Abstract
Background:
Increasing doses of oral antiparkinson medications are indicated in advanced Parkinson’s disease (PD), but little is known about sustainment of high-dose regimens.
Objective:
To investigate sustainment of high-dose oral medication regimens in Medicare beneficiaries with incident advanced PD.
Methods:
This retrospective cohort study utilized 100%fee-for-service Medicare claims from 2011–2013. We identified advanced PD using a pharmacy claims-based proxy and selected patients who initiated a new high-dose oral medication regimen (daily levodopa equivalent dose [LED] >1000 mg/day for ≥30 days) in 2012. In the following 12 months, we examined: 1) annual proportion of days covered (PDC)≥0.80 and 2) presence of a ≥ 90 day continuous gap at varying dosage thresholds: the initial >1000 mg/day, >800 mg/day, >500 mg/day, or >0 mg/day.
Results:
We identified 9,405 patients with advanced PD (mean age 77.4 [SD 6.8] years; 53%men). Only 5%maintained a regimen of >1000 mg/day at PDC ≥0.80; 75% had a ≥ 90-day gap in that dosage level. At a dosage threshold of >800 mg/day, 20% had a PDC ≥0.80 and 53% had a ≥ 90-day gap; at >500 mg/day, 56% had a PDC ≥0.80 and 19%had a ≥ 90-day gap; and at >0 mg/day (any dose), 76% had a PDC ≥0.80 and only 10%had a≥90-day gap.
Conclusion:
Few patients with advanced PD sustained a high-dose oral medication regimen in the year following initiation, but most sustained a substantially lower-dose regimen. Strategies to improve advanced PD treatment are needed.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a chronic, progressive neurodegenerative disorder that results in an array of motor and non-motor symptoms and lacks proven disease-modifying treatment options [1]. Evidence-based reviews recommend treatment with levodopa, dopamine agonists, monoamine oxidase B inhibitors, catechol-O-methyltransferase inhibitors, and amantadine to manage PD and offer symptomatic relief [2]. As the disease advances, there is a physiological need for greater dopamine replacement due to progressive neuronal loss, and subsequent motor decline. Thus, patients with advanced PD are likely to require frequent dosing (e.g., 5 or more times per day) and high pill counts (e.g., 10 or more oral pills per day) as well as polypharmacy [3], resulting in complex medication regimens that potentially place patients at risk of suboptimal adherence [4, 5]. There are additional clinical challenges to maintaining dopamine therapy in advanced PD, including a narrow therapeutic window and delayed gastric emptying, which contribute to motor complications [6]. However, little is known about the degree to which patients are able to sustain high-dose oral regimens over time in the real-world setting.
Clinical trials and qualitative studies of medication adherence in PD have offered important insights into patterns of use and have identified barriers to adherence, including cognitive impairment, greater regimen complexity, and having lived longer with PD [7–9]. However, these studies are necessarily limited in sample size and reach and have not focused exclusively on advanced PD. A population-based perspective on pharmacological treatment in advanced PD would be a helpful addition to the current knowledge base. This perspective is particularly important given the increased costs associated with advanced PD [10, 11], as well as evidence that suboptimal adherence in PD is associated with excess costs due to higher rates of physician visits, emergency room visits, and hospitalizations and poorer quality of life [12, 13]. Prescription claims data are a valuable source of information on medication use; they allow for larger-scale investigations of patterns of prescription fills and can provide a window into sustainment of drug regimens over time and complement clinic-based investigations.
For these reasons, we sought to examine sustainment of newly initiated high-dose oral medication regimens among individuals with advanced PD in the year following initiation of the regimen. Because a majority of individuals with advanced PD in the U.S. are older and covered by Medicare [14], we focused on the Medicare population.
METHODS
Data source and study design
This retrospective claims analysis utilized Chronic Conditions Data Warehouse 100%fee-for-service Medicare claims data from 2011–2013. The dataset included comprehensive Part A and Part B medical claims for inpatient care, skilled nursing facility care, home health services (i.e., intermittent skilled nursing care and in-home physical, speech-language, and occupational therapy), outpatient services, durable medical equipment, and hospice services as well as Part D prescription claims files for outpatient prescription drug events. The claims files were linked to a personal summary file with the beneficiary’s demographic and enrollment information.
