Abstract
There is growing recognition in the field of neurodegenerative diseases that mixed proteinopathies are occurring at greater frequency than originally thought. This is particularly true for three amyloid proteins defining most of these neurological disorders, amyloid-beta (Aβ), tau, and alpha-synuclein (
INTRODUCTION
Neurodegenerative disorders including Alzheimer’s disease (AD), Parkinson’s disease (PD), dementia with Lewy body (DLB), multiple system atrophy (MSA), and Huntington’s disease (HD) are characterized by progressive dysfunction and death of neurons. Within each disease type, this degeneration affects specific neural systems implying selective vulnerability and is associated with the abnormal accumulation of distinct proteins, e.g., amyloid-beta (Aβ), tau, and alpha-synuclein (
Recent studies are however providing new evidence for prevalent mixed proteinopathies across these neurodegenerative diseases, with aging and apolipoprotein E (ApoE)
The co-existence of these proteinopathies provides additional support to accumulating observations documenting molecular interactions and cross-seeding between aggregates of Aβ, tau, Htt and
Beyond a potential contribution of
Despite this greater understanding, many fundamental questions remain unanswered: 1) which endogenous forms of
α SYN FUNCTION AND STRUCTURE
In the past 5–7 years, growing evidence indicates that
In search of a mechanism for how exactly
Beyond neurons, larger amounts of
Structure-function relationships of α Syn
Lipid binding
Consistent with its proposed function(s),
Multimeric vs. oligomeric α Syn
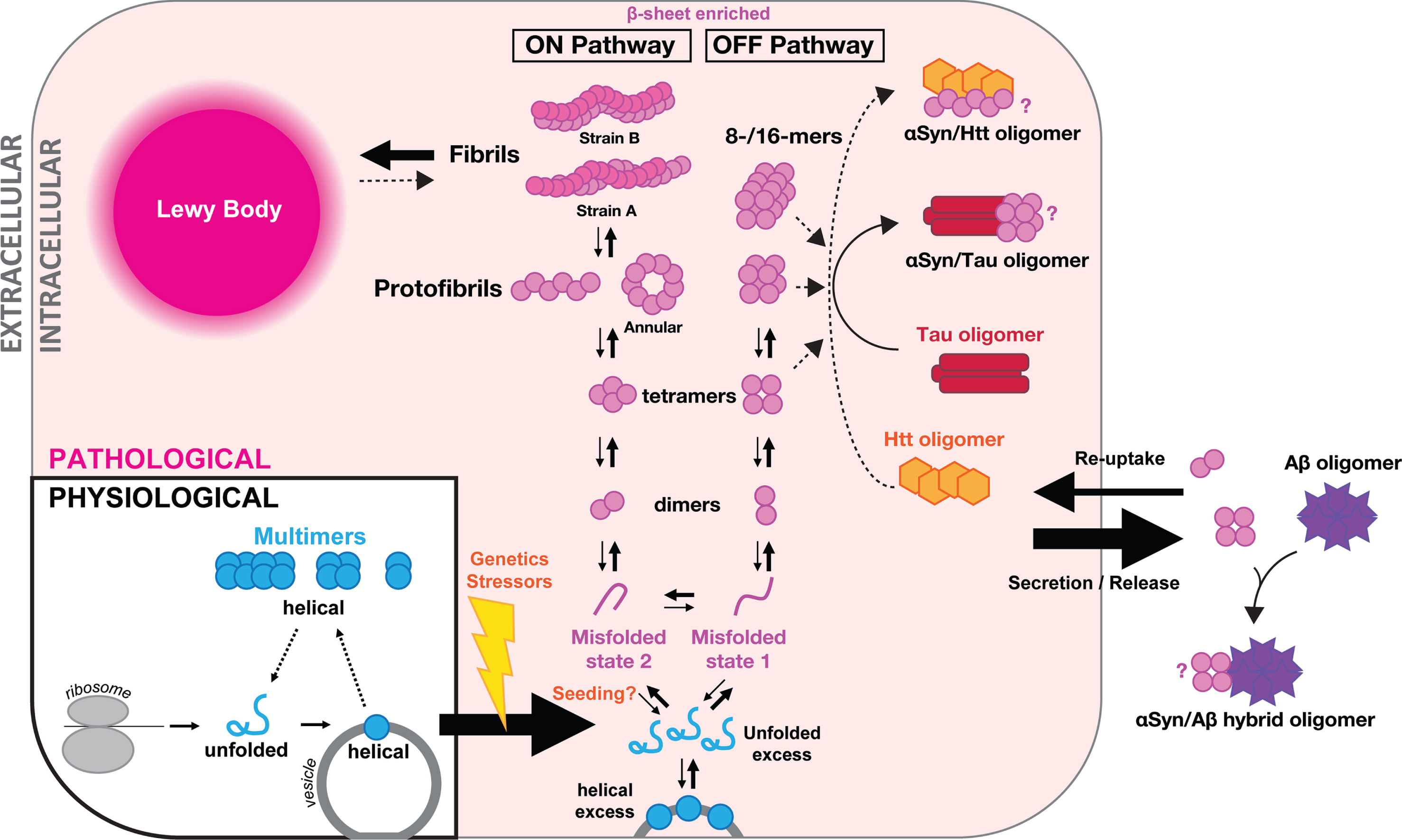
Proposed model of
Given the complexity of purification, intact-cell methods became increasingly important. A live-cell crosslinking study provided a large variety of controls to suggest abundant physiological, not pathological
A key concept to understanding
