Abstract
Background:
Speech disorders are among the most common adverse effects after subthalamic nucleus deep brain stimulation (STN-DBS) in Parkinson’s disease (PD) patients. However, longitudinal speech changes after STN-DBS are not fully understood.
Objective:
We performed a two-year prospective study on PD patients who underwent STN-DBS and analyzed changes in speech function to clarify factors predicting for speech deterioration.
Methods:
Twenty-five PD patients were assessed before and up to two years after STN implantation. Speech function was evaluated in the on-stimulation condition and 30 min after stimulation cessation using auditory-perceptual assessment. Patients who experienced overall worsening in speech intelligibility or naturalness ≥1 point during follow-up were classified into a deteriorated group (n = 16), with the remaining subjects being classified into a stable group (n = 9). Cognitive and motor functions were also assessed.
Results:
The stable group had significantly better values of low volume, monoloudness, and asthenic voice subscores of the auditory-perceptual assessment in the on-stimulation condition compared with the off-stimulation condition. Imprecise consonants, excess loudness variation, and strained voice subscores were improved via cessation of stimulation in both groups. Before surgery, the deteriorated group had significantly lower scores in the Stroop Color-Word Test and Digit Span compared to the stable group.
Conclusions:
During follow-up, some subscores showed significant worsening in the on-stimulation condition in both groups. However, beneficial effects of STN-DBS on speech appeared to counterbalance negative effects of STN-DBS on speech function only in the stable group. Worse cognitive function may be a potential predictor for speech deterioration after STN-DBS in PD patients.
Keywords
INTRODUCTION
Subthalamic nucleus deep brain stimulation (STN-DBS) is a widely accepted surgical treatment for advanced Parkinson’s disease (PD) patients with motor complications. It effectively improves motor function, motor complications, and quality of life, and reduces the levodopa equivalent daily dose (LEDD) [1]. However, speech disorders are one of the most common adverse effects following STN-DBS in PD patients [2], and speech deterioration is a possible obstacle to fully benefiting from STN-DBS therapy in these patients [3]. Nevertheless, detailed prospective studies on perceptual speech change after STN-DBS remain scarce.
Recent prospective studies using auditory-perceptual assessment have demonstrated significant deterioration in speech intelligibility one year after STN-DBS [4–6], and an average deterioration rate of 12.3–16.9% in speech intelligibility was noted [4, 5]. They reported that the lower speech intelligibility before bilateral STN implantation, longer disease duration, and medially-placed active contacts in the left hemisphere were predictive factors for deterioration in speech intelligibility following STN-DBS [4]. A retrospective study for 15 years reported that 52% of PD patients lost their ability to speak over time after STN-DBS [7]. In a previous study, we prospectively evaluated a change in speech function with DBS in 32 PD patients and reported not only significant deterioration in speech intelligibility one year after STN-DBS but also significant deterioration in speech naturalness and subscores constituting speech intelligibility/naturalness [6]. Among these subscores, two items, grade of dysphonia and strained voice, were significantly worsened during one-year follow-up in patients treated with STN-DBS, compared with those treated with medical therapy.
Speech disturbance due to STN stimulation is multifaceted [8]; namely, the degree of speech deterioration after DBS differs among patients, although the factors responsible for the variation in prognosis have not been fully understood. In addition, it remains unknown how speech characteristics change longitudinally in postoperative cases. Therefore, a long-term prospective evaluation of auditory-perceptual assessment subscores in PD patients treated with DBS is needed.
In the present study, we evaluated longitudinal change in speech characteristics by analyzing structured auditory-perceptual analysis subscores to better understand stimulation-induced speech deterioration. Additionally, we aimed to identify factors predicting speech deterioration after STN-DBS.
