Abstract
Background:
There is evidence that both high and low frequency rTMS may have therapeutic effects on motor performance of Parkinson’s disease.
Objective:
The aim of the study was to conduct the first direct comparison of the two approaches.
Methods:
52 PD patients were randomly classified into two groups. The first group received 20 Hz and the 2nd group received 1 Hz rTMS with a total of 2000 pulses over M1of each hemisphere for ten days. Effects were assessed with the Unified Parkinson’s Disease Rating Scale part III (UPDRS), Instrumental Activity of Daily Living (IADL), and a self-assessment score (SA) before, after the last session, and one month later. Cortical excitability was measured before and after the end of sessions.
Results:
There was a significant improvement on all rating scales after either 1 Hz or 20 Hz rTMS, but the effect persisted for longer after 20 Hz (treatment X time interaction for UPDRS and IADL (P = 0.075 and 0.04, respectively). Neither treatment affected motor thresholds, but 20 Hz rTMS increased MEP amplitude and the duration of transcallosal inhibition. In an exploratory analysis, each group was subdivided into akinetic-rigid and tremor dominant subgroups and the effects of 1 Hz and 20 Hz treatment recalculated. There was weak evidence that patients with an akinetic-rigid presentation may respond better than those with predominant tremor.
Conclusion:
Both 20 Hz and 1 Hz rTMS improve motor function in PD, but 20 Hz rTMS is more effective.
INTRODUCTION
The therapeutic usefulness of various forms of non-invasive forms of brain stimulation has been examined in many studies in Parkinson’s disease (PD), and has been the subject of a number of recent reviews and meta-analyses [1–4]. But because of the variation in protocols (e.g., repetitive transcranial magnetic stimulation (rTMS) or transcranial direct current stimulation (TDCS), stimulation parameters (e.g., anodal or cathodal TDCS; high or low frequency rTMS), as well as the targeted cortical sites (e.g., motor cortex (M1), supplementary motor area, dorsolateral prefrontal cortex) it is very difficult to come to a clear conclusion about efficacy.
Zanjani et al. [5] recently conducted a meta-analysis of the effects of one of the commonest protocols and sites of therapeutic stimulation, rTMS on M1. Their analysis concentrated on studies that provided longer-term follow-up data (1 month) as well as assessments of activity of daily living since they regarded both items as essential measures of clinical outcome. The conclusion was that the evidence was suggestive of a small positive effect on clinical outcome. However, they pointed out that in order to have sufficient data to conduct the analysis they had combined outcomes from trials using both low (usually 1 Hz) and high (usually 20 Hz) frequency rTMS. This may have increased the variation in outcome since there is evidence from studies in healthy individuals that these frequencies may have different effects on M1. Indeed, in an early study we [6] had observed that treatment with 25 Hz rTMS was superior to 10 Hz rTMS, consistent with the idea that the effectiveness of rTMS to motor cortex may depend on the stimulus frequency. Zanjani et al. [5] also concluded that in addition to providing overall motor outcomes via UPDRS III scores, further studies should also establish whether rTMS improves specific motor symptoms of PD, since there is some evidence that different symptoms may respond better than others.
Here we address two of the questions posed by Zanjani et al. [5]. First, we conducted a clinical trial to compare simply the effect of “standard” versions of high and low frequency rTMS: 1 Hz vs 20 Hz with no sham group as several other papers have reported positive effects of rTMS at various frequencies (versus sham) (i.e., assuming the previous reports were correct).
Second, we performed an exploratory analysis to examine whether rTMS had different effects on patients with primarily akinetic-rigid versus tremulous PD.
METHODS
Patients
62 consecutive patients (40 males, 22 females) who fulfilled the UK Parkinson’s Disease Brain Bank criteria for idiopathic PD [7] with ages between 50–70 years were recruited from those who attended the Department of Neurology, Aswan University Hospital, Egypt, from December 2016 to October 2017. Patients were divided into two subgroups, tremor-dominant and akinetic-rigid, on the basis of individual items of the UPDRS III scale as described by Lewis et al. [8]. A tremor score was derived from the sum of UPDRS III items 20 (tremor at rest) and 21 (action or postural tremor of hands) divided by 7 [the number of single sub items (for each body region if separated) included]. A non-tremor score was derived from the sum of UPDRS III items 18 (speech), 19 (facial expression), 22 (rigidity), 27 (arising from chair), 28 (posture), 29 (gait), 30 (postural stability), and 31 (body bradykinesia and hypokinesia) divided by 12 (the number of single sub items (for each body region (if separated) included). The patient was classified as tremor-dominant if the tremor score was at least twice the non-tremor score. Conversely, the patient was classified as akinetic-rigid type if the non-tremor score was at least twice the tremor score. The remaining patients, in whom the tremor and non-tremor score differed by less than a factor 2, were classified as mixed type and were excluded.
