Abstract
Background:
Randomized clinical trials (RCTs) in Parkinson’s disease (PD) have historically enrolled a low number of underrepresented minorities, lessening the generalizability of therapeutic developments. Although there are racial disparities in PD, little is known regarding neuropsychiatric symptoms and other nonmotor manifestations across all races/ethnicities.
Objective:
To assess minority participation in PD trials evaluating the treatment of neuropsychiatric symptoms and explore underlying reasons.
Methods:
We systematically searched PubMed and Embase for RCTs with a primary goal of treating neuropsychiatric symptoms in PD patients from 2000-2019. The pooled prevalence and 95% confidence interval (CI) of being white and enrolled in a clinical trial was calculated using the inverse variance method. I-square was calculated as a measure of heterogeneity and meta-regression was used to evaluate temporal trends.
Results:
We included 63 RCTs with a total of 7,973 patients. In pooled analysis, 11 (17.5%) RCTs reported race/ethnicity. Of studies reporting this data, 5 African American (0.2%), 16 Hispanics (0.64%), and 539 Asians (21.44%) were enrolled. The pooled prevalence of being white in clinical trials was 98% (CI 0.97–0.98, p < 0.001), with 1,908 patients (75.8%). NIH-funded studies were most likely to report racial data when compared to non-NIH trials (p = 0.032).
Conclusion:
This large pooled analysis found a small percentage of RCTs reporting race/ethnicity when evaluating treatment of neuropsychiatric symptoms in PD. There was a disproportionally high number of white patients when compared to African Americans and Hispanics. More studies are needed to investigate this discrepancy and improve rates of & minority enrollment in PD trials.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a chronic, neurodegenerative condition that affects people of different ethnicities, races, and socioeconomic backgrounds. It has been estimated that 1–2 per 1000 people are suffering daily from PD around the world and the total number of PD cases is expected to surpass nine million by the year 2030 [1, 2]. Symptoms of motor dysfunction have been well documented, but equally as debilitating are the non-motor manifestations of PD. These include, but are not limited to, mood disorders, cognitive impairment, autonomic dysfunction, and sleep abnormalities [3, 4]. Most specifically, neuropsychiatric symptoms, including fatigue, depression, anxiety, and psychotic symptoms are common in PD, although there are few studies or randomized clinical trials specifically addressing these issues [5]. Disparities in non-motor manifestations including neuropsychiatric comorbidities in PD have not been well documented, however it is known that there are major healthcare disparities in the access and treatment of neuropsychiatric conditions and mental illness in the African American and Hispanic population in the United States and globally [6, 7].
Although there are biological differences in the racial distribution of PD, the inclusion of underrepresented minorities in PD trials has been disproportionately low [8–11]. Inaccurate representation of minority ethnic populations in randomized controlled trials (RCTs) poses issues with generalizability of results and guidelines, especially in the African American and Hispanic communities [12]. The goal of this study was to review the participation of racial and ethnic minorities in PD trials evaluating treatment of neuropsychiatric symptoms.
METHODS
Eligibility criteria and data extraction
We restricted our analysis to studies that met all the following inclusion criteria: 1) RCTs with a 2) primary goal consisting of treatment of neuropsychiatric symptoms, defined as nonmotor symptoms domains 1–5 by the Movement Disorders Society Nonmotor Rating Scale including depression, anxiety, apathy, psychosis, impulse control and cognition [4], and 3) interventions restricted to drugs and neuromodulation therapies. Exclusion criteria were non-controlled studies (absence of comparison group), ongoing studies, duplicate reports or RCTs that included patients with atypical parkinsonism.
Search strategy
We systematically searched PubMed and Embase for RCTs with a primary outcome of treatment of neuropsychiatric symptoms. The search was conducted in October 2019 and included studies from the past 20 years (01/01/2000–10/01/2019). The following medical subject heading terms were included: (Parkinson’s disease AND randomized clinical trial) AND (cognition OR dementia OR psychosis OR apathy OR depression OR mood OR hallucinations OR memory OR attention OR apathy OR impulse OR anxiety). Reference lists of all included studies, meta-analysis and reviews were manually searched. There was no patient population size restriction for the search.
