Abstract
Background:
Little is known about the patients’ view on treatment with medical cannabis (MC) for Parkinson’s disease (PD).
Objective:
To assess the PD community’s perception of MC and patients’ experience with MC.
Methods:
Applying a questionnaire-based survey, we evaluated general knowledge and interest in MC as well as the frequency, modalities, efficacy, and tolerability of application. Questionnaires were distributed nationwide via the membership journal of the German Parkinson Association and locally in our clinic to control for report bias.
Results:
Overall, 1.348 questionnaires (1.123 nationwide, 225 local) were analysed. 51% of participants were aware of the legality of MC application, 28% of various routes of administration (ROA) and 9% of the difference between delta9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD). PD-related cannabis use was reported by 8.4% of patients and associated with younger age, living in large cities and better knowledge about the legal and clinical aspects of MC. Reduction of pain and muscle cramps was reported by more than 40% of cannabis users. Stiffness/akinesia, freezing, tremor, depression, anxiety and restless legs syndrome subjectively improved for more than 20% and overall tolerability was good. Improvement of symptoms was reported by 54% of users applying oral CBD and 68% inhaling THC-containing cannabis. Compared to CBD intake, inhalation of THC was more frequently reported to reduce akinesia and stiffness (50.0% vs. 35.4%; p < 0.05). Interest in using MC was reported by 65% of non-users.
Conclusion:
MC is considered as a therapeutic option by many PD patients. Nevertheless, efficacy and different ROA should further be investigated.
INTRODUCTION
Since 2017, medical cannabis (MC) is legally approved in Germany as a therapeutic option for patients with severe symptoms of Parkinson’s disease (PD), when previous therapies were unsuccessful or not tolerated, and a positive effect of cannabis on disabling symptoms is imaginable. In these cases, MC can be prescribed and is reimbursed by public and private health insurances.
The psychotropic delta9-tetrahydrocannabinol (Δ9-THC) and the non-psychotropic cannabidiol (CBD) are the commonest phytocannabinoids in Cannabis sativa (marijuana) [1] and play a crucial role for medical application. They act via the cannabinoid receptors 1 (CB-1R) and 2 (CB-2R) as most important receptors of the endocannabinoid system (ECS). However, while Δ9-THC activates both CB-1R and CB-2R, recent data suggest that CBD acts as functional antagonist by modulating CB-R1 [2] and CB-R2 [3] function. This might explain the lack of detectable psychoactivity of CBD compared to Δ9-THC. The influence of the ECS has been investigated intensively in parkinsonian models (for review, see [4]). Cannabinoids modulate basal ganglia function on two levels which are especially relevant for levodopa-induced dyskinesia (LID), i.e., the glutamatergic/dopaminergic synaptic neurotransmission and the cortico-striatal plasticity. Furthermore, activation of the ECS might induce neuroprotective effects related to direct receptor-independent mechanisms [5], activation of anti-inflammatory cascades in glial cells via CB-2R [6, 7], and anti-glutamatergic anti-excitotoxic properties [8]. These pathophysiological findings and a few uncontrolled and small controlled clinical studies as well as partly impressive single case reports (reviews [4, 9]) suggest that MC containing THC and/or CBD might have a potential clinical benefit for motor and non-motor symptoms in PD patients. Today, several different cannabis products and formulations are available for medicinal use but there is lack of controlled clinical studies addressing the differential effectiveness in PD (reviews [9, 10]). Cannabis is used by patients either as flowers with a very variable THC content or as extracts (oils or capsules), containing pure THC, pure CBD or both. While in Germany MC containing THC has to be prescribed by the physician, MC with pure CBD can be prescribed but also is freely available at the pharmacy, in internet stores or shops as a food supplement. MC products containing THC and/or CBD vary considerably regarding the percentage of the active ingredient and no standardised dosage is known. Today it is unclear, which type of PD patients and which symptoms might be best treated with MC, whether THC or CBD should be preferred and which route of administration (ROA) should preferentially be used. Furthermore, it is unknown to what extent PD patients are interested in MC at all, whether they are informed about the legal possibility of MC prescription or if they know the difference between cannabis applied as THC or CBD. It is not known, how many patients already used cannabis for PD symptoms relief and how they judged efficiency and tolerability.
This article aims to get some insights into the PD community’s perception of MC, to get an overview which role MC already plays as treatment in daily life, and to evaluate “the patient’s view” regarding the effect of cannabis application on individual motor- and non-motor symptoms.
METHODS
We performed a nationwide, cross-sectional, questionnaire-based survey among the members of the German Parkinson Association (Deutsche Parkinson Vereinigung e.V. [dPV]), which is the largest consortium of PD patients in German-speaking countries with 20,943 members in total (April 2020).
Patients were asked to complete a self-developed questionnaire (see below) which was embedded in the centre of issue no.148 (13th calendar week of 2019) of the dPV membership magazine with a circulation of 24,000 journals per edition. It was created as a separable survey sheet including general information on the study’s background and a prepaid reply envelope (costs were covered by the dPV). Subjects were asked to answer the questions autonomously and to return the survey anonymously by post until recruitment deadline on May 12, 2019.
Additionally, we consecutively recruited local PD patients who visited our movement disorder outpatient clinic within 6 weeks from March 4 to April 21 in 2019. Subjects were supplied with the same information sheet and asked to fill out the same questionnaire on-site and anonymously before putting it into a closed box. These local patients served as a control group to evaluate whether answers given in the nationwide survey are representative or biased, because PD patients with a special interest in treatment with cannabis products might primarily reply to the nationwide query.