Out of 62 patients 12 patients were excluded: 6 patients had mixed type of PD, 2 patients had a history of seizures, 2 patients had a history of cerebrovascular stroke. The remaining 52 patients participated in the clinical trial. All patients were receiving medication that was maintained constant for the duration of the trial. All participants or their caregivers gave informed consent before participation in the test and after full explanation of the study protocol. The local ethical committee of Aswan University Hospital approved the study protocol.
Experimental setup and design
All evaluations were performed by a clinician who was unaware of the treatment group. Patients were assessed clinically with part III of the Unified Parkinson’s Disease Rating Scale (UPDRS III) [9], and the instrumental activities of daily living (IADL) scale [10]. In addition we employed a self-assessment scale (SA) [11] as follows. Each patient was requested to evaluate the following nine parameters in a questionnaire: total body mobility, hand agility, walking, arising from chair, tremors, mood, concentration, sleep, and dreaming. Each of these were scored 1 (best), 2 (no change), or 3 (worst), and the patients were asked to judge the past 24 h.
Electrophysiological assessments
These were performed using transcranial magnetic stimulation (TMS). Patients were seated comfortably in a reclining chair; biphasic rTMS pulses were delivered through a “figure 8” coil (outer diameter of each wing, 7 cm; maximum field strength, 1.9 Tesla) attached to a Magstim stimulator. Details of the methodology are given in Khedr et al. [12]. In brief, we measured resting (rMT) and active (aMT) motor threshold, MEP amplitude at 130% rMT and duration of the ipsilateral silent period of the first dorsal interosseous muscle (FDI) using single TMS pulses with the coil held so as to induce currents approximately perpendicular to the line of the central sulcus.
Repetitive transcranial magnetic stimulation (rTMS)