MATERIALS AND METHODS
Participants
The inclusion criteria were as follows: 1) diagnosis of PD based on the United Kingdom Parkinson’s Disease Society Brain Bank criteria [9]; 2) no further neurological diseases; 3) Japanese as native language; 4) absence of severe cognitive impairment or psychiatric disorders that may hinder speech assessment; and 5) bilateral STN implantation at Nagoya University Hospital. We identified 30 consecutive patients who underwent bilateral STN implantation in the period from 2013 to 2015. Of these, two patients refused on- and off-stimulation assessment, one refused to participate, and two died during the follow-up period of 24 months after STN implantation. Therefore, 25 PD patients (10 males and 15 females) in total were enrolled in the study (Table 1). All participants were taking antiparkinsonian medication. A board-certified neurologist (T.T.) evaluated all participants using the Unified Parkinson’s Disease Rating Scale (UPDRS) [10]. A skilled and certified speech-language-hearing therapist (SLHT; Y.T.) performed speech recordings and cognitive function examinations.
Patient characteristics
Values are mean±SD; Motor and cognitive assessments before DBS implantation were performed in the On-medication condition. UPDRS-III, Unified Parkinson’s Disease Rating Scale motor examination; UPDRS-IV, Unified Parkinson’s Disease Rating Scale motor complications; LEDD, levodopa equivalent daily dose; S & E, Schwab and England Independence Scale; VHI, Voice Handicap Index; MMSE, Mini-Mental State Examination; MoCA-J, Montreal Cognitive Assessment Japanese version; SCWT, The Stroop Color and Word Test; Part 1, naming task of different color patches; Part 2, task of named color-word condition; RCPM: Raven’s Colored Progressive Matrices.
All the PD patients were assessed before bilateral STN implantation (baseline) and at 3, 6, 12, 18, and 24 months after surgery. Baseline assessments of motor, cognitive, and speech functions were done in inpatient settings. Following surgery, each patient was followed regularly for clinical assessments and adjustment of stimulation settings and medication. They were assessed in the on-state under continued medication. The LEDD [11] and DBS parameters in the PD patients are shown in Tables 2 and 3. For auditory-perceptual analyses, speech was first recorded in the on-stimulation condition and then 30 min after stimulation cessation.
Patient characteristics at baseline and during follow-up period after surgery
*p < 0.05, **p < 0.01: significant longitudinal changes within group by Holm post-hoc test. †p < 0.05, ††p < 0.01: significant difference between the on- and off-stimulation conditions by Wilcoxon signed-rank tests. Values are mean±SD; BL, before bilateral STN implantation; M, months after surgery; n/a, not applicable; n.d., not determined; n.s., not significant; UPDRS-III, Unified Parkinson’s Disease Rating Scale motor examination; UPDRS-IV, Unified Parkinson’s Disease Rating Scale motor complications; LEDD, levodopa equivalent daily dose; S & E, Schwab and England Independence Scale; Stim on-condition, Period of STN electrical stimulation on-condition; Stim off-condition, Period of STN electrical stimulation off-condition; VHI, Voice Handicap Index; MMSE, Mini-Mental State Examination; MoCA-J, Montreal Cognitive Assessment Japanese version.
DBS settings
Values are mean±SD; M, months after surgery; p value by Friedman’s nonparametric Two-Way ANOVA.
The study adhered to the Ethical Guidelines for Medical and Health Research Involving Human Subjects endorsed by the Japanese government and was approved by the Ethical Review Committee of Nagoya University Graduate School of Medicine. Written informed consent was obtained from all participants.
Speech and voice analyses
Speech and voice samples were recorded in a sound-treated room and digitized using a voice recorder (ICD-SX813; Sony, Tokyo, Japan) at a sampling rate of 44.1 kHz with 16-bit quantization. A microphone (ECM-MS907; Sony) was positioned to maintain a constant mouth-to-microphone distance of 15 cm during recording. Recorded speech and voice samples were subsequently used in perceptual analyses, for which we used the Assessment of Motor Speech for Dysarthria (AMSD) [12] consisting of analogous variables developed by Darley et al. [13] and GRBAS scale [14]. The overall severity of speech disorders, speech intelligibility and naturalness, are scored from 1 to 5 with a 0.5 increment; a score of 1 indicates normal, a score of 5 indicates severe, and a score of 2 to 4 indicate cases with speech disorders between these two points. Subscores of AMSD and GRBAS scale were scored from 0 to 3; 0 = normal, 1 = mild, 2 = moderate, and 3 = severe. Definitions and interpretations of the variables are summarized in Supplementary Table 1. The speech and voice samples included reading a standard passage (Japanese version of “The North Wind and the Sun”), short conversations, and sustained vowels. All patients were asked to speak in their habitual and comfortable pitch, loudness, and rate. Three certified SLHTs (M.S., R.O., and Y.T.) blindly evaluated the speech samples and a mean value of the three raters was derived for each score. The inter-evaluator reliability kappa coefficient (R, http://www.r-project.org/) was 0.641, representing substantial agreement [15].
