Abstract
Background:
Reduced glutathione and excess free iron within dopaminergic, substantia nigra neurons in Parkinson’s disease (PD) can drive accumulation of toxic hydroxyl radicals resulting in sustained oxidative stress and cellular damage. Factors such as brain penetrance and bioavailability have limited the advancement of potential antioxidant and iron chelator therapies for PD.
Objective:
This study aimed to develop novel nanocarrier delivery systems for the antioxidant curcumin and/or iron chelator deferoxamine (DFO) to protect against rotenone-induced changes in cell viability and oxidative stress in SH-SY5Y cells.
Methods:
Nanocarriers of curcumin and/or DFO were prepared using Pluronic F68 (P68) with or without dequilinium (DQA) by modified thin-film hydration. Cell viability was assessed using an MTT assay and oxidative stress was measured using thiobarbituric acid reactive substances and cellular antioxidant activity assays.
Results:
All formulations demonstrated high encapsulation efficiency (65–96%) and nanocarrier size was <200 nm. 3-h pretreatment with P68 or P68+DQA nanocarriers containing various concentrations of curcumin and/or DFO significantly protected against rotenone-reduced cell viability. The addition of DFO to curcumin-loaded P68+DQA nanocarriers resulted in increased protection by at least 10%. All nanoformulations significantly protected against rotenone-induced lipid peroxidation (p < 0.0001). The addition of DQA, which targets mitochondria, resulted in up to 65% increase in cellular antioxidant activity. In nearly all preparations, the combination of 10 μM curcumin and 100 μM DFO had the most antioxidant activity.
Conclusion:
This study demonstrates for the first time the formulation and delivery using P68 and P68+DQA curcumin and/or DFO nanocarriers to protect against oxidative stress induced by a rotenone PD model. This strategy to combine antioxidants with iron chelators may provide a novel approach to fully utilise their therapeutic benefit for PD.
INTRODUCTION
Postmortem analysis and MRI techniques have highlighted a marked accumulation of free iron within the substantia nigra (SN) in Parkinson’s disease (PD) [1–6]. This, together with reduced levels of the natural antioxidant glutathione, can drive accumulation of toxic hydroxyl radicals within mitochondria resulting in sustained oxidative stress and catastrophic cellular damage [7, 8]. Iron-induced oxidative stress is likely a key contributor to the neurodegeneration observed in PD since intra-nigral injection of iron in rats induces both a PD-like phenotype and pathology [9]. Elevated iron deposition in the SN of people with PD have been significantly correlated with motor symptoms and some non-motor symptoms such as cognitive, sleep and autonomic issues [10].
Iron chelators and antioxidants therefore hold promise as potential neuroprotective therapies for PD. Ward et al. [11] were the first group to demonstrate that bidentate iron chelators like deferiprone (DFP) and deferoxamine (DFO) were able to remove excess brain iron in animal models of brain iron overload. Iron chelators have since been demonstrated to be neuroprotective in 6-hydroxydopamine (6-OHDA) animal models of PD [12, 13]. Furthermore, a recent clinical trial of the iron chelator DFP correlated chelator use with a decrease in the UPDRS score for motor symptoms [14]. Similarly, in both in vitro and in vivo PD models, curcumin has been able to protect against dopaminergic neurodegeneration. For example, in a cellular rotenone-induced neurotoxin model of PD curcumin reduced reactive oxygen species (ROS) and cytotoxicity as well as inhibited caspase 3 and 9 activation, preventing apoptosis [15]. Likewise, in a mutant A53T α-synuclein (α-syn) cellular model of PD curcumin pre-treatment reduced cell death as well as levels of mutant a-syn [16] and in an in vivo drosophila model of PD curcumin not only reduced cell death but also ameliorated PD-like symptoms [15]. More recent evidence suggests that curcumin is also protective against mitochondrial dysfunction. Van der Merwe et al. [17] showed curcumin to be protective in a PINK1 knock down model of PD and PINK1 has an intrinsic role in the removal of dysfunctional mitochondria with mutations in PINK1 resulting in an early-onset autosomal recessive PD. The combination of iron chelators and antioxidant molecules may present a promising potential therapeutic approach for PD as together they are able to completely restore brain function impaired by iron overload in animal models [18].
The free radical scavenging properties of curcumin and the potential of iron chelators such as DFO to limit the availability of detrimental free iron may be a promising mechanism to limit the degenerative process in PD. That said, the therapeutic value of these two compounds is limited as curcumin is unstable with low bioavailability and both are unlikely to access the brain at therapeutic concentrations [19, 20]. Furthermore, long-term effects of continuous iron chelator use are unknown in PD and due to the non-specific nature of such chelators the side effects could be serious since iron is an essential part of numerous cellular processes such as respiration [21] and neurotransmitter synthesis [22, 23].