End points and subgroup analyses
Outcomes of interest included race, ethnicity, year of study, number of included patients, percentage of underrepresented minorities enrolled, country where study was performed and funding source. Although there are different racial and ethnic categories and definitions, we used the most widely accepted classifications by the United States Office of Management and Budget (OMB) described in the Revisions to the Standards for the Classification of Federal Data on Race and Ethnicity [13]. Underrepresented minorities were considered those other than non-Hispanic white patients. Full-text versions of the manuscripts and their supplementary materials were reviewed.
Statistical analysis
Meta-analysis was performed according to recommendations of the Cochrane Collaboration and in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement [14].
We summarized our data in evidence tables and compared categorical variables using chi-square and continuous variables using t-test. P-value ≤0.05 was used to determine statistical significance. We calculated the pooled prevalence and corresponding 95% confidence interval (CI) of being recruited into the trial and being white using the inverse variance method. We pooled proportions from each study with the Freeman-Tuckey double arcsine transformation and used the exact binomial formula to calculate confidence intervals [15]. We also calculated the I square as a measure of heterogeneity. To evaluate the effect of year of publication on the outcome we used meta-regression. For statistical analysis, we used STATA 14.0 (Stata Statistical Software: College Station, TX: StataCorp LLC).
Quality assessment
Quality assessment of RCTs was performed with the Cochrane Collaboration’s tool for assessing risk of bias in randomized trials and with the Jadad score, which evaluates randomization, blinding and follow-up [16, 17]. Publication bias was evaluated by using funnel-plot graphs and checking for symmetrical distribution of trials with similar weights [18].
RESULTS
Study selection and characteristics
Overall, 1547 studies were identified. After removal of duplicate reports, animal studies and non-relevant studies by title or abstract review, 90 articles remained (Fig. 1). These were fully reviewed for satisfaction of inclusion criteria. A total of 63 manuscripts, including data on 9490 patients met all criteria and were included. The main reasons for exclusion were distinct primary goals, no randomization or overlapping population. After further analysis, a total of 7,973 patients were randomized and enrolled according to eligibility criteria related to each individual study.