For detailed understanding of postoperative speech symptoms, we divided the 25 PD patients into two groups per the following criteria. All patients experiencing worsening in overall speech intelligibility or speech naturalness in the on-stimulation condition by ≥1 point during the follow-up period were classified as the “deteriorated group”. The remaining subjects were classified as the “stable group”. The cut-off value for worsening was consistent with the guidelines for perceptual analyses and a level that clearly shows a meaningful difference [12].
Motor function examination
The motor symptoms were assessed using UPDRS motor examination (UPDRS III). To reveal the factors predicting deterioration in speech after STN-DBS, we subdivided UPDRS III score into four subscores [16] and compared them between the deteriorated and stable groups: axial (items 18–19 and 27–30), tremor (items 20–21), rigidity (items 22), and bradykinesia score (items 23–26, 31).
Cognitive function examinations
All patients underwent cognitive assessments using the Mini-Mental State Examination (MMSE), the Montreal Cognitive Assessment (MoCA), verbal fluency (letter: ka, semantic: animal), the Stroop Color-Word Test (SCWT), Digit Span in the Wechsler Adult Intelligence Scale-3rd edition (WAIS–III) [17], Raven’s Colored Progressive Matrices, and judgment of Line-orientation in the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) [18] before operation (baseline). To assess general cognitive function of the patients, we further conducted MMSE, MoCA, and verbal fluency assessments in the on-stimulation condition at 12 and 24 months after surgery (Table 2). We chose these three cognitive examinations to minimize the effect of fatigue on the patients.
Radiological evaluations
Anatomical locations of DBS electrodes were plotted on the standard human brain atlas at the level of 3.5 mm below the anterior commissure–posterior commissure line using the same technique we employed in our previous report [6, 8].
Statistical analysis
We used the software SPSS statistics–24 (IBM, Chicago, IL, USA) for statistical analysis. Longitudinal changes in motor, cognitive, and speech functions were assessed by comparing the data at baseline with those at each timepoint after surgery using Friedman’s non-parametric Two-Way ANOVA with Holm post-hoc test with R (http://www.r-project.org/). The differences in speech functions between the on- and off-stimulation conditions were assessed using Wilcoxon signed-rank tests. Comparison of variables between the deteriorated and the stable group was performed using Mann–Whitney U tests. A p value <0.05 was considered statistically significant.
RESULTS
The effects of STN stimulation on motor and speech functions
Compared with baseline, UPDRS IV, Schwab and England scale off-state significantly improved at 12 and 24 months after DBS implantation along with significantly decreased LEDD (all p < 0.05; Table 2, and Supplementary Table 2). Among the 25 patients who underwent STN-DBS, 16 patients had deterioration in overall speech intelligibility or speech naturalness in the on-stimulation condition by ≥1 point during the follow-up period (the deterioration group). The other nine patients with PD had no such deterioration (the stable group). Although both groups showed worsening speech intelligibility (deteriorated group, p < 0.001; stable group, p < 0.01) and speech naturalness (deteriorated group, p < 0.001; stable group, p < 0.01), the degree was more severe in the deterioration group (Fig. 1). The deteriorated group showed significant worsening from baseline in speech intelligibility at Month 12 or later and in speech naturalness at Months 18 and 24. Neither the deteriorated nor the stable group showed significant longitudinal changes in motor function during the follow-up period (UPDRS III; deteriorated group, p = 0.679; stable group, p = 0.818).
