Abstract
Background:
Availability of validated Parkinson’s disease (PD) questionnaires in languages spoken in Africa will enable the conduct of epidemiological studies.
Objective:
The aims of the current study were to develop cross-cultural translated and validated Arabic and French versions of a PD screening questionnaire, and determine its diagnostic accuracy for recognition of parkinsonism in early and moderate-advanced PD in three countries (Cameroon (French), Egypt (Arabic), and Nigeria (English)).
Methods:
This cross-sectional study screened 159 participants (81 PD and 78 controls) using the PD screening questionnaire. The questionnaire was translated into Arabic and French versions using standard protocols. Cognitive function was assessed using the Montreal Cognitive Assessment and the Identification and Intervention for Dementia in Elderly Africans cognitive screen. Co-morbidity burden was documented using the Charlson Comorbidity Index. PD severity and stage were evaluated using the MDS Unified Parkinson Disease Rating Scale and the Hoehn and Yahr scale respectively.
Results:
Both PD patients and controls were matched regarding age, gender, education, and co-morbidity burden. The PD screening questionnaire scores were significantly higher in PD (median 8.0, IQR 6.0–10.0) in contrast to controls (0.0, IQR 0.0–0.0) (p < 0.0001), with a similar pattern and level of significance across all country sites. In ROC analysis, the questionnaire demonstrated high diagnostic accuracy for PD overall, with an AUC of 0.992 (95% CI 0.981–1.002).
Conclusion:
The Arabic, French, and English versions of this PD screening questionnaire are valid and accurate screening instruments for recognition of Parkinsonism. This paves the way for conducting epidemiological studies in many African countries.
INTRODUCTION
Epidemiologic data on the burden of Parkinson’s disease (PD) in most countries in Africa are sparse, and have been largely based on extrapolation of data derived from studies conducted several decades ago. The current and predicted population expansion occurring globally and boosted by increased life expectancy, including in African countries, has contributed significantly to the growing PD pandemic. Specifically, according to the United Nations, by 2050, the proportion of the population aged 65 and older will reach 7.5% in Africa [1]. The Global Burden of Disease Study data highlight PD as the fastest growing neurological disorder in prevalence, disability and deaths [2]. The estimated global burden of PD in 2017 was 8.5 million cases (range 7–10 million), a figure that is expected to double by 2040 all over the world including Africa [3, 4].
The reported prevalence of PD varies between different geographical regions, and is stated to be higher in Europe and North America than in Africa, with intra-continental variation (higher in North Africa compared to Western Africa for instance) [5, 6]. Methodological differences (e.g., in case definition, identification, and screening methods) in the studies from which these data have been derived may contribute to the discrepancy, although the influence of genetic and environmental factors on disease risk are widely acknowledged [2, 5]. Epidemiological transitions have also occurred since some the studies were conducted. Thus, it is imperative to accurately document and update the prevalence and incidence of PD in Africa. Such data are of interest for their potential to identify risk factors, describe the natural history and outcomes, and guide policy makers in effectively planning and deploying resources for manpower and infrastructure to support care delivery to people with PD.
In conducting large population-based studies capable of generating credible data, validated, sensitive and specific PD screening questionnaires are indispensable in increasing diagnostic accuracy. Although several screening questionnaires have been developed for Parkinsonism, the study design, sample size and inclusion criteria have varied [7]. Differences in the lingua franca across populations has also limited utilization of some of the screening instruments. Availability of translated versions of such questionnaires paves the way for multi-national studies with similar methodologies that will consequently enable direct comparison between populations. The screening questionnaire developed by Tanner and colleagues is widely used, has been translated to several languages and tested in high quality studies [8].
The aims of the current study were to develop cross-cultural translated and validated screening versions of the PD screening questionnaire developed by Tanner et al. in Arabic and French languages for use in Africa, and determine the diagnostic accuracy of the translated French and Arabic and original English PD screening versions in identifying Parkinsonism in early (mild) and moderate-advanced PD in three African countries (Cameroon (French), Egypt (Arabic), and Nigeria (English)).
