Abstract
Background:
Dopaminergic medications in Parkinson’s disease (PD) are usually associated with the development of both levodopa-induced dyskinesias (LID) and impulse control and repetitive behavior disorders (ICRB).
Objective:
To assess the prevalence and the severity of ICRB in a cohort of moderate and advanced PD patients and to investigate the potential interplay between ICRB, LID and dopaminergic therapies.
Methods:
117 PD patients were consecutively recruited. LID were assessed by using the Rush Dyskinesia Rating Scale (RDRS). ICRB were tested by means of Questionnaire for Impulsive Compulsive Disorders in Parkinson’s Disease Rating Scale (QUIP-RS).
Results:
55 patients were affected by LID. Among them, 37 were treated only by oral therapy, OT (LID/OT), while 18 were on treatment with jejunal levodopa infusion, JLI (LID/JLI). 62 patients were not affected by LID (NLID) and all of them were on therapy only with oral drugs. The overall prevalence of clinically significant ICRB was 34% (95% CI = 26% to 43%) and the mean value (±SD) of QUIP-RS total score was 5.4±8.5. Prevalence of clinically significant ICRB, as well as severity of ICRB, was higher in patients with LID compared to NLID patients (p = 0.016 and p < 0.001, respectively). When considering LID/JLI, LID/OT and NLID groups, QUIP-RS total score was significantly higher in LID/OT patients compared to LID/JLI (10.4±11.8 vs. 4.9±6.0, p = 0.019) and NLID (10.4±11.8 vs. 2.5±4.8, p < 0.001) groups.
Conclusion:
PD patients with LID show ICRB more frequently and more severely than patients without LID. Among LID patients, those treated by JLI showed a lower severity of ICRB than those on OT, suggesting a potential protective effect of JLI on ICRB.
Keywords
INTRODUCTION
Dopaminergic replacement therapy is the mainstay of treatment in Parkinson’s disease (PD). Particularly, levodopa is considered the most effective drug to alleviate motor symptoms [1]. However, chronic use of dopaminergic medications is often associated with the development of both motor and behavioral complications, mainly represented by levodopa-induced dyskinesias (LID) and impulse control and repetitive behavior disorders (ICRB), respectively [2].
Pulsatile administration of short-acting dopaminergic drugs such as levodopa, higher cumulative dose and duration of levodopa treatment are reported as strong contributors for LID development [3]. Long half-life dopaminergic agonists seem to have a lower dyskinesiogenic power [4]. Meanwhile, dopaminergic agonists, particularly D2-R and D3-R long-acting drugs, are more involved in the development of ICRB, with associated levodopa treatment further increasing the risk of developing behavior complications [5]. IRCB are a group of neuropsychiatric disturbances which can be defined as the failure to resist a temptation to perform an act which is harmful to either the self or others. They include: 1) impulse control disorders (ICD): gambling, hypersexuality, binge eating and compulsive shopping, 2) repetitive behaviors (RB): punding and hobbism and 3) dopaminergic medication overuse which is strongly related to a condition known as dopaminergic dysregulation syndrome [6]. Different studies found an increasing risk of LID and IRCBs co-occurrence in patients with PD [7, 8], suggesting that motor and behavior complications of dopaminergic replacement therapy might be associated with a common susceptibility.
In this study, we aimed to assess the prevalence and the severity of ICRB in a cohort of moderate and advanced PD patients and to investigate the potential interplay between ICRB, LID and dopaminergic therapy.
METHODS
Patients
This cross-sectional study involved a total number of 117 moderate and advanced PD patients. Patients were recruited in the Movement Disorders Centre of the University Hospital of Perugia from 2016 to 2018. All the enrolled patients met the Movement Disorders Society (MDS) Diagnostic Criteria for clinically established PD [9]. Inclusion criteria for this study were disease duration higher than 5 years and Hoehn & Yahr (H&Y) stage ≥2. Exclusion criteria were diagnosis of atypical parkinsonism, previous or current treatment with Deep Brain Stimulation (DBS) as well as psychiatric comorbidities and dementia, according to the Diagnostic and Statistical Manual fifth edition (DSM-V) criteria [10]. At the time of recruitment, patients were on oral therapy with Levodopa (oral therapy, OT and/or jejunal levodopa infusion, JLI) and/or dopamine agonists, sometimes associated with monoamine oxidase inhibitors, catechol-O-methyltransferase inhibitors and amantadine. The study was reviewed and approved by the local ethic committee (CEAS Umbria).
