Abstract
Background:
The different effects of white matter hyperintensity (WMH) severity and WMH-associated microstructural connectivity on cognition in the early stages of Parkinson’s disease (PD) have not been investigated.
Objective:
To investigate the differential effect of WMH severity and WMH-associated microstructural connectivity on cognition in early stages of PD.
Methods:
A total of 136 de novo PD patients were enrolled and divided into groups based on total WMH visual rating scores as follows: mild, moderate, and severe. Microstructural connectivity was measured using graph theoretical analysis according to WMH severity. Additionally, correlation coefficients between WMH-associated microstructural connectivity or WMH scores and cognitive performance were assessed.
Results:
Patients with severe WMHs demonstrated poorer performance in language function than those with moderate WMHs, and in frontal/executive and visual memory function than those with mild WMHs. Areas of microstructural connectivity were more extensive in patients with severe WMHs compared to those with mild and moderate WMHs, involving frontal and parieto-temporal regions. WMH-associated right fronto-temporo-parietal microstructural disintegration was correlated with cognitive dysfunction in attention, frontal/executive, and memory domains, whereas there was no correlation between WMH scores and any cognitive domains.
Conclusion:
These data suggest that disruption of microstructural networks by WMHs, rather than WMH burden itself, contributed more to cognitive impairment in PD.
Keywords
INTRODUCTION
White matter hyperintensities (WMHs) are commonly observed in virtually every individual aged 60 years or older [1]. As a well-known conventional marker of small vessel disease (SVD), WMHs are associated with an increased risk for cognitive decline and may ultimately lead to dementia in normal aging [2]. Despite the high prevalence of WMHs, however, relatively few individuals develop cognitive decline or dementia, suggesting that other factors may contribute to the development of cognitive deterioration. One of these factors is disrupted microstructural integrity of normal appearing white matter (WM) surrounding WMHs. A postmortem study reported that neuropathological correlates of WMH lesions were also found in normal-appearing WM that were not visible on conventional imaging, but could be investigated using diffusion tensor imaging (DTI) [3].
DTI is a technique that applies a tensor-based model to acquire diffusion-weighted imaging (DWI) scans, which are a magnetic resonance imaging (MRI) sequence sensitive to the diffusion of water molecules, and thereby provide more reliable information about the microstructural organization of WM than conventional MRI techniques, such as fluid-attenuated inversion recovery (FLAIR) [4]. Accordingly, ample evidence has demonstrated that microstructural alterations of WM on DTI demonstrate a robust correlation with cognitive performance associated with SVD compared with conventional markers of SVD, such as WMHs or lacunar volume [5, 6]. These findings suggest that SVD certainly exerts its action outside visible lesions, which contributes to clinical outcomes.
In Parkinson’s disease (PD) patients, the burden of WMHs may also negatively impact cognitive performance by aggravating microstructural disorganization in frontal subcortical circuits and cholinergic pathways [7], and by promoting the aggregation of α-synuclein, a key protein in the pathogenesis of PD [8]. However, the relationship between WMHs and cognitive dysfunction is inconsistent in PD. Some authors have reported that WMH burden is significantly associated with cognitive dysfunction as well as dementia conversion in early PD patients [7], whereas WMH volumes were not a significant predictor of global cognition or specific cognitive domain function in PD [9].
To the best of our knowledge, the different effects of WMH severity and WMH-associated microstructural connectivity on cognition in early stages of PD have not been investigated. As such, we analyzed microstructural connectivity using network-based statistics (NBS) and graph theoretical analysis in de novo PD patients according to the severity of WMHs. In addition, we performed a comparative correlation analysis between WMH-associated microstructural connectivity or WMH severity and cognitive performance in these patients to uncover whether microstructural connectivity is more closely coupled with cognitive deficits relative to simple WMH severity.
