Abstract
Background:
Cognitive impairment and cardiovascular dysautonomia are two major non-motor features of Parkinson’s disease (PD). They have been investigated separately and extensively, but their interactive outcomes have rarely been studied.
Objective:
The purpose of this study was to examine the association between central atrophy and cognition and to assess the influence of cardiovascular lability on this association in PD patients.
Methods:
Out of 151 early PD patients, 47 subjects were ultimately enrolled according to our selection criteria. Their cognitive status was examined by comprehensive neuropsychological tests assessing five domains of cognition. Supine and orthostatic blood pressures were recorded during head-up tilt tests, and orthostatic mean arterial pressure change was calculated. Every patient underwent brain magnetic resonance imaging, and intercaudate nucleus ratio was obtained as a central atrophy surrogate marker. The associations and interactions between central atrophy, cognition, and blood pressure variability were analyzed.
Results:
Among 47 subjects, 20 (42.6%) had orthostatic hypotension. Attention/working memory, executive function, and delayed recall were inversely associated with central atrophy (r = –0.332, p = 0.028; r = –0.314, p = 0.038; r = –0.399, p = 0.024; respectively). In a multiple regression model, only attention/working memory was independently associated with central atrophy when modulated by orthostatic mean arterial pressure change (p < 0.05).
Conclusion:
This study revealed that cardiovascular dysautonomia interacted with the inverse association between cerebral atrophy and cognition, and it reinforced its relationship. Interaction between these two non-motor features should be kept in mind in clinical practice, particularly in PD patients with co-morbid vascular factors.
INTRODUCTION
Diverse non-motor symptoms predate motor features in Parkinson’s disease (PD), and cardiovascular dysautonomia is one of them [1, 2]. Blood pressure lability includes orthostatic hypotension, supine hypertension, or loss of circadian blood pressure rhythm [3].
Cognitive impairment is also recognized at the time of PD diagnosis in as many as 42% of cases, and mild cognitive impairment (MCI) is hypothesized to be a prodromal phase of PD, depending on the spreading pattern of neuropathology [4–6].
Subcortical atrophies such as thalamus, caudate, and nucleus accumbens were associated with cognitive impairment in the domains of attention/working memory and executive function in mild PD [7, 8], and orthostatic hypotension was found to influence trouble concentrating [9]. Cognition in PD could accompany subcortical neural substrate changes, and it appears to be influenced by blood pressure instability. However, how their interactions might affect cognition have not been investigated in PD.
The hypothesis tested in this study was that cognitive dysfunction and subcortical atrophy were related to each other under moderation by cardiovascular autonomic dysfunction. We investigated the association between central atrophy and cognitive domains, and ultimately assessed how the cardiovascular dysautonomia influenced their relationship in de novo PD patients.
METHODS
Patients
This study was approved by the Institutional Review Board of our institution, and all subjects provided written informed consent to participate. All experiments were performed in accordance with relevant guidelines and regulations.
Newly diagnosed patients with PD who visited the movement disorder clinic of a university-affiliated hospital between April 2015 and April 2017 were enrolled. PD was diagnosed based on the UK Parkinson’s Disease Society Brain Bank clinical diagnostic criteria [10], and this diagnosis was supported by positron emission tomography imaging studies using 18F-N-(3-fluoropropyl)-2beta-carbon ethoxy-3beta-(4-iodophenyl) nortropane [11]. Clinical characteristics such as age, sex, disease duration, education, height, weight and histories of hypertension and diabetes mellitus were obtained. Motor features were evaluated with the Unified Parkinson’s Disease Rating Scale (UPDRS) Part III and modified Hoehn and Yahr (H&Y) stage scores.