Clinical assessment
For each patient demographic information including age, gender and disease duration were accurately collected. Data about therapy were recorded, as well, and total Levodopa Equivalent Daily Dose (LEDD) was calculated according to Tomlinson et al. [11]. At the time of enrollment, patients underwent a comprehensive assessment including motor and behavior aspects. Motor impairment and disease-related disability were evaluated by means of MDS-Unified Parkinson’s Disease Rating Scale part III (MDS-UPDRS-III) [12] and Hoehn & Yahr (H&Y) Rating Scale [13], respectively. LID were tested by using Rush Dyskinesia Rating Scale (RDRS) [14]. ICRB assessment was performed by means of Questionnaire for Impulsive Compulsive Disorders in Parkinson Disease Rating Scale (QUIP-RS) [15], including the following items: gambling, hypersexuality, compulsive shopping, binge eating, compulsive hobbies, repeating simple activities (punding) and dopaminergic medication overuse. We considered the QUIP-RS cut-off values according to Weintraub et al. [15] (gambling ≥6; hypersexuality ≥8; shopping ≥8; eating ≥7; punding ≥7; hobbyism ≥7) and the cut-off value ≥7 for medication overuse as proposed by Biundo et al. [16], in order to define the prevalence of ICRB and to distinguish patients with clinically significant ICRB (values above the cut-off) from those with milder symptoms (values below the cut-off).
Statistical analysis
Descriptive statistics were calculated. Continuous variables were summarized as means and standard deviations while categorical data were reported as numbers and percentages. The distribution of continuous variables was tested for normality with Shapiro Wilk test. Between groups comparison were performed by means of Student’s T-test accounting for the multiplicity with Benjiamini-Hochberg procedure. Pearson’s correlation coefficients between clinical parameters were calculated. Whenever appropriate, non-parametric alternatives have been considered. All the comparisons between groups were carried out adjusting for disease duration as potential confounder. The statistical analysis was carried out using R software (http://www.r-project.org), with statistical significance level set at 5%.
RESULTS
Demographic and motor characteristics
We found that 62 out of 117 recruited patients were not affected by LID (NLID), and all of them were on OT. None of the NLID patients were treated by JLI. Meanwhile, 55 patients of our cohort suffered from LID. Among them, 18 were on therapy with JLI (LID/JLI), whereas the remaining part (37 patients) were only on OT (LID/OT).
Results about demographic characteristics (age, gender, disease duration), motor features (H&Y, MDS-UPDRS-III, RDRS scores) and therapy (LEDD) are reported for the whole cohort and for each of the three groups (LID/JLI, LID/OT and NLID) in Table 1. Disease duration and LEDD were significantly higher in LID/JLI patients compared to the other two groups (p < 0.001). The other features were similar across the groups and further significant differences were not found.
Data about demographic features, motor characteristics and therapy in the whole cohort and in each of the three groups (LID/JLI, LID/OT and NLID) of Parkinson’s disease patients. Measures are expressed by means of average (±standard deviation) or number (%)
LID/JLI, dyskinetic patients on jejunal levodopa infusion; LID/OT, dyskinetic patients on oral therapy; NLID, non-dyskinetic patients; DA, dopamine-agonist; H&Y, Hoehn & Yahr rating scale; ICRB, impulse control and repetitive behavior disorders; LEDD, levodopa equivalent daily dose; JLI, jejunal levodopa infusion; LID, Levodopa-induced dyskinesia; MDS-UPDRS-III, Movement Disorders Society-unified Parkinson’s disease rating scale-part III; OT, oral therapy; RDRS, Rush dyskinesia rating scale.