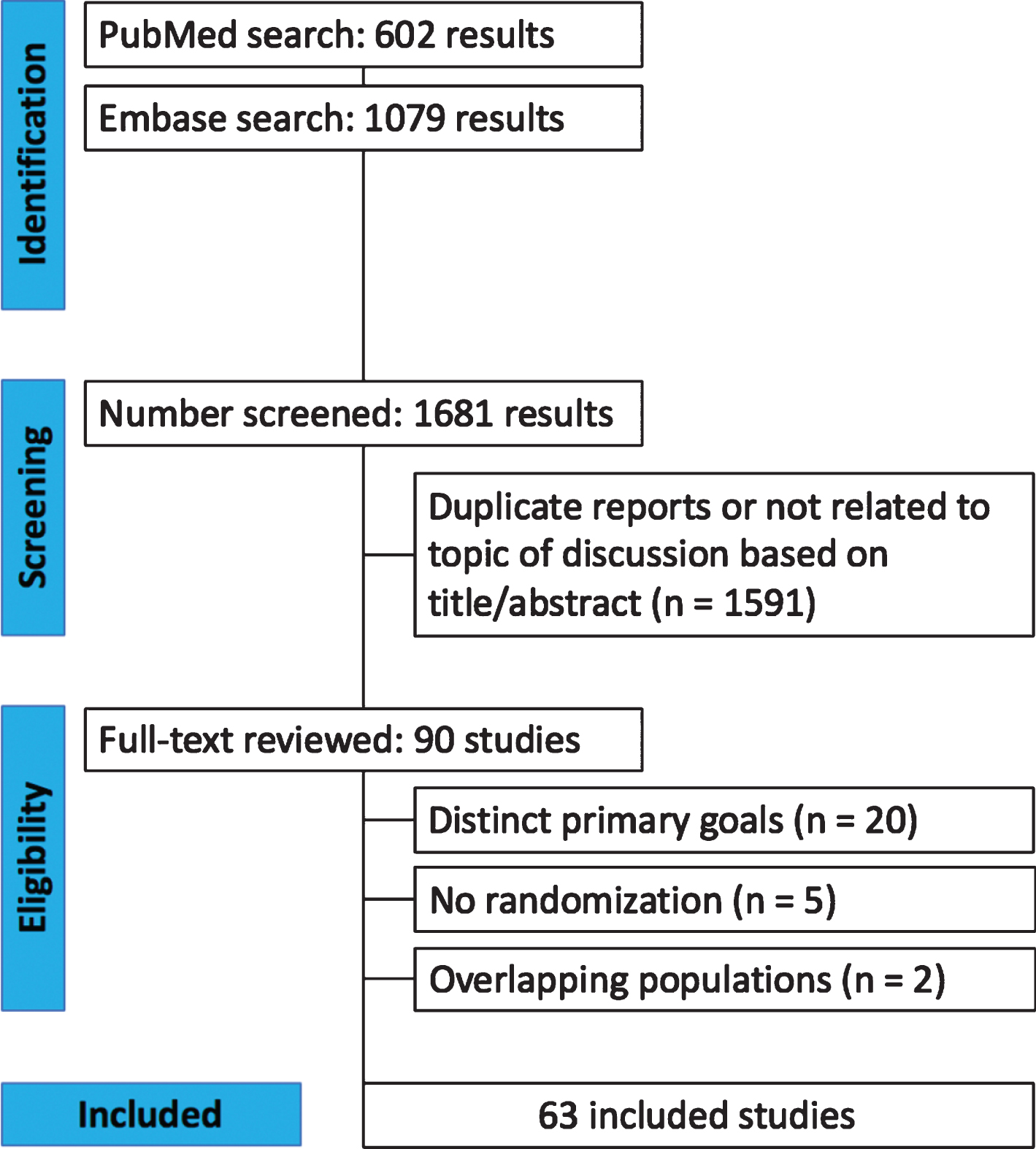
PRISMA flow diagram of study screening and selection.
A summary of the included studies is presented in Table 1 and a detailed description can be found in Supplementary Material 1. The majority of included studies consisted of double-blinded RCTs with a primary goal of evaluating the management of neuropsychiatric symptoms in PD with drugs (56 studies, 88.8% of all RCTs), although 6 evaluated neurostimulation therapies (9.5%) and 1 evaluated bright light therapy (1.6%). Most studies were performed in Europe (42.9%) followed by North America (34.9%) and multinational (9.5%). The most studied psychiatric symptoms were depression (31.7%), cognition/dementia (31.7%) followed by psychosis (17.4%) and apathy (7.8%). The NIH was reported as one of the funding sources in 14.3% of all studies.
Characteristics of studies and participants
The definitions of race and ethnicity were unclear in most studies and these terms were used interchangeably in the majority of the analyzed RCTs. A total of 11 (17.5%; (% CI 9–29%) RCTs reported race/ethnicity of enrolled participants, although in most studies the race and ethnicities mentioned were limited [19–29]. The combined number of patients in these studies resulted in a total of 2515 participants, which represents 31.5% of the patients of all studies. Study characteristics and patient demographics were analyzed (Fig. 1).
Of all studies that included race/ethnicity data, only 5 African American or Black patients were reported to be enrolled (0.2%), 16 Hispanics (0.64%), 539 Asians (21.44%) and 47 (1.86%) other, unspecified or not described by the authors (1.86%). A total of 1,908 (75.8%) white patients were included.
The probability of reporting race or ethnicity was impacted by funding sources although did not significantly differ according to the country where the study was performed. Most studies reporting this specific demographic data were multinational (66.7%) followed by RCTs performed in North America (22.7%), then Europe (7.4%). One-way ANOVA test was performed and did not reveal any statistical significance regarding the location where clinical trial was being performed (Fig. 2) (F = 0.461, p = 0.5) Studies funded by the NIH were more likely to report race and ethnicity when compared to non-NIH funded studies as demonstrated in Fig. 3 (60 versus 30%, F = 4.82, p = 0.032).