Questionnaire
The self-developed German-speaking questionnaire contains 16 categories of questions composed of 25 single questions. 20 questions were constructed as single choice and five as multiple choice questions. The questionnaire was divided into four sections: 1) “Demographics” with patient and disease characteristics (age, gender, population of the patient’s place of residence, type of Parkinson syndrome, disease duration, currently present symptoms). 2) “Information” to evaluate the patient’s knowledge about cannabis and its application in PD; 3) “Experience” to assess whether patients already used cannabis and which type and ROA were applied, and 4) “Efficacy” and “Tolerability” of cannabis application in users (efficacy in general, best type and ROA, efficacy in comparison to dopaminergics, most responsive symptoms, side effects, fear of addiction). Each section was headlined with a short explanation of the general content of the questions. Questions and answers were also partially described in more detail using brief explanations or examples. For the English translation of the questionnaire, please see Supplementary Fig. 1
Statistics
The statistical evaluation was carried out with the IBM™ SPSS™ software (version 23). Tables and figures were created with SPSS™ and Excel 2003 (Microsoft™). The data were evaluated using descriptive statistics and frequency distributions. Pairwise deletion was applied for missing data.
To test whether the nationwide patient sample was representative, answers were compared with the patient data locally collected in our clinic (group comparison). In particular, two hypotheses were tested: that 1) general interest in cannabis use and/or 2) past or present usage of cannabis had led to increased participation in the nationwide study. In order to largely rule out that the local survey in our clinic was not subject to selection bias, a response rate of > 70% of patients consecutively asked for participation was defined as prerequisite.
For group comparisons, the mean values of interval-scaled variables were tested for significance using the t-test. For nominally scaled variables, the frequencies were compared and tested for significance with the Pearson χ2 test. The exact Fisher test was used for expected frequencies < 5.
To assess the influence of patient characteristics on cannabis aspects, binary logistic regression was applied.
For all analyses, levels of significance were set at 5% (p-value < 0.05).
Ethics
The study was approved by the local ethics committee of the Medical Council Hamburg (reference number WF-008/19).
Data availability statement
Anonymized data will be shared by request from any qualified investigator.
RESULTS
Response rates
Until deadline, a total of 1,126 of the 24,000 questionnaires sent nationwide was returned. Three questionnaires were excluded from analysis, one due to missing demographic data, one that reports as free text on an already deceased patient by his wife and one with strongly contradicting combinations of answers. Therefore 1,123 questionnaires (4.7%) were available for statistical analysis.
At our department, 225 of 250 PD patients who were consecutively asked to take part in the study returned the questionnaire (90.0%, i.e., predefined necessary return rate was given). Here, all questionnaires could be used for analysis.
Missing data for different variables of interest was mostly low (<6%), except for questions 12b (10.5%), 13a (21.3%), and 15 (12.4%), but without significant differences between the nationwide and the local cohort. We therefore assume that data was missing at random and used pairwise deletion for statistical analysis.
Demographics
In total, the data from 1,348 study participants (54.7% male, 45.2% female, 0.1% no answer) were evaluated, containing of 83.3% from the “nationwide” survey (n = 1,123) and 16.7% from the “local” survey in our movement disorder outpatient clinic (n = 225). Table 1 shows the detailed patient characteristics of both groups together and separately. Mean age was 71.6 (SD±8.9; range 33-92) years and mean disease duration was 11.6 (SD±7.2; range 1-42) years. Most of the study participants (n = 540) live in places with less than 20,000 inhabitants (40.7%). 879 (65.2%) of subjects reported to suffer from idiopathic Parkinson syndrome (PS). 77 subjects aligned themselves as atypical PS (5.7%), and the other patients did not to know the aetiology of their PS (22.0%), reported “other” aetiology (1.1%) or made no declaration (6.0%).
Patient characteristics
Note: Data of years are given as mean values; SD, standard deviation.
Demographics of the two study groups were significantly different in some aspects: In the local cohort, male patients predominated, they were younger with shorter disease duration and lived more frequently in places with more than 500,000 inhabitants (p < 0.001 for all aspects). Groups did not differ regarding the type of PS.
Clinical symptoms
Figure 1 gives an overview of the participants’ PD symptoms. Akinesia, postural disturbance, tremor, muscle cramps and cognitive impairment were reported mostly and were present in > 50% of participants. Overall, 10 out of 17 default clinical symptoms were reported significantly more frequently in the nationwide group, i.e., postural instability, falls, pain, sleep disturbance, fear/panic (all p≤0.001) and bladder dysfunction, depression, freezing, muscle cramps and slight disorder of memory (all p < 0.05). 140 subjects added symptoms in the field “other”, with bladder dysfunction (n = 21), nightmares (n = 21) and speech disorders (n = 16) being most frequently reported.