METHODS
Subjects
We reviewed the medical records in the Yonsei Parkinson Center database of 513 consecutive PD patients who visited the movement disorders outpatient clinic at Severance Hospital (Seoul, Korea) between January 2015 and May 2018. Details of study participants are illustrated in Fig. 1. Ultimately, 136 de novo PD patients were enrolled. PD was diagnosed according to the clinical diagnostic criteria of the United Kingdom PD Society Brain Bank, and each subject exhibited decreased dopamine transporter (DAT) availability in the posterior putamen on 18F-fluorinated N-3 fluoropropyl-2-beta-carboxy-methoxy-3-beta-(4-iodophenyl) nortropane (18F-FP-CIT) positron emission tomography (PET) scans. All PD subjects underwent brain MRI, 18F-FP-CIT PET, and neuropsychological tests. To assess the severity of parkinsonian motor symptoms, the Unified Parkinson Disease Rating Scale Part III (UPDRS-III) was used. General cognition was assessed using the Korean version of the Mini-Mental State Examination (K-MMSE). Olfactory function was assessed using the Cross-Cultural Smell Identification Test (CCSIT), calculated as the sum of correct responses. The Beck Depression Inventory (BDI) was administered to assess the severity of depression. In addition, baseline vascular risk factors such as hypertension, diabetes mellitus, dyslipidemia, heart disease, and stroke were assessed. The exclusion criteria included atypical parkinsonism, drug-induced parkinsonism, and evidence of focal brain lesions. This study was approved by the Institutional Review Board of Yonsei University Severance Hospital. Written informed consent was obtained from all patients who participated in this study.
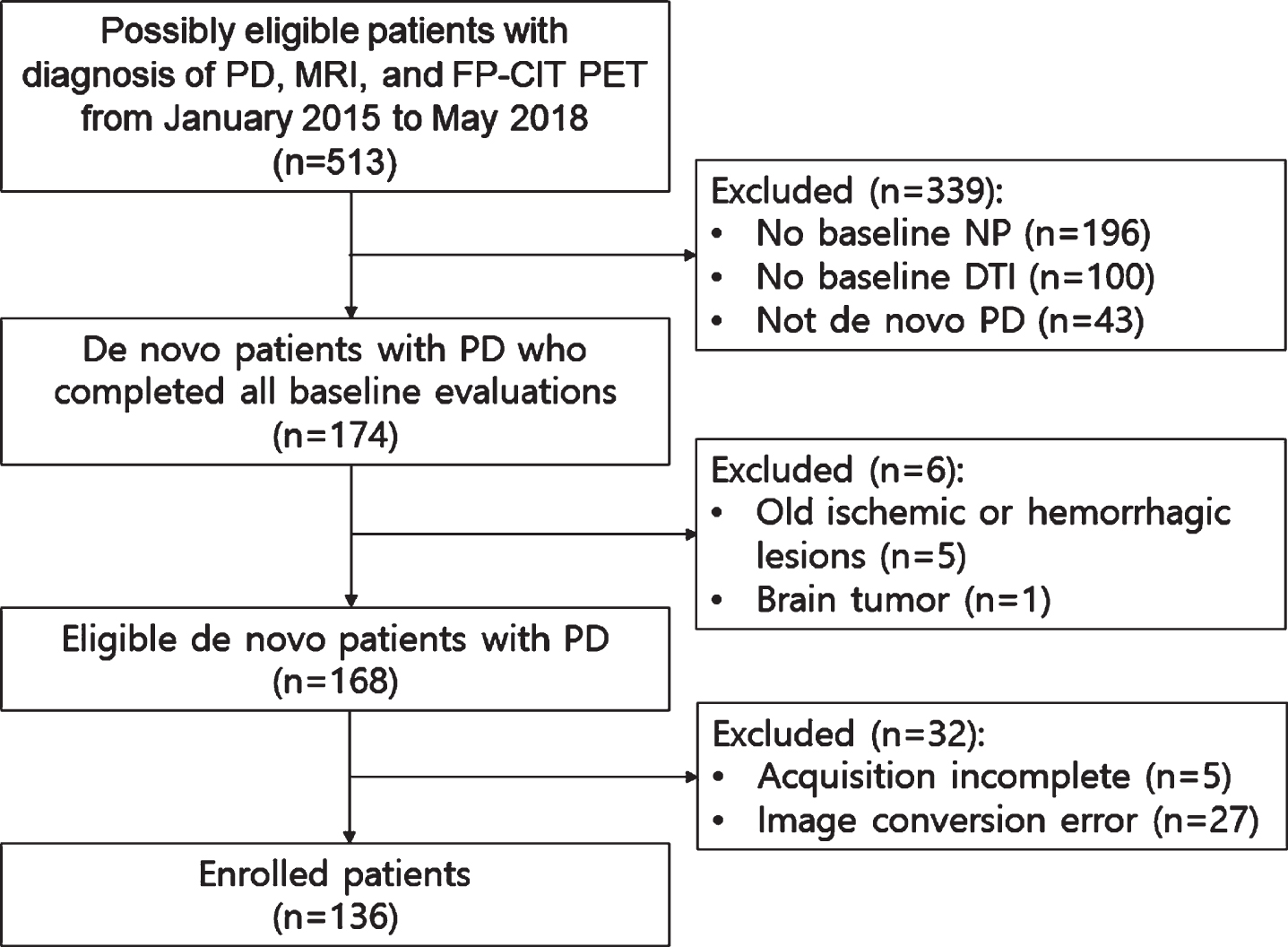
Study flow chart.