Nanocarriers have demonstrated potential as targeted delivery systems to improve stability of labile molecules and enhance delivery across membranes, including the blood-brain barrier (BBB), whilst retaining the potency of entrapped molecules [24, 25]. For example, studies which have altered the formulation of curcumin such as encapsulation in glycerol monooleate have increased the half-life of curcumin [26]. Other nanoformulations of curcumin such as the poly(lactic-co-glycolic acid) (PLGA) nanoformulation have demonstrated BBB penetration and the ability to and enter brain tissues including the cerebral cortex and the hippocampus; however, they are also found in other organs such as the spleen and the liver [27]. Zupančič et al. [25] developed a DQA-some nanoformulation of curcumin using vesicles formed from the amphiphile dequalinium (DQA) which were able to specifically target mitochondria. Mitochondrial targeting of antioxidants in PD would be highly beneficial since mitochondria are the main site of intracellular free radical formation [7, 28]. However, such nanoformulations of curcumin were not developed to gain access into the brain or treat neurodegeneration and therefore need to be assessed in relevant models in order to evaluate the potential benefit for PD. Therefore, this study aimed to develop nanocarrier, nasal delivery systems for curcumin and/or DFO using the amphiphilic polymer, Pluronic F68 (P68) with or without, DQA to protect against rotenone-induced changes in cell viability and oxidative stress in SH-SY5Y neuronal cells.
MATERIALS AND METHODS
Materials
Unless otherwise stated, all chemicals were analytical grade. Dequalinium chloride hydrate (DQA; 95%), Dulbecco’s phosphate buffered saline (PBS), Curcumin from Curcuma longa (Turmeric), powder (≥80%), Deferoxamine mesylate salt (92.5%), Protease inhibitor cocktail (PIC), Thiazolyl Blue Tetrazolium Blue (MTT), Dimethyl sulfoxide (DMSO), 2′,7′-Dichlorofluorescin diacetate (DCFH-DA) and 2,2′-Azobis(2-methylpropionamidine) dihydrochloride (ABAP) were purchased from Sigma-Aldrich, U.K. Methanol (HPLC grade), L-glutamine, fetal bovine serum (FBS), Dulbecco’s Modified Eagle Medium (DMEM) Glutamax®, Minimum Essential Media (MEM) and 100×antibiotic-antimycotic were supplied by Fisher Scientific, U.K. The TBARS Parameter Assay Kit was purchased from R&D Systems, Parameter TM, UK. The BCA protein assay kit was from Pierce (Thermo Fisher Scientific, UK). SH-SY5Y cells were purchased from the American Type Culture Collection (ATCC CRL-2266, USA). Flasks were from Nunc, Denmark and culture plates from Corning, UK.
Development and characterisation of nanoformulations
Preparation of curcumin and/or deferoxamine nanoformulations
All nanoformulations were prepared using a modified thin-film hydration method [25, 29]. Briefly, P68 nanocarriers with or without DQA, curcumin and/or deferoxamine (DFO) at different ratios (Table 1) were formulated using a rotary evaporator (Hei-VAP Advantage Rotary Evaporator, Heidolph, Germany) at 200 rpm and 80°C under vacuum, using methanol as the solvent. The resultant thin film was hydrated with 10 ml of distilled water and mixed thoroughly at 80°C for 1-2 min and sonicated using a VWR Ultrasonic cleaner bath USC300T (VWR International Limited, U.K.) for a further 1 min until the film was fully removed and dissolved in the water. In order to remove any unloaded curcumin and/or DFO, the obtained formulation was filtered through a sterile 0.22 μm filter (Millex-MP, Millipore, Carrigtwohill, Ireland). In order to store samples for further analysis, some samples were freeze dried (lyophilized) using a Virtis AdVantage 2.0 BenchTop freezedryer (SP Industries, UK).
Hydrodynamic Diameter (d), Polydispersity Index (PDI), Surface Charge, Drug Loading (DL), and Encapsulation Efficiency (EE) of drug-loaded P68 and P68 + DQA nanoformulations prepared at 80°C (mean±S.D. n = 6)
Size and surface charge of the nanoformulations
The size and surface charge of prepared nanoformulations were measured following filtration using the Zetasizer Nano ZS (Malvern Instruments, U.K.). Size distribution was measured via photon correlation spectroscopy as Z-Ave hydrodynamic diameter and polydispersity index (PDI). Transmission electronmicroscopy (TEM) analysis was performed to analyse morphology (size and shape) of the nanoformulation complexes. The surface charge was measured by laser Doppler micro-electrophoresis. All experiments were performed in triplicate.
Determination of drug loading and encapsulation efficiency
UV-Visible (UV-Vis) spectroscopy was employed to study drug loading and encapsulation efficiency of the nanoformulations based on the calibration curves of free curcumin and DFO. Methanol and water were added in a 1:1 ratio to dissolve the carrier in order to release the drug and achieve the theoretical concentration of 10 μg/ml curcumin or 20 μg/ml DFO. Curcumin and DFO content was calculated using UV-vis spectroscopy at 423 nm and 204 nm respectively. The percentage of drug loading and encapsulation efficiency were calculated using the following equations: Drug loading (%) = (determined mass of drug within nanocarriers / mass of drug-loaded nanocarriers)×100 Encapsulation efficiency (%) = (determined mass of drug within nanocarriers / theoretical mass of drug within nanocarriers)×100
Assessing the therapeutic potential of nanoformulations in a model of PD
Cell culture and lysate harvesting
The human neuroblastoma SH-SY5Y cell line was used to create and in vitro model of PD (reviewed in Xicoy et al. [30]). SH-SY5Y cells were grown in DMEM - Glutamax®, pH 7.4 supplemented with 10% FBS and 1% antibiotic/antimycotic in a 5% CO2 environment at 37°C. SH-SY5Y cells were thawed and left to grow in plastic T75 (75 cm2) flasks until they reached 70% confluence. Adherent cells were then detached from the surface of the flasks via trypsinisation, counted and seeded into well plates at specific numbers according to the bioassay being performed (6-well or 96-well plates). To produce a model of PD, SH-SY5Y cells were treated with 100 μM rotenone for 24 h in order to reduce cell viability by approximately 50% (Supplementary Figure 1).