Subjects with the following findings were excluded: (1) any neurological findings suggestive of atypical PD and secondary parkinsonism, (2) history of diabetic neuropathy or other etiology indicating autonomic neuropathy, (3) history of cardiac problems that affect blood pressure or with any abnormalities on the electrocardiogram or 24 h Holter monitoring and chest roentgenography, (4) history of peripheral arterial disease or symptoms of vascular claudication and warning signs of peripheral arterial blockage, (5) history of medications such as tricyclic antidepressants and benzodiazepines that influence autonomic and cognitive functions or those who were taking medications at the time of diagnosis known to influence central dopaminergic, noradrenergic, and/or serotonergic systems, (6) history of stroke and head trauma that influences cognition, (7) disease duration ≥5 years, Mini-Mental Status Examination (MMSE) <26, Clinical Dementia Rating (CDR) >0.5 [12–14] or impairment of significant functional independence [12, 15] and (8) family history of Alzheimer’s disease or other types of dementia.
One hundred and fifty-one early PD patients were initially recruited according to the inclusion and exclusion criteria. Presynaptic dopamine transporter uptake was deficient in the striatum in all patients. Among the screened patients, we excluded the subjects who did not complete a full battery of neuropsychological tests because of illiteracy or other physical and/or neuropsychological problems (n = 104).
Head-up tilt test
All patients were at a full resting state before the exam. Continuous electrocardiograph leads and non-invasive blood pressure monitoring equipment were applied to the patients (YM6000, Mediana Tech, Redmond, WA, USA). A supine position was maintained for 20 minutes before tilting to 60 degrees (ENRAF NONIUS, Rotterdam, The Netherlands). While in the supine position, blood pressure and heart rate were measured every 5 minutes for 20 minutes. At 60 degrees, blood pressure and heart rate were recorded at 0, 3, 5, 10, 15, and 20 minutes. The first supine blood pressure (at 0 minutes) was discarded, and maximal supine systolic and diastolic blood pressures were selected among the measurements at 5, 10, 15, and 20 minutes. The lowest systolic and diastolic values at 3 or 5 minutes during the tilted position were chosen. The orthostatic blood pressure changes of systole (Δ SBP) and diastole (Δ DBP) were calculated, and they were manipulated to estimate the change in mean arterial pressure while standing (Δ MAP).
Neuropsychological assessments
Global cognitive status was assessed by MMSE and CDR. Five domains of cognition [15, 16] were evaluated using a comprehensive neuropsychological battery, the Seoul Neuropsychological Screening Battery, 2nd edition [17]. The attention and working memory domains were examined with the Digit Span Forward, Korean-Trail Making Test-Elderly version (K-TMT-E) Part A, and the Korean-Color Word Stroop Test (K-CWST). Executive function was assessed with the Digit Span Backward, Controlled Oral Word Association Test (COWAT) appraising phonemic fluency, and the K-TMT-E Part B. Language domain was evaluated with the Korean-Boston Naming Test (K-BNT), and visuospatial domain was investigated with the Rey Complex Figure Test (RCFT). In the domain of memory, the Seoul Verbal Learning Test (SVLT) and RCFT were utilized for assessing its subdomains: immediate recall, delayed recall and recognition. The z-scores of each test were calculated for analyses. In domains with multiple z-scores, the average scores of each domain were defined as the representative values [15, 16]. The neuropsychological evaluations were performed before the dopaminergic treatments.
123I-metaiodobenzylguanidine (123I-MIBG) scintigraphy
123I-MIBG scintigraphy was performed using a dual-head camera equipped with a low-energy, high-resolution collimator (Siemens), and data were collected at 30-minute (early) and 2-hour (late) time points after the injection of 111 MBq of 123I-MIBG. A static image was obtained with a 128×128 matrix. Regions of interest were manually drawn around the heart and mediastinum. Tracer uptake was measured within each region of interest to calculate the heart-to-mediastinum (H/M) ratio. The lower limit of reference values for delayed H/M ratio was calculated to be 1.78 [18].