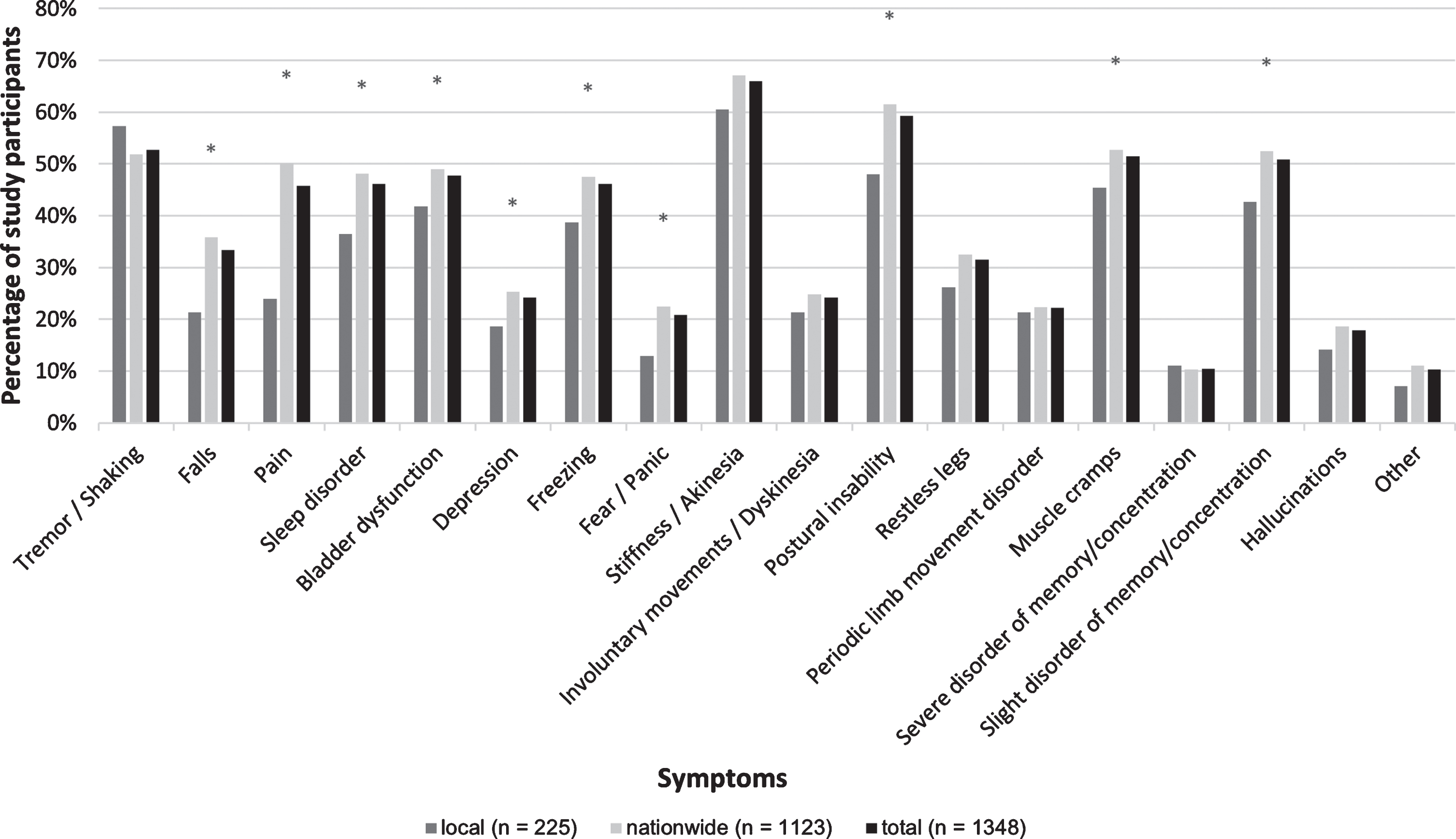
Clinical symptoms. Self-reported symptoms of all study participants (Multiple answers possible). *significant (p < 0.05) for between group comparison (local vs. nationwide).
Information about cannabis treatment in PD
Overall, 689 (51.1%) participants felt to be informed about the possibility of legal applicability and medical prescription of cannabis for PD in Germany. 381 (28.3%) subjects reported knowledge about various ROA of cannabis. Only 118 (8.8%) patients knew the difference between THC and CBD.
In the local group, significantly more participants were informed that there are various medical ROA of cannabis (p = 0.05). The nationwide and local group did not differ regarding relatively good knowledge of approval of MC and lack of knowledge regarding the difference between THC and CBD.
Experience with cannabis
Overall, 202 patients (15.0%) had tried cannabis in their life with 28 regular users (13.9%), 65 occasional appliers (32.2%) and 86 patients (42.6%) who tried it only once (no answers n = 23 [11.4%]).
113 patients (8.4%) had applied cannabis due to PD without gender difference (50.4% male). Here, mean age of users was 66.4 (±10.7) years and mean disease duration 11.6 (±6.5) years. Younger age was associated positively with the prevalence of cannabis consumption (r –0.60, p < 0.001). Cannabis users were on average 5.6 years younger than non-users (mean 66.4 vs. 72.0 years, p < 0.001). Every year of age lowers the likelihood to be a user by about 5.9%. 91% of users were aware of the legal status of medical cannabis and knowledge of legal situation, the various ROA and the difference between THC and CBD were significantly positively related to the frequency of cannabis use (all p < 0.001). Experience with cannabis was higher in patients living in large cities with more than 500.000 inhabitant compared to patients living in small places with less than 20,000 inhabitants (15.0% vs. 6.4%; r = -0.87, p = 0.001) and the odds-ratio for cannabis consume in these patients was 2.4-fold higher.
Study participants in the local cohort had tried cannabis significantly more often in general (p <0.001) as well as due to their PD (p = 0.041) than nationwide participants but without significant differences regarding used active substance (e.g., THC, CBD, or both), ROA, and frequency of daily application (Fig. 2).