Classification of patients with PD according to WMH
WMH scores of all 136 de novo PD patients enrolled in this study were rated on FLAIR images using the Scheltens scale [10]. The WMH rating was performed by two neurologists (CJS and YHS) who were blinded to patients’ clinical information; a final consensus rating was used for the analysis. Based on the distribution of total WMH scores, patients were stratified into the following tertiles: mild, moderate, and severe WMHs.
Neuropsychological assessment
The Seoul Neuropsychological Screening Battery (SNSB), a comprehensive neuropsychological test battery in Korean language, was administered to all subjects. The SNSB includes six cognitive subsets, as described in our previous work [11]. The SNSB includes the cognitive subsets of attention (forward and backward digit span, and Stroop word and color test), language and related functions (Korean version of the Boston Naming Test [K-BNT]), visuospatial function (Rey Complex Figure Test [RCFT]), verbal memory (Seoul Verbal Learning Test [SVLT], immediate recall, 20 min delayed recall, and recognition), visual memory (RCFT, immediate recall, 20 min delayed recall, and recognition), and frontal executive function (semantic and phonemic generative naming using the Controlled Oral Word Association Test [COWAT]). The composite score of each cognitive domain was calculated by averaging the z-scores of the subtests that constitute the domain. Age-, sex-, and education-specific norms of each cognitive test, based on 447 healthy subjects, were available. The scores for each cognitive domain were classified as abnormal when they were at least 1.5 standard deviations below the norms of matched normal subjects.
Quantitative analyses of 18F-FP-CIT PET images
The methodology used to acquire and analyze the 18F-FP-CIT PET data were the same as previously described [12]. The 18F-FP-CIT PET scans were acquired using a GE PET-CT DSTe scanner (GE Discovery STE; GE Healthcare; Milwaukee, WI, USA), which acquires images using three-dimensional (3D) resolution at 2.3 mm full-width at half maximum. After subjects fasted for at least 6 h, they were intravenously injected with 5 mCi (185 MBq) of 18F-FP-CIT. Ninety minutes after the injection, PET images were acquired for 20 min in the 3D mode at 12 kVp and 380 mA. Image processing was performed using SPM8 (Wellcome Department of Imaging Neuroscience, Institute of Neurology, UCL, London, United Kingdom) and Matlab 2013a for Windows (Math Works, Natick, MA, USA). Quantitative analyses were based on volumes of interests (VOIs), which were defined based on a template in standard space. All reconstructed PET images were spatially normalized to the Montreal Neurology Institute template space using a standard 18F-FP-CIT PET template which was generated from 18F-FP-CIT PET and T1-weighted MR images of 13 normal controls. Twelve VOIs of bilateral striatal sub-regions and one occipital VOI were drawn on a co-registered spatially normalized single T1-weighted MR image and 18F-FP-CIT PET template image on MRIcro version 1.37 (Chris Rorden, Columbia, SC, USA). The striatum was divided into the anterior/posterior caudate, anterior/posterior putamen, ventral putamen, and ventral striatum. The boundaries of the striatal sub-regions were defined as described in our previous work [13]. These VOIs were adjusted using a minor translation in our in-house editing software ANIQUE [14]. DAT availability was calculated using the non-displaceable binding potential, which was defined as (mean standardized uptake value of the striatal sub-regions VOI–mean standardized uptake value of the occipital VOI)/(mean standardized uptake of the occipital VOI) [15].
MRI acquisition
MRI scans were acquired using a Philips 3.0 T scanner (Philips Achieva; Philips Medical Systema, Best, The Netherlands) with a SENSE head coil (SENSE factor = 2) as described in our previous work [11]. The high-resolution axial T1-weighted MRI data were obtained using a 3D T1-TFE sequence using the following parameters: 224×224 axial acquisition matrix; 256×256 reconstructed matrix with 170 slices; voxel size, 0.859×0.859×1 mm3; field of view, 220 mm; echo time, 4.6 msec; repetition time, 9.8 msec; flip angle, 8°. The diffusion-weighted MRI data were acquired using a single-shot echo-planar acquisition with the following parameters: 45 non-collinear, non-coplanar diffusion-encoded gradient directions; 128×128 acquisition matrix with 70 slices; voxel size, 1.75×1.75×2 mm3; field of view, 220 mm; b-factor, 600 s/mm2; echo time, 70 msec; repetition time, 7.663 sec; flip angle, 90°. WMHs were determined using FLAIR images (TR/TE/TI, 8502/132/2100 ms, 1 mm section thickness).