Prevalence, severity of ICRB and correlation with LID and dopaminergic therapies
The overall prevalence of ICRB was 41% (95% CI = 32% to 50%), whereas 34% (95% CI = 26% to 43%) of the whole cohort had clinically significant ICRB (with scores above the cut-off values in at least one QUIP-RS item). Punding was the most common disorder with a prevalence of 12% (95% CI = 7% to 19%), whereas compulsive shopping was the least frequent with a prevalence of 4% (95% CI = 2% to 10%). Prevalence of clinically significant ICRB was higher in patients with LID compared to NLID patients (47% vs. 23%, p = 0.016). Eating (18% vs. 5%, p = 0.028) and medication overuse (15% vs. 2%, p = 0.012) were found to be significantly more frequent in patients with LID than in NLID patients. Patients with LID showed multiple ICRB more frequently than NLID patients (22% vs. 3%, p = 0.005), as well. The mean value (±SD) of QUIP-RS total score was 5.4±8.5. Severity of ICRB, expressed by QUIP-RS total score, was significantly higher in patients affected by LID than in NLID patients (8.6±10.5 vs. 2.5±4.8, p < 0.001). Severity of binge eating and medication overuse, expressed by QUIP-RS score for each of the two items, was significantly higher in LID patients than in NLID patients (binge eating: 1.8±3.6 vs. 0.4±1.7, p = 0.010; medication overuse: 1.7±3.5 vs. 0.2±1.2, p < 0.001). Further differences in terms of severity were not found between LID and NLID groups.
When considering the three different groups (LID/JLI, LID/OT, NLID), we found that prevalence of clinically significant ICRB was higher in LID/OT group compared to LID/JLI (57% vs 28%, p = 0.058) and NLID patients (57% vs 23%, p = 0.008), and comparable between LID/JLI and NLID groups (28% vs. 23%, p = 0.895). Table 2 shows the prevalence of clinically significant ICRB and the number of patients showing each disorder with scores above the cut-off values for each of the three groups. QUIP-RS total score was significantly higher in LID/OT patients compared to LID/JLI (10.4±11.8 vs. 4.9±6.0, p = 0.019) and NLID (10.4±11.8 vs. 2.5±4.8, p < 0.001) groups, as shown in Figure 1. Severity of ICRB, as expressed by QUIP-RS score for each item, is represented for each of the three groups in Figure 2. In particular, medication overuse score was significantly higher in LID/OT patients compared to LID/JLI (2.6±4.0 vs. 0.2±0.9, p = 0.004) and NLID (2.6±4.0 vs. 0.2±1.2, p < 0.001) groups. As much, binge eating score was significantly higher in LID/OT patients compared to LID/JLI (2.4±4.0 vs. 0.6±1.9, p = 0.038) and NLID (2.4±4.0 vs. 0.4±1.7, p = 0.004) groups. In the Supplementary Material, we reported further details on the analysis for each item.
Prevalence (%) of clinically significant ICRB and number of patients showing each disorder (with scores above the cut-off values) for the whole cohort and for each of the three groups of Parkinson’s disease patients (LID/JLI, LID/OT, NLID)
LID/JLI, dyskinetic patients on jejunal levodopa infusion; LID/OT, dyskinetic patients on oral therapy; NLID: non-dyskinetic patients; ICRB, impulse control and repetitive behavior disorders; JLI, jejunal levodopa infusion; LID, Levodopa-induced dyskinesia; OT, oral therapy.