The 52 patients were randomly assigned into two equal groups. One group received 2000 pulses of 20 Hz rTMS (delivered in trains of 5 s with 25 s between trains) over the hand area of each motor cortex at 90% of RMT for each hemisphere (5 min between hemispheres). The other group received bilateral stimulation at 1 Hz rTMS (each hemisphere received two trains, separated by 30 s, of 1000 stimuli at 100% RMT).
Follow up of the patients
We followed up the patients clinically after the end of the 10th session and one month later using the same scales the same clinical rating scales (UPDRS part III, IDLA, and SA), as primary outcome. Cortical excitability was only measured after the end of the 10 treatment sessions as a secondary outcome (see Fig. 1).
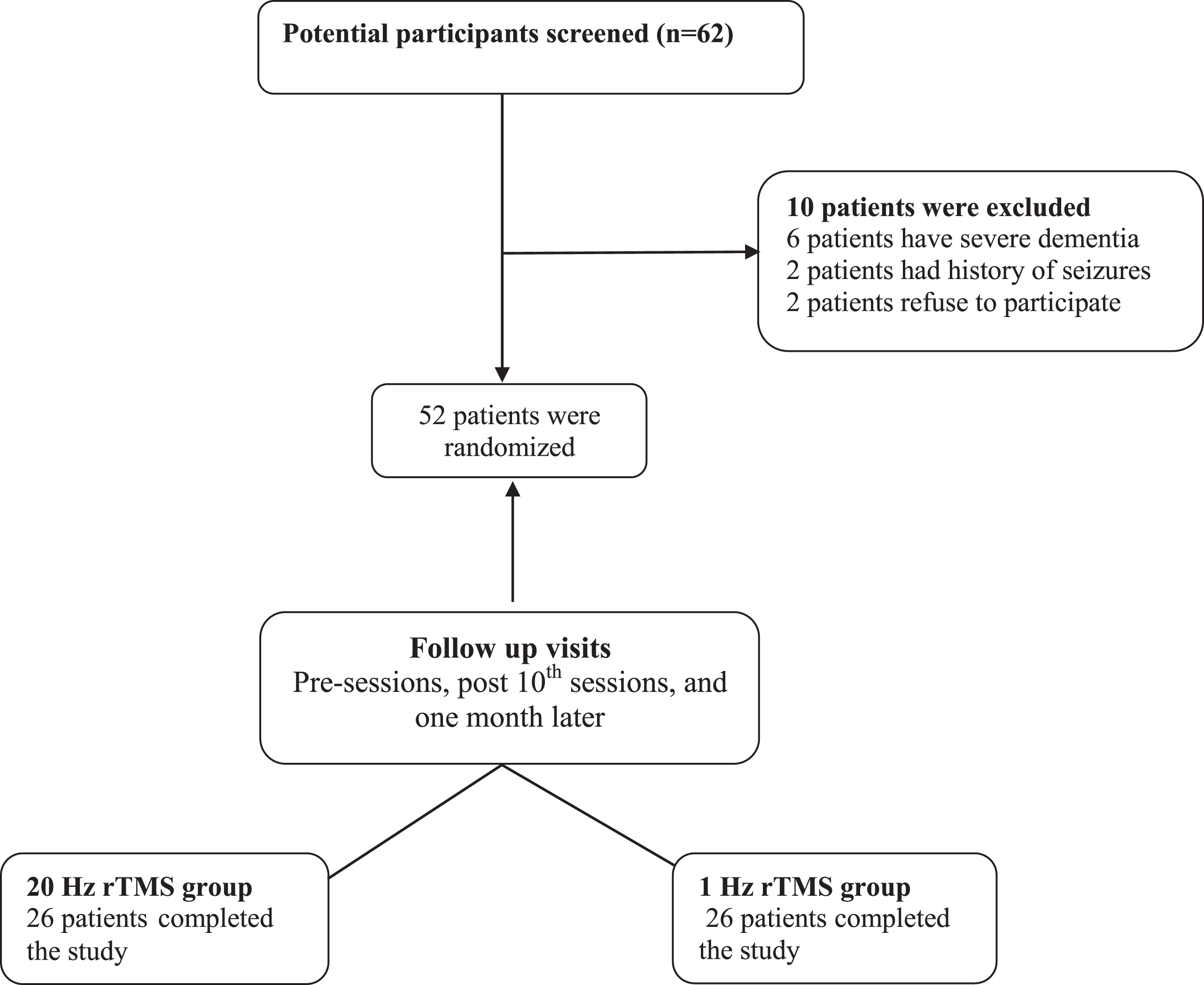
Consort diagram indicating recruitment and treatment staging.
Statistical analysis
All data were analyzed with the aid of the SPSS ver.16. The results were expressed as mean±SD. Since the distribution of the data did not differ statistically from normality, statistical analysis of the scores in each test was performed with repeated measures analysis of variance (ANOVA) with TIME, as the within-subject factor, and treatment condition (20 Hz, and 1 Hz rTMS) as the between subject measure. Greenhouse–Geisser degree of freedom corrections were applied to correct for the non-sphericity of the data. P < 0.05 was considered significant for all statistical analysis.
RESULTS
Clinical characteristics are summarised in Table 1. There were no significant differences between groups as regards age, sex, duration of illness, total score of UPDRS III, IADL, and SA scale. There was also no difference in the proportion of the predominant type of Parkinson’s disease (Akinetic-rigid/tremor 16/10 versus 17/9, respectively).
Demographic, clinical and staging data at baseline assessment
UPDRS III, Unified Parkinson’s Disease Rating Scale; IADL, instrumental activities of daily living; SA, self-assessment scale.
Patients were assessed clinically off medication at baseline (the day before the first rTMS session), the day after the last session, and then 1 month later. Physiological assessments were performed only at baseline and after the last treatment session.