WM structural connectivity analyses
Imaging preprocessing and network construction
The diffusion-weighted MRI data were preprocessed by DTI techniques using the FMRIB’s Software Library (FSL; http://www.fmrib.ox.ac.uk/fsl/). Motion artifacts and eddy-current distortions were corrected using affine registration to a non-diffusion-weighted volume (b0) as a reference volume [16]. The detailed methodology for imaging preprocessing was the same as previously described [17].
Structural connectivity analysis
For the comparison of WM connectivity between each group with different WMH scores, permutation-based NBS was performed [18]. We first calculated edge-by-edge t-statistics between the WM connectivity of the two groups. Age, sex, years of education, and vascular risk factors were used as covariates. Connected components (clusters) were defined as set of edges with coefficients greater than a given threshold (2.5). We tested the significance of each cluster size within the empirical null distribution of maximum cluster sizes estimated by 5000 random permutations of the FA weights in design matrices. P-values were assigned to the original components and 0.05 was the significance level to determine significant clusters. We then identified hub regions from the resultant subnetwork composed of the significant clusters, which had a greater nodal degree than mean plus one standard deviation of all nodal degrees in the subnetwork.
To show detailed associations between the WMH scores and WM networks, we carried out permutation-based cluster-based statistics (CBS) for correlation analyses [19]. Specifically, Spearman’s partial correlation coefficients were computed after controlling for age, sex, years of education, and vascular risk factors. A given threshold (–0.35) identified clusters and significance was tested in the null distribution estimated by 5000 random permutations of WMH scores in the design matrices. Significant clusters were determined at the significance level of 0.05. We also identified hub regions from the extracted subnetworks.
We then showed relationships between neuropsychological functions and WM network weights having meaningful correlation with total WMH scores. Pearson correlation coefficients were computed between neuropsychological scores and the sum of weights in the subnetwork from CBS. We also obtained correlation coefficients between neuropsychological scores and total WMH scores for comparison. Next, differences in correlation coefficients between WMH scores and CBS subnetwork edge sum were analyzed using Wolfe’s test [20], and a false discovery rate controlling method was used for multiple-comparison correction.
Statistical analysis
Data are expressed as mean±standard deviation. Demographic characteristics were compared using a one-way analysis of variance for continuous variables followed by post hoc Bonferroni test, and Pearson’s χ2 test for categorical variables. WMHs rated by the Scheltens scale were compared using the Kruskal-Wallis test followed by post hoc Mann-Whitney U test. To compare DAT availability in each striatal sub-region, analysis of covariance was used while adjusting for age, sex, and PD duration as covariates. Statistical analyses were performed using SPSS (version 23.0; IBM Corporation, Armonk, NY, USA), and results with a two-tailed p-value < 0.05 were considered to be statistically significant.
RESULTS
Baseline clinical characteristics
Demographic characteristics and striatal DAT availability in PD are summarized in Table 1. Patients in moderate and severe WMH groups were older than in mild WMH group. K-MMSE scores were higher in mild WMH group than in severe WMH group. WMH burdens in periventricular, lobar, basal ganglia, and infratentorial regions demonstrated significant differences among groups. Among vascular risk factors, there was a higher incidence of hyperlipidemia in moderate and severe WMH groups than in mild WMH group (p = 0.044), and cardiac disease was more prevalent in severe WMH group than in mild and moderate WMH groups (p = 0.030). DAT availabilities in striatal sub-regions demonstrated no difference among groups.
Demographic characteristics in patients with PD
Values are expressed as mean±standard deviation or number (percentage) for the demographic characteristics, median (Q1, Q3) for the Scheltens scale, and estimated mean (standard error) for the striatal DAT availability. PD, Parkinson’s disease; WMH, white matter hyperintensities; BDI, Beck Depression Inventory; CCSIT, Cross-Cultural Smell Identification Test; K-MMSE, Korean version of Mini Mental Status Examination; UPDRS-III, the Unified Parkinson’s disease Rating Scale Part III; DAT, dopamine transporter. a107 patients (41 with mild WMH, 33 with moderate WMH, and 33 with severe WMH) underwent a CCSIT. bJ Neurol Sci
Neuropsychological assessment
Patients in severe WMH group exhibited poorer performance in language function than those in moderate WMH group (Table 2). In addition, patients in severe WMH group tended to exhibit poorer performance in visual memory and frontal executive function, relative to those in mild WMH group.