The list of relevant publications on the topic of native
All in all, there is accumulating evidence from multiple independent laboratories to support the existence of native
ENDOGENOUS OLIGOMERIC α Syn ASSEMBLIES IN NEURODEGENERATIVE DISEASES
Endogenous oligomeric α Syn assemblies in neurodegenerative diseases
Detection of oligomeric α Syn species by disease
All known
In mice, a transgenic animal model expressing mutant E46K
In both systems, it thus remains clear that additional studies are needed to fully assess the nature of o-
Functionally, soluble
Contrasting with the paucity of studies which used postmortem brain tissue to detect and measure
Despite these issues, a few studies have measured oligomeric
Detection of oligomeric α Syn species by selective brain areas
Neurodegenerative diseases are also defined by the neurodegeneration of distinct brain regions or nuclei, implying a selective vulnerability of neuronal subtypes to the accumulating amyloid protein [1]. Across the studies aforementioned above which have measured o-
Roberts and colleagues detected
In their analyses of AD brain tissues, Larson and coworkers also detected o-
Lastly, the Lucas group observed
Overall, extensive work remains to be done to determine the existence, expression, accumulation and potential spreading of o-
Current understanding of oligomeric α Syn production and toxicity in vivo
Dimers
Studies by several groups showed that chemical or photochemical crosslinking of
Similarly, analysis of protein lysates from human brain tissue also yield equivalent electrophoretic profiles [21, 125]. Amongst those reports, Larson and colleagues readily detected the presence of apparent 35 kDa
Functionally, multivariate regression analyses revealed inferior temporal gyrus levels of putative 28- and 35 kDa
Trimers
Rare studies have documented the existence of putative
Tetramers