Effect on clinical scores (see Fig. 2)
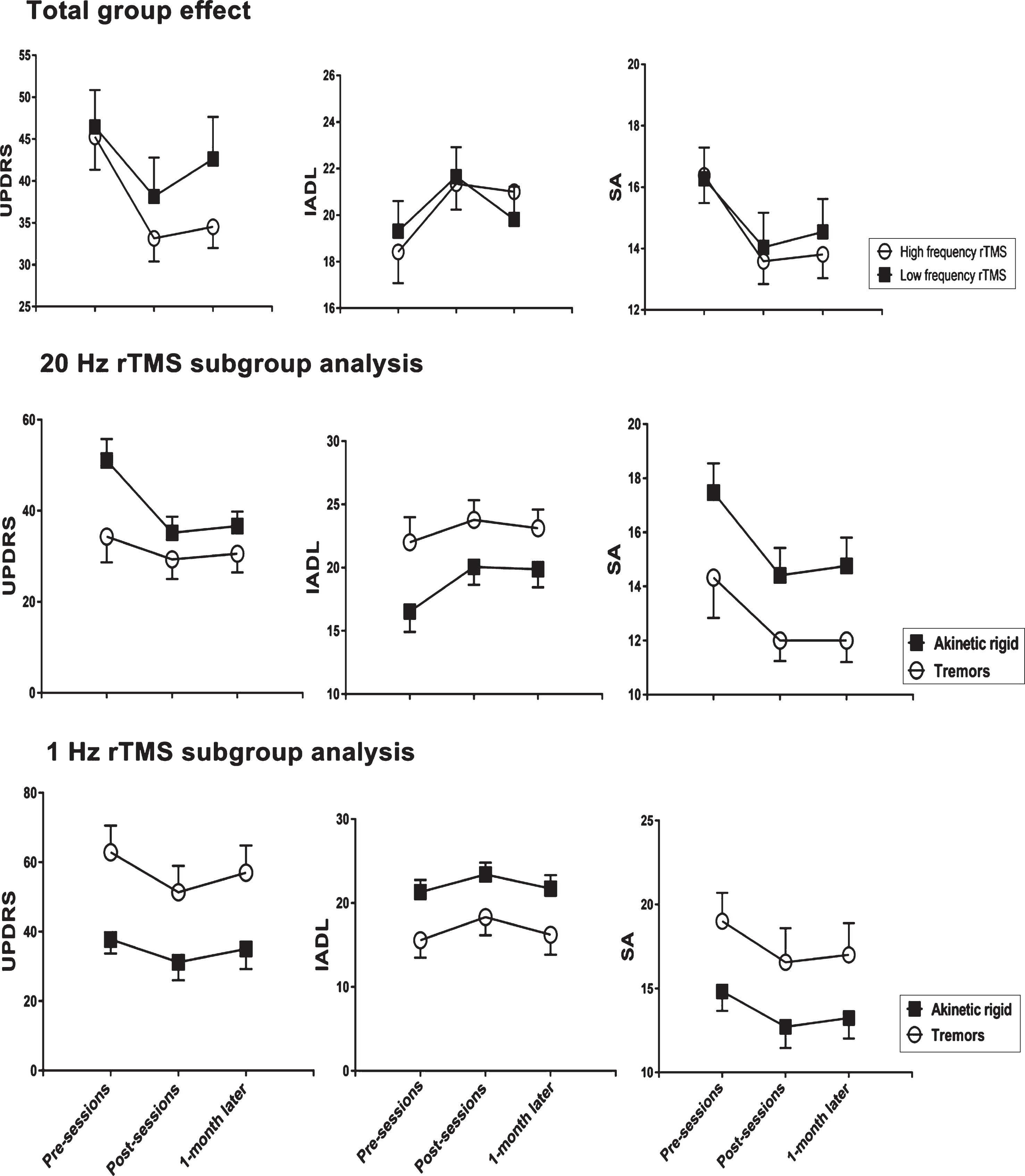
Time course of changes in UPDRS, IADL and self-assessment (SA) scores from before, immediately after 10 day’s rTMS and 1 month later. The top row summarises data from the total group of patients, and compares the effects of treatment with 1 Hz v 20 Hz rTMS. The middle and bottom rows compare the effects of 20 Hz rTMS (middle row) and 1 Hz rTMS (bottom row) on the patient subgroups (akinetic-rigid versus tremor-dominant symptoms). Points are the mean±SD. Statistical details are given in Table 2A.
Total Group Analysis (see Table 2A)
Effect of 20 Hz versus1 Hz rTMS in total group
Effect of 20 Hz rTMS in relation to the dominant type of PD
Effect of 1 Hz rTMS in relation to the dominant type of PD
UPDRS III, Unified Parkinson’s Disease Rating Scale; IADL, instrumental activities of daily living; SA, self-assessment scale.
