Abstract
Background:
Visuospatial skills are impaired in Parkinson’s disease (PD). Other related skills exist, such as spatial orientation have been poorly studied. The egocentric (based on internal cues) and allocentric frameworks (based on external cues) are used in daily spatial orientation. Depending on PD onset, the allocentric framework may have a higher level of impairment in tremor-dominant and the egocentric one in akinetic-rigid.
Objective:
To evaluate spatial orientation and visuospatial functions in PD patients and controls, and to assess whether their performance is related to disease duration and the PD subtype (tremor-dominant and akinetic-rigid).
Methods:
We evaluated egocentric and allocentric spatial orientation (Egocentric and Allocentric Spatial Memory Tasks) and visuospatial abilities, span and working memory in 59 PD patients and 51 healthy controls.
Results:
Visuospatial skills, visuospatial span, and egocentric and allocentric orientation are affected in PD. Visuospatial skills and allocentric orientation undergo deterioration during the first 5 years of the disease progression, while egocentric orientation and visuospatial span do so at later stages (9–11 years). The akinetic-rigid subtype presents worse results in all the spatial abilities that were measured when compared to controls, and worse scores in visuospatial working memory, visuospatial abilities and allocentric orientation when compared to the tremor-dominant group. The tremor-dominant group performed worse than controls in egocentric and allocentric orientation.
Conclusion:
PD patients show deficits in their visuospatial abilities and in their egocentric and allocentric spatial orientation compared to controls, specifically in akinetic-rigid PD. Only spatial orientation are affected in tremor-dominant PD patients. Allocentric orientation is affected earlier in the progression of the disease.
INTRODUCTION
Current diagnosis of Parkinson’s disease (PD) is based on motor symptoms, although other types of alterations, such as cognitive problems, are present in the onset of the disease [1]. Thus, there is a profile of cognitive impairment associated with PD, which includes deterioration of executive functions, attention, long-term memory and visuospatial skills [1]. The low capacity in visuospatial and visuoperceptual processing [2, 3], for example, in copying complex geometric forms, discriminating spatial positions, identifying incomplete letters or estimating spatial relations, together with the motor symptoms characteristic of the disease, seems to be related to difficulties for patients to move through the environment [3]. These difficulties include frequently bumping into different spatial elements or freezing responses when moving through narrow spaces, but also, although less frequent, problems finding their way while driving a car, reading maps, using navigational devices and/or giving directions. All these findings may lead us to believe that these patients present problems in regard to spatial learning, orientation and navigation. These are abilities that allows us to find a path through the environment to reach a target site without getting lost, relying mainly on two frames of reference: the egocentric framework, which assumes taking one’s body as a reference and taking internal cues in which all positions are defined as relative to one’s position, and the allocentric framework, which uses external cues, such as reference points which are located in the surrounding space and are independent from the subject [4]. Classical studies have identified that the egocentric orientation relies on the striatum and the allocentric orientation is more hippocampal dependent [5], but it also seems that both frames of reference involve more extensive networks including occipital, parietal, frontal and temporal areas [6]. There are few studies that have analyzed spatial learning in PD patients. Previous studies have shown worse performance in PD, both with MCI and without it, on a virtual navigation task [7], with PD patients making more heading and distance errors, as well as showing higher response latency. When PD patients were asked to memorize a short route, a task that is more focused on evaluating the self-centered framework, they had difficulty recalling it, both in the short and long term [8]. If both frames of reference are considered, research has proven that PD patients make more errors in navigation tasks by following egocentric cues [9] and studies have focused on how dopaminergic medication improves egocentric and allocentric spatial navigation in PD patients [10]. Furthermore, some studies have concentrated on evaluating specific functions that are related to these navigation strategies. For example, vestibular information and optic flow, both associated not only with a proper locomotor activity and gait, but also with a correct egocentric framework functioning, have been shown to be affected in PD [11–13], especially during navigation and in regard to veering tendency. Nevertheless, and to the best of our knowledge, there are no studies to date that have assessed spatial orientation when the two frames of reference are evaluated with different tasks that include sources of information present in real world environments. Given the progressive and neurodegenerative nature of the disease, further cognitive impairment is thought to be found as the disease progresses, as has already been objected to in other longitudinal studies [14]. Moreover, there have been some attempts to create a phenotype of PD that predicts later and/or quicker progression to dementia. Given the high rates of PD’s dementia prevalence, between 24– 31%, trying to predict its risk of progression seems highly important. Although other variables, such as age, REM sleep behavior disorders, hypotension, gait disturbances or freezing [15, 16] have been proposed as risk factors for dementia progression, cognitive impairment and, specifically, visuospatial functioning in PD could be another one to consider [17]. In this regard, the tremor-dominant phenotype has been found to be related to fronto-striatal dysregulation, while akinetic-rigid has been found to be associated with posterior impairment, related to temporal, occipital and parietal cortexes atrophy as well as reduced cholinergic concentration in these areas, which are presumably associated to visuospatial alterations [18, 19]. This differentiation according to cognitive profiles and their neuroanatomical substratum is interesting due to their different risks associated with the progression to dementia, the posterior impairment having a higher risk than the frontal [17]. Therefore, considering the type of onset-motor dysfunction in PD, we can expect spatial orientation to also be impaired in relation with the brain areas involved, specifically, the egocentric framework in tremor-dominant subtype and the allocentric one in akinetic-rigid. The aim of this study was to test egocentric and allocentric spatial learning in PD in real performance-based tasks, as well as to assess other visuospatial functions, such as visuospatial abilities, span and working memory. We also aimed to verify if any progression of PD exists, that is, if the years of evolution since diagnosis are associated with a worse performance in all spatial functions assessed, as well as to find out if there is any connection with the type of onset (tremor-dominant or akinetic-rigid) and the achievement in all those functions. We hypothesized that we could identify egocentric and allocentric spatial frameworks deficits in PD patients compared to healthy controls. We also hypothesized that a longer course of the disease would be related to poorer performance in all spatial functions. Lastly, according to the brain areas involved in both functions (mainly fronto-striatal for egocentric strategy and temporal for allocentric strategy), we expected patients with akinetic-rigid PD to perform worse in the visuospatial span, visuospatial abilities and allocentric orientation tasks and those with tremor-dominant PD would have more difficulty in the working memory and egocentric orientation tasks.