Cell viability: MTT assay
The protective properties of drug-loaded nanocarriers against rotenone-induced reduction in cell viability was assessed using the MTT Assay. Briefly, SH-SY5Y cells grown at a seeding density of 1,000,000 cells/cm2 in 96 well plates until confluent. The cells were pre-treated for 3 h with either free or nanoformulated curcumin (5 and 10 μM), DFO (100 μM) or combined curcumin (5 or 10 μM) and DFO (50 or 100 μM), or with corresponding unloaded, blank formulations. SH-SY5Y cells were then treated with 100 μM rotenone for 24 h, each well containing a total volume of 200 μl. The cells were then incubated with an additional 20 μl of 5 mg/ml solution of MTT DPBS solution for 4 h at 37°C. Following aspiration, 100 μl of DMSO was added to each well to dissolve the formazan crystals. To ensure DMSO was mixed well, plates were placed on a shaker at 75 rpm for 15 min and the absorbance was read at 570 nm on a spectrophotometer.
Lipid peroxidation: TBARS assay
The thiobarbituric acid-reactive-substances (TBARS) assay was used to assess ROS generated oxidative stress, specifically lipid peroxidation [31, 32]. Briefly, SH-SY5Y cells were grown in 6-well plates until confluent. Following pre-treatment with the relevant free or nanoformulated curcumin and/or DFO, the cells were incubated with 100 μM rotenone for 24 h. Cells were then washed once with DPBS and lysed at 4°C as previously described by Zariwala et al. [33], using 350 μl ice-cold lysis buffer (50 mM NaOH supplemented with 1 μg/ml protease inhibitor cocktail - PIC) whilst rocking gently for 40 min in ice trays on a plate shaker (8 rpm). Cell lysates were then collected using sterile cell scrapers and aliquoted into microcentrifuge tubes ready for further analysis. The lysate total protein concentrations were determined using the Pierce BCA kit with BSA as standard. The TBARS assay was carried out in accordance to manufacturer guidelines (R&D Systems, Parameter TM). Briefly, freshly prepared TBA was added to TBARS acid-treated cell lysate samples (loaded in triplicate) and incubated at 60°C for 2.5 h. The samples were read at absorbance 532 nm before and after incubation to estimate the formation of TBARS.
Cellular antioxidant activity: CAA assay
The cellular antioxidant activity was measured using the method described by Chen et al. [34] which is a slightly modified version of the assay developed by Wolfe et al. [35] and Hu et al. [36]. SH-SY5Y cells were seeded in black-walled, clear-bottom 96-well microplates. Once confluent, cells were washed with DPBS and treated with different concentrations of 200 μl of drug-loaded nanocarriers, free drug solution or MEM only (control) for 1–3 h at 37°C. Cells were then washed with MEM and treated with 200 μl of 100 μM DCFH-DA (dissolved in MEM) and incubated for a further 30 min at 37°C. Following aspiration, each well was treated with 100 μl of 600 μM ABAP (dissolved in MEM). The fluorescence of the cells in the 96-well plate was read every 5 min for 1 h at 528 nm and 485 nm emission and excitation wavelengths (respectively).
Statistical analysis
For all experiments the mean of six replicates was calculated for each treatment, and the data expressed as mean±standard deviation (S.D.). The MTT and TBARS assay results were statistically analysed using one-way analysis of variance (ANOVA) followed by Dunnett’s T3 post hoc test. A two-way ANOVA followed by Tukey’s multiple comparisons post hoc test was used to analyse the CAA assay results (PRISM software package, Version 7, Graphpad Software Inc., San Diego, USA).
RESULTS
All P68 nanoformulations demonstrated high encapsulation efficiency (66–95%), DFO-loaded P68 nanocarriers had the highest mean encapsulation efficiency (95%), curcumin-loaded nanocarriers had 79% and the combined curcumin (67%) and DFO (66%) nanocarriers had the lowest encapsulation efficiencies (Table 1). The mean P68 nanocarrier size was <200 nm in all cases, however curcumin-loaded nanocarriers had the largest particle sizes (Table 1, Fig. 1). All P68 nanoformulations had low polydispersity as represented by mean polydispersity indices <0.22 suggesting that the majority of the nanocarriers within the formulation are of a similar size (Table 1). All P68 nanocarriers had similar low negative surface charges (Table 1). The addition of DQA to formulate the P68+DQA nanocarriers did not significantly change the particle size, however it did result in more positive surface charges (0.02 to 9.29) and in all cases appears to have increased encapsulation efficiency; curcumin-loaded nanocarrier encapsulation efficiency increased by 7%, DFO-loaded nanocarriers increased by 1% and combined curcumin and DFO-loaded nanocarriers had the largest increase of 10–20% in for each drug, from 67% and 66% to 82% and 96%, respectively (Table 1).

TEM images of P68 and P68 DQA drug-loaded nanocarriers. Scale = 500 nm in all cases.