Contrasting with the anecdotal reports related to possible
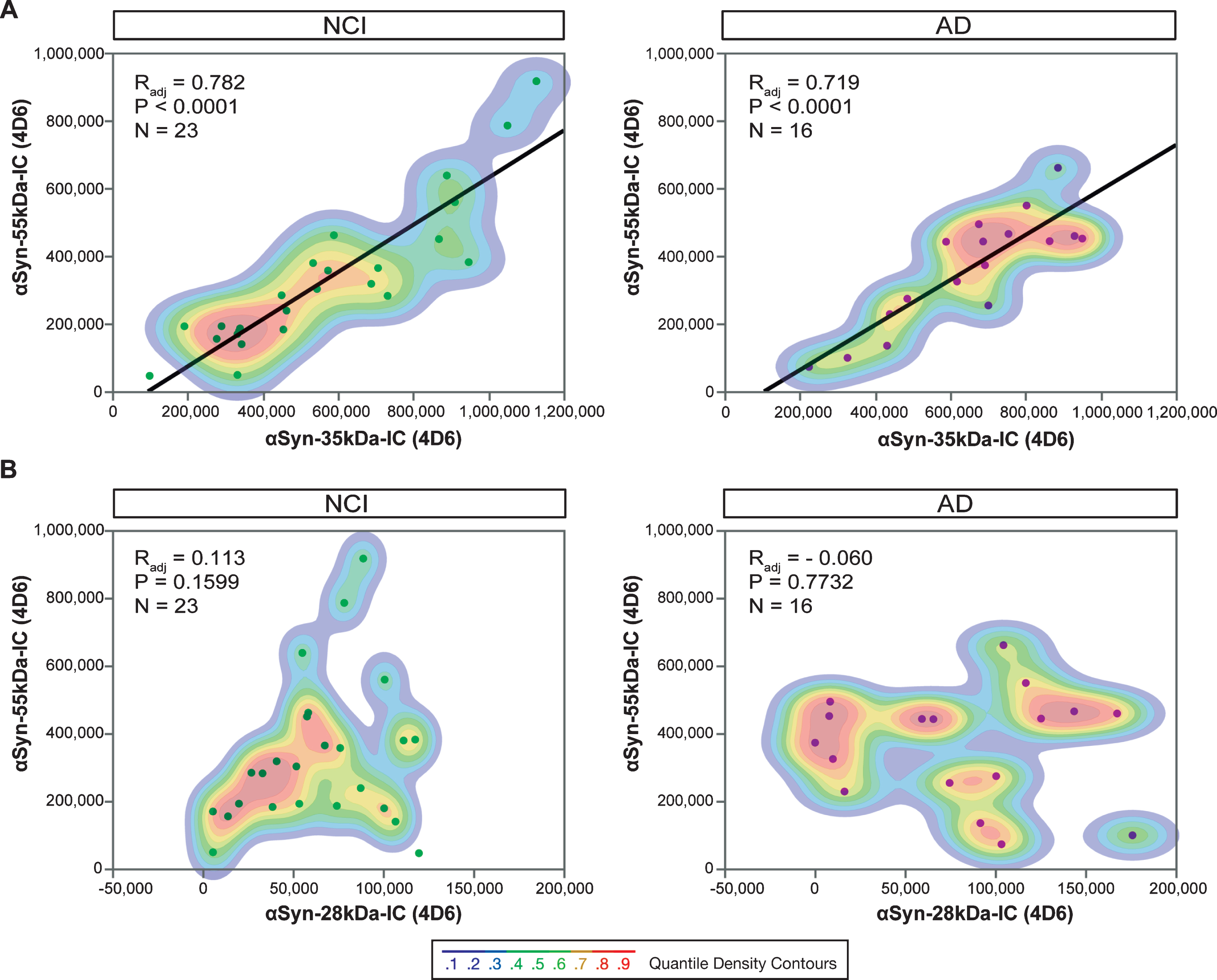
Relationships between 28-, 35- and 55 kDa
Beyond the differences in their respective intrinsic biochemical properties, the SDS-sensitive ∼58–60 kDa
Annular protofibrils
Earlier it was shown that A30P and A53T mutants can form pore-like annular protofibrils faster than
Other assemblies
Large oligomers of
Using newly generated transgenic mice expressing split luciferase
Future directions
As summarized above, many forms of putative oligomeric assemblies of
Post-translational modifications including truncation [139], nitration [140] and phosphorylation [141, 142] might play important roles in
If achieved, these largely technical improvements should allow the field to compare the function of multiple
Lastly, two very recent studies from the Holtzman and Bu groups investigated whether
SPREADING OF OLIGOMERIC α SYN SPECIES
Introduction to prion-like propagation of α Syn
A group of highly infectious neurodegenerative diseases, known as transmissible spongiform encephalopathy (TSE) or prion diseases is caused by the infectious prion protein or PrP [147]. Animals affected with prion diseases exhibit different phenotypes. One of the most astonishing phenomena in prion diseases is that animals infected with the same pathogenic infectious agent displayed different clinical phenotypes [148–150]. Moreover, when such infectious protein aggregates were isolated and inoculated in identical hosts, they induced distinct prion disease specific phenotypes [151]. Growing experimental evidence suggests that many neurodegenerative diseases share common characteristics with prion diseases. Indeed, the pathogenic protein conformers of amyloid proteins defining neurodegenerative diseases can seed and promote the aggregation of their natively-folded counterparts in a prion-like manner [152].