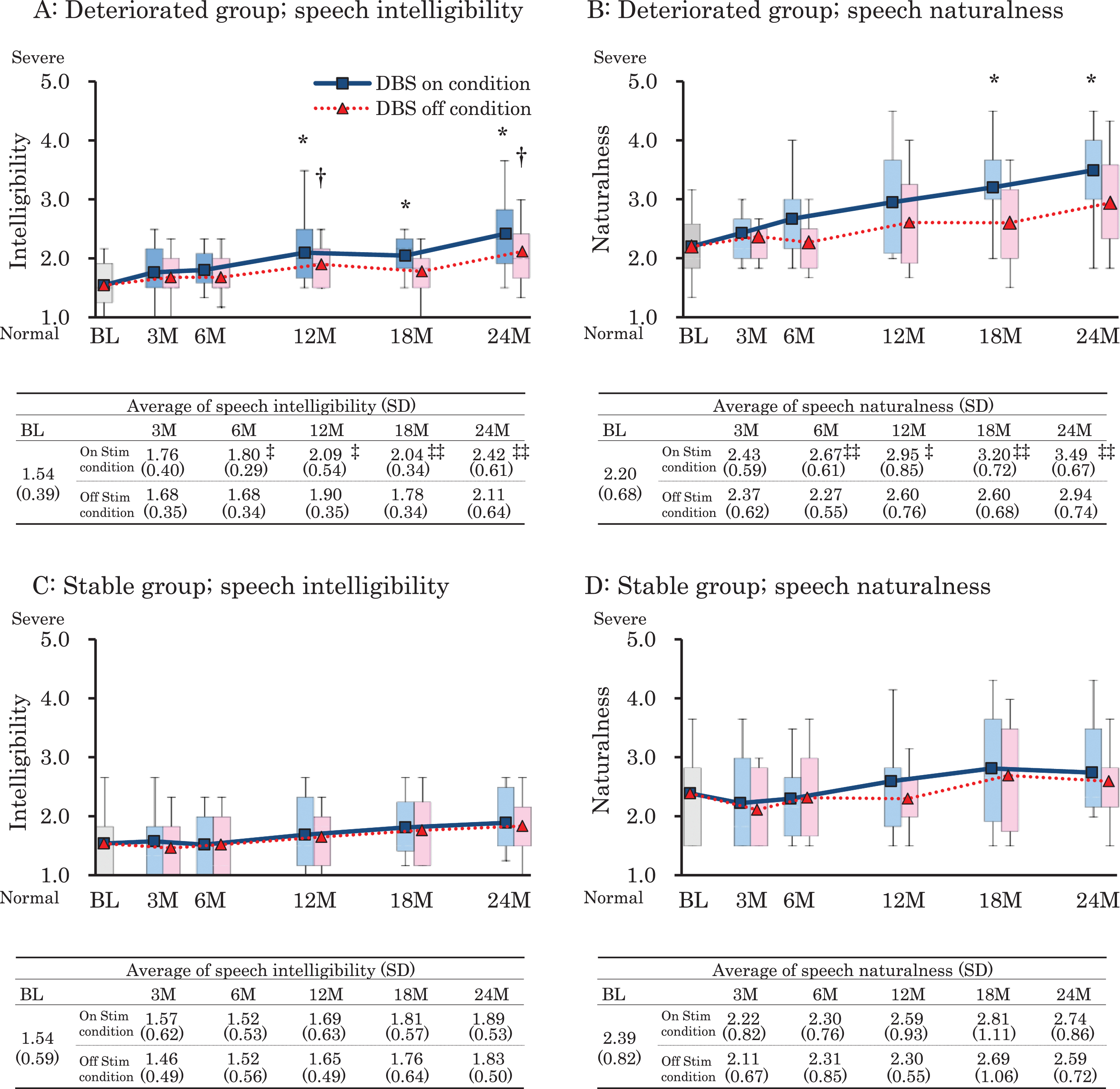
Changes in overall severity of auditory perceptual assessment in the on- and off-stimulation conditions. The deteriorated group showed more severe deterioration in speech intelligibility (A) and naturalness (B) compared with the stable group (C, D). BL, at baseline; M, months after surgery. *p < 0.05; Significant longitudinal changes within a group in on-stimulation conditions. †p < 0.05; Significant longitudinal changes within a group in off-stimulation conditions. ‡p < 0.05, ‡‡p < 0.01; Significant difference between the on- and off-stimulation conditions.