24-h treatment with 100 μM rotenone significantly reduced SH-SY5Y cell viability by 40–50% in all experiments (Fig. 2A, B, Supplementary Figure 1). Cytotoxicity was not observed for the tested concentrations of any nanoformulation ≥80% (n = 6). When comparing the effects of free drug and P68 nanocarriers on cell viability following rotenone treatment, a significant difference in mean cell viability was observed between the different treatments (F(16, 94) = 93.97, p < 0.0001) (Fig. 2A). 3-h pre-treatment of P68 nanocarriers of 5 μM and 10 μM curcumin (p = 0.0039 and p = 0.0078, respectively), 100 μM DFO (p = 0.0002) and combined 5 μM curcumin and 50 μM DFO (p = 0.0029) significantly protected against the reduction in cell viability induced by 24-h treatment with 100 μM rotenone (Fig. 2A). Of the P68 nanoformulation conditions which were protective, no single condition was significantly more protective than another. None of the free drug pre-treatment conditions apart from combined 5 μM curcumin and 50 μM DFO (p = 0.0219) were able to significantly protect against rotenone and pre-treatment with the combination of free curcumin (10 μM) and free DFO (100 μM) followed by rotenone treatment appears to be more toxic than rotenone alone (p = 0.0003) (Fig. 2A). There was no significant difference in cell viability following pre-treatment with P68 nanocarriers compared to the corresponding free drug pre-treatments apart from with 10 μM curcumin + 100 μM DFO where pre-treatment with P68 nanocarriers resulted in higher cell viability (p = 0.0032).
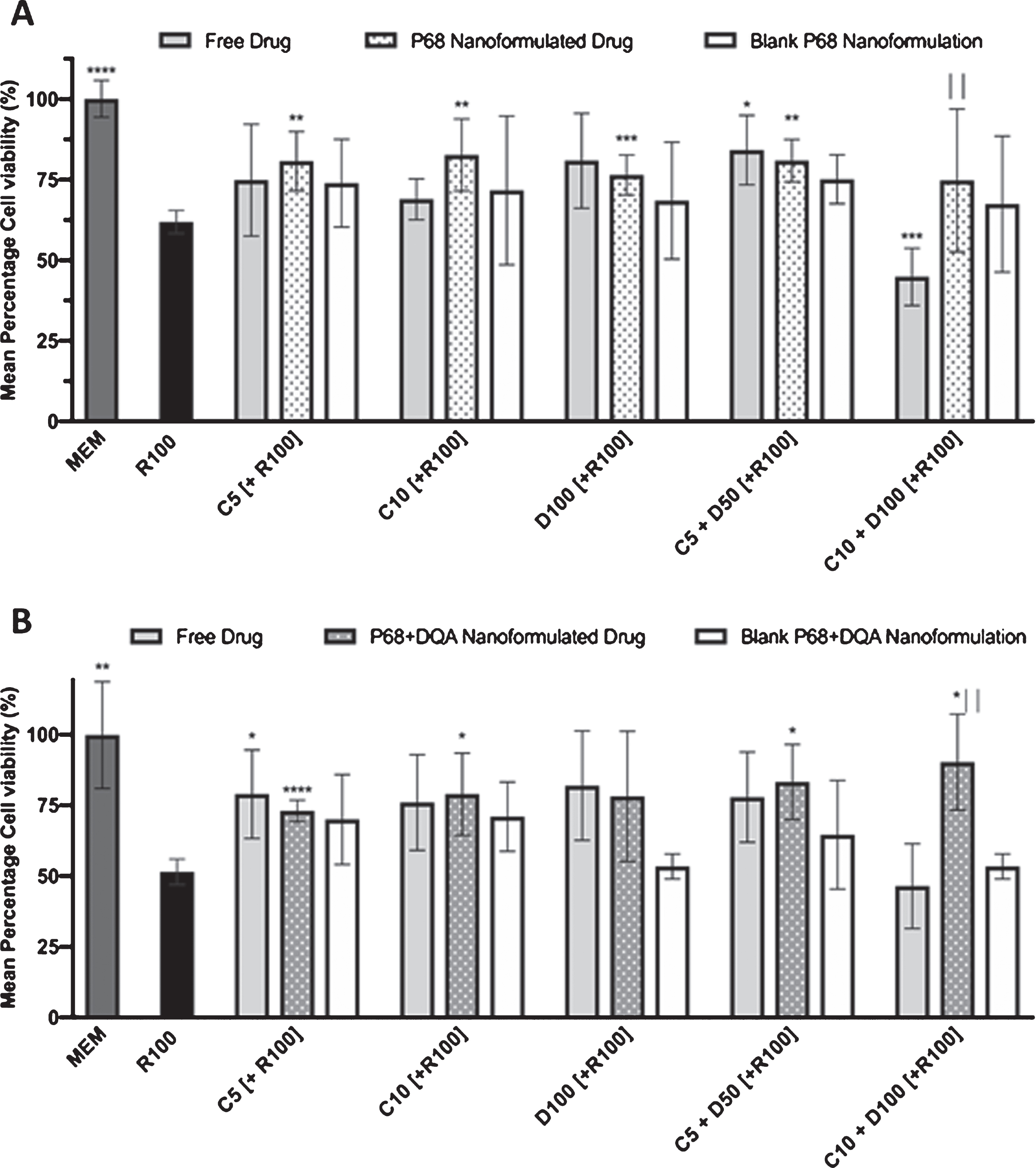
A) MTT assay results of 3-h pre-treatment with free drug, P68 nanoformulated or corresponding blank preparations of either curcumin (C5, C10), deferoxamine (D100) or combined curcumin and deferoxamine (C5+D50, C10+D100) followed by 24-h treatment with 100 μM rotenone (R100) compared to R100 treatment alone. MEM represents the control condition where cells were only treated with media, no pre-treatment nor R100 treatment (n = 6). B) Corresponding MTT assay results for P68+DQA nanoformulated pre-treatments (n = 6). *represents significance values of control or pre-treatment conditions compared to R100 treatment alone (****p < 0.0001, ***p < 0.001, **p < 0.01, *p < 0.05). |represents significance values of nanoformulated drug compared to free drug within the same treatment condition (||p < 0.01).