One way repeated measures ANOVA (pre, post treatment and one month later) showed a significant effect of time on all rating scales (UPDRS III, IADL, self-assessment) in both 1 Hz and 20 Hz treatment groups. Two-way repeated measures ANOVAs on the scores for each rating scale, with treatment CONDITION (1 Hz and 20 Hz) and TIME (baseline, post treatment, one month later) as main factors revealed a borderline significant interaction effect for UPDRS III (P = 0.075), and IADL scores (P = 0.04): 20 Hz rTMS was more effective than 1 Hz rTMS. There was no significant interaction for self-assessment scores. Finally, paired t-tests showed that at 1 month, scores on both the UPDRS III and IADL were significantly improved compared to baseline in the 20 Hz group (P < 0.01 in both cases), but not in the 1 Hz group.
Subgroup analysis (akinetic-rigid versus tremor-dominant groups)
In this exploratory analysis we investigated the possibility that akinetic-rigid patients might respond differently to tremor-dominant patients to the two frequencies of rTMS. An unintended limitation of the randomisation procedure was that the UPDRS III scores in both the akinetic-rigid and the tremor-dominant groups differed between the 1 Hz and 25 Hz treatment protocols (see discussion).
20 Hz rTMS (see Table 2B): one way ANOVA repeated measures analysis (pre, post session and one month later) showed a significant effect of time on all rating scales (UPDR, IADL, self-assessment) in the akinetic-rigid group, while significant improvement was only seen in UPDRS III and self-assessment scores for tremor-dominant patients. Two-way repeated measures ANOVAs on the scores for each rating scale, with condition (akinetic-rigid versus tremor) and time (baseline, post treatment, one month later) as main factors revealed a significant interaction only for UPDRS III (P = 0.013). This was due to the fact that high frequency rTMS was more effective in patients with akinetic-rigid than tremor-dominant PD. (Table 3). Post hoc paired t-tests showed that the UPDRS III score in the akinetic-rigid group was maintained at 1 month follow-up (P = 0.01) but not in the tremor-dominant group.
Effect of rTMS on cortical excitability parameters
1 Hz rTMS (see Table 2C): There were no significant effects of treatment on the UPDRS scores. However, one way repeated measures ANOVA (pre, post session and one month later) showed a significant effect of time on IADL in both akinetic-rigid and tremor-dominant groups and on self-assessment scores in the tremor-dominant group. Two-way repeated measures ANOVAs on the scores for each rating scale, with condition (akinetic-rigid versus tremor-dominant) and time (baseline, post treatment, one month later) as main factors revealed a significant interactions for IADL, with larger effects on the tremor-dominant group. The effects at one month were not different to baseline for either group in any of the scores. The conclusion is that 1 Hz rTMS did not have persisting effects on symptoms in either group of patients.
