Abstract
Combined catechol-O-methyl-transferase-inhibition and Levodopa-Carbidopa intestinal gel (LCIG) infusion has the potential to reduce LCIG daily dose and the costs of this therapy. In this retrospective analysis, we report on Parkinson’s disease (PD) patients on LCIG with concomitant Opicapone. In 11 patients, the introduction of Opicapone led to LCIG daily dose being reduced by 24.8% (p = 0.05) without any significant worsening of dyskinesia. Three patients withdrew from Opicapone due to side effects or inefficacy. LCIG daily dose reduction could lead to cost savings of £142,820.63/year in the United Kingdom while maintaining clinical care.
INTRODUCTION
Levodopa-Carbidopa intestinal gel (LCIG) infusion is one of the available treatments to address motor and non-motor complications in the advanced phase of Parkinson’s disease (PD) [1, 2]. A considerable body of evidence, including the GLORIA registry, EuroInf and EuroInf 2 studies, suggests that LCIG infusion leads to sustained benefits for both motor and non-motor symptoms and is considerably superior to oral therapies in advanced PD [2–4]; however, costs of therapy limit its use, particularly in developing countries. Costs of this device-aided therapy are also a major consideration within the National Health Systems in the United Kingdom (UK).
In the UK, each LCIG cassette costs £77.00, not including Value Added Tax (VAT) [5], and approximately 10% of patients require two cassettes daily, thus substantially increasing costs of treatment. A panel of PD specialists recently highlighted the use of oral enzyme inhibition in combination with LCIG as a strategy to reduce costs [6]. Opicapone, a long-acting, peripheral catechol-O-methyl-transferase (COMT)-inhibitor [7], may be a potential add-on therapy to LCIG infusion as it is likely to improve the efficacy of LCIG by increasing its bioavailability [8]. The latter is also evident from the use of the product Lecigon in Sweden where LCIG is combined with Entacapone [9]. Opicapone is a recently released COMT-inhibitor which appears to have less gastrointestinal side effects compared to Entacapone and only requires a single daily dose as compared to multiple dosing with Entacapone [10, 11]. In this exploratory analysis, we investigated whether the use of Opicapone as an add-on therapy to LCIG infusion in advanced PD patients reduces the LCIG infusion daily dose and potentially the costs associated with this advanced therapy.
MATERIALS AND METHODS
We performed a data analysis of PD patients on LCIG infusion therapy, seen at the Parkinson’s Foundation Centre of Excellence at King’s College Hospital (KCH), London, UK. We included patients in whom Opicapone 50 mg/day was introduced as an add-on therapy to LCIG infusion as has been adopted in the local pathway of care for advanced therapies. Patients were part of the Non-motor Longitudinal International Study (NILS), an international cohort study addressing non-motor symptoms prevalence, treatment effects, and natural history (REC reference: 10/H0808/141; IRAS ID: 62343). Demographics, including age, gender, disease duration, and levodopa equivalent daily dose (LEDD) were taken from NILS. To measure motor symptoms, we used wearable sensor data [12, 13], obtained through the Parkinson’s KinetiGraph™ (PKG) Registry (REC reference: 17/LO/101; IRAS ID: 215965). All patients participating in either study gave written informed consent; both studies were conducted in line with the Declaration of Helsinki.
The primary outcome was to assess the reduction of LCIG daily dose and the potential reduction of LCIG-associated costs after the introduction of Opicapone. We performed a cost savings analysis to determine the potential financial effect of adding Opicapone to LCIG infusion therapy. The analysis was based on the following numbers: according to Healthcare at Home Ltd, the UK’s clinical provider of healthcare out-of-hospital for LCIG, on the first of September 2019, in the UK, a) 335 PD patients were on LCIG infusion therapy, b) of these 335 patients, 23 patients used more than one cassette per day; nine used 1.5 cassette (equalling three cassettes over two days) and 14 used two cassettes per day, c) the average daily amount of LCIG used by 23 patients on more than one cassette a day was 119.7 ml, d) the cost of one cassette of LCIG was £77.00 in the UK [5], e) the cost for Opicapone 50 mg once daily was £93.90 for one month in the UK [14], not including VAT.