Sample selection
To be included in the study, individuals were re-quired to: 1) have an outpatient or inpatient claim in 2012 that included a diagnosis of PD (ICD-9-CM: 332.0); 2) be continuously enrolled in Medicare Parts A and B, as well as a stand-alone Part D prescription drug plan, in 2012; 3) have at least one ph-armaceutical (Part D) claim for levodopa (alone or in combination with other treatment) in 2012; 4) meet study criteria for advanced PD and initiation of a high-dose oral medication regimen, as defined below; 5) be continuously enrolled in Medicare Parts A and B, as well as a stand-alone Part D prescription drug plan, in the 12 months before and after initiating the high-dose oral medication regimen (i.e., the index date); 6) show no evidence of a high-dose oral medication regimen in the 12-month pre-index period (to ensure new initiation of a high-dose oral regimen on the index date); 7) have complete data available for all covariates; and 8) be aged 65 years or older at the start of 2012 (i.e., be eligible for Medicare coverage due to age rather than disability). A sample selection diagram is provided in the Supplementary Material.
Identifying advanced PD and high-dose oral regimen initiation
Because ICD-9-CM codes do not include disease stage, we identified advanced PD with a medication-based proxy (30-day average oral levodopa equivalent dose [LED] >1000 mg/day) that was designed to identify individuals with advanced disease in claims data. The dosing threshold was based on a review of medication dosing reported in clinical trials of advanced PD therapies (e.g., deep brain stimulation). In a previous study of Medicare beneficiaries that classified individuals as having either advanced PD or mild/moderate PD based on this algorithm, the group assigned advanced PD status had higher odds of nine claims-based markers associated with clinical signs of advanced PD (i.e., claims associated with deep brain stimulation, a fall, hallucinations, durable medical equipment [walker, wheelchair, specialty bed], dementia, care in a skilled nursing facility, hospice), as compared to the mild/moderate PD group [15]. A second study using the medication-based proxy to assign advanced PD status found that a prevalent sample of Medicare beneficiaries classified as having advanced PD had higher 12-month all-cause and PD-related direct costs than those classified as having mild/moderate PD [10].
In the current study, the first step in applying the algorithm was to extract all prescription claims for all oral levodopa and non-levodopa PD prescriptions received by each patient in 2012. The second step was to convert the dose of each non-levodopa PD prescription to an LED, based on previously published conversion factors, in order to standardize doses for all PD prescriptions [16]. The third step was to calculate the average daily LED for each PD prescription using the formula for average daily dose (ADD) calculations from prescription claims (i.e., ADD = [dose*pill quantity]/number of days’ supply). Each day covered from the prescription’s start date to the prescription’s end date (i.e., the start date plus the days’ supply) was assigned the average daily LED calculated for that prescription. Since the patient could be on multiple PD prescriptions, the fourth step was to sum the average daily LED available from all available PD prescriptions to estimate the total daily LED available to the patient on each day in 2012. In the fifth step, in order to identify an incident sample of patients with advanced PD and to identify new high-dose oral regimens, we used an algorithm to search chronologically starting from January 1, 2012 for the earliest qualifying consecutive 30-day period in 2012 wherein the total daily LED on the first day of this period exceeded 1000 mg and the mean of the total daily LED over the 30 days also exceeded 1000 mg. Patients with a qualifying 30-day period were identified as having incident advanced PD and initiating a new high-dose oral regimen, with the first day of the period assigned as the date of the high-dose oral regimen initiation (i.e., index date).
Sustainment measures: Proportion of days covered and continuous gap
In order to explore use patterns in the year following initiation of the high-dose oral regimen, we measured whether individuals sustained an adequate total daily LED >1000 mg for the 12-month follow-up. We summarized this information using two traditional claims-based measures of medication use. First, we examined the proportion of days covered (PDC) measure, which calculated the number of days covered by LED >1000 mg divided by the number of days in the measurement period (i.e., 365, or the year following initiation of the regimen). For our main analyses, we selected PDC ≥0.80 as the threshold to classify patients as sustaining a high-dose oral treatment regimen. This means that patients had to have maintained a total daily LED >1000 mg for at least 292 days of the year. Because the PDC measure does not provide information on whether any days without adequate LED were scattered throughout the year of follow up or adjacent/continuous (or both)—and because these two scenarios may have different clinical implications—our second measure examined whether individuals had the presence of a continuous period of 90 days without daily LED of >1000 mg/day (i.e., a gap in treatment).