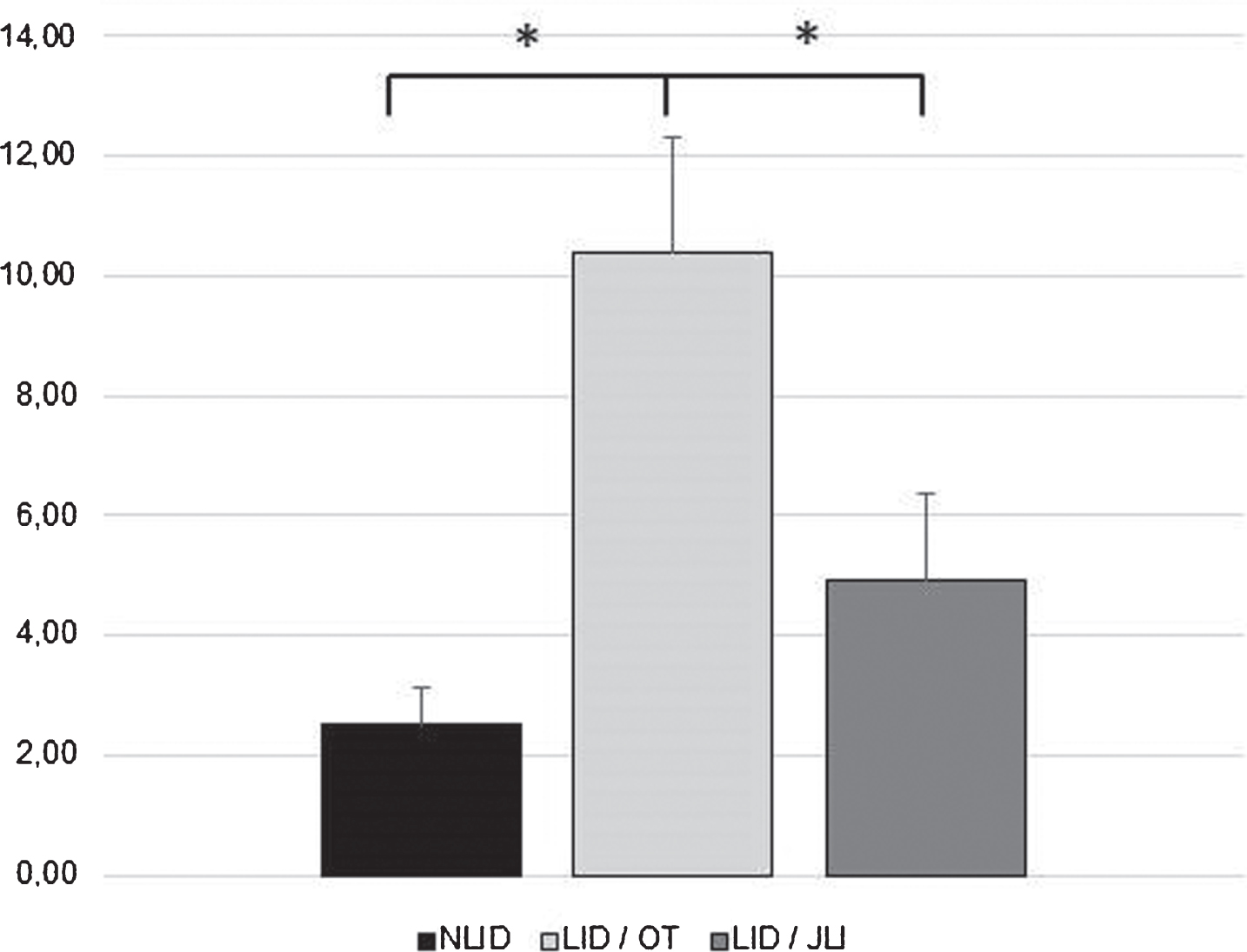
Severity of ICRB, expressed by QUIP-RS total score, in NLID, LID/OT and LID/JLI groups (*significant difference). NLID, non-dyskinetic patients; LID/OT, dyskinetic patients on oral therapy; LID/JLI, dyskinetic patients on jejunal levodopa infusion.