Similarly, there was a significant difference in mean cell viability between the different treatments when comparing free drug and P68+DQA nanocarrier pre-treatments prior to rotenone treatment (F(16, 60) = 5.639, p < 0.0001) (Fig. 2B). The majority of P68+DQA nanoformulation pre-treatments were able to protect against rotenone, (5 μM curcumin (p < 0.0001), 10 μM curcumin (p = 0.0331), 5 μM curcumin + 50 μM DFO (p = 0.0104), 10 μM curcumin + 100 μM DFO (p = 0.014)), including the combination for 10 μM curcumin and 100 μM DFO which was not protective in the P68 or free conditions (Fig. 2B). However, P68+DQA nanocarriers of 100 μM DFO did not significantly protect against rotenone. In these results, free 5 μM curcumin pre-treatment was also able to significantly protect against rotenone (p = 0.0455). Similar to when using P68 nanocarriers, when comparing P68+DQA nanocarriers with the corresponding free drug pre-treatments, there was only a significant difference found for 10 μM curcumin + 100 μM DFO where pre-treatment with P68+DQA nanocarriers resulted in higher cell viability (p = 0.004).
A significant difference in mean TBARS concentrations was observed between the different treatments when testing both P68 (F(16, 84) = 35.36, p < 0.0001) and P68+DQA (F(16, 17) = 41.55, p < 0.0001) nanocarriers compared to the corresponding free drugs (Fig. 3A, B). 3-h pre-treatment of all preparations (free drug, P68 and P68+DQA nanoformulations) of all conditions (5 μM curcumin, 10 μM curcumin, 100 μM DFO, 5 μM curcumin + 50 μM DFO, 10 μM curcumin + 100 μM DFO) significantly protected against rotenone-induced lipid peroxidation, measured by TBARS concentration (Figs. 3A and 4B). 3-h pre-treatment with P68 nanocarriers of 5 μM and 10 μM curcumin (p = 0.0012 and p = 0.0135, respectively), 100 μM DFO (p = 0.0012) and the combination of 5 μM curcumin and 50 μM DFO (p = 0.0007) were significantly more protective against rotenone-induced increase in TBARS concentration compared to the corresponding free drug conditions (Fig. 3A). There was no significant difference in the ability of P68+DQA nanocarriers and the corresponding free drug pre-treatments to protect against rotenone-induced lipid peroxidation (Fig. 3B).
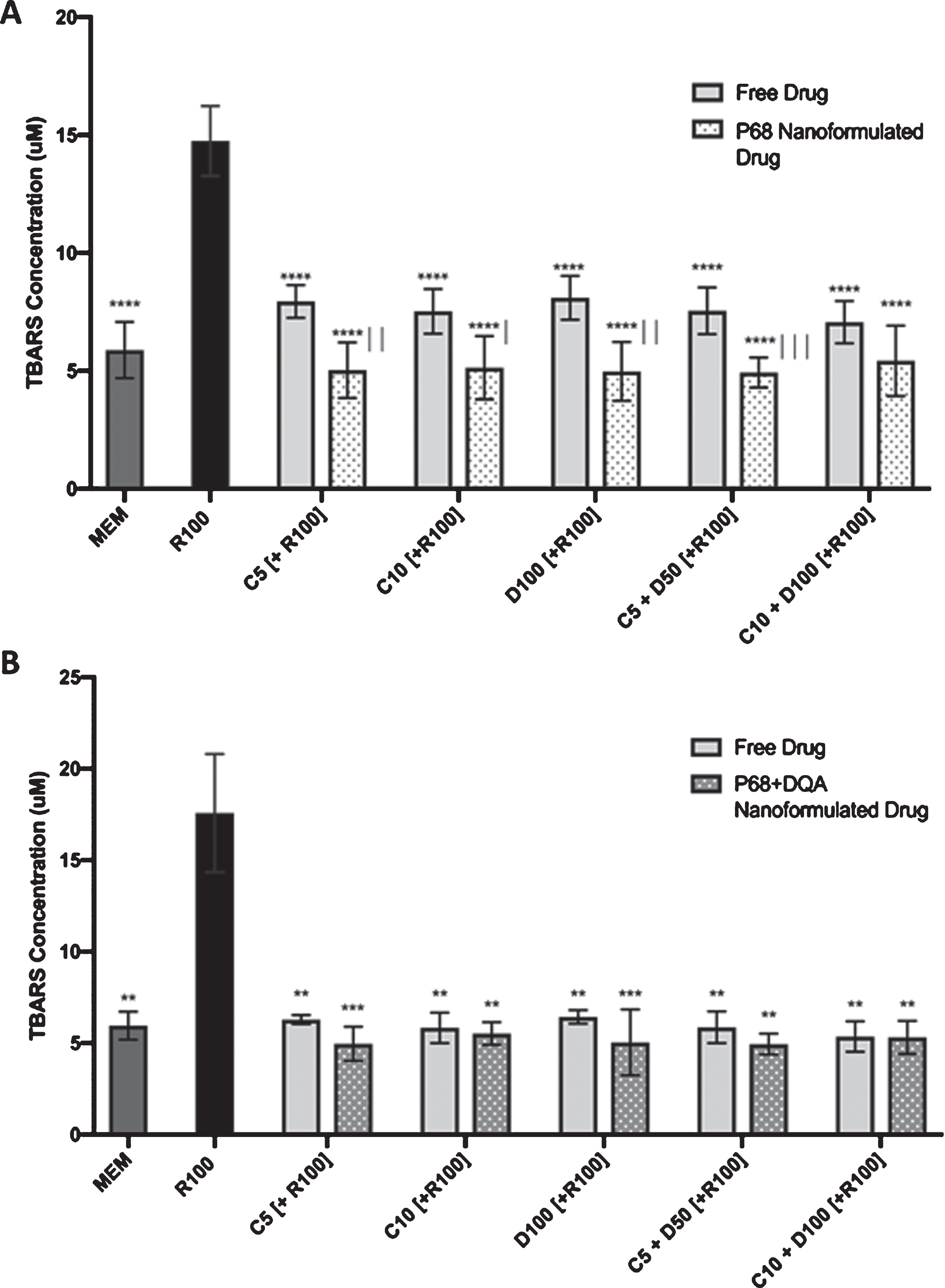
A) TBARS assays results of 3-h pre-treatment with free drug or P68 nanoformulated preparations of either curcumin (C5, C10), deferoxamine (D100) or combined curcumin and deferoxamine (C5+D50, C10+D100) followed by 24-h treatment with 100 μM rotenone (R100) compared to R100 treatment alone. MEM represents the control condition where cells were only treated with media, no pre-treatment nor R100 treatment (n = 6). B) Corresponding TBARS assay results for P68+DQA nanoformulated pre-treatments (n = 6). *represents significance values of control or pre-treatment conditions compared to R100 treatment alone (****p < 0.0001, ***p < 0.001, **p < 0.01). |represents significance values of nanoformulated drug compared to free drug within the same treatment condition (|||p < 0.001, ||p < 0.01, |p < 0.05).