To identify stimulation-induced speech disturbances, we also evaluated speech intelligibility and naturalness 30 minutes after stimulation cessation. The deteriorated group showed significantly worse scores in the on-stimulation condition than in the off-stimulation condition at every interval after Month 6, while the stable group showed no significant difference at any timepoints (Fig. 1). The stimulation conditions (amplitude, frequency, and pulse width) showed no significant differences between the deteriorated and stable groups (Table 3).
The effects of STN stimulation on speech subscores
The differences in subscores of auditory-perceptual assessment between the on- and off-condition are summarized in Fig. 2. In the stable group, three subscores were significantly better in the on-stimulation condition than in the off-stimulation condition (p < 0.05); low volume at Months 18 and 24, monoloudness at Months 18 and 24, and asthenic voice at Months 12 and 24. In contrast, in the deteriorated group, no subscores were improved due to the stimulation.
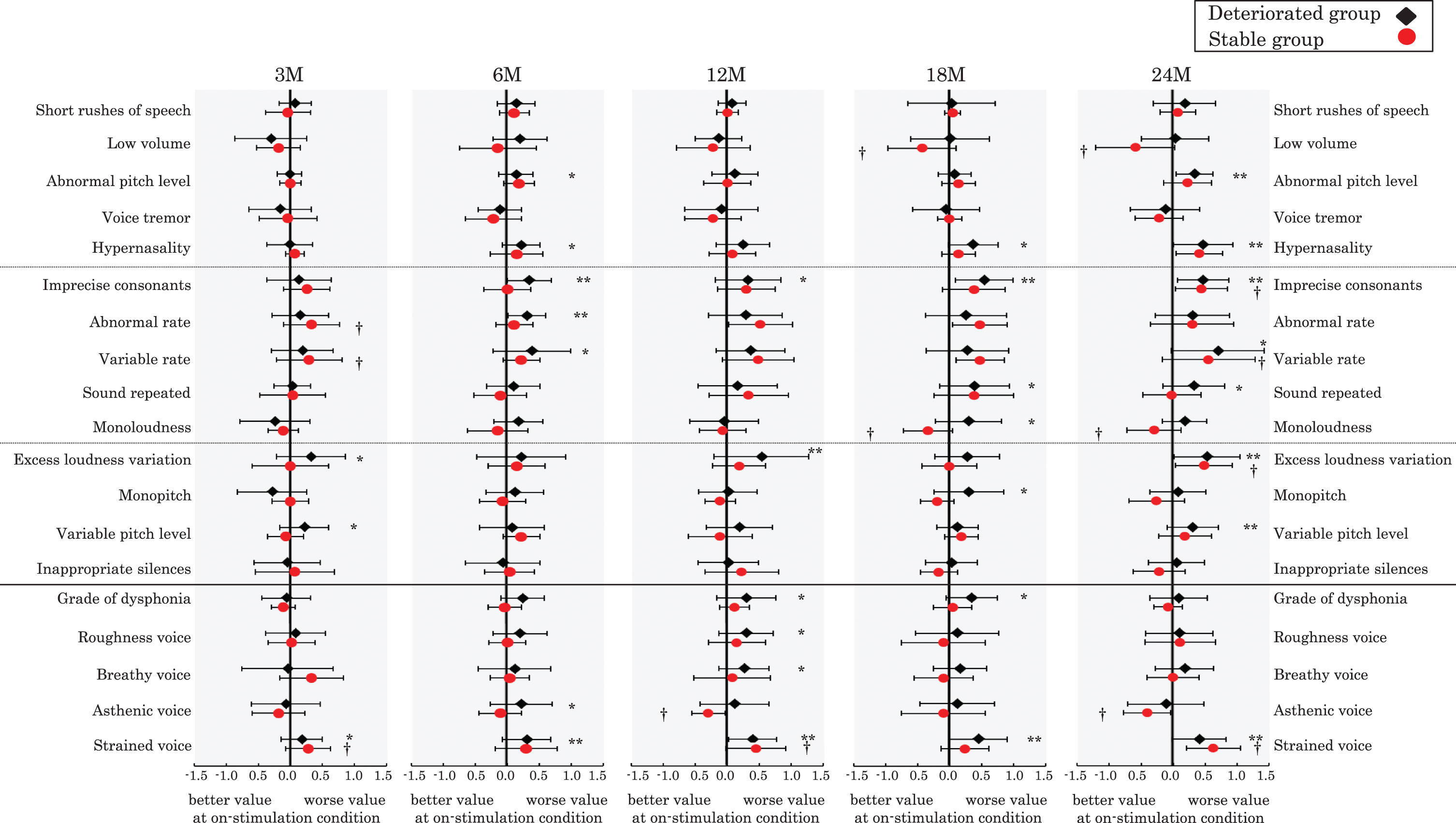
The longitudinal change of subscores of auditory-perceptual analyses. The difference in subscores of auditory-perceptual analyses between the on- and off-conditions during 3 to 24 months after the surgery. M, months after surgery. *p < 0.05 or **p < 0.01, significant difference between the on- and off-stimulation conditions in the deteriorated group; †p < 0.05: significant difference between the on- and off-stimulation conditions in the stable group.