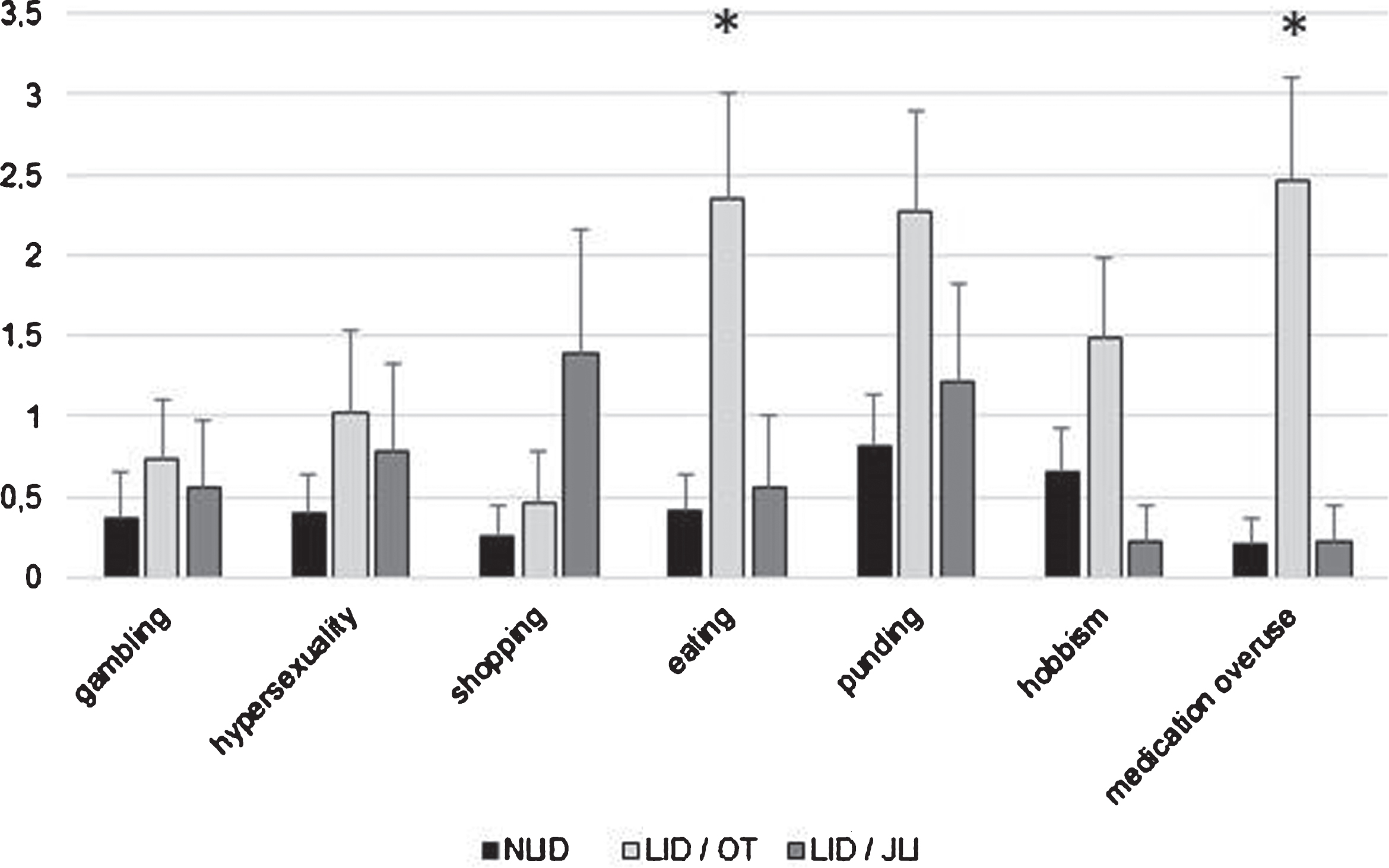
Severity of different ICRB, expressed by QUIP-RS score for each item, in NLID, LID/OT and LID/JLI groups (*significant difference in the QUIP-RS score between LID/OT and the other two groups). NLID, non-dyskinetic patients; LID/OT, dyskinetic patients on oral therapy; LID/JLI, dyskinetic patients on jejunal levodopa infusion.