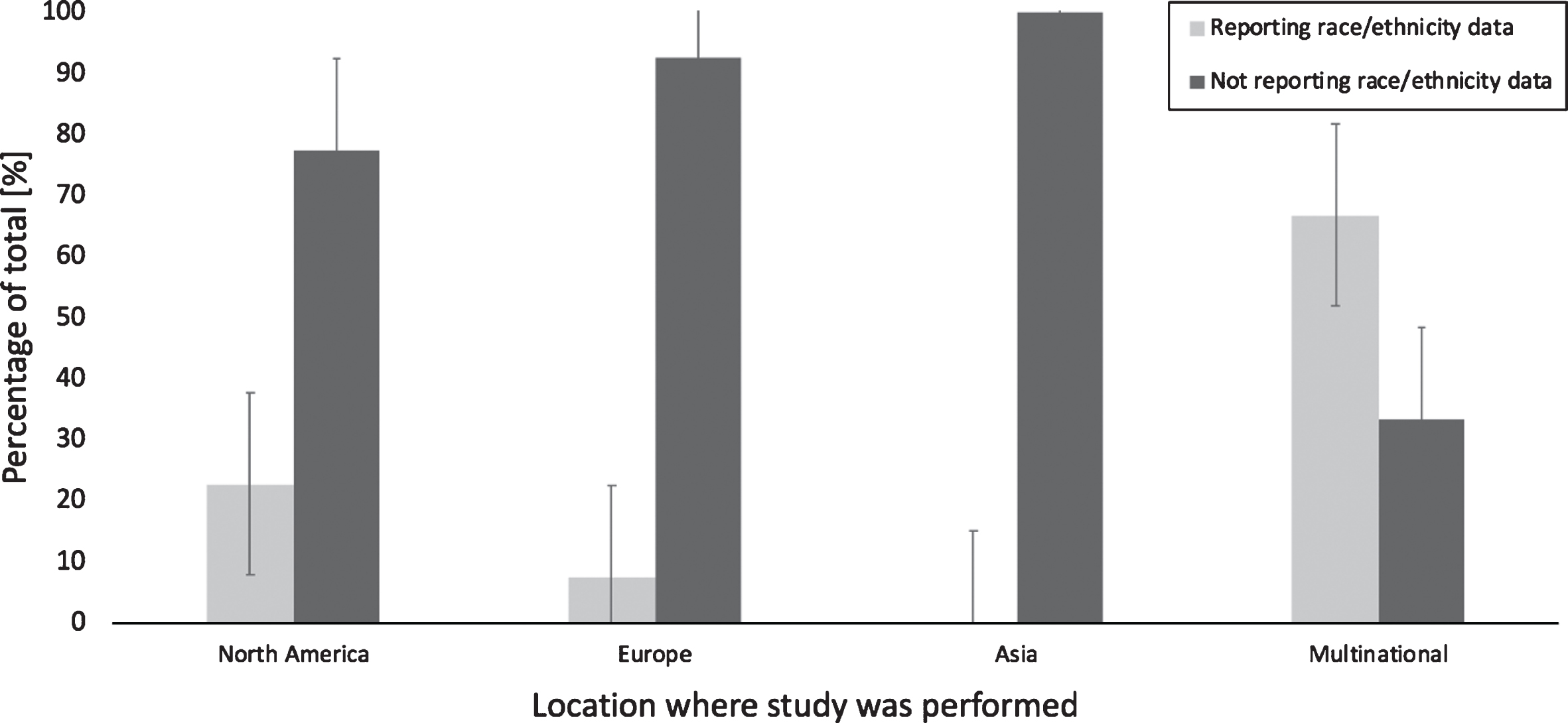
Reporting of race or ethnicity in RCTs by location. Data presented comparing the percentage of studies that reported race and or ethnicity vs. those which did not according to geographic distribution. There was no statistically significant difference comparing the different locations. Error bars show the standard deviation.