MATERIALS AND METHODS
In this observational, cross-sectional, international multi-center study, consecutive PD patients were recruited from Neurology outpatient clinics in three African countries (Nigeria, Egypt, and Cameron) between March and September 2019. The study was approved by the institutional ethics committees at Hospital Laquintinie in Douala (Cameroon), Lagos University Teaching Hospital (Nigeria), and Ain Shams University (Egypt), consistent with the ethical standards of the Declaration of Helsinki. Informed consent was obtained from all participants.
The PD diagnosis was made by a neurologist with expertise in movement disorders at each site according to the Movement Disorders Society (MDS) clinical diagnostic criteria [9]. The study included patients with both early (mild) PD (Hoehn and Yahr (HY) stages 1 and 2) and moderate-advanced PD (HY stage ≥3) [10], with a pre-planned recruitment ratio of 1 to 1 for each stage category. PD patients with dementia based on the Identification and Intervention for Dementia in Elderly Africans (IDEA) cognitive screening questionnaire (score < 10) [11], the Montreal Cognitive Assessment (MoCA) (score < 21) [12], and/or the DSM-V criteria were excluded. Control participants were unrelated community-based age-, gender-, and educationally-matched healthy volunteers. Other exclusion criteria were atypical or secondary Parkinsonism, presence of comorbidities that might impair participation or confound screening, or presence of other neurological disorders such as stroke.
The demographic data (including age, gender, and years of education) were documented for all study participants. Each PD participant underwent comprehensive clinical assessment by a movement disorders expert and was evaluated using the MDS Unified Parkinson Disease Rating Scale (MDS-UPDRS), the HY scale, and the Charlson Comorbidity Index (CCI) to measure associated comorbidities [13–15]. The MoCA (the existing Arabic, French and English versions) [16, 17] and IDEA screening questionnaires (the origional English and current translated Arabic and French versions) were used for cognitive assessment. The English version was employed in Nigeria, whilst the Arabic and French translations were used in Egypt and Cameroon, respectively. The IDEA cognitive screen has been previously validated in Nigeria, and the method of administration (interviewer-administered in the local language with the words from the 10-word list translated into the local equivalent) was utilized [18].
Subsequently, the PD screening questionnaire developed by Tanner and colleagues was adminstered by trained physicians following translation as described below [8]. This screening approach has also successfully identified parkinsonism in other populations [7, 19]. The screening questionnaire developed by Tanner et al. comprises 11 items that ask about difficulty arising from chair, smaller handwriting, difficulty buttoning, softer voice, change in facial expression, lack of balance, presence of shaking (tremor), presence of freezing, slowness of movements, falls and shuffling gait. In addition, a question regarding the previous diagnosis of PD is included. The range of responses are ‘yes’, ‘no’, or ‘uncertain’, with each response scored correspondingly as 1, 0, or recorded as a missing answer. The maximum score attainable is 11.
Arabic and French Translation of PD screening and IDEA questionnaires
Arabic and French translations and linguistic validation were performed according to the standard methodology of translation and cultural adaptation in Egypt and Cameroon, respectively [20]. Two bilingual professionals (native Arabic-English and French-English) from each group translated the original English version of both questionnaires independently, and considered clarity, cultural applicability and concise formulation. Subsequently, the two copies of the translated language versions were compared and reconciled into one Arabic and one French version. Back translation (to English) was carried out by two different bilingual experts, for each unified language version, independently and without prior exposure to the original questionnaire. Thereafter, all translations for each language and the original version were reviewed by the whole panel to resolve any discrepancies and develop the pre-final harmonized Arabic and French copies of the questionnaire. This final copy of each language was tested in ten PD patients by the interviewers to detect any difficulties and probe their understanding of all questions (cognitive debriefing). The Egyptian and Cameroon teams approved the final copy of the Arabic and French translations of the screening questionnaire respectively, to be applied in the current study.