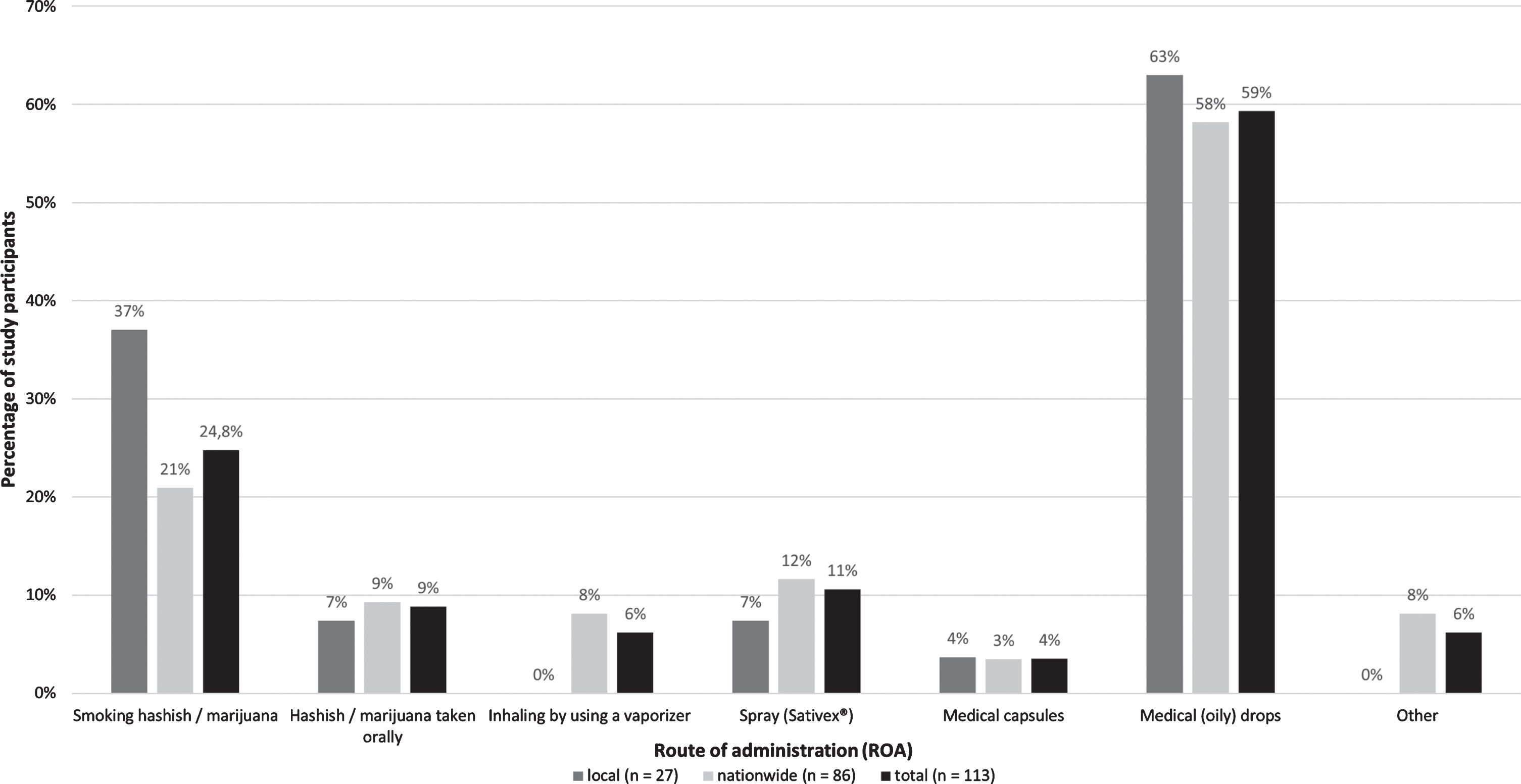
Route of administration (ROA). ROA reported by all participants who used cannabis because of their PD (multiple answers possible).
Among the 113 PD-related cannabis users, 11 (9.7%) reported application of THC only, 45 (39.8%) of CBD only and 23 (20.4%) of both THC and CBD. 20 (17.7%) users did not know which type of cannabis they had taken and 14 (12.4%) gave no information on the active ingredient used. In summary, 79 (69.9%) users could specify the type of cannabis they applied but only 56/113 (49.6%) users reported to know the difference between THC and CBD.
Mean disease duration of cannabis users was 11.6 years and was comparable to that of non-users. Tremor, but not other motor- or non-motor symptoms, was reported significantly more often by cannabis users compared to non-users (61.9% vs. 52.0%, p = 0.043). On the other hand, non-users reported more frequently slight problems with memory and concentration (51.8% vs.41.6%, p = 0.039) and hallucinations (18.5% vs. 10.6%, p = 0.036).
Cannabis was most frequently applied exclusively as medical liquids/drops (n = 54/113; 47.8%) or in combination with hashish/marijuana (67/113; 59.3%). The latter was applied by 28 PD patients (28/113; 24.8%).
Interest in applying MC was reported by 808/1235 (65.4%) of subjects who had not used cannabis due to PD symptoms yet. They would prefer medical capsules (44%) or liquids/drops (31%) rather than a spray (24%) or leaves via a vaporizer (11%) [multiple answers possible].
Fear of physical and/or psychological addiction was affirmed by 6.7% of cannabis users. Although not intended in the query, 90 of the non-users also replied to this question and 41 of them (45.6%) stated fear of addiction.
Efficacy and tolerability
General aspects
Overall, more than half of users (n = 61/113 [54.0%]) reported a clinical benefit due to cannabis use (these patients are classified as “therapy responders” in the following paragraphs). The success rate correlated significantly positively with the frequency of use (χ2 = 16.3, p < 0.001). Users with frequent application reported more frequently (79%) efficacy compared to users with occasional (67%) or single (25%) use. In successful applications, the effect was rated by half of the patients (n = 31/61; 50.8%) to be better than that of levodopa/dopamine agonists and an equal effect was reported by 14/61 of patients (23.0%). Three of four MC users with atypical PS reported general improvement of symptoms (75%). Cannabis use was generally tolerated in 96/113 users (85%) with side effects reported in 41 (36.3%) of subjects. The most common side effects included fatigue (54%), dizziness (37%) and ravenous appetite (22%) (Fig. 3). Hallucinations were mentioned as free text by four patients. Nine patients discontinued using cannabis due to side effects (fatigue n = 2, dizziness n = 2, nausea n = 1, not specified n = 4).