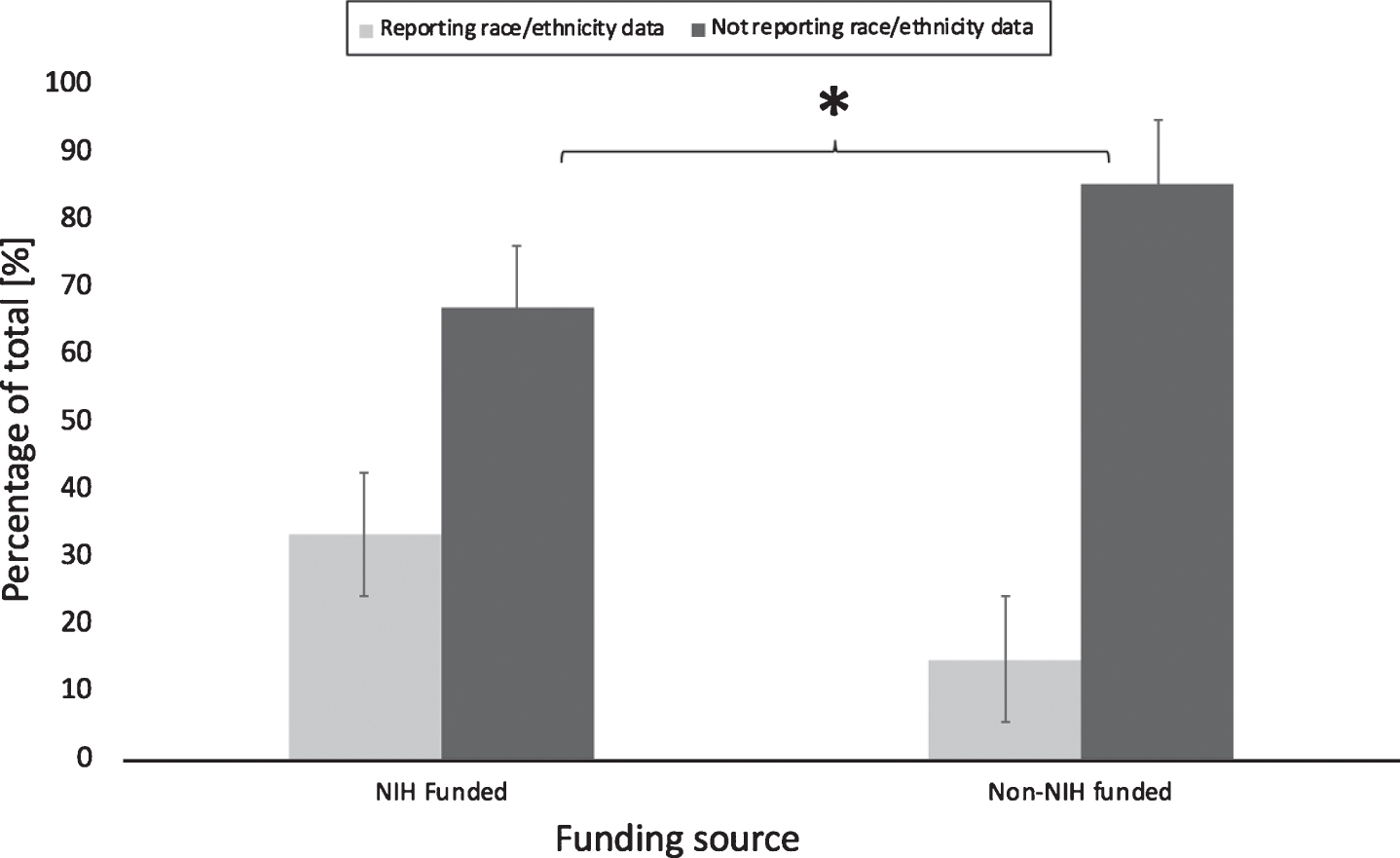
Reporting of race or ethnicity in RCTs by funding source. Comparison of the percentage of studies that reported race and or ethnicity vs. those which did not according to funding source. NIH funded studies presented a statistically significant higher percentage compared to the non-NIH funded ones t(61) = –1.35, p = 0.032. Error bars show the standard error deviation.