Neuropsychological data in patients with PD
Values are expressed as mean±standard deviation or number (percentage) for the demographic characteristics and composite scores of each cognitive domain. For the neuropsychological data, the values are expressed as estimated mean (standard error) white adjusting for age, sex, and years of education as covariates. PD, Parkinson’s disease; WMH, white matter hyperintensities; K-BNT, the Korean version of the Boston Naming test; RCFT, the Rey Complex Figure Test; SVLT, the Seoul Verbal Learning Test; COWAT, the Controlled Oral Word Association Test. aGroup comparison with composite scores. bSignificantly different (p < 0.05) between mild and moderate WMH groups by post hoc Bonferroni correction. cSignificantly different (p < 0.05) between moderate and severe WMH groups by post hoc Bonferroni correction. dSignificantly different (p < 0.05) between mild and severe WMH groups by post hoc Bonferroni correction.
Between-group comparison of microstructural connectivity according to WMH scores
A subnetwork whose microstructural connectivity was significantly disrupted in severe WMH group compared with mild WMH group was identified (Fig. 2A). The subnetwork consisted of 92 edges (p = 0.005) located in fronto-temporal regions and were more dominant in right hemisphere. These regions were heavily connected to six hub nodes in the identified subnetwork, including right superior frontal gyrus, right inferior frontal gyrus (triangular part), right and left inferior frontal gyrus (orbital part), left precuneus, and right middle temporal gyrus. The hub nodes were heavily connected with disrupted connections more than average; therefore, they represented brain regions most affected by abnormal microstructural connectivity. A subnetwork whose microstructural connectivity was more impaired in severe WMH group relative to moderate WMH group was also found (Fig. 2B). The subnetwork consisted of 32 edges (p = 0.021) located in