The secondary outcomes were changes in motor symptoms, as measured by the PKG bradykinesia and dyskinesia scores, and tolerability of Opicapone. Data were summarised descriptively and the differences pre- to post- Opicapone initiation were tested using the Wilcoxon signed-rank test (p≤0.05 was considered significant).
RESULTS
Thus far, data on 11 PD patients are presented in Table 1. Patients’ assessment data were available at baseline and at a median follow-up duration of 4.4 (2.7–5.2) months after Opicapone introduction. In these 11 PD patients, LCIG daily dose was reduced by 24.8% (p = 0.05) (Table 1). The addition of Opicapone and the subsequent reduction in LCIG daily dose did not result in a worsening of motor symptoms as measured by a wearable sensor (PKG bradykinesia and dyskinesia scores (p≥0.53)). Two out of 11 PD patients stopped Opicapone due to the development of side effects (hallucinations or dizziness) and an additional one for lack of efficacy.
Demographics and outcome measures before and after initiation of Opicapone
LCIG, levodopa-carbidopa intestinal gel; LEDD, levodopa equivalent daily dose; M/F, Male/Female; PKG, Parkinson’s KinetiGraph; NA, Not Applicable. Data are presented as median (25th and 75th percentiles) or percentage. Differences between pre- and post- Opicapone initiation were tested using the Wilcoxon signed-rank test (p≤0.05 was considered significant).
Based on the above-mentioned information, the annual costs associated with LCIG infusion in the UK (not including care with home nurses and other associated costs) was £9,941,922.38.
DISCUSSION
Given the reduction of 24.8% of LCIG daily dose after the introduction of Opicapone in our cohort, all 23 patients on more than one cassette a day in the UK, with an average daily amount of LCIG of 119.7 ml, would be able to use only one cassette on a daily basis with subsequent gross average cost savings of £22,621.68. After including costs related to Opicapone introduction, net average cost savings of £21,494.88 per patient per year could be achieved (Fig. 1).