Magnetic resonance imaging acquisition and measurements
Brain magnetic resonance imaging (MRI) was acquired using a 3.0-T scanner (Magnetom Verio, Siemens, Erlangen, Germany). Axial T1-weighted MR images (FOV 220×220 mm2; ET 1.0; FA 70.0; TR 250.0; TE 3.1; Thickness, 5.0 mm) were obtained to measure intercaudate nucleus ratio (ICR) [19, 20] (Fig. 1). Fluid attenuated inversion recovery images (FOV 220×220 mm2; ET 13; FA 150.0; TR 9000.0; TE 99.0; Thickness, 5.0 mm) were utilized to score the severity of white matter hyperintensities [21]. The images were rated by an experienced radiologist who was blinded to patient clinical data.
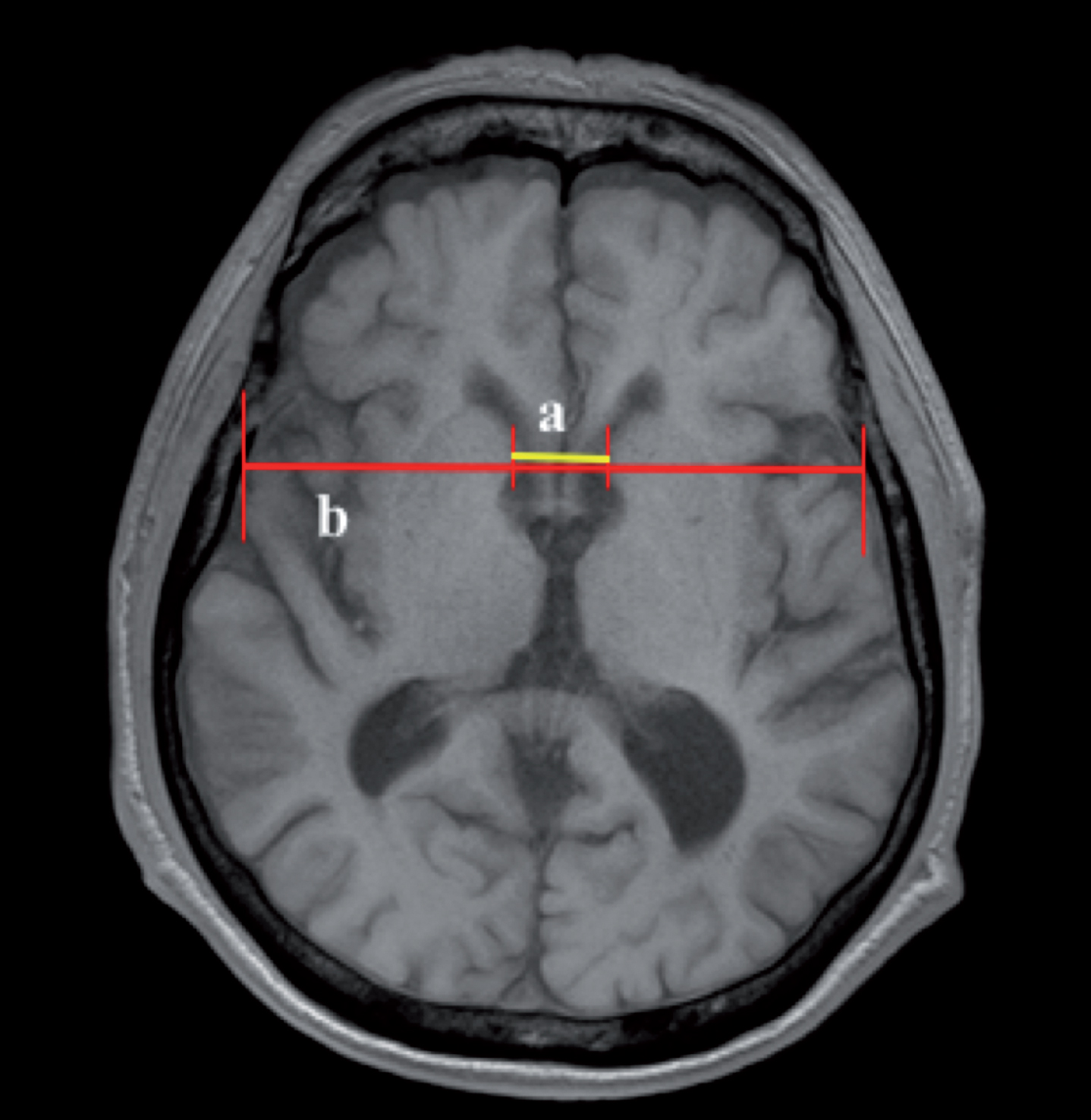
Axial T1-weighted image was obtained. On the caudal-most axial cut, the distance between the medial border of caudate nuclei (a) and the transverse width of the inner skull (b) were measured at the same level. Intercaudate nucleus ratio was calculated by dividing the intercaudate distance with the transverse width of the skull.