In order to gain a clearer picture of overall PD med-ication dosage and use patterns, we also explored whether individuals in the sample showed evidence of sustaining a daily LED that met lower dose thresholds: 1) >800 mg/day, representing a slightly lower dose that still may adequately control motor sympt-oms in this advanced PD cohort; 2) >500 mg/day, representing half the dose initially prescribed by the provider; and 3) >0 mg, representing treatment with at least some PD medications. We examined the percentage of individuals who met the PDC cutoff of ≥0.80 for each of these daily dose levels and the percentage who had a continuous period of 90 days without LED >800 mg/day, LED >500 mg/day, and LED >0 mg/day. Individuals who did not sustain a da-ily LED >0 mg for a continuous 90-day period were deemed to have discontinued all PD medications.
Statistical analyses
Descriptive statistics were used to examine sample characteristics and outcomes. Sociodemographic data included age, sex, race, and region of residence. Clinical and treatment data were captured during the pre-index period and included whether the individual had a claim indicating selected PD-related comorbidities or potential side effects of therapy (anxiety, depression, dementia, psychosis, impulse control disorder, orthostatic hypotension, syncope, leg edema); whether the individual had claims for an outpatient neurologist visit, physical therapy, occupational therapy, deep brain stimulation, and/or durable medical equipment (all yes/no); the number of 30-day prescriptions per month (for any indication); and the number of unique PD drugs for which the individual had medication supply on the index date (i.e., at initiation of the high-dose oral regimen). Finally, a zip code-level variable capturing the median household income in the individual’s area of residence was also included.
In sensitivity analyses, we examined alternate definitions of our study outcomes. For the PDC measure, we opted to look at a slightly lower threshold (PDC ≥0.70, meaning an individual maintained the specified dose for ∼256 or more days in the year) as well as a slightly higher threshold (PDC ≥0.90, meaning an individual maintained the specified dose for ∼329 or more days in the year). Similarly, we explored presence of continuous gaps at both a lower threshold (i.e., 60 days) and a higher threshold (i.e., 120 days). In additional sensitivity analyses, we also examined two alternative dosage thresholds to identify advanced PD and high-dose oral regimens according to the methods described above: 1) LED >800 mg/day (instead of LED >1000 mg/day) and 2) levodopa (instead of LED) >1000 mg/day.
The University of Pennsylvania Institutional Review Board approved the project and deemed the study exempt from informed consent procedures because no data were collected directly from patients.
RESULTS
The study sample (N = 9,405) was 53%male, 90%white, and had a mean age of 77.4 years (SD = 6.8) (Table 1). Dementia (34%) and depression (29%) were the most common comorbid conditions among those we examined. In the year prior to initiating their high-dose (LED >1000 mg/day) oral regimen, 84%of patients had an outpatient visit with a neurologist. Less than half the sample had a claim for physical therapy (42%), and 3%had a claim for deep brain stimulation. On average, patients were on six 30-day prescriptions each month (SD = 3.3) during the pre-index period. Patients were receiving an average of 1.5 unique PD drugs (SD = 0.7) on their index date.
Sample characteristics (N = 9,405)
PD, Parkinson’s disease aIncludes assistive devices (unilateral, bilateral, wheelchair, specialty bed).
As shown in Fig. 1, very few patients (∼5%) sustained a high dose regimen of LED >1000 mg/day at PDC >0.80 in the 12 months after initiation and 75%had a continuous period of 90 days without a total LED >1000 mg/day. Even when LED threshold levels were lowered to 800 mg/day, only 20%of patients had a PDC >0.80 and over half (53%) had a conti-nuous gap of 90 days without LED >800 mg/day. At a threshold of LED >500 mg/day, about 56%of patients had a PDC >0.80 and 19%had a continuous 90-day gap. Finally, examination of availability of any PD medication (i.e., LED >0 mg/day) resulted in 76%of patients meeting the PDC >0.80 threshold and 10%of patients having a continuous 90-day gap without any PD medication.