Effects on cortical excitability (see Fig. 3)
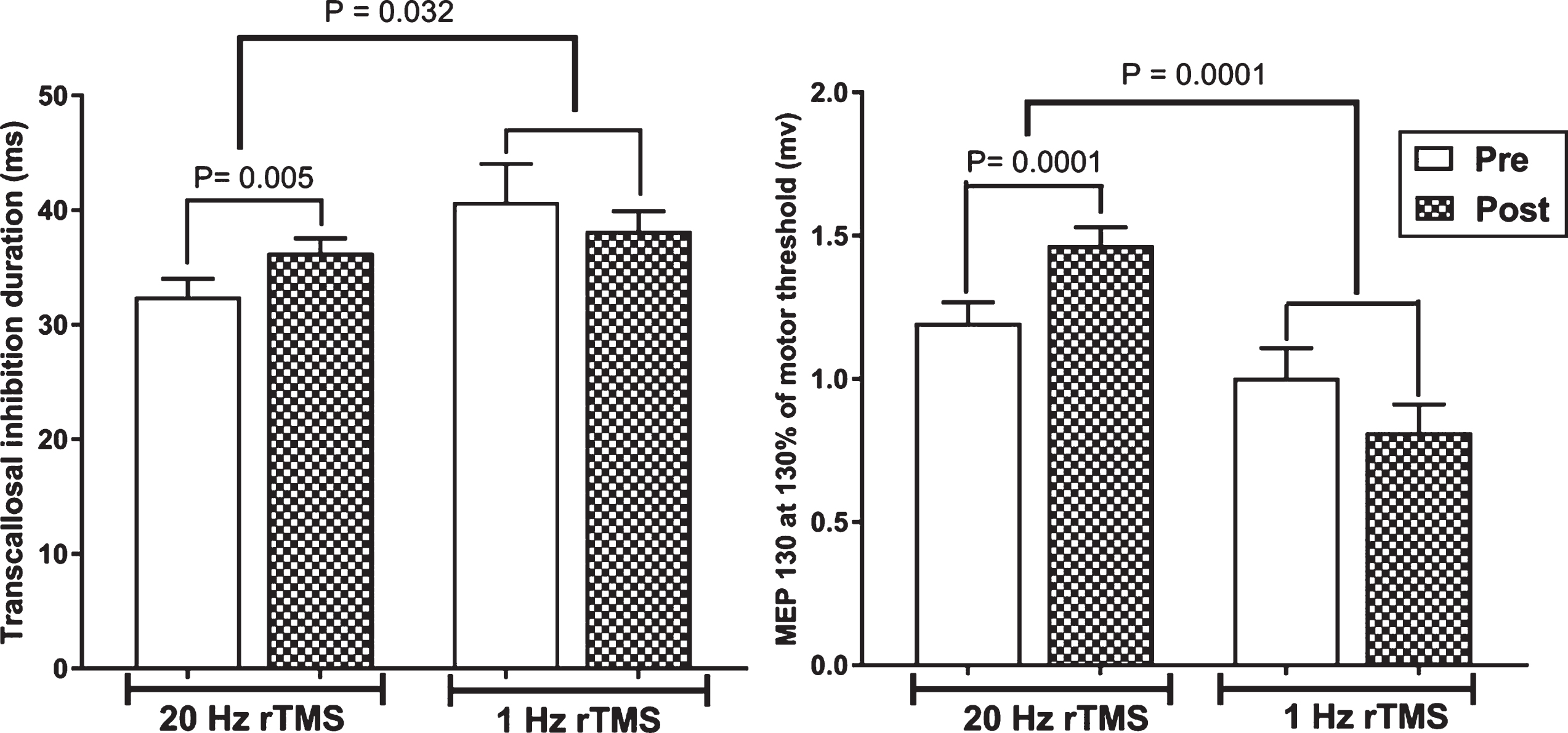
The effect of 20 Hz and 1 Hz rTMS on the duration of transcallosal inhibition (left panel) and MEP amplitude (right panel) following a TMS pulse of 130% rMT.
Given the differences in baseline scores, this analysis was only performed on the total group data. Neither frequency of rTMS had any effect on motor thresholds. However, 20 Hz rTMS increased MEP amplitude at 130% RMT and prolonged the duration of TCI, whereas there was no effect after 1 Hz rTMS. (See Table 3 for numerical data and statistical details.)
DISCUSSION
The present study is the first to compare directly the effects of 10 consecutive day’s treatment with 1 Hz and 20 Hz rTMS over M1 in patients with Parkinson’s disease. Although both treatments transiently improved UPDRS III scores, there was a borderline tendency for a greater effect after 20 Hz. In addition, post hoc tests showed that only the group receiving 20 Hz rTMS maintained a significant effect at 1 month.
Because of the evidence that there may be different clinical subtypes of patients with Parkinson’s disease who may potentially respond differently to treatment, we also conducted a sub-analysis of the data by examining separately the effects on patients with an akinetic-rigid versus a tremor-dominant form of the condition. However, randomization resulted in different baseline UPDRS III scores in both the akinetic-rigid and tremor-dominant groups: the akinetic-rigid scores were higher at baseline than tremor-dominant scores in those treated with 20 Hz, whereas the opposite was true for treatment with 1 Hz. It is thus difficult to make a definitive conclusion about possible differential effects on clinical subtypes. Nevertheless, the data suggested that akinetic-rigid patients benefit from 20 Hz treatment whereas there was no significant effect on the tremor-dominant form. Treatment with 1 Hz rTMS had no significant effect on either group. Finally the effects of treatment on electrophysiological measures were consistent with the clinical effects and showed that MEP amplitude increased and cortical silent period duration decreased only after 20 Hz rTMS.
Our conclusion is that 20 Hz rTMS over M1 may be superior to 1 Hz rTMS, particularly in patients with predominantly akinetic-rigid symptoms.
Khedr et al. [13] had measured the serum dopamine concentration before and after six sessions of 25 Hz rTMS over motor cortex in 20 PD patients. They found that the improvement in UPDRS was paralleled by an increase in plasma levels of dopamine. Although they only measured dopamine in plasma, it seems likely that the changes observed reflected changes in cerebral dopamine levels. Our results support the meta-analysis that confirmed the superiority of high-frequency stimulation over low-frequency stimulation [1].
