Abstract
Background:
A wide variety of conversion factors for a levodopa-equivalent-dose (LED) have been proposed for each Parkinson’s disease (PD) medication. The currently-used set of conversion factors is based on studies that relied on subjective experience or theoretical assumptions. This set was never validated in patients receiving polytherapy.
Objectives:
To use real-life data to identify an optimal set of conversion factors independent of prior assumptions regarding clinical efficacy of different medications.
Methods:
Retrospective analysis of data from 206 cognitively-preserved patients with advanced PD receiving polytherapy before deep brain stimulation (DBS) surgery. A nonlinear automated problem solver was used to find a set of conversion factors that, when applied, minimized the coefficient of variation of LEDs in a relatively homogenous cohort of patients.
Results:
Independent and model-free evaluation of a wide range of possible sets of conversion factors to LED suggested a set of normalized conversion factors for immediate release levodopa (1.00), controlled release levodopa (0.88), and amantadine (1.23). A minimal clinical benefit of entacapone was observed for patients with motor fluctuations. Our analysis could not detect conversion factors for dopamine agonists and MAO-B inhibitors, possibly because their clinical contribution when added to levodopa is limited.
Conclusions:
Independent from previous studies and prior assumptions we show that the currently-used LED conversion factors for immediate release levodopa, controlled release levodopa and amantadine are largely correct and that dopamine agonists, MAO-B inhibitors and entacapone, given in addition to levodopa, have little additional clinical value for PD patients with motor fluctuations.
Keywords
INTRODUCTION
Dosage conversion of different anti-parkinsonian agents into a single unit of measurement, namely levodopa-equivalent-dose (LED), guides smooth switching between medications for the individual patient and enables generalizing the outcomes of clinical trials. The most widely used table of conversion factors to LED [1] is based on the calculated mode statistics of previously suggested values of conversion factors. In the majority of cases, the values which have been used to generate this table have not relied on direct head-to-head comparison of agents, but rather on theoretical assumptions or on the clinical experience of investigators (for example, see [2, 3]). This approach resulted in a range of suggested values for each single conversion factor. For example, a range of values between 0.25 mg [4] and 1.67 mg [5] of pramipexole have been suggested as equal to 100 mg of immediate-release levodopa. This wide range of conversion values indicates that the methods used to find these values were not sufficiently robust or consistent. In few cases, In addition, the currently used table of conversion factors was never validated in a real-life setting or with patients on polytherapy.
In order to search, in a real-life setting, for the optimal values of conversion factors, we studied a homogenous cohort of patients with advanced Parkinson’s disease (PD) without associated dementia. We used model-free methodology to search for an optimal set of conversion factors for the different anti-parkinsonian agents without relying on prior assumptions regarding pharmacological properties of these agents or on our clinical experience.
MATERIALS AND METHODS
Retrospectively analyzed data were extracted from the medical records of all PD patients who underwent deep brain stimulation (DBS) surgery at Hadassah Medical center (Jerusalem, Israel) between July 2008 and April 2018. Data sets included the doses of all oral anti-parkinsonian drugs used by the individual patients, and the clinical characteristic (age, age at symptoms onset, UPDRS III on and off medications, Addenbrooke’s cognitive examination), as they were documented by the neurologist on the day of clinical evaluation for DBS surgery. The study was approved by the local IRB committee and participants signed informed consent (0545-08-HMO or 0064-12 HMO).
Based on predefined criteria, data sets were excluded from analysis if: 1) detailed record of medication dosages was incomplete, 2) the patient received less than 150 mg/day of levodopa due to personal preference or side effects, 3) the patient was treated with additional non-oral anti-parkinsonian therapy (continuous intestinal carbidopa and levodopa or subcutaneous apomorphine) or 4) the patient received dopamine receptor blocking agents other than quetiapine or clozapine. In cases where UPDRS values were not fully documented these records were still incorporated in the analysis.