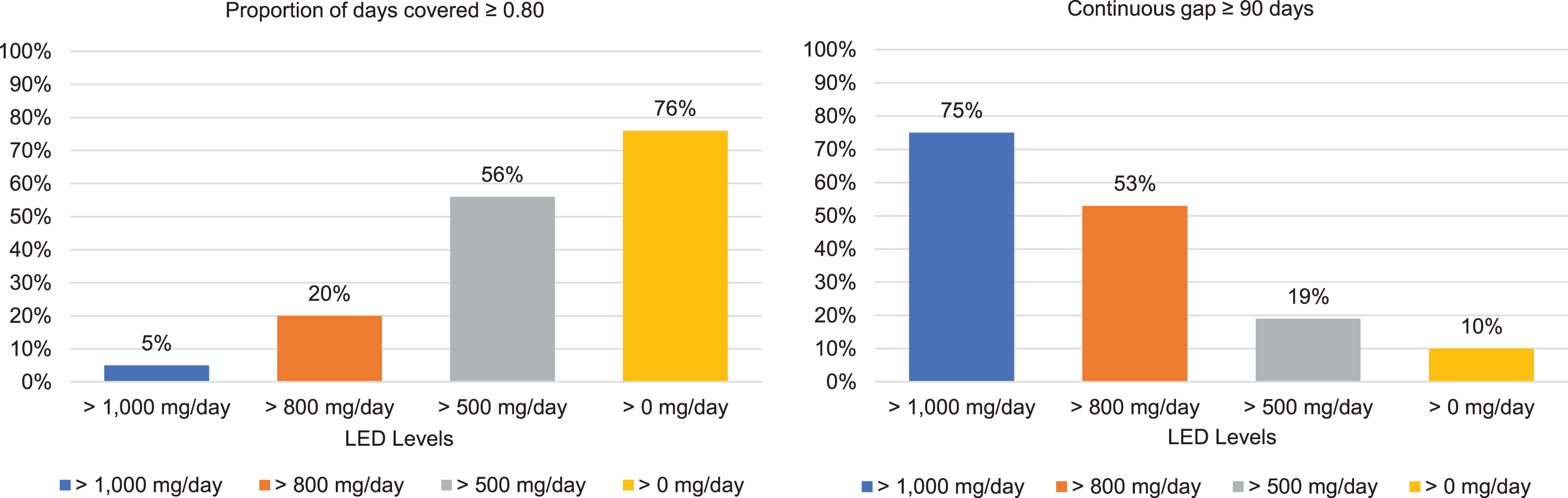
Outcomes in the 12 months following initiation of a high-dose regimen (LED >1,000 mg/day) of oral antiparkinson medication. Bars represent the percentage of patients who subsequently met criteria for each outcome measure at the original dosage level (LED >1000 mg/day) and at various lower dosage thresholds. Individuals who did not sustain a dosage level of LED >0 mg/day for a continuous period of 90 days were considered to have discontinued all PD medications.
In sensitivity analyses examining a lower PDC threshold (≥0.70), the percentage of patients sustaining regimens meeting the various LED dosage levels (i.e., >1000 mg/day, >800 mg/day, >500 mg day, >0 mg/day) increased but remained very low for the original high-dose LED >1000 mg/day oral regimen (i.e., 7%of patients had a PDC≥0.70; Fig. 2A). As expected, even fewer patients sustained dosages corresponding to the specified thresholds when the PDC cut-off was raised to≥0.90. Similarly, higher proportions of patients had a continuous gap in availability of the various LED dosage levels when a gap was defined as a shorter time window of 60 days (instead of 90 days) and a lower proportions had evidence of a gap when the time window was extended to 120 days (Fig. 2B).
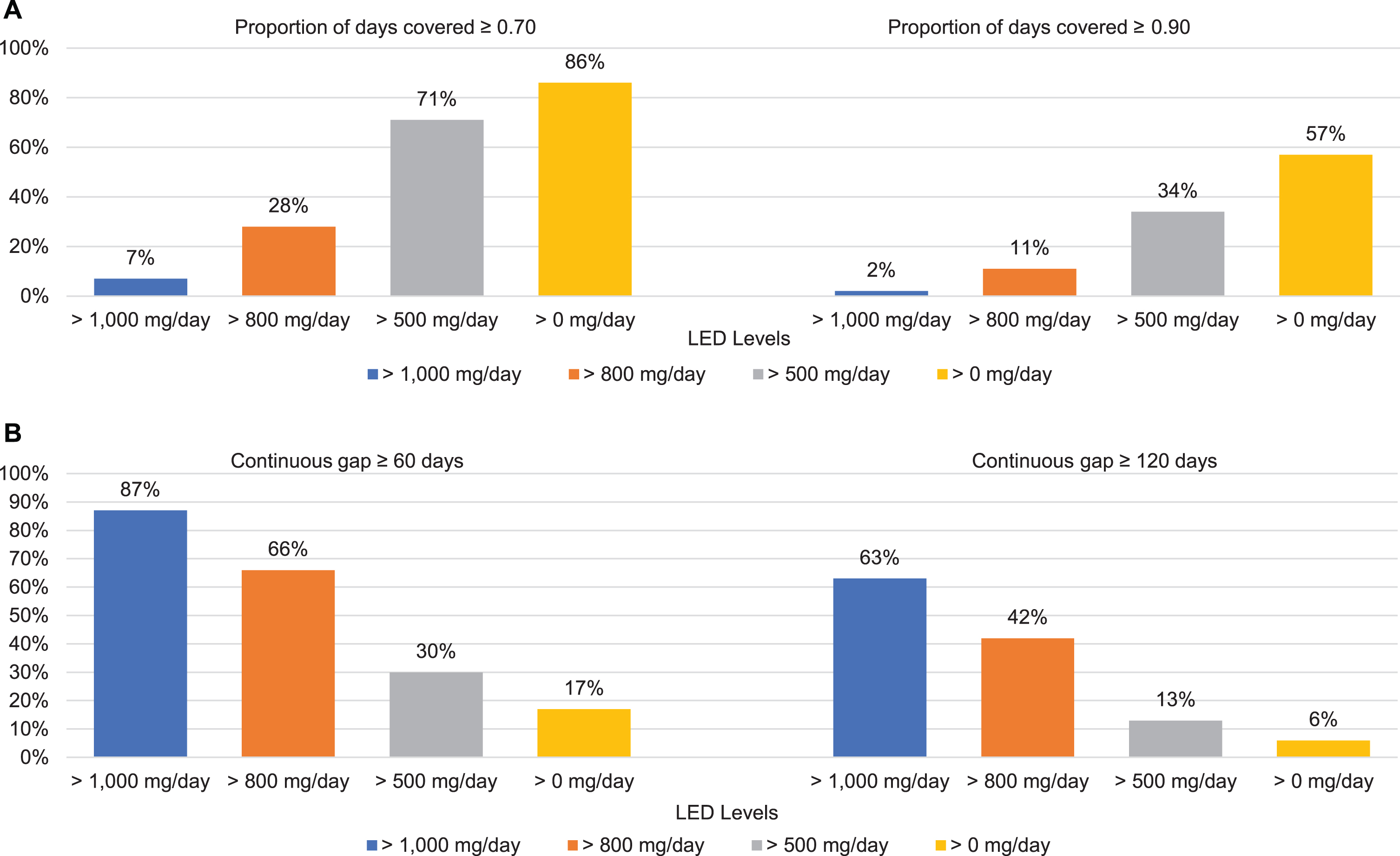
Sensitivity analyses: Outcomes in the year following initiation of a high-dose regimen (LED >1,000 mg/day) of oral antiparkinson medication, using alternate thresholds for proportion of days covered and continuous gap measures. A) Bars represent the percentage of patients who met criteria for the specified PDC outcome at the original dosage level (LED >1000 mg/day) and at various lower dosage thresholds. B) Bars represent the percentage of patients who had the specified gap in medication supply at the original dosage level (LED >1000 mg/day) and at various lower dosage thresholds. Individuals who did not sustain a dosage level of LED >0 mg/day for a continuous period of 60 or 120 days were considered to have discontinued all PD medications.
While the specific percentages varied somewhat, the pattern of results was generally similar in additional sensitivity analyses examining alternative dosage thresholds to identify advanced PD and to define a high-dose oral regimen (Fig. 3A, B). For example, in the sample defined based on a threshold of LED >800 mg/day (i.e., a lower dosage in the algorithm used to detect advanced PD), very few patients (7%) sustained that regimen at PDC >0.80 in the subsequent year. In the sample defined based on a threshold of levodopa >1000 mg/day, only 3%sustained that dosage level at PDC >0.80 in the following year.