Laboratory investigations
Apolipoprotein E (ApoE) polymorphism was evaluated, and the subjects were categorized depending on the presence of the ApoE ɛ4 allele.
Statistical analyses
All statistical analyses were performed with the Statistical Package for the Social Sciences, version 24.0 for the Mac (SPSS, Inc.; Chicago, IL, USA). Descriptive statistics were applied to describe the baseline characteristics of forty-seven PD patients. Associations between the ICR and each neuropsychological test result were evaluated by partial correlation analysis after adjusting for age, disease duration and severity of white matter hyperintensities. In a moderation regression model [22], the moderator effect, Δ MAP, was investigated to determine how it influenced the central atrophy’s association with the cognitive domains. Cognitive domains were separately explored. ICR (step 1), Δ MAP (step 2), and their interaction (ICR×Δ MAP; step 3) were included in the regression model sequentially. When ICR and the interaction were of statistical significance, the Δ MAP was considered to have moderating effect on association between cognitive domain and central atrophy. Covariates of age, disease duration and severity of white matter hyperintensities were controlled in every step. Mean centering method was utilized to deal with multicollinearity. Statistical significance was defined as a two-tailed p-value <0.05. Multiple comparisons were not corrected because of the small number of study subjects and exploratory nature of the outcomes.
RESULTS
Clinical characteristics of the study population are described in Table 1. The mean age was 69.5±8.2 years and 22 (46.8%) were female. The median disease duration was 1.0 year (interquartile range [IQR]: 1.5 years). The UPDRS Part III score was 14.1±7.3 and the median modified H&Y score was 1.5 (IQR: 1.0). Forty subjects did not have any ApoE4 alleles (85.1%).
Clinical characteristics
PD, Parkinson’s disease; BMI, body mass index; MMSE, mini-mental status examination; CDR, clinical dementia rating; UPDRS, Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn and Yahr; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; H/M, heart-to-mediastinum; ApoE, apolipoprotein E.
The mean orthostatic Δ MAP and ICR were 8.9±6.4 mmHg and 0.129±0.029, respectively. Twenty patients (42.6%) had orthostatic hypotension, according to the 20/10 mmHg criteria [23], and 38 (80.9%) also had cardiac sympathetic denervation with delayed H/M < 1.78 (mean delayed H/M ratio: 1.46±0.35). The median total Scheltens’ score was 6.0 (IQR: 8.0).
The associations between ICR and neuropsychological tests are summarized in Table 2. Attention and working memory domains, the executive domain and delayed recall were negatively associated with increasing ICR (r = –0.332, p = 0.028; r = –0.314, p = 0.038 and r = –0.339, p = 0.024, respectively). The UPDRS Part III and Δ MAP were not associated with ICR (r = 0.176, p = 0.253 and r = –0.133, p = 0.391, respectively).
Partial correlation between intercaudate nucleus ratio and cognitive subtests
Adjusted for age, disease duration, and white matter change burden. K-TMT-E, Korean-Trail Making Test-Elderly version; K-CWST, Korean-Color Word Stroop Test; COWAT, Controlled Oral Word Association Test; K-BNT, Korean-Boston Naming Test; SVLT-E, Seoul Verbal Learning Test-Elderly version; RCFT, Rey Complex Figure Test. 1Average z-scores of Digit Span Forward, K-TMT-E: Part A and K-CWST. 2Average z-scores of Digit Span Backward, COWAT: Phonemic and K-TMT-E: Part B. 3Average z-scores of SVLT-E: Delayed recall and RCFT: Delayed recall. 4Average z-scores of SVLT-E: Recognition and RCFT: Recognition.