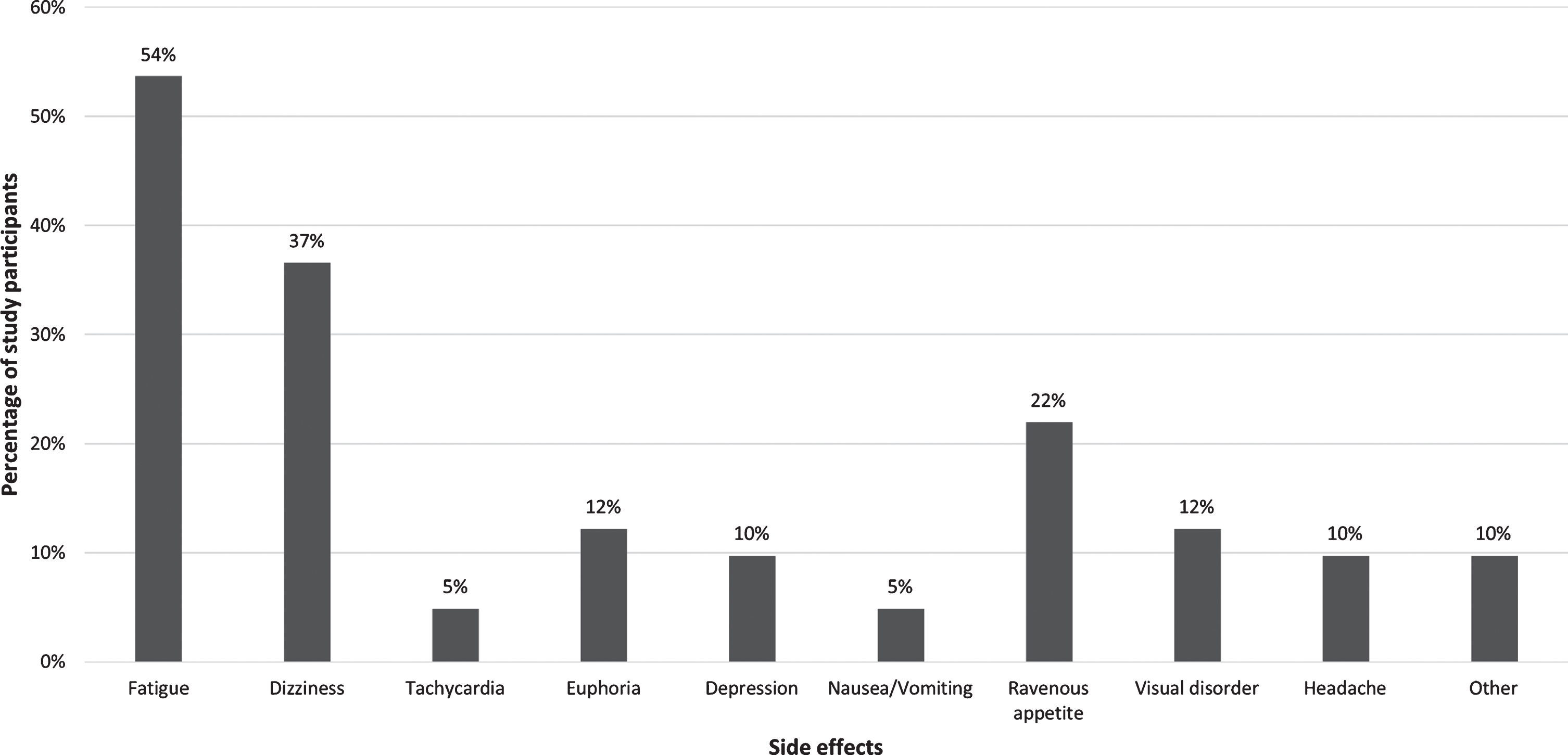
Side effects of cannabis use. Side effects reported by participants who used cannabis to relief PD symptoms. Variables are reported as percentage of the total of n = 41 cases in both the local and nationwide group who reported side effects (multiple answers possible).
The local and the nationwide study cohorts did not differ significantly in any of the aspects regarding efficacy and tolerability.
Efficacy of cannabis on PD symptoms
Table 2 provides an overview of how typical PD symptoms responded to cannabis. The symptoms “pain” and “muscle cramps” were most often reported to be improved (43.9% and 41.4%, respectively). Overall, nine different motor and non-motor symptoms were referred to be relieved in at least 20% of patients applying cannabis.
Efficacy of cannabinoid therapy on different PD symptoms
Note: The percentages given refer to the total number of participants who used cannabis to relief the given symptom. Accordingly, the number differs from the total number of cannabis users due to PD (n =113).
Comparison of THC and CBD on efficacy and tolerability
To approximately assess whether general efficacy and tolerability are more related to THC or CBD, we compared the effects of the two most frequently applied therapies of inhaled THC as content in “hashish” (as gum extract) or “marijuana” (as leaves) with the oral administration of pure CBD. Using exclusively THC, ROA mainly was inhalation via a “joint” (n = 18) and less frequently via a vaporizer (n = 2) or applied as a combination (“joint” and vaporizer; n = 2). Pure CBD was applied mainly as oily liquid/drops (n = 34/54; 63.0%) and less frequently as capsules (n = 3/3).