MATERIALS AND METHODS
Subjects
The study involved 59 subjects with PD (32 females and 27 males), mean age 69.7±7.9 (range 50– 86), mean duration of disease since diagnosis of 8.5±4.6 years (range 2– 19), mean age at onset of PD of 63.91±9.02 (range 37– 82), mean UPDRS part III score 16.8±8.4 and Hoehn& Yahr range 1– 3. Pharmacological treatment included Levodopa (N = 44, 74.60%), dopaminergic agonist (N = 25, 42.40%), MAO inhibitor (N = 35, 59.31%), cholinergic inhibitor (N = 2; 3.42%), and deep brain stimulation procedures (N = 4; 6.80%). Eight patients received monotherapy, while 51 patients received two or more drugs. PD subjects met the UK Parkinson’s Disease Society Brain Bank clinical diagnostic criteria [20] and were classified in akinetic-rigid or tremor-dominant by an expert neurologist in PD according to the motor symptoms These symptoms had been reported by a neurological examination at the beginning of their diagnosis. Exclusion criteria for PD group were to have a Hoehn & Yahr range above 3 and to have suffered from another neurological disease in the past or the present. Fifty-one healthy subjects served as controls (33 females and 18 males), mean age 66.7±8.6 (range 50– 82). Exclusion criteria for control group were to obtain a MoCA score <21 according to the normative values of our country [21] and to have a diagnosis of any neurological of psychological disease. All participants, from both PD or control group, had normal or corrected vision. All subjects provided written informed consent and the study was approved by the institutional review board. Sample characteristics of both groups are shown in Table 1.
Sample description of tremor-dominant (TD) and akinetic-rigid (AK) PD and control groups in sociodemographic and disease-related variables
*No education refers to those participants who did not complete primary school.
Assessments
All participants completed the Montreal Cognitive Assessment test (MoCA) [22] for cognitive screening, the Benton’s Judge of Line Orientation Test (JoLO) TF 2/3 H11-30 [23] for the evaluation of visuospatial abilities and the Spatial Span from Cambridge Neuropsychological Assessment Battery (CANTAB) (SSP-F, forward variant, and SSP-B, backward variant) [24] for visuospatial short-term and working memory capacity. These tests were selected as they involve a lesser motor component than others designed to measure similar functions.
Egocentric spatial memory task
In this task, based on the Card Placing test to assess heading disorientation [25], we examined the ability to represent spatial locations of objects placed on walls around the subject [26]. Each participant was seated in a swivel chair and placed between 4 opaque panels to avoid environmental cues, forcing the participant to use their own body position as a reference. The task consists of two parts. In part A (Ego A), the participant stands in the center of a square surrounded by 4 panels and is instructed to remember the location of three cards (circle, triangle and cross), which are placed in one of the eight positions surrounding the subject. After 10 s, the time exposure proposed by the original task, the examiner takes the cards away, and asks the participant to put them back in their original location (10 s delay). In part B (Ego B), the participant has to memorize the location of the same three cards. Immediately after the cards have been removed, the subject is rotated to the right or to the left by 90 or 180 as determined, and then asked to restore the three cards to the same position as before. Part B measures the egocentric strategy, given that the participant must monitor his or her own viewpoint and how it changes throughout the trial so as to properly place the cards. Meanwhile, part A evaluates visuospatial short-term memory in three-dimensional environments, given that the egocentric point of view remains stable as in the majority of memory tasks, and serves as a control for the execution of Part B. Both part A and B share the same trials, the only variation being whether the subject is rotated or not. To minimize the impact of the motor and cognitive slowness typical of PD, the participant had no time limit for positioning the cards. For each part, a subject undergoes 10 consecutive trials, getting 1 point for each correctly placed card (full scores for each part are 30 points) (Fig. 1).