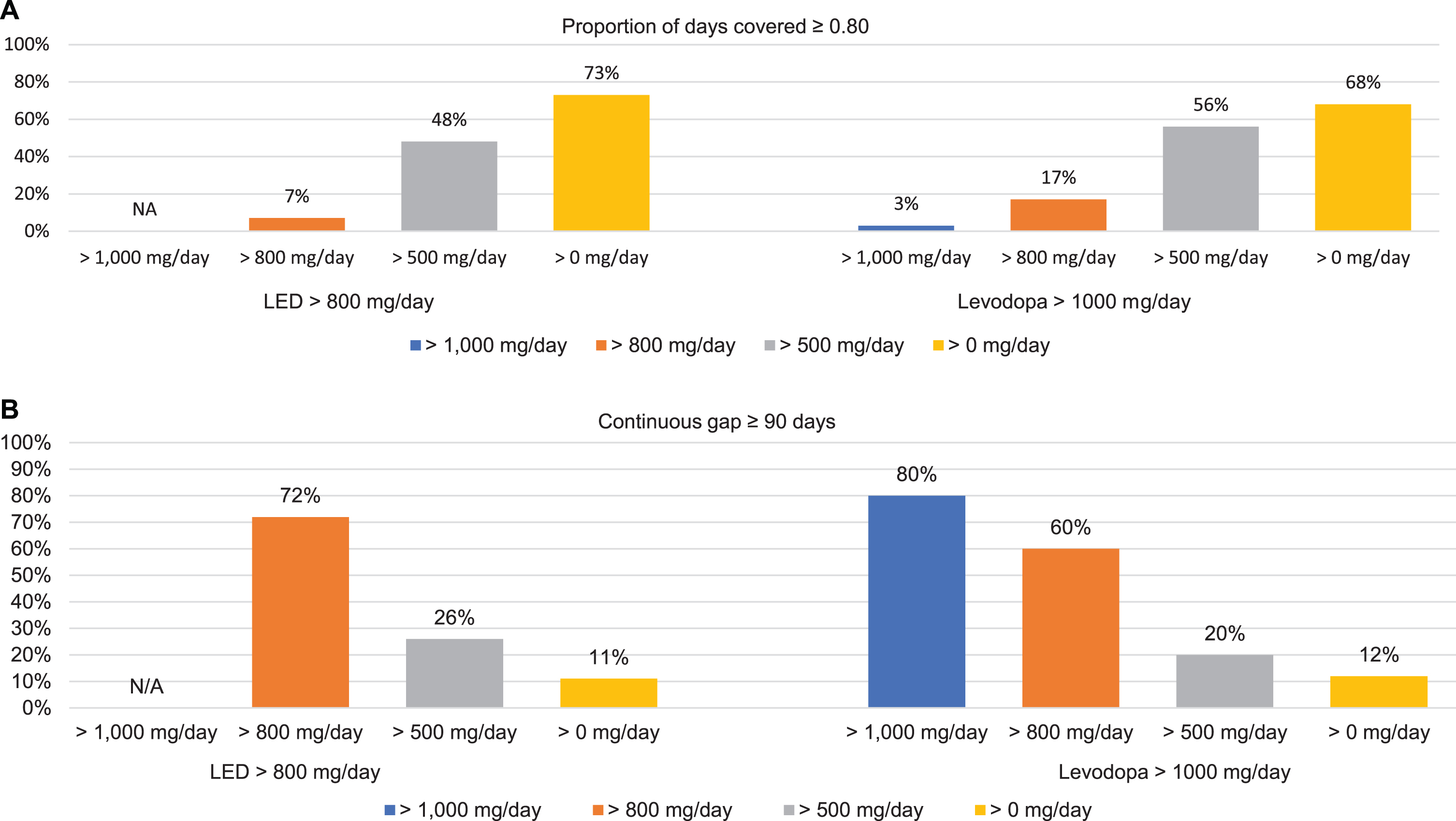
Sensitivity analyses: Outcomes in the 12 months following initiation of a high-dose oral regimen, using alternate definitions of high-dose regimen for advanced PD. High-dose regimens were defined as LED >800 mg/day (left figures) or levodopa >1,000 mg/day (right figures). A) Bars represent the percentage of patients who met criteria for the specified PDC outcome at the original dosage level (LED >1000 mg/day) and at various lower dosage thresholds. B) Bars represent the percentage of patients who had the specified gap in medication supply at the original dosage level (LED >1000 mg/day) and at various lower dosage thresholds. Individuals who did not sustain a dosage level of LED >0 mg/day or levodopa >0 md/day for a continuous period of 90 days were considered to have discontinued all PD medications.
DISCUSSION
To our knowledge, this is the first study to investigate maintenance of high-dose oral medication regimens for individuals with advanced PD. Pharmacy claims revealed that only 1 in 20 older Medicare beneficiaries who initiated a new high-dose (LED >1000 mg/day) oral medication regimen sustained that regimen during the following year, and only 1 in 5 sustained a dosage level that would be likely to adequately control the motor symptoms associated with advanced disease (i.e., LED >800 mg/day). Significant gaps in adequate dosage levels were also common, with three quarters of the sample showing evidence of a 90-day continuous gap at the LED >1000 mg/day level and half showing evidence of a 90-day continuous gap at the LED >800 mg/day level. Findings were robust across various thresholds for defining our sustainment outcomes (i.e., lower and higher cutoffs for proportion of days covered and shorter or longer continuous gaps in medication). These findings highlight widespread difficulty in maintenance of high-dose regimens and raise questions about whether current oral treatments are meeting the needs of patients with advanced PD.
