [27], show diurnal fluctuation in patients with PD [8, 9], suggesting a circadian influence on the expression of non-motor features of PD. Interestingly, acute psychological distress (anxiety) [28], excessive daytime sleepiness [29] and visual performance [30] have been shown to worsen as the day progresses in patients with PD, consistent with the motor pattern that we observed. This suggest that the intrinsic 24 h master clock orchestrates both motor and non-motor fluctuations throughout the day. However, as this remains a relatively underexplored area of PD, further studies are warranted to characterize the daytime dependency of the whole spectrum of PD features, including motor and non-motor symptom severity. Here, the use of wearable sensor data might prove a useful and objective means of not only exploring motor, but also non-motor, features [31].
Some limitations to our study need to be acknowledged. The main limitation is related to the cross-sectional design, and preferably studies looking at diurnal patterns should deploy a longitudinal design. We feel, however, because of the large sample size our results are still meaningful. Moreover, we corroborated our findings in a small cohort using a longitudinal assessment with objective wearable sensor data, overcoming the inherent limitations of the subjective character of clinical assessments [32], even though the wearable sensor cohort had a small sample size. After omission of the earliest morning epoch, which may be interpreted as confounding the diurnal patterns as some patients may still have been asleep, bradykinesia scores still worsened as the day progressed. We were unable to obtain information regarding the time when patients woke up and when they would get out of bed, which is another limitation in the current study. However, we only included de novo PD patients ruling out the possible influence of dopaminergic medication on motor performance. It is, therefore, likely, despite some limitations, that the observed diurnal pattern is in fact due a rhythm imposed by the biological master clock, although future studies should aim to directly assess the role of this clock on PD symptoms.
In summary, we observed that motor scores, as measured by the MDS-UPDRS III and through a wearable sensor, appeared to worsen of the course of a day and are dependent on time of assessment. In the cross-sectional study, the most important driver for these differences were the bradykinesia items. This pattern in motor functioning implicates the biological clock in the SCN as the driving force behind these differences. The worsening of motor symptoms over the course of a day should be taken into account in the interpretation of clinical studies and trials, and could be clinically relevant for the delivery of personalised care.
CONFLICT OF INTEREST
DvW has received travel grants, and advisory board and speaker fees from Britannia Pharmaceuticals; KRC has received speaker and advisory board fees from Britannia Pharmaceuticals; DU reports no conflicts of interest.
Footnotes
ACKNOWLEDGMENTS
Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/data). For up-to-date information on the study, visit ![]() . PPMI (a public–private partnership) is funded by the Michael J Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB. The PKG Registry is partly funded by a grant from Britannia Pharmaceuticals. The current data analyses were not supported by funding. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
. PPMI (a public–private partnership) is funded by the Michael J Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB. The PKG Registry is partly funded by a grant from Britannia Pharmaceuticals. The current data analyses were not supported by funding. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