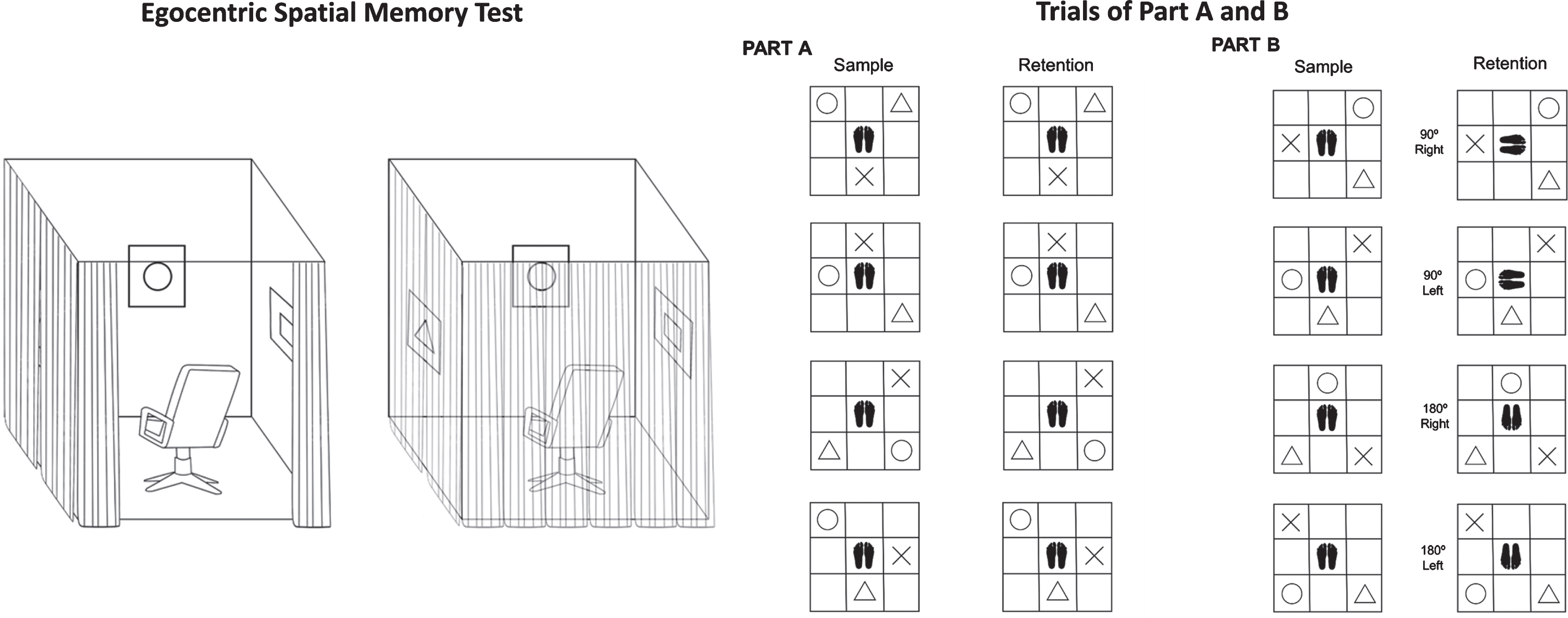
Representation of experimental conditions of the Egocentric Spatial Memory Task.
Allocentric spatial memory task
To assess allocentric strategy (Allo), a task designed to examine the ability to represent spatial locations of objects using distal spatial cues located in the room was used [26], including a door and some windows, tables, chairs and wardrobes. The participant is seated in a swivel chair at a round table with 8 possible locations. The subject is instructed to remember the location of the three cards. After 10 s, the participant is blindfolded and the examiner moves the subject around the table to another position. From this new position, the participant is asked to restore the three cards to their original positions, also without a time limit. Errors are corrected, showing the subject the correct position. The task consists of 5 blocks of 4 trials. The position of the 3 cards on the table is the same in each block and repeated throughout the 4 trials, but the participant is moved to a different position in each trial. In each trial, the subject obtains 1 point for each correctly placed card (full score for each block is 12 points, 60 points in total) (Fig. 2).
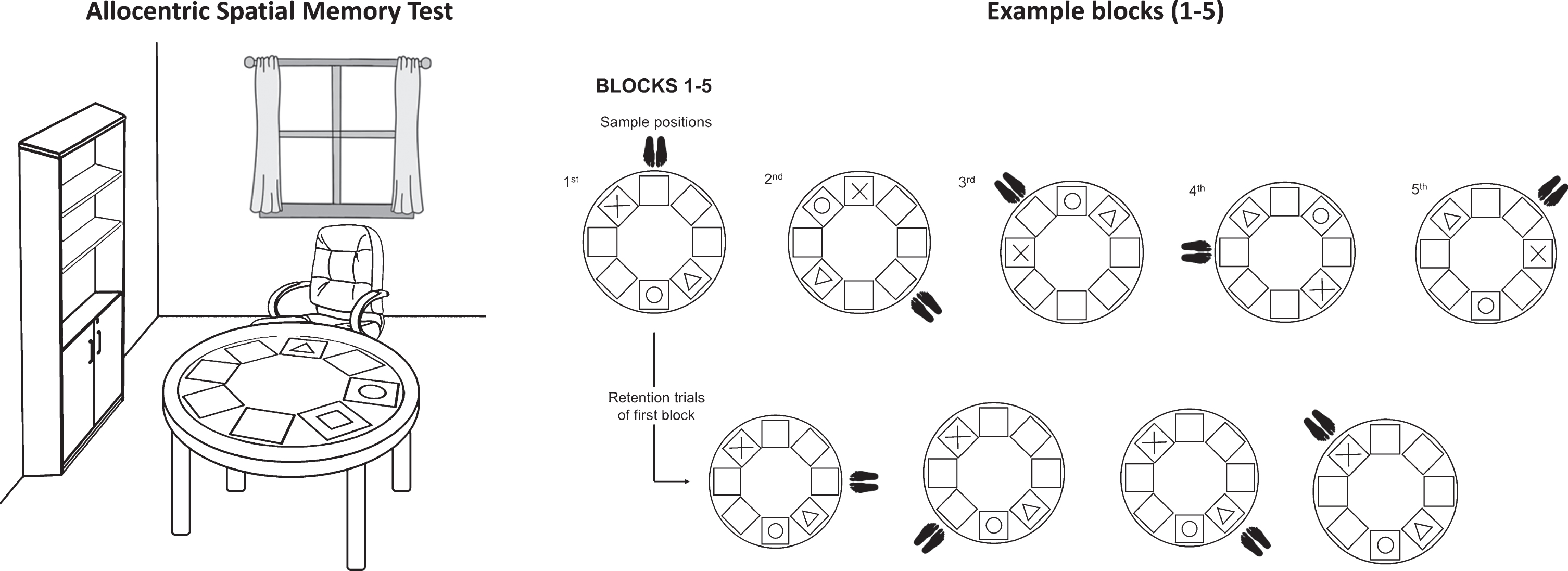
Representation of experimental conditions of the Allocentric Spatial Memory Task.