Pooled analysis of studies reporting racial and ethnic data
White patients were significantly more likely to be enrolled in clinical trials evaluating neuropsychiatric symptoms. The pooled prevalence of being white in a clinical trial was 98% (CI 0.97 to 0.98, p < 0.001) as seen in Fig. 4. I-square was 0%, meaning no heterogeneity for all analyzed studies. Using meta-regression, we evaluated the effect of year on the pooled prevalence of being white and included in a clinical trial. Year (from 2002 to 2015) had no effect on the prevalence of being white and being included in a clinical trial (p = 0.31).
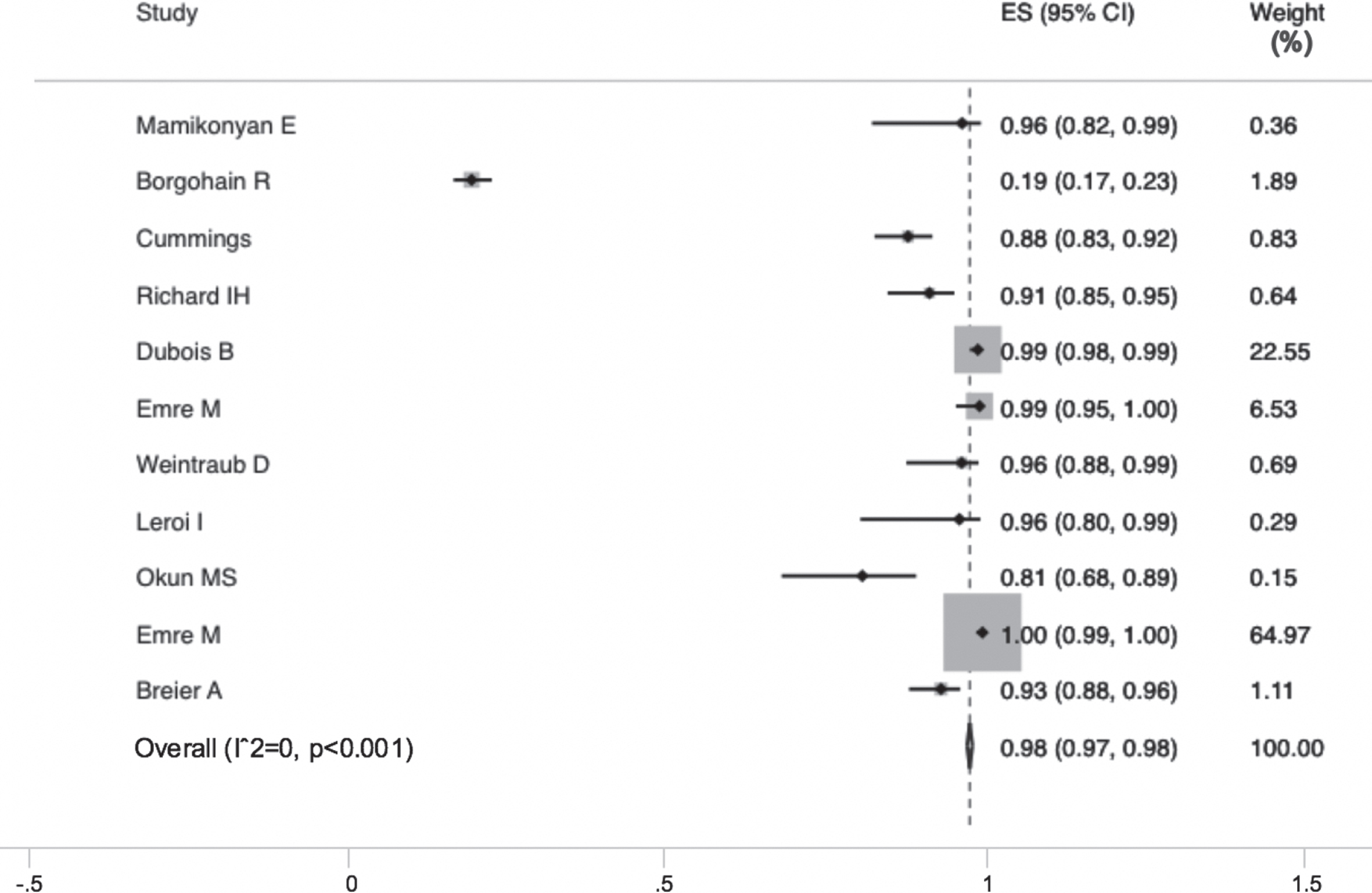
Forest plot of randomized controlled trials and prevalence of enrolled white patients.