Based on the course of the Lewy pathology in the enteric nervous system (ENS), peripheral nervous system (PNS) and central nervous system (CNS) together with clinical symptoms of patients with sporadic PD, Del Tredici and Braak have proposed the following staging procedure [153]. At stage 1, the Lewy pathology initially develops somewhere in ENS, PNS or CNS, then is detected in olfactory structures (bulb and nucleus) and in the medulla oblongata, specifically in the dorsal motor nucleus of the vagal nerve (CN X) and/or intermediate reticular zone. At stage 2, LNs and LBs are found in peripheral parasympathetic and sympathetic nerves, medullary nuclei including the locus coeruleus and lower raphe nuclei. Stage 3 is characterized by
In the late 2000s, several reports documented host-to-graft propagation of
Spreading of endogenous oligomeric α Syn species in vitro
Although the exact mechanism by which o-
Spreading of endogenous oligomeric α Syn species in vivo
To our knowledge, experimental evidence demonstrating a spreading of endogenous o-
INTERACTIONS BETWEEN OLIGOMERIC α SYN ASSEMBLIES AND OTHER AMYLOID PROTEINS
Although amyloidogenic proteins might follow unique cascades of pathological events, the majority of them shares certain common mechanisms for toxicity. Membrane permeabilization is one of such mechanisms that has been suggested for many amyloid proteins [170]. Co-occurrence of multiple protein pathologies represents a wide range of neurodegenerative disorders where comorbidity and overlap between the diseases are frequently observed [171]. In addition to LB, senile plaques of Aβ, neurofibrillary tangles (NFTs) and neuropil threads composed of tau were observed in the cerebral cortex of PD brain tissues [172]. Senile plaques and NFTs are often found together in DLB cases [173]. Thus, there is a considerable overlap between
For the context of this review, we will differentiate the terms “cross-talk” as defined by direct or indirect molecular interactions in absence of enhanced fibrillization from “cross-seeding” which occurs when oligomers composed by one misfolded amyloid protein induce the oligomerization of a distinct amyloid protein.
α Syn/Aβ hybrid species
Cross-talk
While many studies have suggested that
To assess whether Aβ and
α Syn/tau species
While
There is also accumulating evidence that spreading of pathogenic protein aggregates is a necessary event in the progression of neurodegenerative diseases [190–193]. To this end, studies have also been directed to dissecting out the mechanisms by which amyloidogenic protein aggregates spread from cell to cell [190, 191]. Walker et al. have suggested a templating mechanism of misfolded protein in which once a small amount of aggregate is formed, it serves as “seed” for further aggregation. Such seeds can recruit newly-formed protein by templating their conformation to the newly added proteins [194], including
Cross-talk
Using human brain cytosol from unspecified source and characterization, early studies reported that
However, these seminal studies focused on fibril formation as opposed to oligomerization per se. Consequently, the functional role of oligomeric
Furthermore, the role of tau in mediating impairment of hippocampal neurotransmission and memory deficits in a similar transgenic animal model, TgA53T mice overexpressing mutant A53T human
Combined, these findings thus suggest the existence of hybrid o-
Beyond a potentially direct effect of
Overall, these studies strongly support the importance of functional crosstalk between
Cross-seeding
As overlapping protein pathologies are frequently observed in multiple neurodegenerative diseases, cross-seeding between disparate proteins has been suggested as a possible mechanism underlying the detection of mixed pathologies in the same brain tissue [207, 208]. While it has been proposed that both tau and
In more recent studies however, the bilateral intracerebroventricular injection of o-
Together, these
α Syn/HTT species
Cross-talk
As discussed earlier, an aberrant interaction of mutant Htt with
Overall, further investigation is needed to better understand the molecular inter-relationship between
OLIGOMERIC α SYN SPECIES AS DRUG TARGET(S)
Monotherapy
With the discovery of cell-to-cell spreading of
Molecules that can modulate the aggregation process by decreasing the load of
Multi-therapy with other amyloid proteins
Most neurodegenerative diseases, including synucleinopathies, encompass various protein pathologies. Passive TOMA immunotherapy targeting tau oligomers in the M83 Tg53T animal model of PD prevents cognitive and motor deficits as well as brain protein pathology [204]. The importance of synergistic effects of
CONCLUSIONS
Aggregation of
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by grants from the National Institutes of Health (NIH) to SEL (RF1-AG044342, R21-AG065693, R01-NS092918, R01-AG062135 and R56-NS113549), to RK (R01-AG054025, R01-NS094557 and RF1-AG055771) and to UD (R01-NS099328). Additional support included start-up funds from the University of Minnesota Foundation and bridge funds from the Institute of Translational Neuroscience to SEL, grants from Gilson Longenbaugh Foundation and Mitchell Center for Neurodegenerative Diseases to RK. We thank the study participants and staff of the Rush Alzheimer’s Disease Center and its director Dr. David Bennett at Rush University.