Imprecise consonants, excess loudness variation, and strained voice subscores were significantly improved after cessation of stimulation in both groups. Furthermore, in the deteriorated group, abnormal pitch level and variable pitch level were worse in the on-stimulation condition than in the off-stimulation condition. Albeit not significant, a similar trend was found in the stable group.
Clinical backgrounds before STN implantation
The clinical backgrounds of the patients in the deteriorated and stable groups were compared to identify factors predicting speech deterioration after STN-DBS. At baseline, the deteriorated group had significantly lower SCWT and Digit Span scores compared with the stable group (p < 0.05), although there were no significant differences in speech and motor function, other cognitive tests, LEDD, age, or disease duration between the two groups (Table 4).
Clinical backgrounds in deteriorated group and stable group before surgery
Values are mean±SD; UPDRS-III, Unified Parkinson’s Disease Rating Scale motor examination; Axial score, items 18-19 and 27–30 of UPDRS III; Tremor score, items 20-21 of UPDRS III; Rigidity score, items 22 of UPDRS III; Bradykinesia score, items 23–26 and 31 of UPDRS III; UPDRS-IV, Unified Parkinson’s Disease Rating Scale motor complications; LEDD, levodopa equivalent daily dose; S & E, Schwab and England Independence Scale; VHI, Voice Handicap Index; MMSE, Mini-Mental State Examination; MoCA-J, Montreal Cognitive Assessment Japanese version; SCWT, The Stroop Color and Word Test; Part 1, naming task of different color patches; Part 2, task of named color-word condition; RCPM, Raven’s Colored Progressive Matrices; p value by Mann–Whitney U tests.
Electrode positions
The electrode positions were available for 24 of 25 PD patients. The overall positions of the deteriorated group tended to locate laterally to STN compared with the stable group (Supplementary Figure 1).
DISCUSSION
The present study classified 25 PD patients into two groups (the deteriorated and stable groups) based on change in overall severity of speech intelligibility and naturalness during the two-year follow-up after STN-DBS. In the deteriorated group, overall speech function significantly worsened at Month 12 or later, whereas motor function remained stable throughout the two-year follow-up period. In addition, speech intelligibility and naturalness in the deteriorated group were significantly worse in the on-stimulation condition than in the off-stimulation condition at Month 6 or later, suggesting that electrical stimulation negatively affected speech functions as early as six months after the surgery. Tripoliti et al. [4] reported that significant speech deterioration in PD patients treated with STN-DBS emerged between six months and one year, consistent with our findings. Therefore, regular and detailed assessments of speech function are recommended along with careful DBS adjustments to maintain the good communication abilities of PD patients.