The orthostatic Δ MAP moderated both the central atrophy and cognitive dysfunction, adjusted for age, disease duration, and white matter burden (Table 3). ICR negatively predicted the cognition in the domains of attention and working memory, executive function and memory delayed recall (step 1, B: –11.868, –14.498 and –10.335, respectively; p < 0.05). However, when Δ MAP was included in the regression model (steps 2 and 3), the moderator (Δ MAP) and its interaction with ICR (Δ MAP×ICR) were only inversely related to attention and working memory domain at a statistically significant level (Δ R Square step 2, 0.131 and step 3, 0.073, respectively; p < 0.05). As the moderator increased, it gradually intensified the negative association between subcortical atrophy and attention/working memory function (Fig. 2).
Moderating effects of orthostatic Δ MAP on the association between ICR and cognition
*p < 0.05; **p < 0.01. step 1: covariates + ICR. step 2: step 1 +ΔMAP. step 3: step 2 + ICR×ΔMAP. ICR, intercaudate nucleus ratio; ΔMAP, orthostatic mean arterial pressure change; K-TMT-E, Korean-Trail Making Test-Elderly version; K-CWST, Korean-Color Word Stroop Test; COWAT, Controlled Oral Word Association Test; K-BNT, Korean-Boston Naming Test; SVLT-E, Seoul Verbal Learning Test-Elderly version; RCFT, Rey Complex Figure Test. Variables are controlled by covariates of age, disease duration and white matter change burden. Multicollinearity was controlled by the mean centering method. 1Average z-scores of Digit Span Forward, K-TMT-E: Part A and K-CWST. 2Average z-scores of Digit Span Backward, COWAT: Phonemic and K-TMT-E: Part B. 3Z-scores of K-BNT. 4Z-scores of RCFT. 5Average z-scores of SVLT-E: Delayed recall and RCFT: Delayed recall. 6Average z-scores of SVLT-E: Recognition and RCFT: Recognition.
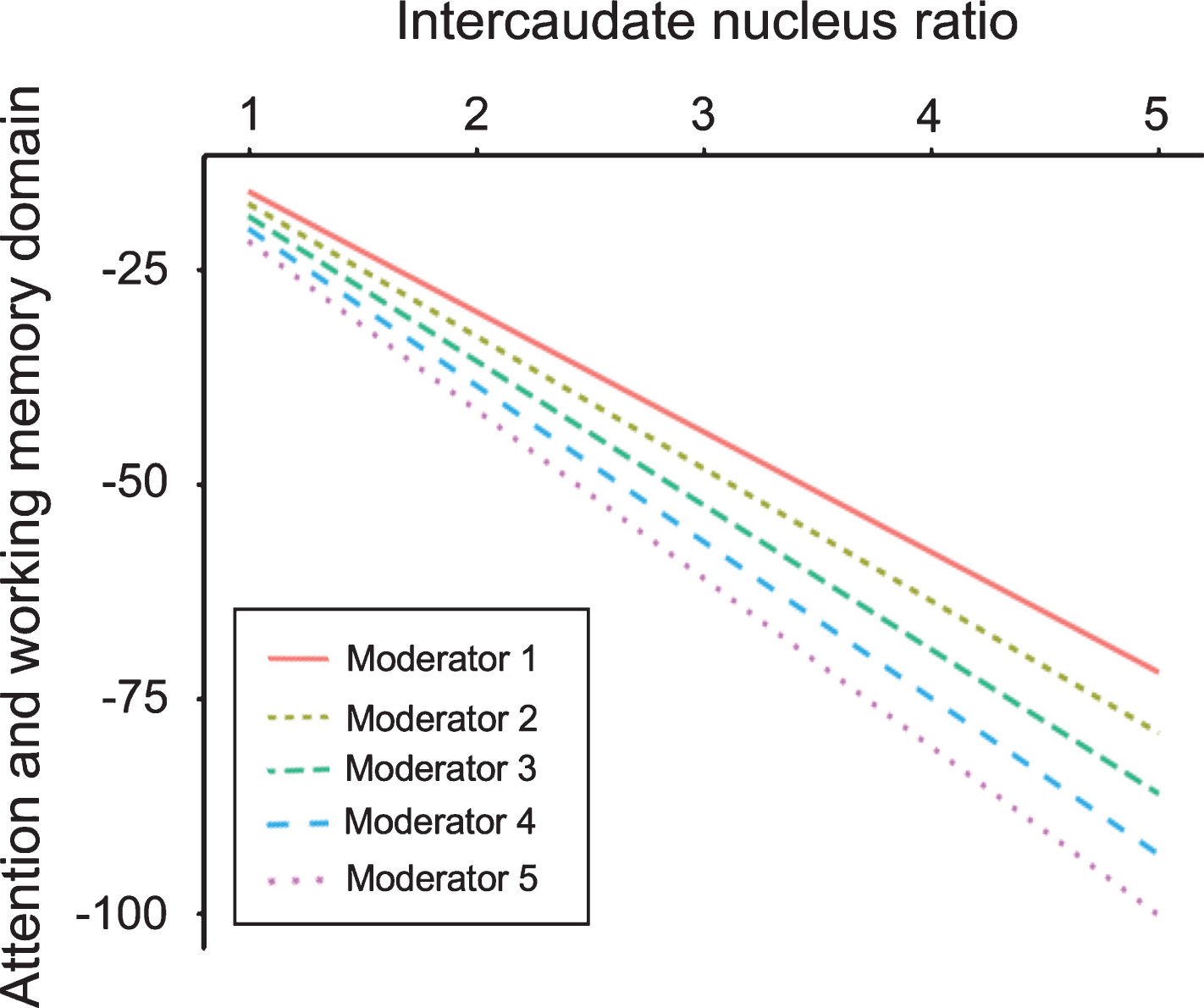