Statistical analysis
Data analysis was conducted using the Statistical Package for Social Sciences (SPSS) ® version 25.0 (IBM, San Francisco, CA). Normal distribution of the variables was analyzed using the Kolmogorov-Smirnov test. Descriptive data are provided in terms of frequencies (percentage), mean and median values with standard deviation or interquartile range, as appropriate, and 95% confidence intervals. Differences were compared using Analysis of Variance (ANOVA) and non-parametric tests respectively. The significance of inter-group differences in categorical variables was determined using χ2 test. Correlations were calculated using the Pearson (parametric) and Spearman (non-parametric) correlation coefficients. Linear regression analysis was used to estimate the association of PD screening questionnaire scores after adjusting for age, gender, comorbidity, education, and cognitive status. Using Receiver Operating Curve characteristics (ROC) analysis, cut-off value scores against the clinical diagnosis of PD (yes versus no), sensitivity, specificity, positive predictive values (PPV) and negative predictive values (NPV) were determined for each question and the total number of positive answers that effectively identified PD. In addition, we performed post-hoc analyses. In these analyses, we stratified the sample according to mild versus moderate- advanced PD [HY stage <or ≥3, respectively], old versus young (based on the mean age of the PD sample), and gender. Statistical significance was set at a p value <0.05.
RESULTS
Baseline characteristics of study participants
A total of 159 participants (105 male, 66% and 54 female, 34%) were enrolled in the study, comprised of 81 PD and 78 healthy controls. The demographic characteristics overall and by country of study are compared in Tables 1 and 2. The PD screening questionnaire was fully completed in 65 (80.2%) of the PD cases, and 73 (93.6%) of controls. When PD patients and controls were compared, there was no statistically significant difference in the age at study, gender distribution, years of formal education, and burden of co-morbidities. PD cases had significantly lower cognitive screening scores on both the IDEA and MoCA tests (p < 0.0001 for both intergroup comparisons). In country-specific comparisons, the pattern was similar except for a higher median number of formal years of education in controls versus PD in the Egypt cohort (p = 0.01), and a significantly higher co-morbidity burden index in PD compared to controls in Cameroon (p = 0.02) (Table 1).
Demographic and clinical characteristics of PD cases and controls; total and country-specific distribution
*Values are reported in means (standard deviation); **Values are reported in median (interquartile range); y, years; m, male; CCI, Charlson Comorbity Index; IDEA, Identification and Intervention for Dementia in Elderly Africans; MoCA, Montreal Cognitive Assessment; PD, Parkinson’s disease; n, number.
The median duration of PD was 4.0 years (2.0–7.0), and was longest in the Cameroon cohort [6.0 years (2.5–9.0)] (p = 0.01). The median HY stage, levodopa equivalent daily dosage (LEDD), and total and motor MDS UPDRS scores were also similar across the study sites (p = 0.32 and 0.21, respectively) (Table 2).
Parkinson’s disease specific characteristics compared by country of study
*Duration based on earliest PD-defining motor symptom; *Median (IQR); LEDD, levodopa equivalent daily dosage; PD, Parkinson’s disease; HY, Hoehn and Yahr stage.
Overall, higher PD screening questionnaire scores were correlated with older age (r s = 0.65, p = 0.02), longer PD duration (r s = 0.48, p < 0.0001), higher Charlson Comorbidity Index (r s = 0.25, p = 0.02), higher MDS-UPDRS total score (r s = 0.65, p < 0.0001), lower number of years of education (r s = –0.24, p = 0.02), and lower total IDEA score (r s = –0.41, p < 0.0001), but not with the total MoCA score (r s = –0.15, p = 0.20). In the linear regression analysis, the presence of PD (β= 6.99, 95% CI 0.35–5.99, p < 0.0001), higher comorbidity (β= 0.31, 95% CI 0.04–0.59, p = 0.02), and lower cognitive scores (lower IDEA total score) (β= –1.15, 95% CI –1.85–0.45, p = 0.001) contributed to 79% of the variance of the PD screening score.