Procedure
All participants took the tests in a single session of approximately 60 min, carried out by expert psychologists in neuropsychological evaluation. The order in which the tests were applied was: Allocentric Spatial Memory Task, MoCA, JoLO, SSP-F, SSP-B, and Egocentric Spatial Memory Task.
Statistical analysis
Data were analyzed using the software program SPSS v.19. One-way ANOVA, two-way ANOVA and ANCOVAR were used to compared between different groups. Holm-Sidak was used as post-hoc test. Effect size were reported employing Eta squared. Significance level was set at p < 0.05 (two-tailed test).
RESULTS
An ANOVA analysis to examine group differences revealed that the PD group showed significantly lower performance than the control group in MoCA (F = 11.717; gl1 = 101; p = 0.001; η2 = 0.104), JoLO (F = 17.901; gl2 = 99; p = 0.000; η2 = 0.153), SSP-F (F = 9.341; gl2 = 102; p = 0.003; η2 = 0.084), Ego A (F = 16.459; gl2 = 98; p = 0.000; η2 = 0.144), Ego B (F = 22.872; gl2 = 98; p = 0.000; η2 = 0.189), and Allo (F = 34.236; gl2 = 97; p = 0.000; η2 = 0.261] (Fig. 3). High effect sizes were found in Ego A and B, Allo, and JoLO. An ANCOVA analysis revealed that differences between groups in Ego B remained significant after Ego A covariation, although the effect size was reduced [F = 7.549; gl2 = 97; p = 0.007; η2 = 0.072]. An ANOVA analysis (Group×Gender) was conducted. Neither Gender, nor Group×Gender presented significant effects (p > 0.05).
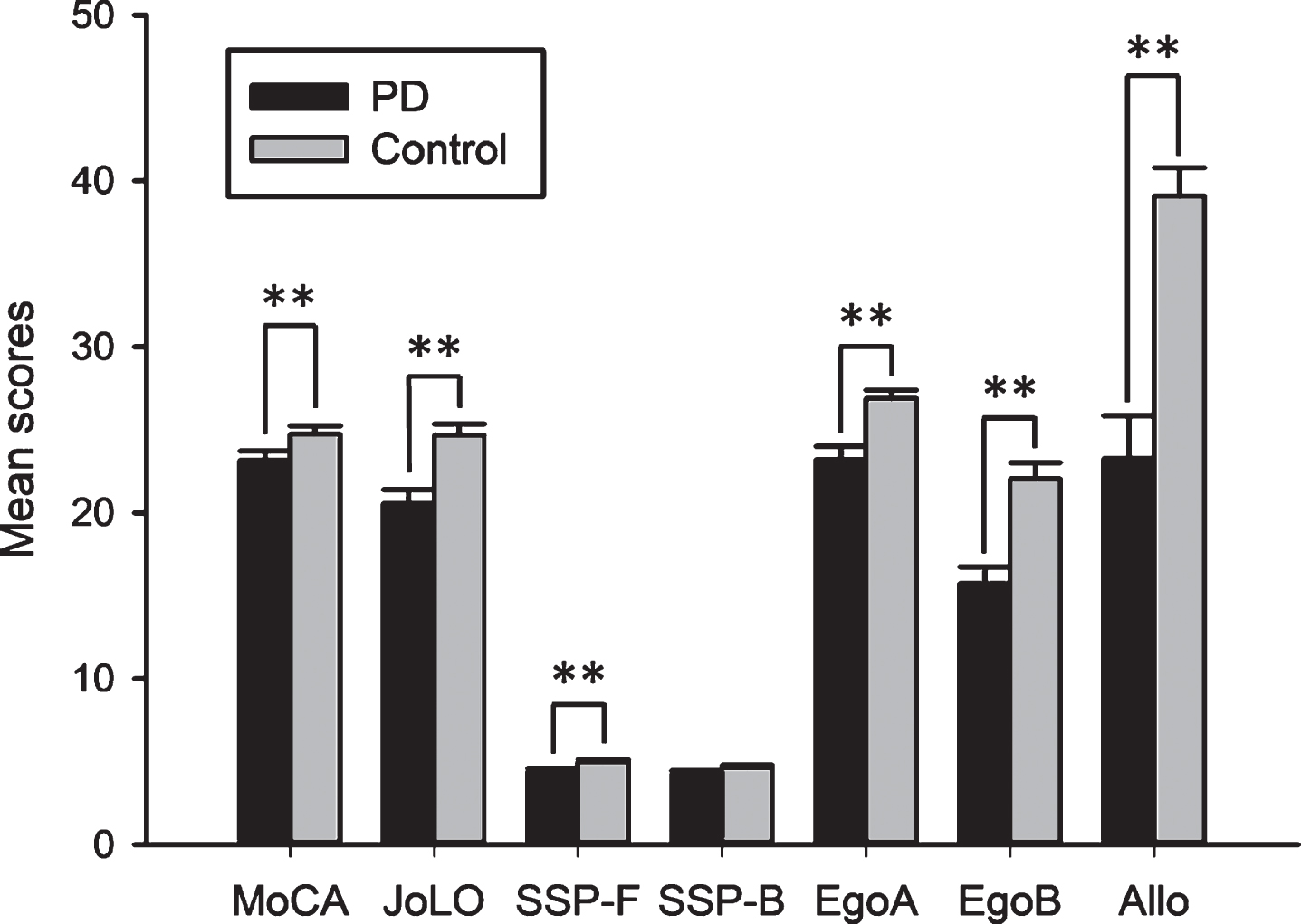
Comparison between PD patients and controls in neuropsychological tests. Significant differences were found in MoCA, JoLO, SSP-F, EgoA, EgoB and Allo. **p<0.01. PD, Parkinson’s disease; MoCA, Montreal Cognitive Assessment; JoLO, Benton’s Judge of Line Orientation Test; SSP-F, Spatial Span forward; SSP-B, Spatial Span backwards; EgoA, Egocentric Spatial Memory Task A; EgoB, Egocentric Spatial Memory Task B; Allo, Allocentric Spatial Memory Task.