The CAA results showed significant differences in cellular antioxidant activity between the different treatment preparation types (F(4, 100) = 27.78, p < 0.0001) and the different concentrations of treatments (F(3, 100) = 26.78, p = p<0.0001) (Fig. 4A, B). P68+DQA drug loaded nanocarriers had significantly higher antioxidant capability than P68 nanocarriers in all corresponding conditions except 100 μM DFO where there was no significant difference (Fig. 4A). The highest difference in antioxidant activity between these formulations was at 10 μM curcumin concentrations, where P68+DQA curcumin-loaded nanocarriers had 63% higher antioxidant activity compared to P68 curcumin-loaded nanocarriers (p < 0.0001) (Fig. 4A). Likewise, P68+DQA drug loaded nanocarriers had significantly higher cellular antioxidant activity compared to P68 drug loaded nanocarriers at 5 μM curcumin (48%, p = 0.001), 5 μM curcumin + 50 μM DFO (48%, p = 0.0012), 10 μM curcumin + 100 μM DFO (30%, p = 0.0063) concentrations. 100 μM DFO P68+DQA nanocarriers also had higher antioxidant activity, 21% higher than P68 DFO-loaded nanocarriers at this concentration, although not significant (Fig. 4A). P68+DQA nanocarriers were also significantly superior in cellular antioxidant activity compared to the corresponding curcumin free drug+DQA conditions (5 μM and 10 μM curcumin (p = 0.0003 and p = 0.0022 respectively) (Fig. 4A). There was no significant difference in cellular antioxidant activity of P68+DQA nanocarriers compared to any of the corresponding free drug alone conditions. There was also no significant difference between P68 nanocarriers and free drug+DQA for any of the conditions apart from 10 μM curcumin + 100 μM DFO where free drug+DQA was superior (p = 0.0337) (Fig. 4A). However, in the majority of cases the free drug conditions had a significantly higher cellular antioxidant unit than the corresponding P68 nanocarriers (10 μM curcumin (p = 0.0083), 100 μM DFO (p = 0.0343), combination of curcumin and DFO for both 5 μM + 50 μM (p = 0.0203) and 10 μM + 100 μM (p = 0.0082), respectively) (Fig. 4A). In all preparations, free drug, free drug+DQA, P68 and P68+DQA, the combination of 10 μM curcumin and 100 μM DFO had the most antioxidant potential compared to the majority of other conditions (Fig. 4B).