Moderating effects of orthostatic ΔMAP on the association between ICR and the attention/working memory domain. Y = –1.867766 + (–12.572745×ICR) + (–0.048694× ΔMAP) + [–1.401558× (ICR×ΔMAP)]. Y, attention and working memory domain; ICR, intercaudate nucleus ratio; ΔMAP, orthostatic mean arterial pressure change. Predictive variables were controlled by covariates of age, disease duration, and white matter change burden. Multicollinearity was handled by the mean centering method.
DISCUSSION
In this study, central atrophy was inversely correlated with attention/working memory, executive function and delayed recall domains, and independently predicted a negative influence on cognition. A steepening blood pressure drop was associated with worsening of attention and working memory through its moderation of the relationship between subcortical atrophy and cognition.
In accordance with previous studies [7, 8], central atrophy was correlated with the particular domains of cognition, when controlling for age, disease duration, and white matter hyperintensities. In early nondemented PD, the exact neural substrates for cognitive impairment are poorly understood [5, 24]. The “bottom-up” theory of the Braak schema [25] could be ascribed as dopamine deafferentiation was reflected in caudate atrophy [6, 26]. Thalamic involvement in PD could also explain the inverse relationship between central atrophy and cognition [27].
This study was designed to include only early nondemented PD cases to minimize the effects of advanced neuropathology on cortices. This selection was intended to assess the theoretical link between subcortical cognition and orthostatic blood pressure change. Despite controversies, central atrophy’s negative relationship with cognition implicates subcortical cognitive impairment in this study [7, 27].
This cohort revealed that attention/working memory, executive function, and delayed recall impairments were independently associated with central atrophy. All of these domains fall mainly into the frontostriatal/executive network [6, 29], which may be impaired at an early stage of PD. This outcome also corresponds to the results of a large longitudinal study that subtyped PD-MCI into “frontal executive” and “posterior cortical” impairments [30].