The PD group was subdivided into percentiles according to the disease duration (25, 50 and 75): <5 (9 women and 6 men; MoCA mean (SD): 24.24 (4.45)); 6– 8 (5 women and 10 men; MoCA mean (SD): 24.26 (3.34)); 9– 11 (5 women and 5 men: MoCA mean (SD): 21 (2.98)); and >12 years (7 women and 4 men; MoCA mean (SD): 20 (6.15)). An ANOVA analysis was performed comparing the performance of these subgroups and the control group. This analysis showed significant differences in performance for MoCA (F = 5.306; gl2 = 90; p = 0.001; η2 = 0.191), JoLO (F = 7.542; gl2 = 93; p = 0.000; η2 = 0.255), Ego A (F = 6.597; gl2 = 93; p = 0.000; η2 = 0.231), Ego B (F = 7.799; gl2 = 93; p = 0.000; η2 = 0.262), and Allo (F = 8.625; gl2 = 92; p = 0.000; η2 = 0.284). Calculating the covariance for Ego A and age of participants, differences by disease duration remain significant in Ego B, with a lower size of effect (F = 3.633; gl2 = 93; p = 0.009; η2 = 0.145). In MoCA, the control group outperformed the 9– 11 (p = 0.014) and >12 (p = 0.002) PD groups. In JoLO, the control group presented better scores than the <5 years PD (p = 0.002), 9– 11 years PD (p = 0.011), and >12 years PD groups (p = 0.003). A better performance of the control group was also found in Ego A with respect to the 9– 11 (p = 0.030) and >12 years PD group (p = 0.000). Similarly, the control group presented a better score in Ego B than the 9– 11 years PD (p = 0.000) and the >12 years PD groups (p = 0.007). Finally, in Allo, the control group performed better than the >5 years PD (p = 0.015), 6– 8 years PD (p = 0.005), 9– 11 years PD (p = 0.003), and >12 years PD groups (p = 0.001) (Fig. 4). These significant differences were maintained when including age and UPDRS as covariates and none of the PD groups differed significantly in their age, gender, Hoehn & Yahr stages, UPDRS, or educational level.
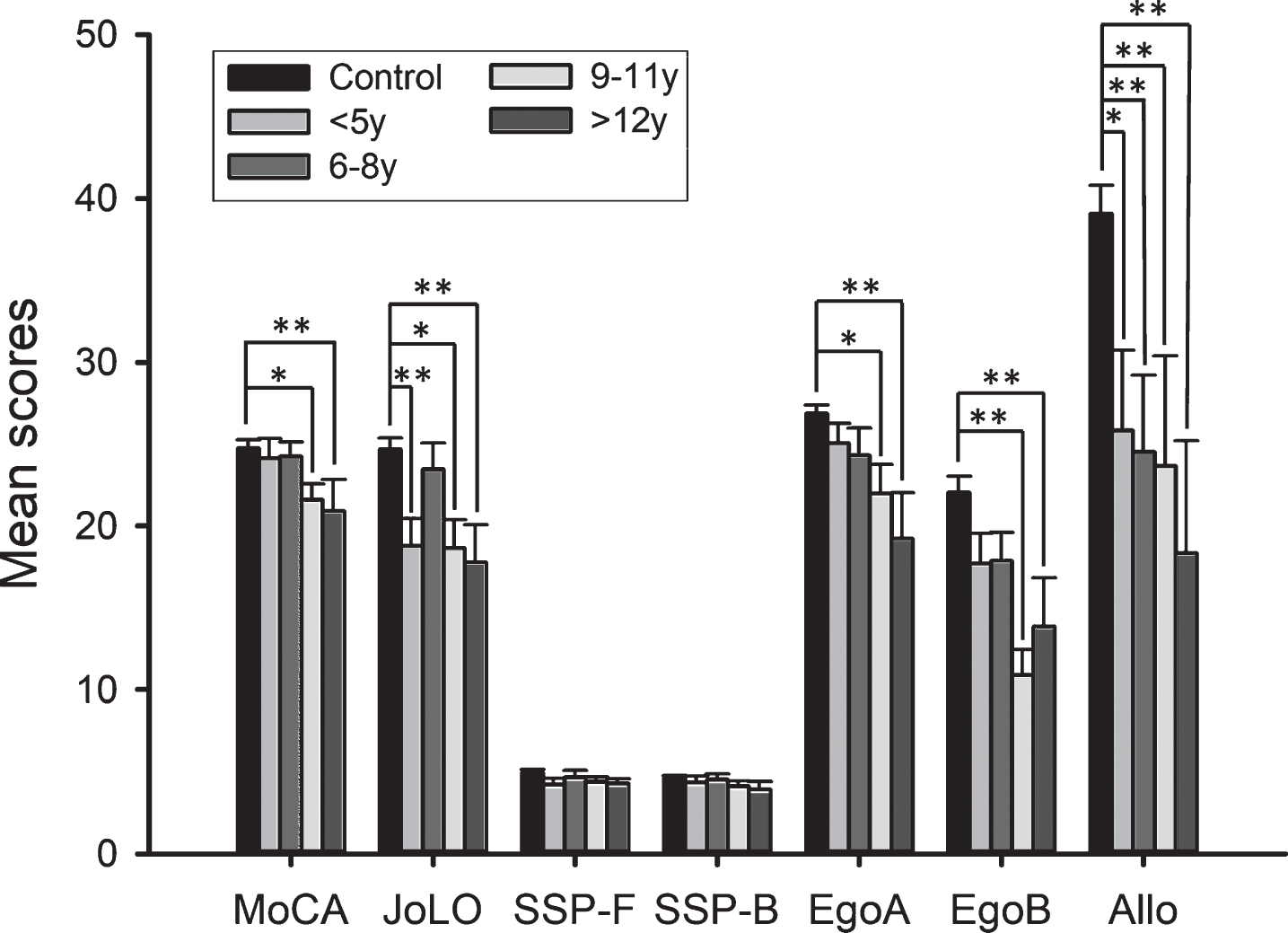
Comparison between PD subgroups by years of evolution (<5 years, 6–8 years, 9–11 years, and >12 years) and controls in neuropsychological tests. Significant differences were found in MoCA, JoLO, EgoA, EgoB, and Allo. *p < 0.05; **p < 0.01. PD, Parkinson’s disease; MoCA, Montreal Cognitive Assessment; JoLO, Benton’s Judge of Line Orientation Test; SSP-F, Spatial Span forward; SSP-B, Spatial Span backwards; EgoA, Egocentric Spatial Memory Task A; EgoB, Egocentric Spatial Memory Task B; Allo, Allocentric Spatial Memory Task.