PD screening questionnaire performance; diagnostic accuracy and predictive values
The PD screening questionnaire (score range 0 –11) scores were significantly higher in PD compared to controls [8.0 (6.0 –10.0) versus 0.0 (0.0–0.0)] (p < 0.0001), with a similar pattern and level of significance across all country sites as shown in Tables 1 and 2. In ROC analysis, the PD screening instrument showed high diagnostic accuracy for PD overall, with an AUC of 0.99 (95% CI 0.98–1.00), and similarly high sensitivity (0.95, 95% CI 0.87–0.99), specificity (0.97, 95% CI 0.90–0.99), PPV (0.96, 95% CI 0.89–0.99) and NPV (0.95, 95% CI 0.88–0.99). This discriminant accuracy, based on the AUC, was maintained irrespective of age (≤63 or >63 years), gender, and stage of PD. The specificity and positive predictive values were lower in mild PD in comparison, although the values were still high (0.89 and 0.81) (Table 3 and Fig. 1).
Diagnostic accuracy and predictive value of the PD screening questionnaire
AUC, Area Under Curve; PV, predictive value; PD, Parkinson’s disease; HY, Hoehn and Yahr stage.
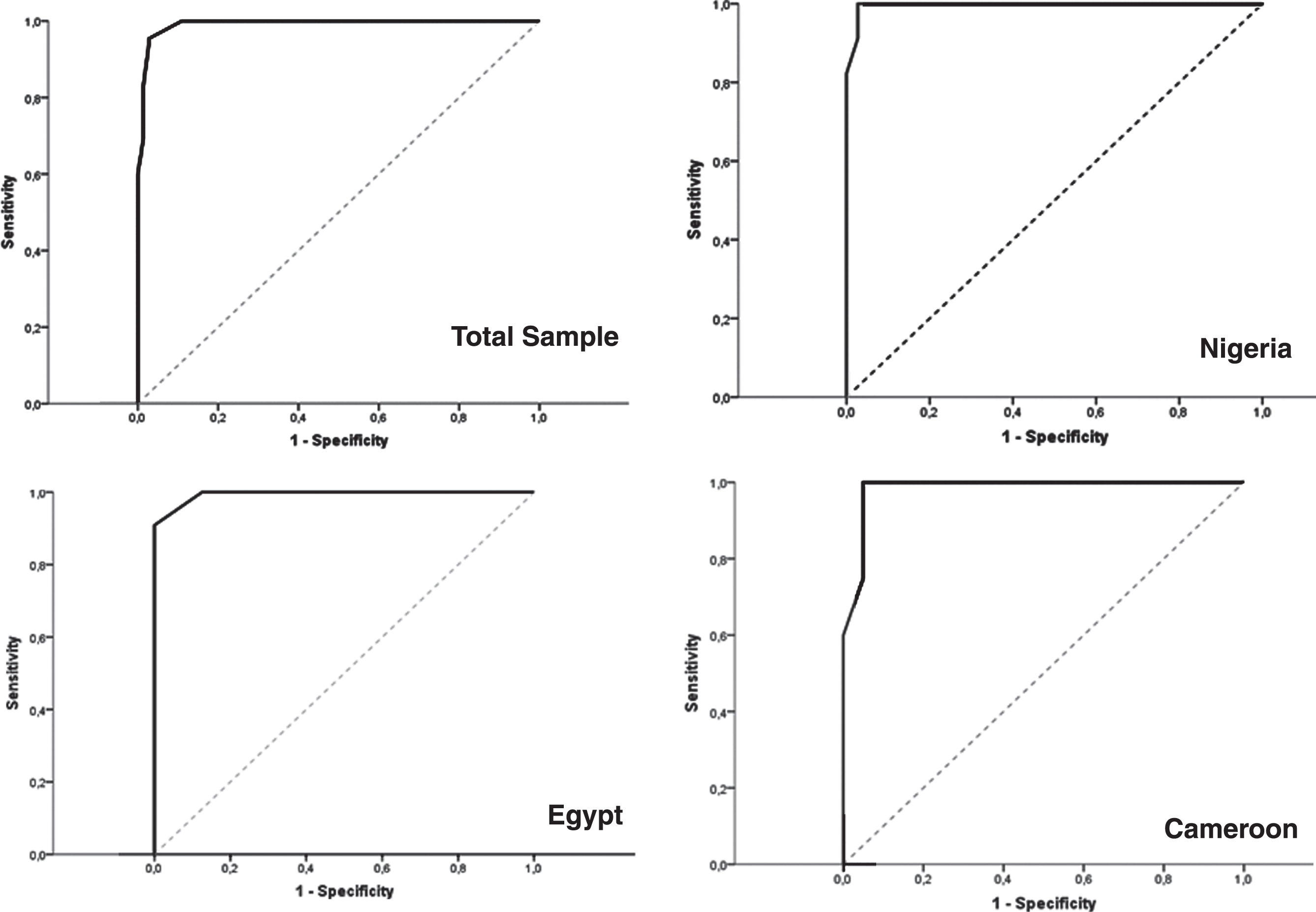
ROC curves of specificity and sensitivity of PD screening questionnaire for the total sample and different populations.
In country-specific analyses (Supplementary Table 1), all the PD screening questionnaire versions exhibited high discriminant value for diagnosing PD (AUC≥0.98). The original English version (used in Nigeria) had an AUC≥0.99 for all comparisons, and sensitivity, specificity, PPV and NPV≥0.93. The Arabic version (Egypt) had an AUC≥0.99 for all comparisons, sensitivity, specificity, PPV and NPV≥0.90 except for mild PD (specificity 0.87 and PPV 0.75). The French translation (Cameroon) had an AUC≥0.96 overall in discriminating PD from controls, and specificity, sensitivity, PPV and NPV≥0.95.
In evaluation of the item-specific performance, falls, freezing of gait and slowness of movement were most sensitive (100%), while shaking (tremor) was the most specific item (99%). This pattern was maintained across the three countries and when sub-analyzed by age, gender, and disease severity. Moreover, shaking, buttoning and facial expression had high sensitivity in early and moderate-advanced groups, while other symptoms had lower sensitivity among early PD (especially falling, arising from chairs and voice change). Slowness of movement and facial expression had high specificity in both PD groups, while other symptoms had high specificity only in moderate-advanced PD (Supplementary Table 2).