Currently used conversion factors [1] were applied for each data set, to calculate the total daily LED. For all patients taking entacapone, the dose of levodopa was multiplied by 0.33 for calculating LED for entacapone and was added to the total LED, irrespective of the dose of entacapone. In any case, in our cohort all patients on entacapone took this agent multiple times per day. We did not include anti-cholinergic agents in total LED calculation since this agent is no longer in common use for this indication. Rotigotine is not in common use in Israel and therefore none of the participants in this study was using it.
Without prior assumptions regarding the potency of the different anti-parkinsonian agents, we searched for an alternative set of seven conversion factors (X1 to X8, for the anti-parkinsonian agents: immediate release levodopa, controlled release levodopa, pramipexole, ropinirole, entacapone, amantadine, selegiline and rasagiline respectively), using real-life data from a cohort of patients receiving different combinations of oral therapies.
We analyzed a range of possible sets using an automated problem solver (see below). For each studied set of 8 conversion factors, the automated algorithm calculated a newly-suggested total daily LED, for each patient (yielding a total of 206 values for each possible set of 8 conversion factors), using the following formula:
For each of the studied sets of conversion factors, the automated algorithm then calculated the coefficient of variation using all newly-suggested total daily LEDs (that were calculated based on this specific set). The set that yielded the minimal coefficient of variation was considered as the optimal test. A minimal coefficient of variation was used as an endpoint since we expected that patients at a similar stage of progression of their diseases received therapies with comparable efficacy. We did not use standard deviation as an endpoint since such an endpoint would have been artificially minimized by a set of conversion factors equaling zero.
The MATLAB (Massachusetts, USA) command ‘fmincon’ was used to explore the range of possible values of each conversion factor and to identify the optimal set within this range. This command uses a nonlinear problem solver to examine numerous alternative number sets in order to find the best set of numbers (here representing conversion factors to LED) that would minimize a predefined calculated value (coefficient of variation in our case). For each conversion factor, we used the values of 0 and 1000 as the lower and upper possible bounds, respectively. A range of values of 0–40 were used as the starting point to avoid local minima. The range of starting point that yielded stable values of conversion factors was determined by visual inspection. Each calculated set of conversion factors were normalized to X1 so that the conversion factor of immediate release levodopa would always be 1.
RESULTS
We reviewed the medical charts of 262 patients who underwent DBS surgery for PD. Based on our predefined criteria we excluded the data sets of 37 patients with insufficient documentation of their pre-DBS regime of medications, 12 patients who did not receive an optimal dose of levodopa due to their personal preference or side effects (11 patients without levodopa therapy and a single patient on 75 mg of levodopa once daily), four patients on non-oral anti-parkinsonian therapies and three patients on dopamine receptor blocking agents other than quetiapine or clozapine. After excluding the above participants, 206 patients were included in our analysis.
Our decision to focus on a sub-population of PD patients who were considered good candidates for DBS surgery resulted in a relatively homogenous group of patients. The patients in this cohort all had advance PD, a good levodopa response, received multiple anti-parkinsonian types medications (89% had more than a single type) that were optimized by a movement disorder specialist, and did not have significant cognitive deficits that limited therapy. These characteristics are summarized in Table 1.
Characteristics of 206 patients included in analysis
UPDRS, Unified Parkinson’s disease rating scale; LED, levodopa-equivalent-dose.
Conversion factors for LED calculation
Without any prior assumptions (other than the linear additive clinical benefit of different anti-parkinsonian drugs), we then looked for an optimal set of 8 conversion factors (for immediate release levodopa, controlled release levodopa, pramipexole, ropinirole, entacapone, amantadine, selegiline and rasagiline). We hypothesize that by using this set to calculate 206 newly-suggested LEDs (one for each participant) we would be able to minimize the coefficient of variation calculated over these 206 newly-suggested LEDs.