Analyzing PD onset (Tremor-dominant PD vs. Akinetic-rigid PD vs. Control) and gender with a two-way-ANOVA (PD onset×Gender), we found significant differences in all neuropsychological variables regarding PD onset: MoCA [F = 8.039; gl2 = 95; p = 0.001; η2 = 0.153], SSP-F [F = 5.325; gl2 = 96; p = 0.007; η2 = 0.106], SSP-B [F = 5.521; gl2 = 96; p = 0.005; η2 = 0.109], JoLO [F = 14.710; gl2 = 93; p = 0.000; η2 = 0.253], Ego A [F = 7.409; gl2 = 93; p = 0.001; η2 = 0.146], Ego B [F = 9.636; gl2 = 93; p = 0.000; η2 = 0.181], and Allo [F = 20.911; gl2 = 92; p = 0.000; η2 = 0.327]. Akinetic-rigid PD group showed a worse overall performance than controls in MoCA (p = 0.000), SSP-F (p = 0.004), SSP-B (p = 0.002), JoLO (p = 0.000), Ego A (p = 0.003), Ego B (p = 0.001), and Allo (p = 0.000). When comparing controls and the tremor-dominant group, these differences were observed in Ego A (p = 0.011), Ego B (p = 0.001), and Allo (p = 0.009), with the tremor-dominant group obtaining lower scores. Finally, when controlling for the age of the participants and the disease duration, the tremor-dominant PD group showed better performance than the akinetic-rigid PD group in MoCA (p = 0.029), SSP-B (p = 0.016), JoLO (p = 0.011), and Allo (p = 0.015) (Fig. 5). No significant differences (p > 0.05) in other variables, such as age, UPDRS scale, Hoehn & Yahr stages, gender, or educational level appear between the tremor-dominant and akinetic-rigid PD groups, and therefore, we assume that the above results are not due to any of these variables. No significant differences were found according to Gender or Gender×PD onset.
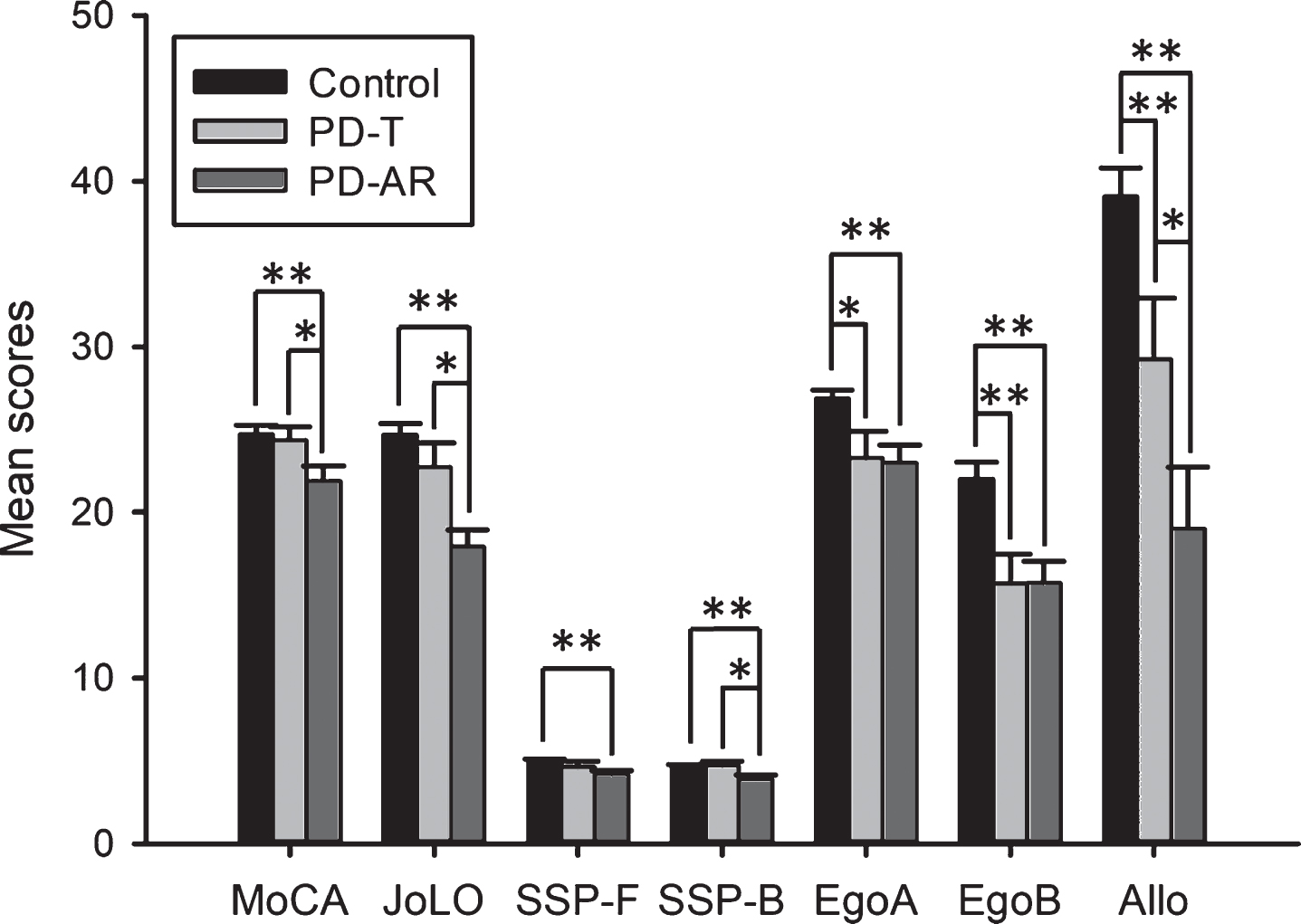
Comparison between PD subgroups by type of onset (tremoric and akinetic-rigid) and controls in neuropsychological tests. Significant differences were found in MoCA, JoLO, SSP-F, SSP-B, EgoA, EgoB, and Allo. *p < 0.05; **p < 0.01. PD, Parkinson’s disease; T, Tremoric; AR, Akinetic-rigid; MoCA, Montreal Cognitive Assessment; JoLO, Benton’s Judge of Line Orientation Test; SSP-F, Spatial Span forward; SSP-B, Spatial Span backwards; EgoA, Egocentric Spatial Memory Task A; EgoB, Egocentric Spatial Memory Task B; Allo, Allocentric Spatial Memory Task.
DISCUSSION
In the present study, we observed a worse overall performance of PD patients compared to controls in visuospatial skills (JoLO), visuospatial span (SSP-F), and egocentric (Ego A and B) and allocentric (Allo) spatial orientation, as well as lower cognitive ability (MoCA). Visuospatial skills have also been found to be altered in PD patients, with the JoLO test being particularly sensitive to these differences [27] showing these patients a some directional bias [28]. Difficulties in visuospatial span and short-term memory in two- (SSP-F) and three- dimensions (Ego A) are consistent with previous research, although contrary to previous literature, we did not find any alteration of visuospatial working memory (SSP-B) in our PD sample [27]. Two types of cognitive impairment profiles characterize PD patients: one with a greater frontal dysfunction and one with a greater posterior involvement [17–19]. In this way, and given that our sample has shown a generalized deterioration of spatial functions but not visuospatial working memory, these patients could generally fit into the cognitive deterioration profile related to posterior brain areas, and therefore, differences in their working memory, more associated to frontal dysfunctions, could not be detected. However, given our study does not include a protocol for evaluating frontal functions, we cannot draw definite conclusions in this regard.