DISCUSSION
The main purpose of this study was to enable the future conduct of epidemiological research of Parkinsonism, including PD, across more sites within Africa, by linguistically translating and validating the existing widely used and highly recommended screening questionnaire developed by Tanner et al. [7, 22]. This screening questionnaire has been tested in communities and clinics, and translated from English to several languages, but not to Arabic or French languages [7, 23]. Therefore, our approach was to validate the original version in a country in which English is the lingua franca (Nigeria), and translate the questionnaire in two countries in which the official language is Arabic (Egypt) and French (Cameroon). With this strategy, the screening instrument would potentially find application for epidemiological studies of Parkinsonism in the majority of African countries where it can be applied as an initial screening tool for Parkinsonism, with verification of the diagnosis of PD or other causes of Parkinsonism using established clinical diagnostic criteria. We estimate that this provides coverage of approximately 78% of African countries, considering that these three languages are spoken collectively in at least 42 of the 54 countries. The current distribution of official languages in African countries indicates the use of English in 24, French in 21, Arabic in 13, Portuguese in 6, Spanish in 1, and Swahili in 5 (with some countries having a bilingual or multilingual official language system). (https://en.wikipedia.org/wiki/List_of_official_languages_by_country_and_territory). A large number of indigenous languages (nearly 3000) are spoken in Africa, with most belonging to the three largest language groups (Niger-Congo, Nilo-Saharan, and Afroasiatic) [24]. As such, the use of ‘official’ languages is logical and commonplace (including in clinical settings) to simplify communication. The disadvantage is the need to use an interpreter in some situations with the slight possibility of misinterpretation. We however anticipate that expanding the opportunity of using the screening questionnaire in the official languages through these translations will provide the impetus and foundation for and facilitate further translation into the indigenous languages in each country.