Whereas total discontinuation of oral PD medications was less common, it was concerning to find 1 in 10 advanced PD patients with a 90-day continuous gap in availability of any oral PD medication supply. It is possible that some discontinuation was deliberate due to non-response (e.g., in individuals with medication-refractory tremor), or due to revised diagnosis (e.g., in individuals who did not ultimately have idiopathic PD), and some individuals with advanced, end-stage PD may have stopped medications in favor of palliative care because side effects were greater than the benefits. Individuals with 90-day gaps could also have been taking their medication irregularly and accumulated extra medication supply. Nonetheless, these findings warrant additional investigation.
Our findings offer a unique, population-based view of dosage patterns in advanced PD. Traditio-nal studies of adherence, which typically examine smaller groups of patients and seek to assess “the degree to which a person’s behavior corresponds with the agreed recommendations from a health care provider” [17], are challenging in advanced PD due to the complexity and variability of symptoms and regimens, and very little is known about adherence in the later stages of illness. Individuals with PD are often on more than one medication and may ex-perience frequent medication changes such as augmentation and/or switching [5, 18], making it difficult to interpret observed changes in medication supply or behavior. Our approach used a daily LED calculation that combined the different prescribed PD medications into one metric. This allowed us to evaluate whether those with advanced PD maintained high-dose oral regimens over time, regardless of whether the specifics of the regimen changed, and to gather valuable information about what dosage levels individuals did maintain when their use dropped below the initial high-dose threshold. In this way, we were able to obtain a more complete picture of medication use that incorporates traditional notions of medication adherence (given that adequate medication supply is a prerequisite of adhering to a prescribed regimen) but would also reflect deliberate changes in medication prescribing or use due to efficacy, tolerability, and/or other barriers such as costs.
Prior studies examining PD prescription fills in claims data have also found that many patients (46%to 61%) do not maintain adequate oral medication supply [12, 20]. Tarrants and colleagues, using a methodology similar to ours, examined pharmacy claims obtained via a proprietary database and found that less than 20 percent of individuals persisted with a variety of prescribed PD medications for an entire year, defined as not having a gap of 45 days or changing to another medication [21]. The study observed a large drop in persistence between the 30-day and 60-day marks, with the rate of persistence in the group who passed this milestone also continuing to decline over the remainder of the study year [22]. Our findings suggest that these challenges in sustaining medication regimens are amplified in the high-dose oral regimens used in advanced PD. Our findings may be understood in the context of several qualitative studies exploring medication use in PD, which have found that nonadherence is multifaceted, including both accidental deviation from a regimen (e.g., forgetting to take a tablet) and deliberate modification (e.g., skipping a dose to avoid unwanted side effects) [22]. These issues are likely to be intensified in advanced disease, where cognitive impairment, dexterity issues, the need to take medication on an empty stomach, the need for careful timing of dosing, balancing symptom relief with the risk of dyskinesia, and troubling side effects of multiple medications are all issues that may complicate an individual’s ability or willingness to follow a recommended regimen [23]. In addition, patients may misattribute discomfort associated with disease symptoms to medication side effects, leading them to adjust dosages in an effort to obtain relief, despite the fact that treatment with lower doses of medications likely leads to greater ‘off’ time and motor disability. Finally, there is evidence of rising drug costs in PD, on top of the overall financial burden faced by individuals with advanced PD [24]. Difficulty affording medications is common and has been shown to interfere with adherence [25].
Our findings offer several implications and insights for clinicians. First, for older patients with advanced PD, difficulty sustaining new high-dose oral regimens appears to be the norm rather than the exception. Second, although we were unable to determine whether patients discussed any medication difficulties they may have had with their clinicians, reticence about adherence challenges is not uncommon [17, 26]. Proactive conversations that acknowledge the reality of adherence difficulties and explore specific concerns or obstacles are likely to help providers assist patients in troubleshooting their specific challenges. Elderly patients, particularly those supported by elderly partners who may have their own chronic conditions to manage [27], may be more easily overwhelmed by complicated regimens and especially likely to benefit from treatment plans that are simplified as much as possible and that incorporate medication reminders [28]. Medication therapy management offered by pharmacists has been explored [29], and there is also growing interest in the use of technology to assist in disease management. One study demonstrated the feasibility of SMS text messaging alerts to support medication-taking behavior in a sample of individuals with advanced PD [30], and both smartphone apps and programs that utilize remote monitoring via wearable devices are being studied [31–33]. Discussion of the full range of tre-atment options for advanced PD, including device-aided therapies that may be better tolerated and more effective while reducing adherence demands [34, 35]—as well as new treatments that may become available in the future—will help to ensure that individuals are receiving optimal care. Finally, we observed that 16%of the individuals with advanced PD in our sample had not seen a neurologist in the year prior to initiation of their high-dose oral regimen. Regular monitoring by a provider with expertise in PD management will likely improve outcomes in advanced PD [36].