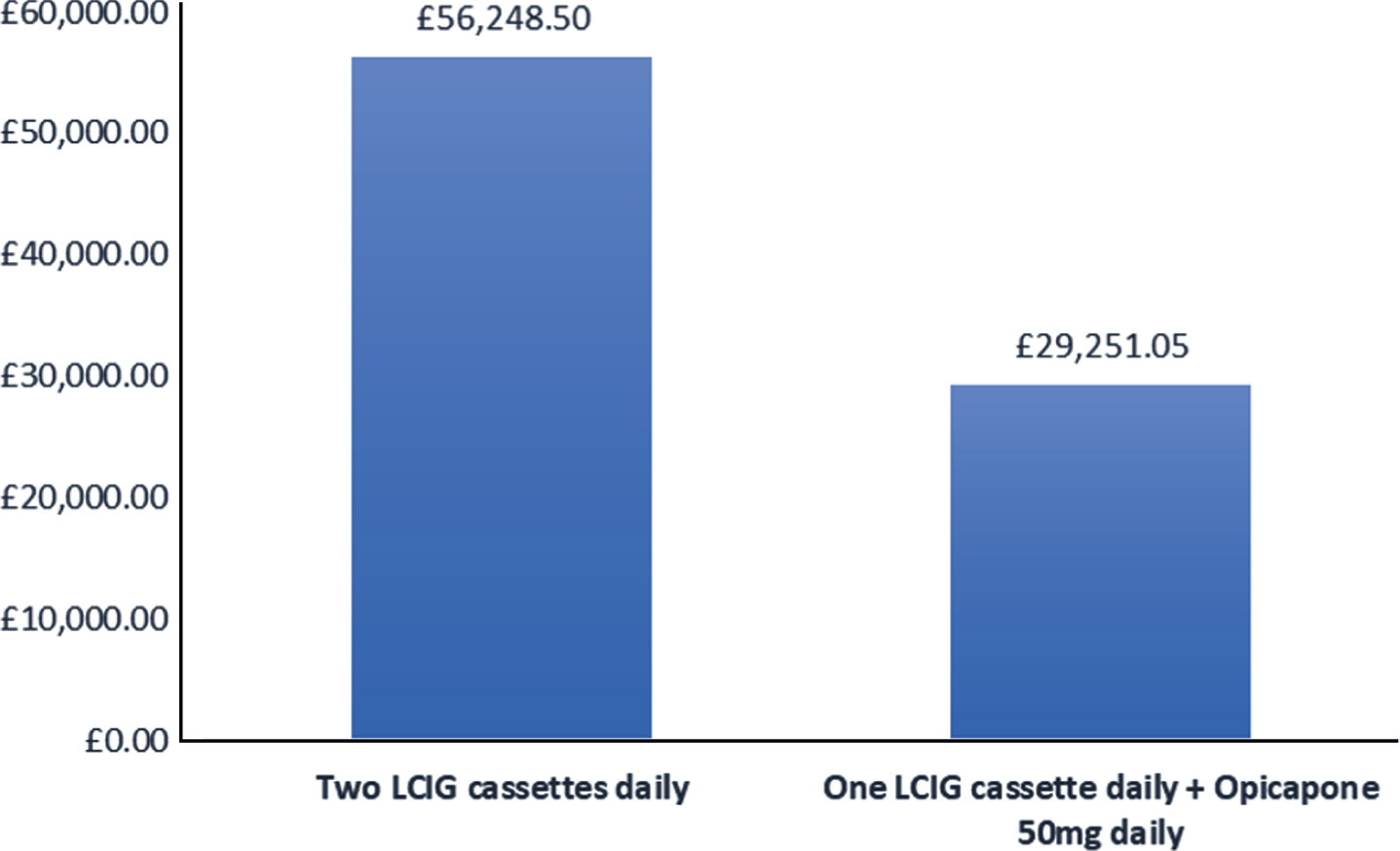
Example of yearly costs per Parkinson’s disease patient in the United Kingdom on two cassettes of Levodopa-carbidopa intestinal gel and costs after the introduction of Opicapone. LCIG: levodopa-carbidopa intestinal gel.
On a national level this could result in gross savings of £520,298.63 per year (5.2% reduction in annual costs for LCIG infusion therapy) in the UK. The costs associated with introducing all patients on LCIG infusion on Opicapone would amount to £377,478.00 with subsequent net savings per year of £142,820.63 (1.4% reduction in annual costs for LCIG infusion therapy). Clinically the introduction of Opicapone in this small series did not result in a worsening of dyskinesia as also measured by a wearable sensor. As such, the combination of these two drugs deserves further research for long-term tolerability and safety.
Based on our findings, further cost savings could also be achieved by considering smaller cassette sizes for LCIG infusion. When taking into account that 57% of patients on LCIG infusion in our cohort after the introduction of Opicapone used 50 ml or less of LCIG a day, one could reason that these patients would no longer require one cassette a day, but instead could manage with half a cassette, provided that the cassettes could be produced in half size. These results set the scene for the commercial use of LCIG with COMT-inhibition, as is now licenced in Sweden (intrajejunal Levodopa-Carbidopa-Entacapone gel infusion, Lecigon) [9] or an add-on COMT-inhibitor within centres already performing standard LCIG infusion therapy. Further longitudinal follow-up is needed to monitor the effect of this therapeutic strategy, although we are encouraged to see no significant worsening of dyskinesia. Additional investigations are also required to determine how oral long-acting add-on COMT-inhibition may improve night-time symptoms of PD as has been shown with drugs, such as rotigotine transdermal patch, which provides sustained dopaminergic stimulation overnight [15].