One of the major strengths of the study is that the cross-cultural translations into Arabic and French was conducted in accordance with standard international guidelines [20]. Furthermore, we applied the most recent MDS clinical criteria for diagnosis of PD with inherent high sensitivity and specificity. Our study has demonstrated that the original English version and the Arabic and French translations are diagnostically accurate in distinguishing parkinsonism in neurologist-diagnosed early stage and moderate-advanced PD from controls, irrespective of age and gender. However, further studies are warranted to evaluate the performance of the screening instrument in other parkinsonian syndromes. Our findings were similar between the three studied groups/languages, including the original English questionnaire. Pramstaller et al. also found no differences between validation versions of the Tanner questionnaire in Italian and German cohorts [25]. Previous studies showed variable sensitivity and specificity of this screening questionnaire that could be attributed to different characteristics of the studied population and the method of administration (self-administered, via mail or interview) or by physician [7]. Remarkably, most of the previous studies used the 9 items Tanner questionnaire whereas we used the 11-item version.
The screening questionnaire performance was expectedly high because of its inclusion of several items specific to Parkinsonism, and more so in the population source of the study being previously diagnosed PD [7, 26]. Rather than the proposition of a bias relating to participants prior knowledge of PD symptoms, and taking cognizance of the fact that our study included recently diagnosed (but late presenting) PD, we suggest that the high sensitivity may be due to the discriminatory quality of questions included in the original questionnaire. Furthermore, to simulate real-world application of the questionnaires, although PD and controls were matched for co-morbidity burden and age (within 3 years mean age), the questionnaire maintained its discriminatory ability in identifying Parkinsonism.
In addition, our study identified three items (falls, freezing of gait. and slowness of movement), that were most sensitive among different subgroups across all three versions and one item that was most specific (tremor). Sarangmath et al. identified the tremor question as having the highest specificity, and slowness of movement and shuffling of gait as the most sensitive [22]. Duarte et al. used the 9 item questionnaire and found that low voice, shaking, shuffling gait and buttoning had most positive predictive value, but compared to ophthalmology patients [26]. A note of caution regarding the item on buttoning would be that some cultures have commonly worn apparel completely devoid of buttons and even zips, and this may inadvertently reduce the utility of the question as a surrogate for impaired dexterity based on the peculiarity of this dressing task. Falls showed lowest specificity (61%) in the current study, especially in the Nigerian cohort. This could be attributed to the shorter disease duration and lower HY staging (milder PD cohort), compared to the other countries. As expected, the countries with a higher proportion of moderate-advanced PD within the cohort showed higher fall rates and higher specificity for falls (83%). Furthermore, the current study showed differences in questionnaire performance based on PD severity. Identifying these differences is important to determine the most sensitive and specific questions for screening patients with early stages of Parkinsonism.
The application of the questionnaire by a neurologist, neurologist in training, or other trained physician as occurred in this study may have contributed to the performance. Therefore, the results should be viewed in that context. In field studies, trained non-physician interviewers are conventionally employed due to the logistic difficulties and resource constraints that preclude engaging physicians for initial surveys. On this basis, we suggest that in utilizing these questionnaires in the context of community based epidemiological surveys, that a phased/multi-stage strategy (such as is described in the World Health Organization research protocol for measuring prevalence of neurological disorders in developing countries) with follow-up standardized clinical diagnostic verification by trained physicians or neurologists be employed [27–29]. The a priori demonstration of higher sensitivity and specificity would be an advantage and potentially improve the likelihood of detecting Parkinsonism even in early stage or milder PD as a part of multistage strategy, thereby increasing the accuracy of prevalence and incidence data from Africa. Considering the current initiative for prodromal diagnosis of PD however, this instrument would only be applied in clinically overt PD where motor manifestations such as are captured in the questionnaire are present. Thinking futuristically, there may be other applications for the questionnaire in early detection of PD, including incorporation of prodromal symptoms and integration into prediction models using machine learning techniques [23, 30].