According to our results, PD patients have shown egocentric and allocentric spatial orientation memory impairment. Although previous studies on spatial orientation and memory and orientation in PD are scarce, some have shown that patients express subjective complaints in their everyday spatial activities, such as reading a map, using navigation devices, or finding their way while driving [3], while others have been shown to have problems related to allocentric [7] and egocentric [8, 9] memory performance, as well as in regard to abilities related to the vestibular system and optic flow [11– 13, 29]. Thus, our study manages to expand the previous literature alluding to the fact that both strategies, egocentric and allocentric, evaluated separately, seem to be affected in this population. Two main reasons may explain why spatial orientation could be affected in PD. On the one hand, it is possible that an impairment in some related capacities, such as the mentioned before (visual and spatial processing skills [1–3] optic flow [12, 29], vestibular signals [11]) may have specifically affected the egocentric orientation in PD patients and difficulties in visually exploring complex environments may have affected their allocentric orientation, possibly impacting patients’ visual fixations on less prominent elements of the room [30] instead of on the more distinctive landmarks. On the other hand, the reason may have to do with two of the main brain areas involved in allocentric and egocentric spatial orientation, the hippocampus and the striatum, respectively, progressively lose their volume [31] and are less active during spatial activities in PD [32].
In this study, the tasks that showed the more marked differences between controls and PD patients were the spatial orientation and visuospatial skills tasks. In addition, some of them were sensitive to the number of years a patient had been suffering PD, irrespective of their age or motor status. In this way, the functions that are altered in the first years of the disease are visuospatial abilities (JoLO) and allocentric orientation (Allo). These were followed by egocentric orientation (Ego B), that worsens between the 9- and 11-year time frame, and finally, the three-dimensional visuospatial span (Ego A) which is impaired in patients with more than 12 years of PD evolution. The early alteration of these spatial functions is consistent with previous findings, which point out that deficits in visuospatial skills can be considered a prodromal symptom of PD [19]. Indeed, visuospatial deficits were reported in a 5-year longitudinal follow up of PD patients [17]. Regarding orientation frameworks, these early deficits specifically regarding the allocentric strategy, in comparison with egocentric, could make PD patients, as their disease progresses, tend to orient themselves worse in large spaces and when guided by environmental cues, as already reported in previous studies in relation to navigation capacity [3], but preserve their orientation for longer when in known smaller spaces and guided by internal cues. These progressive alterations in PD may be related to cortical thinning of the occipital, parietal and temporal areas [34]. Thus, it seems that these early alterations are due to a progressive cortical loss in the first years of the disease.