Table 3 shows the efficacy and tolerability of treatment with THC (n = 22) compared to CBD (n = 37). A beneficial clinical effect was reported by 68.2% of subjects in the THC and 54.1% of subjects in the CBD group (between group difference n.s.). Among therapy responders in both cohorts, subjects in the THC group rated the efficacy by trend more frequently better compared to levodopa/dopamine agonists (12/15 [80.0%] vs. 7/20 [35.0%], p = 0.06) Efficacy on stiffness/immobility/akinesia was more frequently reported in the THC group (8/16 [50%] vs. 4/26 [15.4%], p = 0.03). Rate of improvement of other PD symptoms was not different between groups. Noteworthy, freezing improved in 4/5 (80.0%) of patients inhaling THC-containing cannabis using joint but not when applying a vaporizer (0/2, 0%) and only in 5/21 (23.8%) of subjects taking CBD orally.
Comparison of THC and CBD on efficacy and tolerability
Smoking/inhaling THC was by trend less frequently tolerated than orally taken CBD (81.8% % vs. 91.9%, p = 0.07) and provoked significantly more often side effects (54.5% vs. 18.9%, p = 0.01). Mainly, fatigue was reported in 27% and 16% and dizziness in 18% and 8% of THC and CBD users, respectively. Ravenous appetite occurred in 18% of THC but only 3% of CBD users. Among patients with side effects, 33% in the THC and 29% in the CBD group stopped cannabis application.
DISCUSSION
This work presents a large questionnaire-based survey about the “real-life” situation regarding the therapy with medical cannabis (MC) in PD patients in Germany. We assessed the patients’ general knowledge and interest in this treatment as well as the frequency, modalities, and subjective efficacy and tolerability of application. We found that half of study participants are well-informed about the option of a legal prescription of MC but less than one-third about its different ROA, and even less than 10% know the difference between THC and CBD. Only a minority of 8.4% of participants had used cannabis due to PD, almost two-thirds orally as oily liquid (CBD) and one quarter inhaled as hashish/marijuana (THC). Remarkably, half of the cannabis users reported clinical benefit with mainly pain and muscle cramp relieve in 40% of users and motor (stiffness/akinesia, freezing, tremor, restless legs syndrome) as well as non-motor symptom reduction (depression, anxiety) in about 20% of users. Overall tolerability was reported to be good, but slightly better for pure oral CBD compared to inhalative THC-containing cannabis. However, the latter was reported more frequently to improve akinesia. Noteworthy, two-thirds of non-users are interested in applying MC.
Representativeness of the nationwide survey
With a total of 1,348 participants, this is, to our knowledge, the largest study investigating the patients’ view on cannabis therapy in PD. Interestingly, 22% of respondents reported not to know which type of Parkinson syndrome they have and 6% made no declaration. We interpret that more likely as a lack of knowledge of the asked medical terms “idiopathic” or “atypical” than of incomprehension of the personal PS type. Because the nationwide survey was addressed to members of the German Parkinson Association, we assume that the majority of these 28% of patients suffer from idiopathic PS (PD).
Although 1,123 patients responded to the nationwide survey, this was only a return rate of 4.7% with respect to the vast circulation number of the membership journal. In contrast to surveys that were directly addressed to patients and which showed average response rates of 45±20% [10–89%] for paper surveys and 34±22% [range 7–88%] for web-surveys [11], our embedded questionnaire in the journal likely has been missed by many patients.
Because the study design of the nationwide survey includes a risk of a “report bias”, we added a consecutively recruited local PD patient cohort with a response rate of 90% as control. The high response rate confirms that personally addressed surveys result in higher response rates than unselected, non-directly addressed surveys. Furthermore, the high local participation rate and epidemiological data suggest that our local cohort reflects an average outpatient urban PD population with a wide range of age and disease severity. Compared to the nationwide group, patients in the local group were about 20% more male, in average 5 years younger, had a disease duration which was 2.2 years shorter, reported less often problems regarding typical PD symptoms and had higher experience with cannabis (26.7% vs. 12.6%). The slightly younger age in our local cohort can possibly be explained by the fact that only outpatient patients—often with better mobility compared to older patients—took part in our survey whereas the questionnaire which was embedded in the nationwide journal might have also reached the elderly, less mobile clientele. Furthermore, our clinic is located in a large city and federal state with 1.8 million inhabitants and has the lowest proportion of the age group “65 years and older” compared to all other 15 federal states in Germany (Statista Research Department, 06.02.2020).
As crucial result, answers of the local and the nationwide cohort did not differ significantly in any item regarding interest in cannabis therapy in non-users, route and frequency of administration or efficacy and tolerability of cannabis application. We therefore assume that our results are representative for the average outpatient PD patient collective in Germany. To increase the study power, we finally analysed both cohorts combined together as one sample.
Interest in cannabis and experience with cannabis application
Half of participants were informed that cannabis can be used legally when medically prescribed. Knowledge about various ROA or the difference between THC and CBD was slim among all study participants. This lack of knowledge and a possibly associated fear of these substances could discourage patients from considering cannabis as therapeutic option. Accordingly, despite two-thirds of non-users are interested in applying MC, almost half of 90 non-users were concerned about addiction in case of using MC.