Quality assessment
The 11 RCTs describing race or ethnicity were analyzed. All studies were considered high quality by the Jadad score. One of the RCTs consisted of a surgical trial and therefore double blinding was not applicable. Nevertheless, it was still considered a high-quality study according to the Jadad criteria. No studies were stopped prematurely. The risk of selection, attrition, and reporting biases was considered low in all 11 RCTs. On funnel plot analysis, studies occupied a symmetrical distribution according to weight and converged toward the pooled effect as the weight increased (p = 0.22) (Supplementary Material 2). Therefore, there was no evidence of publication bias.
DISCUSSION
A total of 7,973 patients from 63 studies were included in the analysis of ethnicity and race reporting rates in trials investigating neuropsychiatric symptoms in PD. Our results demonstrate a strikingly low report of ethnicity and race in RCTs evaluating neuropsychiatric symptoms in PD. Among those who did report race and ethnicity data, there was a decreased representation of patients who identified with a minority population, specifically African Americans and Hispanics.
Few studies have previously investigated the enrollment of minorities in PD trials. For instance, Schneider et al. found that only 17% percent of all PD trials conducted in the US from 1985 to 2007 reported racial and ethnic characteristics of enrolled patients. Among the studies that provided this information, only 8% of all participants were non-white [12]. In accordance with the literature, our analysis demonstrated that only a minority of RCTs provided racial and ethnic data regarding their patients or had a much lower enrollment of underrepresented minorities.
To our knowledge, this is the first study evaluating minority enrollment in trials for non-motor symptoms in PD. Although non-motor symptoms, including neuropsychiatric manifestations, may be severely debilitating, there is an overwhelming predominance of studies evaluating the motor manifestations of PD [5]. Additionally, there is a high disparity in neuropsychiatric symptoms and treatment amongst different races or ethnicities in the general population. For instance, underrepresented minorities are less likely to receive guideline-based treatments for depression and anxiety care compared to the white population and African American patients are less likely to be treated with newer antipsychotics in the United States [30, 31]. Although most studies addressing this issue originate from North America, we believe this is a global issue that may occur in other continents. The disproportionally low participation of racial/ethnic minorities in clinical trials evaluating neuropsychiatric symptoms may contribute to worsening disparities, as it may lessen the generalizability of studies of new therapeutics for patients with PD. In our meta-analysis, none of the studies acknowledged the skewed representation of race and ethnicity in their study populations, disregarding possible differences in efficacy and side effects of the tested interventions among different racial/ethnic groups.
Multinational studies were more likely to report race and ethnicity when compared to North American or European studies. Nevertheless, the comparison may be slightly inconsistent as some racial and ethnic designations are not universally used. Although the total number of Asian patients was high in our meta-analysis (21%), only one RCT reported the rate of enrolled Asians [19]. As the study was mostly performed in Asia, it enrolled a disproportionately high number of this underrepresented population (539 patients). We believe that the inclusion of this study might have skewed the final analysis. According to the last US census in 2010, 73% of the population was white, 12.7% Black or African American, 5.4% Asian and 16.3% Hispanic, although about one-fifth of all patients with PD in the US may be part of an underrepresented minority [32, 33]. Nevertheless, when analyzing RCTs performed in North America only, the mean enrollment of white population was of 94.72%, Hispanics 2.08% and no African American patients were reported to be enrolled.