fronto-parietal and temporo-occipital regions, and more dominantly in right and posterior parts of the brain. Four hub regions with stronger contributions to the identified subnetwork were discovered: right middle occipital gyrus; right superior parietal gyrus; left precuneus; and right middle temporal gyrus. There was no difference in microstructural connectivity between low and moderate WMH groups.
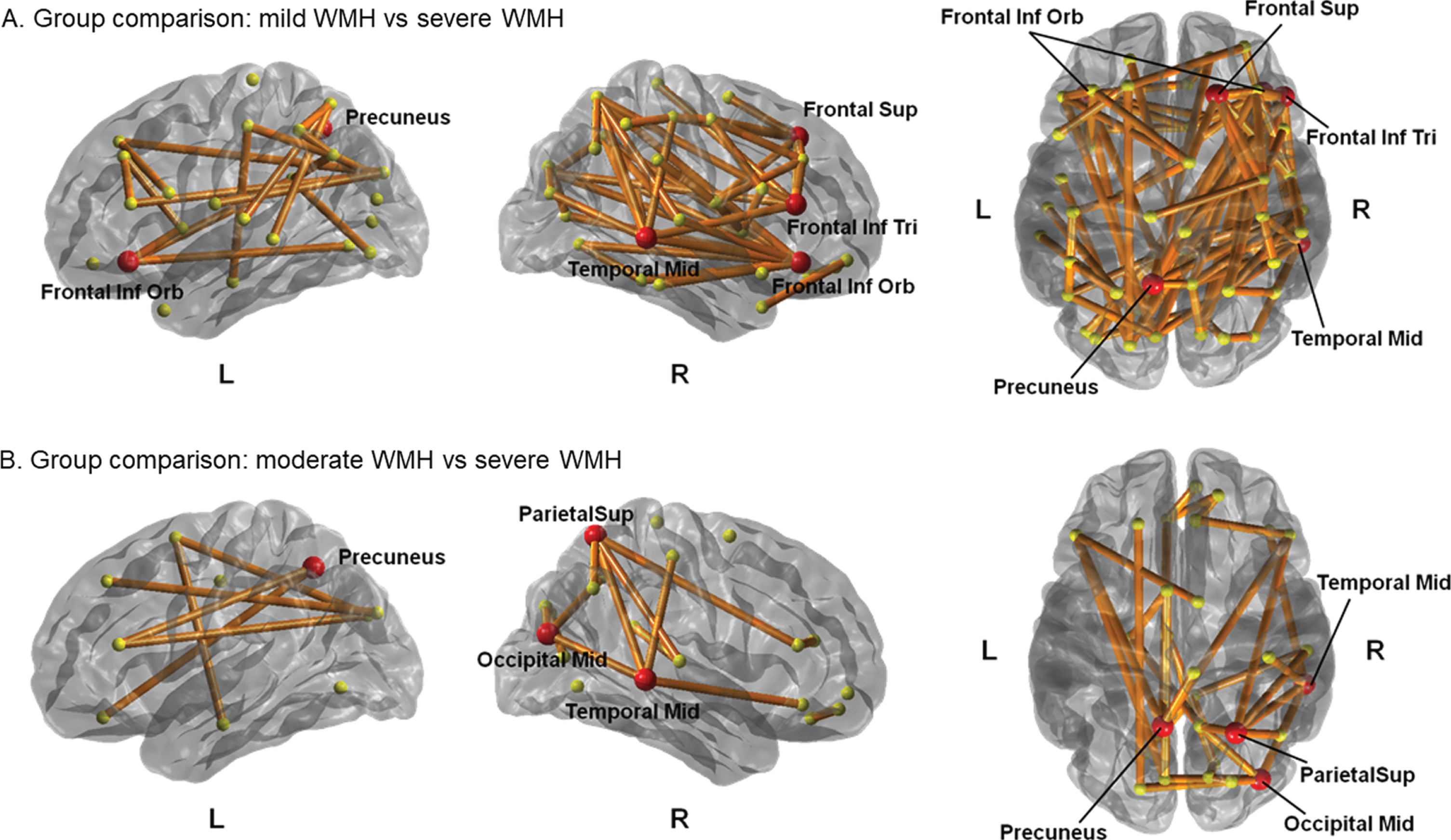
Between-group comparison of microstructural connectivity according to WMH scores. (A) Group comparison between PD group with mild WMHs and severe WMHs. (B) Group comparison between PD group with moderate WMHs and severe WMHs. PD group with mild and moderate WMHs exhibited decreased microstructural connectivity compared with PD group with severe WMHs. The red circles represent their hub regions representing the brain regions most affected by the white matter disruption. Frontal Sup, superior frontal gyrus; Frontal Inf Tri, inferior frontal gyrus (triangular part); Frontal Inf Orb, inferior frontal gyrus (orbital part); Temporal Mid, middle temporal gyrus; Occipital mid, middle occipital gyrus; Parietal Sup, superior parietal gyrus.
Correlation between WMH scores and microstructural connectivity
A subnetwork in which the severity of WMH was negatively associated with microstructural connectivity was found, thus demonstrating that higher WMH scores were associated with lower edge weights in PD (Fig. 3). This subnetwork comprised five edges (p < 0.001) that were interconnected and three hub nodes that were more highly interconnected than other nodes in right precentral gyrus, right superior parietal gyrus, and right middle temporal gyrus.
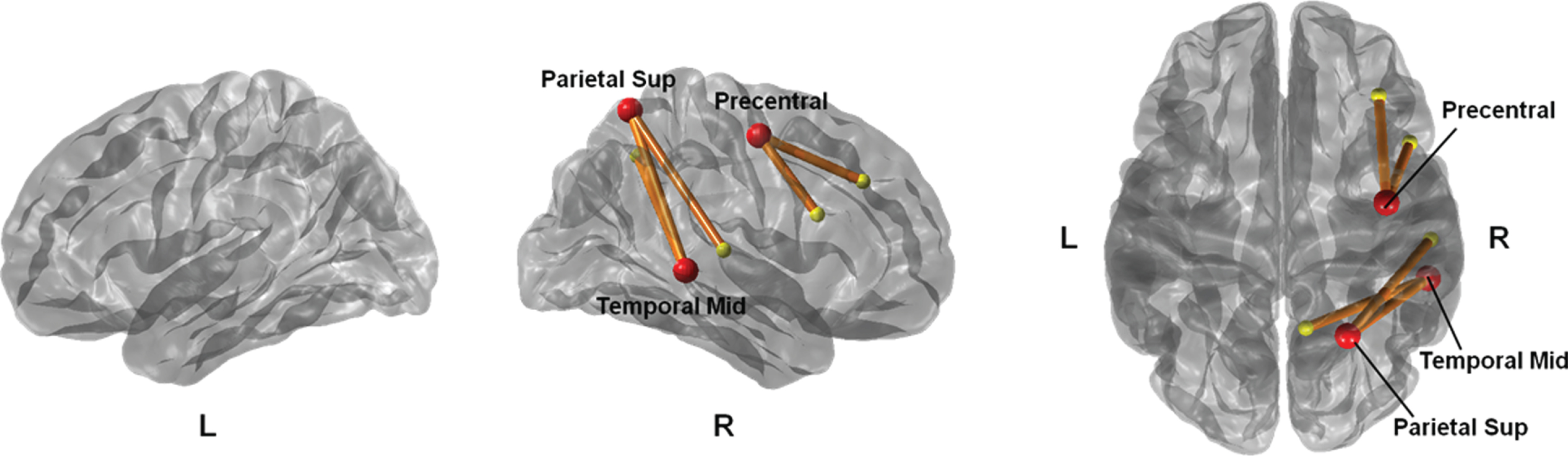
Correlation analysis between WMH scores and microstructural connectivity. The subnetwork consists of five edges of which weights negatively correlated with WMH scores. It contains three hub regions with greater connections with other nodes.