Fifteen percent of all subjects had experience with cannabis and more than half of them (8.4%) tried cannabis to diminish PD symptoms. Knowledge about the difference between THC and CBD was much higher in PD-related cannabis users compared to all study participants (49.6% vs. 9.1%). However, even more cannabis users could specify the type of cannabinoid they used (69.9%). This suggests that knowledge about which substance has been applied (THC or CBD) does not automatically mean that users know about the different clinical effects of both cannabinoids. This seems not exceptional because, e.g., many patients can name their specific dopamine agonist but do not know the differences between dopamine agonists in general. However, it is remarkable especially with view to differences regarding the psychoactive effects of THC but not of CBD.
Almost two-thirds of cannabis users (59.3%) applied medical liquids or drops and one quarter (24.8%) hashish or marijuana without gender difference. Subjects taking exclusively medical liquids/drops (n = 54/113; 47.8%) applied mainly pure CBD (n = 34/54; 63.0%). CBD therefore seems to be the mostly applied cannabinoid.
The quite long and comparable mean disease duration of more than 11 years in cannabis users and non-users contradicts the assumption that cannabis use might be related to the youth or a potential “life style” aspect. Since the symptom “tremor” was found significantly more often among users, this symptom seems to be a reason for PD patients to try cannabis, while disorders of memory/concentration and hallucinations might discourage PD patients from trying cannabis. The overall burden of other symptoms does not seem to play a significant role in the decision to take cannabis.
Interestingly, only 10% applied cannabis as spray which, in Germany, contains identical portions of THC and CBD (each 50%) and has already been approved for years against spasticity in patients with multiple sclerosis.
Efficacy
More than half of users (54.0%) reported a beneficial clinical effect due to cannabis application. This is less than reported in a previous structured observational telephone survey in 47 PD patients with an overall improvement in 82.2% of users [12]. The authors assessed similar aspects of cannabis application as in our study, but patients there were more frequently men (85%) and cannabis was consumed in most patients as THC containing “joint” (81.0%).
Half of users in our study who reported relief of symptoms rated the efficiency of cannabis better than that of levodopa or dopamine agonists and 23.0% as at least comparable. However, cannabis intake might be related to a relevant placebo effect because of high patient expectations and conditioning but even than it can be considered as therapeutic effect [13].
In our study, improvement of the motor symptoms akinesia, freezing and tremor and the non-motor symptoms sleep disturbance, depression, anxiety and restless legs syndrome was reported by more than 20% of participants applying cannabis. Pain and muscle cramp relief was reported by even more than 40% of subjects. Currently, only limited and inconclusive data are available regarding the efficacy of cannabis products on single motor- and non-motor symptoms in PD (for review see [4]). Three out of six studies found a positive [12, 15], and three a negative [16–18] effect of cannabis on rigor, tremor and bradykinesia.
Assessing motor-symptoms, improvement of dyskinesia was reported only in 2/29 of our patients with dyskinesia (6.9%). This is noteworthy because the worldwide mostly spread video (“Ride with Larry” with 3.7. million views on YouTube) shows a tremendous effect of cannabis oil on dyskinesia. Again, available data assessing levodopa-induced dyskinesia (LID) are inconclusive with two positive (one randomized controlled trial, RCT) [15, 19] and 3 negative (all RCT) [17, 20] studies. Interestingly, 13/52 of our patients (25.0%) reported improvement of freezing of gait (FOG). This is of special interest because there is no proven medication against FOG which frequently occurs in advanced disease stages despite optimized medication and increases the risks for falls. Noteworthy, Balash et al. found that mainly smoked MC reduced significantly complaints of falling from 22/47 (46.8%) to 6/18 (33.3%) of patients.
Regarding non-motor symptoms in PD, pain relief was found in two open observational studies after mainly smoking cannabis [12, 14] but was not seen in one RCT after oral intake of combined THC/CBD [17]. However, two recent large meta-analyses suggest that cannabinoid-based pharmacotherapy may serve as effective replacement/adjunctive option against pain in general [21] and in multiple sclerosis in which combined THC/CBD improved pain [22] and is approved for the treatment of moderate to severe (painful) spasticity [23, 24]. In line with our results, others also found improvement of sleep due use of MC. REM-sleep behaviour disturbance was improved in 4/4 PD patients (100%) taking CBD [25] and improvement of sleep quality was found in 12 out of 22 PD patients (54.5%) [14] and 33 out of 46 (71.7%) [12] exclusively or mainly smoking cannabis, respectively. However, the cross-over RCT in 19 patients by Carroll et al. [17] did not confirm a positive effect of orally administrated THC/CBD on sleep. In our study, 9 of 32 patients (28.1%) reported improvement of depression, which is less than reported before with improvement of mood in 35/46 (76.1%) patients in a previous investigation [12].
Symptoms relief were described mainly in studies with long-term cannabis application for at least eight [14], twelve [12] or six [18] weeks. In line with that, success rate in our study correlated significantly with the frequency of cannabis use. However, it is unclear whether frequent application is the reason or the result of reported symptom relief.
Tolerability
In the majority of patients (85%) cannabis was well tolerated, but one-third reported unwanted or side effects, mainly fatigue, dizziness and ravenous appetite. These are well known adverse effects of THC [4]. Nevertheless, only 9/113 patients (8.0%) discontinued therapy because of intolerance which is in accordance to other studies describing discontinuation of medical cannabis due to side effects in 5/47 patients (10.6%) [12] or generally as rare [26, 27]. In our study, hallucinations were mentioned as free text by only four patients but we did not ask for hallucinations as preselected question. Rate of hallucinations might have been higher as has been reported before with 17% [12]. However, hallucinations likely are related to THC because clinical studies did not reveal psychotic side effects for CBD [18], whereas vice versa CBD might even have some anti-psychotic effects in PD patients [28].