We noted the difference in the range of years of formal education of the participants (matched in Nigeria and Cameroon and significantly higher in Egypt controls compared to PD cases). In spite of the latter, this had no measurable negative effect on the performance of the translations, as, if anything, the lower educational level of PD cases did not unduly impact on the accuracy of the screening instrument in terms of the sensitivity, specificity, and predictive values in between-country comparisons. Although the report published by Sarangmath et al. suggested an effect of literacy level of participants on the performance of the modified and translated version of the Tanner questionnaire used in their study, there was a wide variation in reported specificity and sensitivity depending on the administrator (medical personnel, each non-medical assistant). This suggests the possibility of an administrator rather than a participant confounder as has been alluded to in other studies [22]. This is perhaps indicative of the need for careful training of interviewers (physician or non-medical personnel) prior to administration of any survey questionnaire in order to enhance inter-rater reliability.
The less restrictive inclusion of PD cases and controls with co-morbidities is regarded as an advantage in that it enabled us test the questionnaire performance in a diverse real world scenario. The prevalence of PD increases with aging, occurring at a time when other co-morbid non-communicable diseases are also prevalent and often co-existent, and despite which the questionnaires diagnostic accuracy was acceptable.
The current study has some limitations. We highlighted the difference in educational levels across the study sites and the possible implications to the screening results in general, although we did not observe this effect in the present study. Our study did not evaluate inter-rater reliability, and we did not study differences in the performance of the instrument if it were to be administered by other personnel that may be employed in research studies, including nurses, non-medical research assistants, and non-neurologist physicians. We thus suggest that future studies include a wider variety of interviewers to further explore the utility in the broader context. Furthermore, our cohorts had some differences in the clinical characteristics of the PD cases compared across the three countries, specifically longer duration of PD and higher scores in the non-motor and motor aspects of experiences of daily living (MDS UPDRS Part I and II scores) in the Cameroon cohort. This did not translate into any consistent difference in the overall and item-specific performance of the questionnaires particularly as relates to the most specific and most sensitive items highlighted previously.
In conclusion, our study reiterates the validity and diagnostic accuracy of the original English version and provides valid linguistic translations of the Tanner screening instrument for recognizing Parkinsonism. We propose adoption of these Arabic and French translations and wide use of the original English version in epidemiological studies depending on the official language in use in different African countries. We also recommend additional linguistic translation (and validation) into other languages such as Swahili, to extend its utility to other African countries.
CONFLICT OF INTEREST
Caroline M. Tanner is an employee of the University of California – San Francisco and the San Francisco Veterans Affairs Health Care System. She receives grants from the Michael J. Fox Foundation, the Parkinson’s Foundation, the Department of Defense, Bio Electron, Roche/Genentech, Biogen Idec and the National Institutes of Health, compensation for serving on Data Monitoring Committees from Voyager Therapeutics, Intec Pharma and Cadent Therapeutics and personal fees for consulting from Neurocrine Biosciences, Adamas Therapeutics, Grey Matter, Acorda, Acadia, Amneal and CNS Ratings.
Esther Cubo: Spanish government grants (Fondo de Investigación Sanitaria 2020-2023), Travel grants (Allergan, Boston, Abbvie).
Njideka Okubadejo: Michael J. Fox Foundation grant (2019 – 2021)
Other authors declare no relevant financial disclosures.