In the stable group, there were no significant differences in overall speech intelligibility or naturalness between the on- and off-stimulation conditions at any follow-up timepoint. However, the speech function tended to deteriorate with time even when electrical stimulation was stopped. This may be related to disease progression [19] and/or residual stimulation effects. In terms of auditory-perceptual analysis, low volume, monoloudness, and asthenic voice subscores, the stable group showed better scores in the on-stimulation condition than in the off-stimulation condition, suggesting that STN stimulation had beneficial effects on these items in certain populations. In support of our findings, a previous report also documented that loudness of PD patients significantly improved after STN-DBS [4]. These speech aspects are thought to be mainly due to parkinsonian symptoms, i.e., rigidity and akinesia [13, 16]. STN electrical stimulations are well known for their effectiveness in improving rigidity and akinesia symptoms of PD [19–23]. Our previous study demonstrated that STN stimulation improves movement of articulation structures including the mouth/jaw and tongue by improving their symptoms using an acoustic analysis [26]. In addition, we also reported that a minority of PD patients treated with STN-DBS became almost speechless due to severe rigidity and akinesia after stimulation cessation [8]. Therefore, improvements in speech symptoms observed in the present study appear to stem from the beneficial effects of the stimulations on improving rigidity and akinesia of speech-related muscles. Speech functions other than low volume, monoloudness, and asthenic voice showed similar changes between the deteriorated and stable groups. In particular, imprecise consonants, excess loudness variation, and strained voice were significantly improved due to cessation of stimulation in both groups, as we previously documented [27, 28]. Taken together, these findings suggest that negative effects of STN-DBS on speech function commonly appear in PD patients, but significant beneficial effects of DBS may be obtained only in a subset of population, leading to better prognosis of speech function after DBS.
To clarify factors predicting deterioration in speech after STN-DBS, we compared preoperative clinical backgrounds of patients between the deteriorated and stable groups. We found that SCWT and Digit Span Test scores in the deteriorated group were significantly worse in comparison with those in the stable group. Moreover, the patients in the deteriorated group tended to be older and to have longer disease duration and poorer UPDRS III total and bradykinesia scores and speech functions compared with the stable group. A recent prospective study reported that in 54 PD patients, lower speech intelligibility before bilateral STN implantation, longer disease duration, and medially-placed active contacts in the left hemisphere were predictive factors for deterioration in speech intelligibility following STN-DBS although cognitive function were not analyzed [5]. Other previous studies have documented these cognitive functions before bilateral STN implantation may be predictive factors for motor function after STN implantation in patients with PD [29, 30]. Considering the above-mentioned results, we speculate that patients with worse cognitive function might be vulnerable to detrimental effects of the stimulation on speech function because of less effective compensatory mechanisms in the brain with more advanced disease. Furthermore, we found that the electrode positions of the deteriorated group tended to be laterally to STN compared with the stable group, suggesting the current spread to the surrounding structures, such as the corticobulbar fibers [8]. However, we were unable to assess the correlations between the electrode positions and speech outcomes with our method. Future imaging-driven analyses of brain connectivity or diffusion tensor imaging should allow us to reveal the contributions of the current spread and cognitive dysfunction to speech deterioration after DBS.
In interpreting our results, several limitations must be considered. First, our cohort had a relatively small sample size. Second, the changes over time were not compared with a control group (i.e., patients treated only with medication). Third, patients were assessed only in the on-state under continued medication, and thus the impact of medication on speech function was not assessed. Fourth, the off-stimulation assessment was done 30 min after stimulation cessation. Although we have shown significant changes after stimulation cessation for 30 min in our previous studies [6, 26–28], stimulation-related changes may become larger with a longer off-stimulation period. Fifth, we plotted the electrode positions of the two groups but could not assess the relationship with the electrode positions and speech outcomes as noted above. Finally, we adjusted the STN stimulation settings for maximal improvement in patient status including motor and speech functions during follow-up. Despite such conditions, we were still able to identify a group that easily developed speech disorders, providing an important insight for clinical practice.
In conclusion, we found a differential pattern of longitudinal speech changes in PD patients treated with STN-DBS. Our results suggest that beneficial effects of STN-DBS on speech deficits, particularly those due to parkinsonism, appear to compensate for the negative effects of the procedure on speech ineligibility and naturalness. The beneficial effects of STN-DBD might be attenuated in PD patients with impaired cognition Novel devices with new programming techniques that adjust the area and direction of stimulation [31–35] may have the potential to resolve these critical problems. Future studies are needed to examine these possibilities and to elucidate relevant mechanisms.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