In the current analysis we used Opicapone due to its 24-hour activity [7, 8], but a similar strategy may also involve other COMT-inhibitors, such as Entacapone or Tolcapone [16]; however, given the relatively short half-life of the latter two COMT-inhibitors, these would need to be taken more than once a day, which could be difficult for PD patients in an advanced stage of the disease with concomitant dysphagia [17]. Furthermore, a single daily dose could increase patient’s adherence to the treatment [18]. In addition, Tolcapone use is clinically restricted in many countries and requires regular liver function monitoring. Interestingly, a combined infusion of levodopa carbidopa with entacapone has been developed in Sweden and is now licensed in some European countries as Lecigon. Two out of the 11 patients in our cohort stopped the intake of Opicapone because of hallucinations or dizziness. These side effects are commonly reported in patients recruited for clinical trials assessing the efficacy of Opicapone as an add-on to Levodopa therapy [10, 19]. On the other hand, dyskinesia, which is commonly reported as a side effect in these clinical trials, did not significantly increase in our cohort of patients. This was probably due to the concurrent reduction in LCIG daily dose which allowed retention of similar levodopa plasma levels.
Limitations of this report include a small sample size and retrospective data collection rather than a specific prospective study. As such, our findings need to be confirmed in larger and preferably randomised studies. Despite this, we feel that our results are meaningful, given we were still able to show a significant reduction in LCIG daily dose without a worsening of motor symptoms in a small cohort, representative of a real-life scenario.
In summary, COMT-inhibition, and in this case Opicapone, in combination with LCIG infusion is a promising therapeutic strategy with potential to reduce the costs associated with this advanced therapy and increase the effectiveness of LCIG treatment. Future studies with larger cohorts and a randomised design should focus on replicating the results of this observation.
CONFLICT OF INTEREST
Valentina Leta reports grants from BRC, Parkinson’s UK, a travel and congress grant from Bial UK Ltd, speaker-related activities fees from Britannia pharmaceuticals, and consultancy fees from Invisio Pharmaceuticals, outside the submitted work. Daniel J. van Wamelen reports grants and speaker-related activities fees from Britannia pharmaceuticals, and consultancy fees from Invisio Pharmaceuticals, outside the submitted work. Anna Sauerbier reports grants from Parkinson’s UK and Kirby Laing, and consultancy and speaker-related fees from Britannia, UCB and Bial outside the submitted work. Shelley Jones has nothing to disclose. Miriam Parry reports speaker-related activities fees and travels grant from Britannia pharmaceuticals, Bial UK Ltd and AbbVie, outside the submitted work. Alexandra Rizos reports speaker-related activities fees from Britannia pharmaceuticals and salary funding from the NIHR Clinical Research Network South London, outside the submitted work. K. Ray Chaudhuri reports personal fees from AbbVie, Britannia Pharmaceuticals, UCB, Mundipharma, Zambon, Global Kinetics, Bial; grants from Parkinson’s UK, NIHR, PDNMG, Kirby Laing, NPF; other from AbbVie, UCB, Sunovion, Pfizer, Jazz Pharma, Bial, Global Kinetics, outside the submitted work.
Footnotes
ACKNOWLEDGMENTS
We acknowledge data collection efforts by all contributors of the NILS. We acknowledge Prof P. Odin, Prof A. Storch, Prof Z. Pirtošek, and Prof A. Antonini for their advice. The views expressed are those of the authors and not necessarily those of the NHS, NIHR or Department of Health. This article represents independent collaborative research part funded by the NIHR Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. Rachel Parratt, PhD (Cello Health Med Ergy), funded by Bial Pharma UK Ltd, participated in writing the first draft of the manuscript. Bial UK funded the publication costs. Bial UK Ltd neither funded nor had any control of the research project, collection and analysis of data here described. Bial UK did not have access to any subject level data. The manuscript was produced by the authors and collaborators and, therefore, represents entirely their personal view without any input by Bial UK Ltd.