Additionally, as expected, NIH-funded studies reported racial and ethnic data more consistently than non-NIH funded trials. Although unclear, we believe the larger proportion of underrepresented minorities in trials funded by the NIH may be due to the implementation of stricter and more comprehensive guidelines by this institute supporting the recruitment and enrollment of minorities [34]. Moreover, recent NIH-funded trials in PD have been encouraged by this institute to recruit more minority patients in an effort to increase generalizability [35]. In our meta-analysis, most trials collected racial/ethnic data by self-report, which is in accordance to the most recent recommendations by the FDA [36]. As noted by several studies, obtaining this information from administrative data may result in incorrect classifications when compared to self-reported data [37].
Possible explanations for the low minority enrollment are still unclear. Recruitment methods may play an integral role in the deficit of minority enrollment. Williams et al. reported that inconvenience and reputation of the research institution within the minority community affected the ability to recruit African Americans into their trials [38]. Due to overall limited access to healthcare within minority communities, this may lead to a sampling of a very homogenous population. Difficulties in recruitment of minorities to enroll in specialty clinics are likely rooted in the degree of overall health literacy, frequency of referrals to specialty clinics, and overall lack of representation of their own ethnicity/race amongst healthcare providers [39–41].
Additionally, the cultural acceptance of symptoms varies amongst certain ethnicities and races. A study by Dahodwala et al. reported that African Americans are more likely to report disability at later stages in PD than Caucasians [42]. Reduced reporting of symptoms results in delayed PD diagnosis and therefore may partially explain the differences in demographic distribution of PD and inclusion into randomized trials. Williams et al. reported that mistrust, insufficient and information dissemination led to decreased enrollment rates of African American patients in Alzheimer’s biomarker research [38]. We believe similar barriers are present in enrollment of African American patients in PD trials. One other potential hypothesis is that the lack of acceptance for psychiatric symptoms amongst specific cultures may lead to a decrease in reporting rates and enrollment rates in these patient populations. Britt-Spells et al. highlights how perceived discrimination from expressing depressive symptoms is a barrier to seeking psychiatric care in the African American community [43].
Possible strategies to increase minority enrollment in PD trials may include a standardized requirement to include racial and ethnic data in all studies. Identifying this health disparity would be the first step in establishing appropriate measures to address this issue. To support this recommendation, the FDA published a guideline in 2005 that strongly recommended the inclusion of underrepresented minorities, “Guidance for Industry: Collection of Race and Ethnicity Data in Clinical Trials” [44]. Additionally, there are also some ongoing studies and clinical trials to evaluate the best strategies to improve minority recruitment.
Our study has limitations. First, most studies did not include race and ethnicity data, which may not directly reflect a low inclusion of this underrepresented minorities but only a lack of collecting or reporting this information. Second, many of the included RCTs were performed outside North America, including countries in Europe and Asia, where some ethnic and racial designations such as African American may not be consistently used or even exist. Racial and ethnic identities are considered a complex phenomenon that may be shaped by social and cultural experiences [45]. Therefore, we acknowledge the complexity of such designations and the possibility of selection bias when analyzing trials that did report race and ethnic data. Moreover, given the inclusion of worldwide studies where minorities may not exist, there may be a chance that our study is not fully representative of the current situation regarding the inclusion of underrepresented populations in RCTs around the world. Nevertheless, we consider the fact that most studies did not even collect or include any racial or demographic characteristics in their manuscript a relevant finding and potential sign of health disparity by itself.
Conclusions
The results of this meta-analysis including more than 7,500 patients found that only a small percentage of RCTs evaluating the treatment of neuropsychiatric symptoms in PD reported race and ethnicity from 2000–2019. Consistent with previous studies, minorities have been underrepresented in PD trials, where there is an overwhelming predominance of white patients. Despite some of the known differences in terms of diagnosis and treatment of neuropsychiatric symptoms in underrepresented groups, clinical trials in PD did not consistently describe their ethnic representation. The exclusion of race and ethnicity data from clinical trials, or the under enrollment of certain racial/ethnic groups may limit our knowledge of the management of neuropsychiatric symptoms in PD in these populations. Finding solutions to the barriers detailed above are needed to improve minority enrollment in RCTs and improve generalizability of new therapies.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