Because the optimal set of conversion factors, as identified by a non-linear problem solver, such as the one we used, could change when the search starting point is changed we first explored a wide range (0–40) of possible starting points (Fig. 1). This analysis showed that our method yielded replicable results over a wide range of starting points (0–20), for immediate release levodopa (mean±sd, 0.56±0.01 in 41 equally-spread starting points in the relevant range, normalized to 1 in all sets), controlled released levodopa (normalized conversion factor of 0.88 relative to immediate release levodopa, Fig. 1A), amantadine (normalized conversion factor of 1.23 relative to immediate release levodopa, Fig. 1B) and entacapone (normalized conversion factor of 0 relative to immediate release levodopa, Fig. 1E). Parallel steady state at this range of starting points was not reached for ropinirole, pramipexole, rasagiline and selegiline (Fig. 1C, D). Starting point values in the range of 20–40 seemed to yield stable results for the two agonists’ and for the two MAO-B inhibitors’ conversion values. These values, however, were identical to both agonists and to both MAO-B inhibitors and, therefore, probably represent the mathematical instability of the used function.
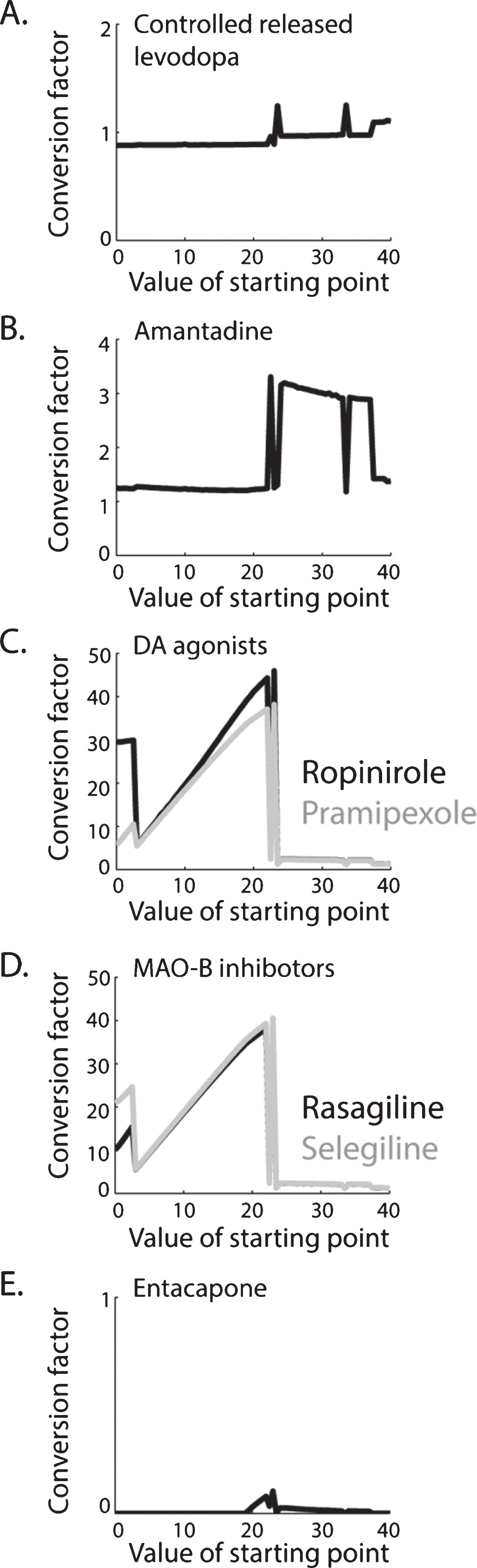
Newly suggested conversion factors varying with the set value of the starting point of the nonlinear problem solver. For immediate release levodopa (not shown), controlled release levodopa (A), amantadine (B), and entacapone (E) new conversion factors are suggested based on replicable results over a wide range (0–20) of starting points. For dopamine agonists (C) and MAO-B inhibitors (D), no biologically-relevant steady-state was reached and therefore new conversion factors are not suggested.