In regard to the type of disease onset (tremor-dominant or akinetic-rigid), the akinetic-rigid group has shown more marked affectation and in more functions, compared to tremor-dominant. Although attending to the neuroanatomical bases that have been proposed for the akinetic-rigid onset we hypothesized that the affected functions would be mainly the allocentric orientation and the visuospatial span and working memory, this group showed not only a worse overall performance in all measured functions compared to controls, but also a worse performance compared to the tremor-dominant group in visuospatial working memory (SSP-B), visuospatial skills (JoLO), and allocentric framework (Allo). The greater alteration found in the akinetic-rigid PD patients is consistent with previous literature, which indicates that patients with this subtype are most likely to develop cognitive dysfunctions later on [35], with their visuospatial functions being especially affected [36]. Furthermore, this type of onset is related to posterior and temporal cortical deterioration, while the tremor-dominant phenotype is associated with frontostriatal dysregulations [18]. As previously discussed, two profiles of cognitive dysfunction have been described in PD regarding neuroanatomical alterations, neuropsychological profiles, type of onset of motor symptoms, deficits in neurotransmission and risk of progression to dementia [19]. On the one hand, there is a profile of prefrontal deterioration, which is related to frontostriatal and dopaminergic alterations, and is associated with tremor-dominant onset and a tendency to a very slow or even stable progression of cognitive impairment, with patients presenting problems in planning and/or working memory. On the other hand, there is a profile of posterior affectation, mainly of the parietal and temporal cortex, which involves acetylcholine deficits and problems in semantic fluency and visuospatial abilities, and is related to gait impairment, akinetic-rigid onset and a higher risk of mild cognitive impairment or dementia progression. Therefore, when considering the potential risk of developing cognitive impairment, it seems vital to take the PD onset into account. The akinetic-rigid type offers a worse prognosis and will also present a further deterioration in regard to spatial functions.
In addition, the tremor-dominant group appears to have some difficulties when compared to controls in 3-D visuospatial span (Ego A) and egocentric and allocentric (Ego B and Allo) spatial orientation. The alteration of both egocentric and allocentric spatial orientation in the tremor group is unexpected regarding our initial hypothesis, the alteration of both egocentric and allocentric spatial orientation in the tremor group is unexpected, given that we predicted that this group would show impairment in working memory and egocentric framework, and if we consider the two profiles previously discussed, considering that we would expect that the difficulties stemming from the allocentric framework would emerge especially in the rigid-akinetic group. However, we have to consider that although there are main areas in charge of orientation, such as the striatum in the egocentric strategy and the hippocampus in the allocentric strategy, we have found that in humans, widely distributed brain networks participate in these functions. Both strategies seem to involve bilateral fronto-parietal networks, involving the fusiform gyrus, insula, lingual gyrus, precuneus, cuneus, superior occipital gyrus, and the superior frontal lobe [6, 37], being the frontal areas the ones involved in planning [6]. Therefore, this involvement of the frontal lobe, in connection with other areas of the brain, could explain the difficulties shown by the tremor-dominant group in visuospatial span and egocentric and allocentric spatial orientation. Thus, we observed a correspondence between each of the orientation strategies and the type of PD onset, while considering the neuroanatomical basis involved in each orientation function and phenotype.
Some limitations of this study should be mentioned. Firstly, it is difficult to ensure that in each of the tasks, egocentric and allocentric, the participants only implement the hypothetically required strategy (i.e., egocentric strategy in the egocentric task, and allocentric strategy in the allocentric task). These tasks have already demonstrated independence in their execution in healthy young people, but not in the elderly [38]. Thus, it is possible that some participants from this study may have used egocentric sources of information, such as proprioceptive and vestibular signals, in the resolution of the allocentric test. In this sense, a deeper analysis of the methodology of both tasks would be necessary to assure their construct validity. Furthermore, given the absence of a complete neuropsychological evaluation protocol, we cannot assure that the control group subjects did not present any cognitive deficit in any neuropsychological dimension. To characterize the cognitive status of this sample, we only used the MoCA, which does not fully guarantee that all participants were cognitively healthy. Likewise, the absence of a complete cognitive assessment prevents us from characterizing the PD group in all neuropsychological dimensions, nor have we considered how taking medication can affect the neuropsychological profile. In addition, even though we tried to carry out tasks that included some of the characteristics found in real space navigation, the fact that these are still laboratory tasks should be considered. Therefore, future research could focus on using other tasks that measure spatial orientation in a functional way. Employing augmented or real reality, or even spatial navigation tasks in real environments already used for older people could be of special interest to draw stronger conclusions about spatial orientation in PD.
In conclusion, our study shows the existence of a deterioration of different spatial abilities in PD, focusing on visuospatial skill and egocentric and allocentric spatial orientation difficulties. According to the duration of the disease, the functions that seem to deteriorate earlier are the visuospatial abilities and the allocentric orientation, followed by the egocentric orientation. In addition, we have been able to verify that the akinetic-rigid onset is associated with a greater neuropsychological deterioration in comparison with the tremor-dominant onset. However, this last subtype seems to present specific difficulties with orientation. All of this highlights the importance of assessing visuospatial functions in this disease and including spatial orientation evaluation.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by Project grants of Gobierno de España (PSI2017-83893-R and PSI2017-90806-REDT) and Programa “Severo Ochoa” CONSEJERÍA DE CULTURA Y DEPORTE del Principado de Asturias (PA-17-PF BP16090) to C F-B. We would like to thank “Asociación Parkinson Asturias” and “Fundación AINDACE” (Ayuda a la Investigación del Daño y Enfermedades Cerebrales) for their collaboration.