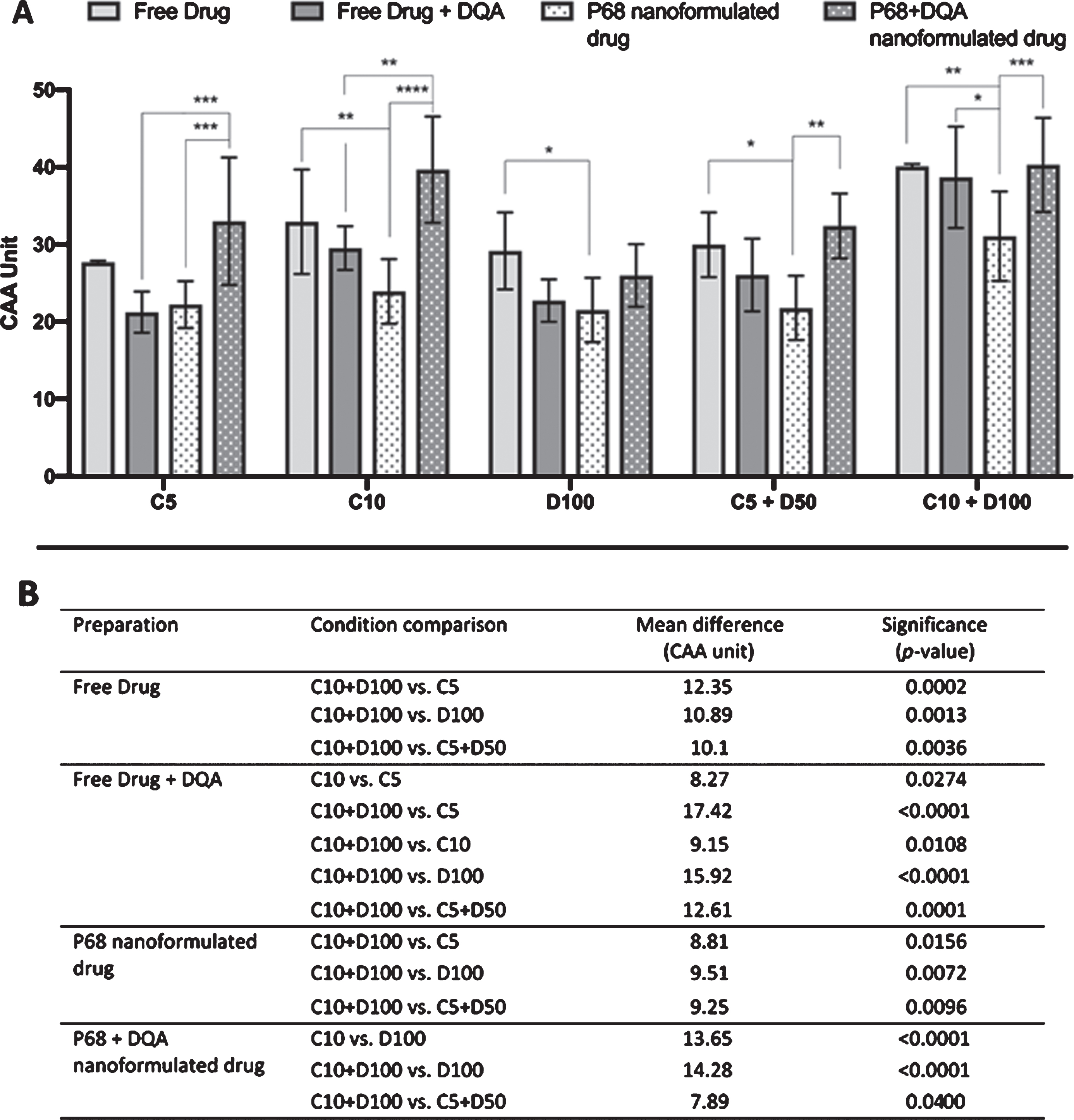
A) CAA assay results for free drug, free drug + DQA, P68 & P68 DQA preparations of curcumin (C5, C10), deferoxamine (D100) and combined curcumin and deferoxamine (C5+D50, C10+D100). *represent significance values within drug conditions (****p < 0.0001, ***p < 0.001, **p < 0.01, *p < 0.05). B) A table to show the corresponding significant CAA results between drug conditions of the same treatment preparation.
DISCUSSION
There is much evidence suggesting that curcumin and DFO are protective in numerous models of PD [12–17] and that the combination of such antioxidants and iron chelators may have even more potent effects [18]. However, the potential of these compounds as possible therapies for PD has been limited due to numerous factors such as brain penetrance, bioavailability and stability [19, 20]. Advances in nanotechnology have provided a possible solution to these issues as nanocarriers can be developed to retain the potency of the entrapped molecule while enhancing targeted delivery and passage across biological membranes [24, 38]. The aim of this study was to develop nanocarrier, nasal delivery systems for curcumin and DFO as well as the combination of the two and assess the ability of such nanocarriers to protect against reduced cell viability and increased oxidative stress in a cellular rotenone model of PD.
Nanocarriers were developed using the amphiphilic polymer P68 as it has been successfully used to develop micellar nanocarriers of numerous compounds [39–41] including curcumin for other indications [42–45]. Mitochondrial targeting of nanocarriers using dequilinium has been established [25, 46] therefore, the addition of dequilinium to the nanoformulation was used to assess whether mitochondrial targeting would result in increased potency. Curcumin and DFO were successfully incorporated into both P68 and P68+DQA nanocarriers (Table 1). The mean size of both P68 and P68+DQA nanocarriers was fairly consistent, in both cases curcumin-loaded nanocarriers had a larger mean size than DFO-loaded nanocarriers, and the addition of DFO to curcumin further increased nanocarrier size but all were under 200 nm (Table 1, Fig. 1). Such results are consistent with the ability to cross the BBB [37, 47].
The mean surface charge of the P68 nanocarriers was relatively neutral, between –0.44 and –5.88 mV (Table 1). This may also be beneficial for brain penetrance because most nanocarriers with a low to moderate surface charge between –1 mV and –15 mV have been shown to pass the BBB [48–51]. The addition of DQA to the formulations resulted in a moderate increase in charge to a more positive state (+0.02 to +9.29 mV), consistent with previous use of DQA in lipid-based formulations [25], however to a much lesser extent as such studies reported charges up to +50 mV. This only slight-moderate increase in charge is more suitable for brain delivery as overall such charges are relatively neutral, not so positive to cause toxicity to the BBB, known to occur at high positive charges [52], but a large enough shift in charge to potentially improve physical stability [25]. Further to this, drugs are more likely to be able to pass the BBB if they are not rapidly cleared from the bloodstream [38] and neutral nanocarriers have been shown to have longer circulation time in the bloodstream compared to those that are very negatively or very positively charged [53].