Our study had several limitations. As noted, pharmacy refill records have the advantage of enabling the study of large samples, but claims do not capture full details of the prescribed medication regimen (e.g., medication timing, number of doses in a day) and do not necessarily reflect actual medication intake. In light of our population-based approach, we were not able to examine the degree to which any individual’s dosage variations were due to deliberate cha-nges in prescribing versus nonadherence, and we examined broad patterns (e.g., utilizing a PDC cutoff and capturing major gaps in treatment) [9], which do not offer insight into important nuances and specific deviations from prescribed regimens (such as missing doses every day, not taking any medication for two days every week, or overuse of medications) [37] that can lead to adverse consequences such as dyskinesias, psychosis, and dopamine dysregulation syndrome. We also lacked detailed clinical data on patient factors relevant to sustainment, including cognitive ability, presence of motor complications such as dyskinesias, motor improvement with medication (‘on’ and ‘off’ difference), and medication side effects. Antiparkinson medications are often taken alongside other medications used to treat non-motor PD symptoms such as depression or anxiety, and this is likely to influence regimen complexity and associated challenges. Finally, we examined an older population with fee-for-service Medicare and our definition of advanced PD captured only individuals who were initiating a new high-dose regimen, due to the lack of an alternate gold standard strategy to identify individuals with advanced disease in claims data. As discussed in detail elsewhere [15], this strategy would not detect older or medically complex patients with advanced PD who were already unable to tolerate higher doses of dopaminergic therapy at the start of our study period. Medication patterns in younger patients and those with Medicare Advantage plans or different insurance arrangements may differ. Future research that includes these variables will help to improve our understanding of specific barriers to sustaining high-dose oral regimens in advanced PD and be better positioned to identify whether there are specific risk factors for low sustainment or discontinuation.
CONCLUSIONS
Very few patients with advanced PD who initiated a high-dose oral medication regimen sustained that regimen during the following year, suggesting that patients may have struggled to manage or tolerate it. Our findings highlight that ongoing, proactive prescriber monitoring is important throughout the course of treatment with high-dose oral PD medication regimens in advanced PD. Further investigation into understanding the principal drivers of low sustainment of high-dose oral regimens are needed to identify and develop improved treatment strategies for advanced PD.
CONFLICT OF INTEREST
Drs. Dahodwala, Pettit, Li, and Doshi, Mr. Jahnke, and Mr. Ladage received research grant support from AbbVie for this study. Dr. Dahodwala also receives grant funding from Roche (clinical trial site investigator), Eli Lilly (clinical trial site investigator), CalaHealth (clinical trial site investigator) and Medtronic (training grant). Dr. Li has consul-tancy relationships with HealthStatistics LLC, Avalon Health Economics LLC, and Robert Ohsfeldt LLC. Drs. Kandukuri, Bao, Zamudio, and Jalundhwala are employees of AbbVie and may own stock/shares in AbbVie Inc. Dr. Doshi reports serving as an advisory board member or consultant for Allergan, Ironwood Pharmaceuticals, Janssen, Kite Pharma, Merck, Otsuka, Regeneron, Sarepta, Sage Therapeutics, Sanofi, Shire, and Vertex; and has received research funding from Biogen, Humana, Janssen, Novartis, PhRMA, Regeneron, Sanofi, and Valeant to support her and/or her coauthors (Mr. Jahnke, Drs. Pettit and Li, and Mr. Ladage), all unrelated to this manuscript. Her spouse holds stock in Merck and Pfizer.
Footnotes
ACKNOWLEDGMENTS
Financial support for the study was provided by AbbVie. AbbVie participated in the interpretation of data, review, and approval of the publication. All authors contributed to the development of the publication and maintained control over the final content. Amrita Singh, BS and Rudmila Rashid, BA, both research assistants at the University of Pennsylvania, provided assistance with formatting the manuscript. Ms. Singh’s assistance was supported by AbbVie. Ms. Rashid was not compensated by AbbVie.