Comparison of correlation coefficients between WMH-associated microstructural connectivity or WMH scores and cognitive subsets in PD patients
Next, we assessed the association between subnetwork edge sum of WMH-associated microstructural connectivity or WMH scores and cognitive performance in PD (Table 3). The subnetwork edge sum was positively correlated with z-scores of cognitive subsets including Stroop color, semantic and phonemic generative naming using Controlled Oral Word Association Test (COWAT), Korean version of the Boston Naming Test (K-BNT), verbal memory (immediate memory, delayed memory, and recognition), and visual memory (immediate and delayed memory). Additionally, the subnetwork edge sum tended to be positively associated with attention function. However, none of cognitive subsets were significantly correlated with WMH scores. A comparative correlation analysis between subnetwork edge sum or WMH scores and individual cognitive subsets revealed that correlation coefficients of the subnetwork edge sum were significantly higher in supermarket COWAT (p = 0.013), delayed verbal memory (p = 0.036), and recognition of verbal memory (p = 0.036) than those of WMH scores (Table 3).
Correlations of white matter hyperintensity scores and white matter hyperintensity-associated structural connectivity with cognitive performance
Data are correlation coefficients (r). pa indicates the difference in the correlate coefficients between CBS and WMH scores with individual cognitive subdomains, and FDR-controlling method was used for multiple comparisons correction. CBS, cluster-based statistics; WMH, white matter hyperintensities; K-BNT, the Korean version of the Boston Naming test; RCFT, the Rey Complex Figure Test; SVLT, the Seoul Verbal Learning Test; COWAT, the Controlled Oral Word Association Test.
DISCUSSION
The present study investigated the relationship between WMH severity and microstructural connectivity and the association between cognitive performance and WMH severity or WMH-associated microstructural connectivity in de novo PD patients. Major findings were as follows: (1) PD patients with severe WMH exhibited poorer performance in language function than those with moderate WMH and in frontal/executive and visual memory function than those with mild WMH. (2) PD patients with severe WMH exhibited microstructural network disruption in both frontal, left parietal, and right temporal regions compared to those with mild WMH, and in both parietal and right temporo-occipital regions compared to those with moderate WMH. (3) There was a correlation between the severity of WMHs and microstructural connectivity in right fronto-parieto-temporal regions. (4) Disintegration of WMH-associated microstructural connectivity was correlated with dysfunction in attention, frontal/executive, and memory domains. However, there was no correlation between the severity of WMHs and any cognitive domain. These findings suggest that in PD patients, WMHs may contribute to changes in microstructural connectivity, specifically in right parieto-temporal regions and may lead to declines in semantic fluency and verbal memory function. In addition, this study reveals that the association between the presence of WMHs and cognitive dysfunction is not mediated by WMH lesions themselves, but through network disruption by WMH lesions in de novo PD patients.
WMHs appear homogeneous on conventional imaging markers; however, a previous post-mortem pathological study investigating WMHs in PD patients revealed different degrees of gliosis or demyelination in similarly appearing WMHs on FLAIR imaging [21]. These heterogeneous pathological findings of WMHs indicate that a radiologically similar appearance cannot reliably reflect the degree of tissue damage or demyelination, whereas quantitative imaging techniques including DTI, are more sensitive to tissue alterations than WMH lesions [22]. Ample evidences have demonstrated that diffusion metrics better reflect ongoing changes in WM injury over time, while visible WMH lesions on FLAIR imaging fail to capture continuous WM injury processes [23, 24]. Furthermore, in line with the present study, diffusion metrics rather than total WMH burden assessed using visual rating scales have yielded more consistent evidence linking cognition and WMHs [25, 26]. In addition, given that DAT availability in all striatal sub-regions did not differ among PD groups with WMHs, our study provides evidence that total WMH burdens contribute to dysfunction in cortico-cortical or cortico-subcortical microstructural networks in PD patients. Therefore, it is crucial to examine individual diffusion metrics when evaluating WM to better understand the role of WMHs in cognitive changes in PD.