The results of the present study are in contrast with the results of Mally et al. [14]. In their study, sixty-six patients with PD were included and randomly divided into three groups. The effects of 1 Hz, 5 Hz and 5 + 1 Hz frequency at low intensity over each DLPFC and the brain stem for 7 days were compared. Patients were followed for six months. They found that only 1 Hz had an effect on motor scores. Five Hertz and 5 + 1 Hz did not cause improvement. However, this was an open study trial and used a lower intensity of rTMS than in the present study (25% of maximum output).
Although the UPDRS III was our primary outcome measure, we also evaluated patients with the IADL and SA scores. The IADL scores followed a similar pattern to the UPDRS III scores in the total group analysis, and confirmed that rTMS was an effective form of treatment that improved overall activity as well as specific motor signs. In the subanalysis there was a larger effect on the tremor-dominant group after 1 Hz rTMS. Siebner et al. [15] also noted an effect on tremor after 5 Hz rTMS which may indicate that lower frequencies of rTMS could be more effective for symptoms of tremor although more data is needed to substantiate this point. Self-assessment scores did not correlate well with the objective UPDRS III and IADL scores. Patients tended to respond that both forms of treatment were beneficial, even when the other scores indicated little overall change.
It was interesting to note that in the total group analysis, the immediate effects of both 1 Hz and 20 Hz rTMS were similar, but that the effect at 1 month follow-up was only maintained after 20 Hz rTMS. Strafella et al. [16] showed that a single session of 10 Hz rTMS to M1 caused dopamine release in the striatum. Although Strafella et al. [16] did not explore the effects of different frequencies of rTMS, it could be that both frequencies of stimulation used here lead to immediate increases in dopamine release and that this is responsible for the initial effect. The implication would be that the longer term benefit at one month was due to a different mechanism, presumably involving some more permanent changes in neural network activity.
Although the major objective of the study was to examine the clinical effectiveness of rTMS therapy, we also included outcome measures of motor cortical excitability. These are useful because they provide an independent and objective measure of the response to rTMS that is not compromised by behavior. Previous work in healthy individuals has suggested that low frequencies (such as 1 Hz) of rTMS are likely to reduce cortical excitability by effects on synaptic plasticity whereas higher frequencies do the opposite. This was true here: 20 Hz rTMS increased excitability as measured by the larger response to single TMS pulses, while there was a tendency for excitability to be reduced after 1 Hz rTMS. We conclude that rTMS did have an objective effect on the cortical motor system in our participants. Thresholds were unchanged as expected since these depend on the properties of ion channels in the neural membrane which are unaffected by rTMS. The combination of unchanged threshold with increased MEP amplitudes is compatible with the idea that the input-output relationship of cortical excitability is steeper after 20 Hz rTMS, which may be advantageous for movement initiation. There was also a significant increase in the duration of TCI after 20 Hz rTMS, but the significance of this is unclear since the amplitude of the MEP also increased and this in itself could affect TCI.
As noted in many previous studies, there may be no direct relationship between these physiological measures and the clinical outcome. Importantly, though, 20 Hz rTMS had a more significant effect on excitability measures than 1 Hz rTMS, which did reflect our overall clinical conclusion.
Conclusion and recommendation
20 Hz rTMS over M1 appears to be more effective than 1 Hz rTMS particularly at longer follow-up. Our exploratory analysis suggested that the effects of rTMS might be slightly better in patients with a predominantly akinetic-rigid clinical presentation. However, the evidence is weak and studies with larger (i.e., >26 per treatment group) numbers of patients are need to confirm this work.
CONFLICT OF INTEREST
The authors have no conflict of interest for report.
Contributors
Eman M. Khedr designed the study and wrote the protocol., analysis the data, wrote the final revision of the manuscript, Mostafa Saber conducted the clinical study, Asmaa Hasan performed data entry, and wrote the first draft, Bastawy Al-Fawal, Ahmed Abdel Wraith, and Ahmed Bassiony conducted literature searches, prepared the figures and provided summaries of previous research studies and Ahmed Nasr Eldein wrote the first draft of the manuscript, data analysis. John Rothwell wrote the interpretation of data, and revising the manuscript critically for important intellectual content. All authors contributed to and have approved the final manuscript.
Footnotes
ACKNOWLEDGMENTS
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