Comparing the efficacy of smoked THC and orally administered CBD
MC can be applied as pure THC, CBD or mixed forms with different ratios of both ingredients. To make it even more complex, it has to be considered that inhaled cannabis types contain different phytocannabinoids with a THC content between 4% and 28% in hashish and 3% and 22% in marijuana [29] or that Cannabis sativa used as leaves or hashish contains more than 100 other phytocannabinoids with unknown influence on PD symptoms. The ROA vary tremendously. Leaves can be smoked with and without tobacco, inhaled via bong or vaporizer as well as boiled with water and applied as tea. Furthermore, illegal and medical leaves contain different ratios of THC/CBD. Oral administration of cannabis is possible as capsules, liquid oils, leaves, hashish or spray, again with different ratios and concentrations of CBD and/or THC. This makes it difficult to compare studies. Overall, the available clinical data on the effects of THC and CBD in the literature are very limited.
Noteworthy, studies or observations reporting responsiveness of PD symptoms to cannabis often assessed patients who smoked cannabis containing THC [12, 14] while negative reports were often obtained in studies investigating oral ROA [17, 18]. To evaluate whether the efficacy of THC compared to CBD or the application of inhalation compared to oral administration might have substantially different effects, we compared these two main groups of users. Despite a comparable response rate to THC and CBD regarding general improvement of PD symptoms, level of efficacy seems to be higher when inhaling THC compared to applying CBD orally, because patients in the THC group reported more frequently improvement of akinesia and higher efficacy compared to dopaminergics. Noteworthy, freezing improved in 4/5 (80%) of patients inhaling cannabis as joint but only in 5/21 (23.8%) of subjects taking CBD orally. Because freezing often is not adequately controlled with usual medication, smoking cannabis leaves might be a therapeutic option. Only 3 participants reported to take pure THC orally as Dronabinol drops, so that a comparison of orally applied THC and CBD was not meaningful.
Limitations of the study
The study has some limitations. The response rate of the nationwide survey likely could have been increased by addressing all patients personally via mail and drawing more attention to the study. However, relevant additional mailing costs did not allow this procedure.
We applied a German-speaking self-developed questionnaire which has not been validated before. However, so far there is no validated questionnaire addressing the subjective patient evaluation of prevalence, efficacy or tolerability of cannabis application in PD. The high rates of completed returned questionnaires indicate a good understandability of the questions. However, it was intended that the last question (#16) on fear to get physically and/or psychologically addicted to cannabis was only answered by patients who already used cannabis due to PD. Nevertheless, 90 subjects without cannabis experience also answered this question. A second version of the questionnaire should state this intention more clearly. Because it is not uninteresting whether also non-users might be afraid of addiction, we included the answers of these patients into our analysis. Furthermore, we had to translate the questionnaire into English for this publication without language validation of the translation. Future studies should focus on more objective ways to examine the therapeutic effect of MC.
We did not evaluate whether the individual symptom-related burden of disease of cannabis users and non-users differed or if a certain bothersome symptom led to the use of cannabis. Also we did not ask how many years after the onset of the disease the participants first used cannabis and whether patients felt a vanishing effect of standard dopaminergic drugs. These aspects can affect cannabis usage and more studies addressing these points are needed.
We intended to evaluate the patient’s view and knowledge and to assess the patients’ very subjective opinion about effects of cannabis on their PD symptoms. It was not intended and due to the study design and the highly variable modalities of cannabis application not possible to objectively differentiate between the treatment effects of different cannabis products or degree and duration of subjective treatment effects. Accordingly, we did not ask for doses used or duration of treatment.
However, the study faces general limitations that come along with a subjective self-report. Especially participants with a positive effect on their PD symptoms may tend to exaggerate the overall positive effects. This includes a possible pronounced placebo effect due to high expectations of patients on the efficiency of cannabis.
Furthermore, the low response rate may bias the findings toward those who had a positive treatment effect. However, our consecutively recruited and highly responsive control group, which was set up to be devoid of that bias, showed highly comparable results which makes a relevant selection bias unlikely.
The study design did not allow to distinguish patients with and without medical prescription of THC-containing leaves or CBD extracts. We assume, that both cannabis formulations were mainly applied without prescription, because doctors are reluctant to prescribe THC as leaves and CBD extracts are freely available and only in a few states reimbursed by health insurers.
Conclusion
Our study offers insights into the PD community perception of MC and shows that cannabis is applied in almost 10% of patients against motor- and non-motor symptoms. Results suggest that MC might be helpful for selected PD patients with insufficient symptom relief despite their usual anti-parkinsonian medication. Controlled clinical studies investigating the efficiency, tolerability and best ROA of MC therapy in PD are desirable.
CONFLICT OF INTEREST
Ferhat Yenilmez reports no disclosures.
Dr. Odette Fründt, MD: Congress attendance fees: AbbVie, Abbott/St. Jude, Lecture fee: Daiichi-Sankyo
Dr. Ute Hidding reports no disclosures
Prof. Carsten Buhmann: Fees for advisory board participation: UCB Pharma, Zambon. Lecture fees: AbbVie, BIAL, Desitin, GE Healthcare, Grünenthal, Licher, Medtronic, Novartis, TAD Pharma, UCB Pharma, Zambon
Footnotes
ACKNOWLEDGMENTS
We thank the National German Parkinson Association (Deutsche Parkinson Vereinigung e.V. [dPV]), especially its chairman Dr. Friedrich Wilhelm Mehrhoff, for the support of this study.
We also thank Eik Vettorazzi (Institute of Medical Biometry and Epidemiology, University Medical Center Hamburg-Eppendorf, Martinistraße 52, 20246 Hamburg, Germany) for statistical support.