P68 and P68+DQA nanocarriers were protective against rotenone-induced reductions in cell viability suggesting that they may be able to protect against cell death induced by rotenone. It was expected that the combination of the highest tolerable concentrations of curcumin and DFO would be the most effective at protecting against rotenone due to the combination of reduced free iron and increased antioxidants which together should prevent the accumulation of toxic free radicals which drive oxidative stress. However, pre-treatment with the combination of these free drugs appeared to result in toxicity following rotenone treatment (Fig. 2A, B). That said, following the combined formulation of 10 μM curcumin and 100 μM DFO into P68 and P68+DQA nanocarriers, pre-treatment with the combined drugs not only removed the toxicity previously observed but also significantly increased cell viability (Fig. 2A, B). P68 nanocarriers of 10 μM curcumin and 5 μM curcumin + 50 μM DFO were the most successful at protecting cell viability against rotenone compared to the other drug-loaded P68 nanocarriers, retaining cell viability over 80% (Fig. 2A). Whereas, increasing concentrations of P68+DQA curcumin and combined curcumin + DFO nanocarriers increasingly protected cell viability, with the pre-treatment of 10 μM curcumin + 100 μM DFO retaining 90% cell viability (Fig. 2B). Although DFO-loaded P68+DQA nanocarriers did not significantly protect cell viability against rotenone, the addition of 50 μM and 100 μM DFO to 5 μM and 10 μM curcumin-loaded P68+DQA nanocarriers resulted in a modest increase in protection, by 10% and 11%, respectively.
Similarly, all P68 and P68+DQA drug-loaded nanocarriers protected against rotenone-induced lipid peroxidation, in all cases maintaining TBARS levels equivalent to control conditions (Fig. 3A, B). The results suggest that P68 but not P68+DQA drug-loaded nanocarriers are significantly more protective than the corresponding free drug pre-treatments (Fig. 3A, B). However, it appears that the free drug preparation of all conditions resulted in lower TBARS concentrations in the experiment using the P68+DQA nanocarriers compared to when using P68 nanocarriers. This could be due to the cells having a higher starting TBARS concentration in the P68 experiment as measured by the control cells which did not receive any pre-treatment or rotenone treatment.
The cellular antioxidant activity results suggest that P68+DQA drug-loaded nanocarriers of all conditions have more potent antioxidant potential than P68 drug-loaded nanocarriers. The addition of DQA to the formulations resulted in a significant increase from 48% to 65% in cellular antioxidant activity in all conditions apart from 100 μM DFO where 20% higher antioxidant activity was observed but did not reach statistical significance (Fig. 4A). This suggests that the addition of DQA, which is known to target mitochondria [25, 46], significantly improves the antioxidant ability of the formulations. This result was anticipated as mitochondria are the main site of intracellular free radical formation [7, 28]. Furthermore, in nearly all preparations, the combination of 10 μM curcumin and 100 μM DFO had the most antioxidant activity (Fig. 4B), suggesting that the combination of increasing antioxidants and decreasing free iron may be the most successful strategy for reducing ROS production.
Overall, these results suggest that all drug-loaded nanocarriers were at least as capable as the corresponding free drug conditions at protecting against reduced cell viability and increased lipid peroxidation induced by rotenone. As the potential therapeutic value of the free curcumin and DFO is limited due to issues such as bioavailability and brain penetrance [19, 20], the nanocarrier delivery systems may provide a viable solution as the particle size and charge data indicated that these formulations would be able to pass the BBB. Moreover, the ability to target these non-specific drugs to mitochondria using P68+DQA nanocarriers seemed to result in increased cellular antioxidant activity. The outcome measures of these experiments focused mostly on antioxidant capability therefore it is unsurprising that the curcumin formulations were generally superior to DFO as curcumin is known to obtain antioxidant properties. However, for most preparations the addition of DFO to curcumin resulted in increased cellular antioxidant activity suggesting that a combination therapy may provide a good approach to combat oxidative stress in PD models. This is another benefit of using nanocarriers as, unlike combination delivery of free drugs which is challenging due to the distinct pharmacokinetic profiles of each drug, nanocarriers can ensure that drugs are delivered together for synergistic treatment [54].
In summary, this study demonstrates for the first time the formulation and delivery of P68 and P68+DQA curcumin and/or DFO nanocarriers to protect against oxidative stress induced by a cellular rotenone model of PD. This strategy may thus provide a novel approach to fully utilise their therapeutic benefit for PD. The next steps will be to assess additional parameters such as iron, dopamine and alpha synuclein prior to and following pre-treatment with these formulations. As these results are from a neurotoxin model, in a neuroblastoma cell line that do not only contain dopaminergic neurons, further experiments will be carried out to determine if the effects are seen in more accurate models of PD, for example using patient derived induced pluripotent stem cells and in vivo alpha synuclein models. Such additional data would be valuable in providing the evidence needed as to whether these formulations can move forward into clinical testing as potential disease-modifying therapies for PD.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
Leah Mursaleen was supported by the FST studentship grant, University of Westminster.
The authors wish to thank Andrew Weston and Stefanie Chan from University College London for their assistance in characterising the nanocarriers.