DISCUSSION
This is the first cross-sectional study aimed at exploring the prevalence and severity of ICRB in a cohort of PD patients in which advanced patients treated by JLI were considered.
We found that overall prevalence of clinically significant ICRB was 34%, greatly higher than frequency (13.6%) reported in the largest multi-center study assessing ICD in a cohort of PD patients (see the DOMINION study) [5]. Our prevalence was also higher than frequency found in more recent studies including PD patients treated by dopamine agonists [17, 18]. Different hypothesis can be considered to explain such differences in terms of prevalence. First, it is probably due to the longer disease duration of our patients compared to that of previous works, since cumulative levodopa time exposure can influence the risk of ICRB development. Furthermore, it could depend also on different scales to assess ICRB. Compared to QUIP-RS, Minnesota Impulsivity Disorders Interview [19] and Massachusetts Gambling Screen [20] of the DOMINION are not specifically validated for PD, probably resulting less reliable to identify subtle behavioral symptoms in PD. Moreover, RB (punding and hobbism) and dopaminergic medication overuse had not been considered in the DOMINION. Conversely, we investigated not only ICD, but also RB and dopaminergic medication overuse and we found that punding was the most frequent disorder, thus impacting greatly on the overall prevalence of ICRB. As much, our prevalence of clinically significant ICRB was similar to that of the recent ALTHEA study (36%) in which a population of dyskinetic PD patients was tested for ICRB by means of QUIP-RS [16]. Even when ICRB were assessed by using the self-administered QUIP scale in a cohort of elderly PD patents, the prevalence of ICRB was higher than 40% [21].
In the DOMINION study [5] both dopamine agonists and Levodopa were independently associated with ICRB, with a stronger correlation with dopamine agonists compared to levodopa. However, the association was dose-dependent for Levodopa, but not for dopamine agonists. Otherwise, we found no correlation between ICRB and LEDD in our cohort. Indeed, dyskinetic patients on JLI had higher LEDD, simultaneously showing lower QUIP-RS total score, as compared to dyskinetic patients on OT. Similarly, the ALTHEA study did not reveal any correlations between QUIP-RS total score and LEDD in patients with multiple ICRB [16].
Focusing on relationship between ICRB and LID, our results strengthen the idea that LID and ICRB are strictly related in PD patients. We found that patients affected by LID not only showed ICRB more frequently but also exhibited higher scores at QUIP-RS as compared to patients without LID. Previously, Voon et al. assessed factors associated with ICD in a retrospective case-control study based on the results from the DOMINION. They found that patients with multiple ICD showed greater LID scores (UPDRS items 32-33) than patients with single ICD [22]. Conversely, Biundo et al. showed no significant differences in terms of LID severity between patients with and without ICRB in the ALTHEA study. They found that patients with multiple ICRB showed higher Unified Dyskinesia Rating Scale (UDyRS) scores than patients without ICRB, but this difference was related to the historical scores of off dystonia [16]. However, it should be considered that the aim of the ALTHEA study was to assess ICRB in a cohort of PD patients with different degrees of LID severity without considering patients without LID, differently from our study. Overall, our results support the view that LID and ICRB are pathophysiologically linked and share common molecular substrates. Particularly, previous imaging studies showed decreased midbrain D2-R and D3-R autoreceptors levels with higher striatal dopamine release during a gambling task in PD patients with ICRB than in patients without ICRB [23]. Furthermore, lower dopamine transporters levels were found in the ventral striatum with subsequent greater synaptic dopamine accumulation in PD patients with ICRB compared to those without ICRB [24]. Meanwhile, it is known that an identical dose of levodopa induces higher synaptic dopamine concentrations in the dorsal striatum of PD patients with LID compared to those without LID [25]. Thus, it seems that striatal dopaminergic overstimulation is a common feature of ICRB and LID, even if with a different topographic distribution (ventral versus dorsal striatum, respectively). A series of neurotransmitter systems other than dopaminergic, especially glutamatergic and serotoninergic, might play a role in the development of both LID and ICRB as consequence of repetitive dopaminergic stimulation [2, 24]. When we considered different types of ICRB, we found that binge eating and medication overuse were both more frequent and more severe in patients with LID compared to those without LID. Previously, a strong relationship between specific types of ICRB and LID was reported for punding, which was associated with both greater dyskinesia severity and more frequent interventions to reduce dyskinesia in a cohort of 45 PD patients [27].