Assuming that our approach did not yield reliable new conversion factors for those medications which do not significantly affect the clinical state of a patient on polytherapy, we repeated our analysis after omitting pramipexole, ropinirole, rasagiline and selegiline from our data (so that a set of only 4 conversion factors was now evaluated). This analysis yielded similar results to the previous analysis with suggested normalized conversion factors of 1, 0.89, 1.32, and 0.05 to immediate release levodopa, controlled released levodopa, amantadine, and entacapone, respectively.
DISCUSSION
We applied a method that does not depend on assumptions regarding the pharmacological properties of the relevant agents or on clinical observations in order to find the optimal set of conversion factors to LED. LED calculation in practice relies on the unproven assumption that the effects of different pharmacological agents are linearly summed. Our study did not aim to test this assumption, however, we are aware that it is oversimplified and was never rigorously evaluated. It is reasonable to expect, for example, that levodopa and a dopamine agonist compete for the same striatal receptors [6] and, therefore, that their additive value would be sub-linear.
We chose to apply our approach to a cohort of patients at the time of successful evaluation for DBS surgery. All patients in this cohort fulfilled the criteria for DBS [7–9] and therefore did not have significant cognitive decline, had Hoehn and Yahr score ≤3 during the ‘On’ state, had clinically significant motor fluctuations that required multiple dosing of medications and had a good response to levodopa. Based on our pre-defined criteria we excluded patients who were evaluated at an earlier stage of their disease.
PD is a heterogeneous disease. Our method of minimizing LED variability in order to find the optimal set of conversion factors is based on assuming homogenous clinical characteristics in our study cohort. This assumption, however, was only partially fulfilled. Patients were assessed for a DBS surgery following a variable interval of time from the onset of their disease and from the onset of motor fluctuations. Part of this variability could be attributed to the fact that 65% of the participants in this study were referred to us by movement disorder specialists from other medical centers. Patients also varied widely in a range of other clinical characteristics (Table 1), such as total daily LED at the time of DBS evaluation. While we attempted to homogenize our cohort by selecting patients based on the above criteria, our success was partial. Variability between patients, at the time of DBS evaluation, is a possible cause for our failure to identify conversion factors for some of the pharmacological agents examined.
The currently used conversion factor for controlled release levodopa [1] is based on a meta-analysis of a large number of studies that, in many cases, used an identical set of conversion factors. Our suggestion of using a conversion factor slightly higher (0.88) than the one in current use (0.75) is compatible with reduced bioavailability (85–90%) of this drug relative to the bioavailability of immediate release levodopa [10]. The currently-used conversion factor for amantadine is based on a single study [1]. Our findings suggest a slightly higher value for an amantadine conversion factor.
Our results suggest that the clinical benefit of entacapone is negligible when LED at the time of DBS surgery is considered as an endpoint. It seems that the 33% increment of a levodopa serum concentration, when administrated together with entacapone [11], is not translated into a clinically-significant improvement at this advanced stage of the disease [12]. In addition, our method failed to suggest conversion factors for MAO-B inhibitors and for dopamine agonists. We suspect that this failure resulted in part from the limited clinical benefit of these agents given in addition to more potent agents (levodopa and amantadine). This hypothesis is compatible with our observation that when we omitted MAO-B inhibitors and dopamine agonists from our analysis the conversion factors identified by our problem-solver procedure did not change for levodopa and amantadine.
Finally, our results emphasize the need to distinguish between two important considerations. The first is the relative potency of anti-parkinsonian medication that can be measured based on the observed clinical benefit, following a single dose of any pharmacological agent (and can be expressed in the LED of a single dose). The second, which we evaluated in this study, is the total-daily LED, currently calculated by summing the LEDs of all daily single doses. Establishing this second consideration as a clinically useful and reliable procedure would require future studies on the complex competitive and synergistic interactions between drugs.
CONFLICT OF INTERESTS
The authors have no conflict of interest to report.