As expected, the present study demonstrated that areas of disrupted microstructural connectivity were more extensive in PD with severe WMHs than those with mild and moderate WMHs, and involved bilateral frontal regions, right parieto-temporo-occipital regions and left precuneus. Moreover, the severity of total WMHs in PD was strongly correlated with disrupted microstructural connectivity, especially in right fronto-parieto-temporal regions. With regard to cognitive performance, there was no correlation between WMH severity and any cognitive domains. However, disintegration of WMH-microstructural connectivity was strongly correlated with cognitive dysfunction in working memory, frontal/executive, language, and memory domains. These findings were consistent with our previous morphometric study demonstrating that PD patients with high grade WMHs exhibited cortical thinning in the bilateral frontal cortex, right medial temporal gyri, and right parietal regions compared to patients with low-grade WMHs, and that WMH-related mean cortical thickness was significantly correlated with attention and executive function [27]. Along with the ventral stream of the fronto-parietal pathways, parietal and middle temporal gyri have been demonstrated to play an important role in episodic memory, working memory, and executive function [28, 29]. Therefore, the present study suggests that in PD patients, WMH-associated microstructural alterations in parieto-middle temporal areas may act as a key determinant of cognitive dysfunction in executive and memory performance via disruption of the ventral fronto-parietal pathway rather than a direct lesional effect of WMHs.
The precise mechanism describing the relationship between poorer verbal memory function and WMH-associated microstructural abnormal connectivity within right cortical areas is unclear. From a pathological perspective, a possible explanation for the relationship between cognition and altered WMHs-associated microstructural connectivity in this PD cohort is that the presence of WMHs may reflect more extensive Lewy body pathology in diffuse brain areas. In this regard, a previous study support that ischemic insults promote the oligomerization and aggregation of alpha-synuclein, a key protein attributed to the pathogenesis of PD, thus increasing neuronal toxicity [8]. In addition, other processes including low-grade inflammation, Wallerian degeneration, and axonal transport disruption may exert their action outside visible lesions by remote effects, which leads to cognitive dysfunction in WMH-independent cognitive domains [30]. Finally, vascular pathology itself contributes to impairment in the capillary network within multiple brain regions in PD. This damage to the capillary network can make brain tissue more vulnerable to the neurodegenerative process via increased oxidative stress, inflammatory responses, and blood-brain barrier instability in widespread brain areas [31]. Accordingly, it is possible that higher pathological burden related to WMHs may disturb cortical functions in extensive brain areas, thus contributing to cognitive impairment in remote cognitive domains.
Another interesting finding of the current study is that alteration in WMH-associated microstructural connectivity was greater in temporo-parietal regions lateralized in right hemisphere of PD patients. Similar findings of atrophy in these areas have been described and linked to cognitive impairment in PD [32]. It is not clear whether right hemisphere is inherently more vulnerable than the left in PD. However, because the ventral frontoparietal network is localized predominantly in the right hemisphere, neurodegeneration of this network may be responsible for right lateralized microstructural alterations in PD. Furthermore, a previous study reported that areas of cortical thinning would differ according to the side of symptom dominance in early and nondemented PD patients, specifically in the hemisphere contralateral to the symptomatic side in left-sided disease onset patients [33]. Similarly, because the current study included more patients with left-sided symptom onset than right-sided, it may have influenced results of hemispheric predilection.
This study had several limitations. First, although the scoring method for WMHs is widely used and was conducted in a blinded manner, this visual scaling method is less objective than volumetric analysis. Second, our study was limited by the absence of PD patients with dementia and heathy controls for comparison. This constraint prevented us from exploring associations between microstructural dysconnectivity and cognitive dysfunction across different cognitive stages. Third, the region-specific influence of WMHs on microstructural connectivity may differ depending on the anatomical distribution of WMHs; however, the current study lacked this consideration. Finally, further longitudinal research is necessary to examine whether these regions continue to contribute to cognitive dysfunction as the disease progresses.
In conclusion, our findings demonstrated that microstructural network disruption by WMHs rather than the WMH burden itself, contributed more to cognitive impairment in patients with PD, especially in association with microstructural dysconnectivity in parieto-temporal areas. The current data suggest that individual diffusion metrics when investigating WMHs are crucial in explaining the role of WMHs in cognitive dysfunction in PD.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2016R1A2A2A05920131). Phil Hyu Lee receives funding from NRF-2016R1A2A2A05920131. This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number : HI19C1132).