Of more interest, we found that severity of ICRB was significantly higher in dyskinetic patients on OT than in dyskinetic patients on JLI and non-dyskinetic patients. Dyskinetic patients on JLI were affected by ICRB to a similar level of patients without dyskinesia and less severely than dyskinetic patients treated by only OT. Prevalence of ICRB showed a similar trend among three different groups, with higher value in dyskinetic patients on OT vs. non-dyskinetic patients, and in dyskinetic patients on OT vs. dyskinetic patients on JLI, however without reaching a significant difference in the latter comparison. It seems to be ascertained that JLI is accompanied by positive effects on motor complications, resulting in a decreasing off time and increasing on time without troublesome LID [28–30]. Meanwhile, influence of JLI on behavioral disturbances is still a matter of debate. Chang and colleagues identified ICRB in 27% (4/15) advanced-PD patients at a mean of 8.5 months after commencement of JLI, reporting ICRB as one of the main adverse events of JLI [31]. However, findings resulting from different prospective studies suggested a beneficial effect of JLI on behavioral disorders. Catalán and colleagues assessed the effects of JLI on behavioral symptoms in 8 PD patients with severe impulsivity and dopamine dysregulation syndrome (DDS), finding that behavioral symptoms dramatically improved with nearly complete resolution after JLI commencement [32]. Particularly, it seems that JLI is associated with longer lasting resolution of DDS when compared to other advanced treatments such as apomorphine and DBS [33]. The multi-centric open-label study EuroInf revealed that JLI led to the resolution of ICRB in all the recruited patients with pre-existing ICRB without observing new cases of behavioral disorders [34]. More recently, a multi-center prospective study focusing on advanced PD patients on JLI showed a significant reduction of QUIP-RS score (64.4%) 6 months after JLI commencement [35]. In line with our results, findings deriving from previous longitudinal studies suggest that continuous administration of levodopa, differently from pulsatile administration, is able to ameliorate behavioral disturbances related to impulsivity and compulsivity. The underlying mechanisms are still under investigations and not completely understood. However, synaptic plasticity and dopamine receptors sensitization-desensitization mechanisms depending on levodopa continuous stimulation have been considered [36].
Our work has some limits. Firstly, we were not able to properly define the onset time of both LID and ICRB, since the study was cross-sectional and the patients were relatively advanced. Secondly, we did not consider the total exposure time on dopaminergic OT before the beginning of levodopa infusion in LID/JLI group. Third, RDRS reflects the severity of LID only at the time of examination and it does not take into account the presence and impact of LID on patients’ daily-life functioning. Finally, the relatively small sample size did not allow us to provide further information deriving from multivariate analysis. However, future larger cohort studies could be helpful.
Conclusion
To the best of our knowledge, our work is the first one in which the role of JLI on behavior complications had been tested throughout a cross-sectional study. Our results indicate that both prevalence and severity of ICRB were higher in patients with LID as compared to patients without LID, in line with previous findings. When considering the effect of dopaminergic therapy, dyskinetic patients on JLI had higher LEDD compared to other groups, but they showed ICRB less severely than dyskinetic patients on OT, suggesting a potential protective effect of JLI on ICRB. Further and larger studies, both cross-sectional and longitudinal, are critically needed to elucidate the effects of JLI on behavioral complications resulting from dopaminergic replacement therapy.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The Movement Disorders Center of the University of Perugia was supported by a grant from New York University School of Medicine and The Marlene and Paolo Fresco Institute for Parkinson’s and Movement Disorders, which was made possible with support from Marlene and Paolo Fresco. This research was also supported by a Grant from the Italian Ministry of Education, University and Research - PRIN - Prot. 2017ENN4FY to P.C.