Autonomic failures in PD occur when blood pressure fluctuation exceeds its physiologic limits during orthostasis [3, 31], and cardiac sympathetic denervation plays a part in blood pressure instability [1, 32]. This blood pressure lability is reported to be related to cerebral structural changes [33–36]. Mean arterial pressure is an indirect measure of the cerebral perfusion pressure that determines cerebral flow, and its orthostatic change reflects the cerebral hemodynamic fluctuations that trigger cerebral changes in PD. In this study, the majority of the population had cardiac sympathetic denervation, implying unsteady perfusion to the brain. Amidst acquired inconstant blood perfusion, Δ MAP was analyzed.
This study demonstrated a null association between subcortical atrophy and orthostatic blood pressure change. It alternatively emphasized the role of the modulating effect of Δ MAP, which was established by the significant interaction between ICR and Δ MAP in the attention/working memory domain. The result could be interpreted such that Δ MAP modulated the functionality of subcortical structures within frontostriatal circuitry [28, 29]. This intensified the inverse relationship between central atrophy and cognition. Monoaminergic brainstem structures such as the locus coeruleus, pedunculopontine nucleus, ventral tegmental area, and substantia nigra are interconnected with the striatum, thalamus, and cortex to form a cognitive network [6, 28]. Their functional deficits on the striatum and thalamus become involved preferentially according to the Braak schema [25] and relate to frontal dysexecutive syndrome, as shown in this study. The already malfunctioning striatum and thalamus may become prone to cerebral perfusion fluctuations in PD, thus resulting in its independent negative moderating effect. This corroborates with a previous study reporting that trouble concentrating is related to the magnitude of the orthostatic blood pressure drop [9].
This retrospective study has several limitations. First, in order to select early PD patients without dementia and those who completed the comprehensive neuropsychological battery, many patients were excluded. The large number of exclusions might cause a selection bias. However, the stringent restriction allows a less contaminated assessment of associations between subcortical structures and cognition. Second, 85.1% (40/47) of subjects in this study were non-ApoE ɛ4 allelic. The ApoE ɛ4 genotype is strongly related to Alzheimer’s disease and it carries the risk of cognitive impairment in PD [6]. Because only a small portion of patients (7/47) had the ApoE ɛ4 allele, its effect was assumed to be minimal, limiting cortical involvement in the overall analysis in this study. Further studies would be necessary to investigate the potential effects of ApoE polymorphism on cognition and blood pressure. Third, the analyses would be more elaborate if the individual interactions of subregions such as the caudate, putamen, and thalamus with hemodynamics were investigated separately [8]. However, cognition is a result of the neural network that includes the thalamus and basal ganglia [27, 28], rather than each distinct neural substrate functioning alone. The intercaudate nucleus ratio is a broad measure of the central region [20]. A collective, easily measurable scale such as ICR might be a suitable alternative for evaluating the role of subcortical interconnectivity in cognition. Finally, to demonstrate a significant causal interaction between subcortical substrates and blood pressure lability on cognition, a longitudinal study with a larger sample such as the CamPaIGN cohort is required [30]. The results of this study could be expanded by a large prospective design.
In conclusion, it was confirmed that even from an earlier stage of PD, blood pressure lability influences cognition. Non-motor features do not act separately, but intermingle with each other, which complicates the clinical picture. In clinical practice, PD patients with comorbid hypertension are on anti-hypertensive medications. No consensus has been reached on how to treat blood pressure in PD patients. This result suggests that antihypertensives have potential to aggravate cognition in patients with PD who have blood pressure lability. The interaction of cognition and blood pressure should be incorporated into treatment decisions.
CONFLICTS OF INTEREST
The authors declare no financial or other conflicts of interest.
Footnotes
ACKNOWLEDGMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2017R1D1A1B06028086). The funder had no role in study design, data collection and analysis, the decision to publish or manuscript preparation.
