Abstract
Background:
Impaired gait and postural stability are cardinal motor symptoms in Parkinson’s disease (PD). Treadmill training improves gait characteristics in PD.
Objective:
This study investigates if postural perturbations during treadmill training improve motor performance and particularly gait and postural stability in PD.
Methods:
This work presents secondary outcome measures of a pilot randomized controlled trial. PD patients (n = 43) recruited at the University Hospital Erlangen were randomly allocated to the experimental (perturbation treadmill training, PTT, n = 21) or control group (conventional treadmill training, CTT, n = 22). Outcome measures were collected at baseline, after 8 weeks of intervention, and 3 months follow-up. Motor impairment was assessed by the Unified Parkinson Disease Rating Scale part-III (UPDRS-III), Postural Instability and Gait Difficulty score (PIGD), and subitems ‘Gait’ and ‘Postural stability’ by an observer blinded to the randomization. Intervention effects were additionally compared to progression rates of a matched PD cohort (n = 20) receiving best medical treatment (BMT).
Results:
Treadmill training significantly improved UPDRS-III motor symptoms in both groups with larger effect sizes for PTT (–38%) compared to CTT (–20%). In the PTT group solely, PIGD –34%, and items ‘Gait’ –50%, and ‘Postural stability’ –40% improved significantly in comparison to CTT (PIGD -24%, ‘Gait’ –22%, ‘Postural stability’ –33%). Positive effects persisted in PTT after 3 months and appeared to be beneficial compared to BMT.
Conclusions:
Eight weeks of PTT showed superior improvements of motor symptoms, particularly gait and postural stability. Sustainable effects indicate that PTT may be an additive therapy option for gait and balance deficits in PD.
INTRODUCTION
Gait and balance deficits are cardinal motor symptoms in Parkinson’s disease (PD), deteriorate with disease progression, and lead to reduced mobility and quality of life [1]. These motor symptoms substantially contribute to increased risk of falling in this population [2]. The ability to maintain a stable and steady walking pattern have been shown to be impaired in PD [3]. Deficits in regularity of gait reflect the disease specific impairment of dynamic postural control [4]. With respect to stable walking, postural stability and regularity of gait are pre-defined gait domains in PD [5] and play an important role in falls [6]. Motor symptoms in PD including deficits of gait and postural stability are clinically rated by the widely used clinical scale, the Unified Parkinson Disease Rating Scale, part III (UPDRS-III) [7, 8]. The dynamic postural control aspects in PD are represented by the Postural Instability and Gait Difficulty subscore (PIGD) of the UPDRS-III which particularly focusses on gait and balance symptoms in PD [9].
Pharmacological therapy to improve gait and balance deficits is limited in PD. Therefore, non-pharmacological therapies, especially exercise interventions are increasingly recognized as both effective and additional treatment [10]. Mehrholz and colleagues [11] observed in a recent meta-analysis of 18 trials that treadmill therapy is effective in improving gait speed and stride length in PD. Furthermore, this form of training may improve dynamic postural control and regularity of gait [12]. However, conventional treadmill training (CTT) does not reflect natural walking conditions as they appear in everyday life - including challenging situations for balance control. Increasing postural challenge by adding perturbations to the training has been suggested to be a very promising strategy to improve gait stability and to decrease fall risk in older adults [13]. In a first study in PD, we recently showed that treadmill training with perturbative stimulations (PTT) to the treadmill surface lead to significant improvements of gait performance (walking capacity in the 2-Minute Walk Test) and dynamic balance control (regularity of gait) compared to CTT [14–16]. The effect of PTT on clinical motor symptoms, in particular ‘Gait’ and ‘Postural stability’ has not yet been established.
Some studies investigated the effects of standard treadmill training on clinical motor symptoms in PD using UPDRS-III. These studies reported that treadmill training improved global UPDRS-III scores after the intervention phase [17–21]. In most of these studies, treadmill training conditions (duration of training session: 30 mins, frequency: 3–5 days per week, duration of intervention: 4–8 weeks) were similar. The largest study conducted by Schenkman et al. [22] in 128 PD patients revealed that high-intensity treadmill training (up to 50 mins, 4 days per week, intensity: 80–85% maximum heart rate, 6 months) stabilized global motor symptoms (UPDRS-III) over 16 months. Besides the general positive effect of treadmill training, no study has addressed the question whether adding postural perturbations during treadmill training has a specific additive effect on gait and postural stability in PD.
The aim of this pilot randomized controlled trial was to investigate the effect of PTT on clinical motor symptoms assessed by the UPDRS-III (secondary outcome measure of this clinical trial; primary outcomes were reported by Steib et al. [16]), and in particular on gait and balance evaluated by the PIGD score, and the subitems ‘Gait’ and ‘Postural stability’ in comparison to CTT. In addition, we compared the observed substantial effects of physical exercise on motor symptoms to an independent group of patients receiving best medical treatment (BMT) and to historical studies assessing pharmacological therapy. The BMT group was selected from our population-based Franconian PD patient cohort. We hypothesized that PTT results in additive improvements in gait and postural stability compared to CTT and that both groups improve general clinical motor symptoms after treadmill intervention.
MATERIALS AND METHODS
Participants
Forty-three PD patients were included in this single-blind, randomized controlled trial (trial registration ID: NCT01856244). Primary endpoints (overground gait speed and balance (assessed by the Mini-BESTest)) have been presented by Steib et al. [16]. In this manuscript, the secondary outcome measures (change in UPDRS-III and its subitems) are reported. PD patients were recruited at the Movement Disorders Outpatient Unit (MDU), Department of Molecular Neurology, University Hospital Erlangen, Germany from a larger stratified patient cohort (n = 406) described below. Sporadic PD was defined according to the Guidelines of the German Association for Neurology (DGN) similar to the UK PD Society Brain Bank criteria [23]. Beside the diagnosis of sporadic PD, inclusion criteria consisted of Hoehn & Yahr disease stage (H&Y) between 1 and 3.5 (the ability to walk independently without a walking aid), and UPDRS-III subscore ‘Gait’ and/or ‘Postural stability’ ≥1. PD patients were excluded if other neurological disorders, severe cardiovascular or orthopedic comorbidities were present, potentially affecting the intervention or assessments. Parkinson-related medication was maintained stable during the intervention period. Data were collected between May 2014 and July 2016 at the MDU and the Department of Sport Science and Sport (DSS), FAU Erlangen-Nürnberg, Erlangen, Germany. This study was approved by the local ethics committee (reference numbers: 4208 and 181_12B, FAU Erlangen-Nürnberg, Germany), and participants gave written informed consent according to the Declaration of Helsinki.
Participants were randomized to either an experimental (PTT) or control group (CTT) using a computer-generated block-randomization with a block size of 6 and a stratification by H&Y disease stage (H&Y 1–2 and H&Y 2.5–3.5) (Fig. 1) [16]. Patient characteristics are presented in Table 1 and do not show significant differences between groups regarding age, gender, height, weight, and disease duration.
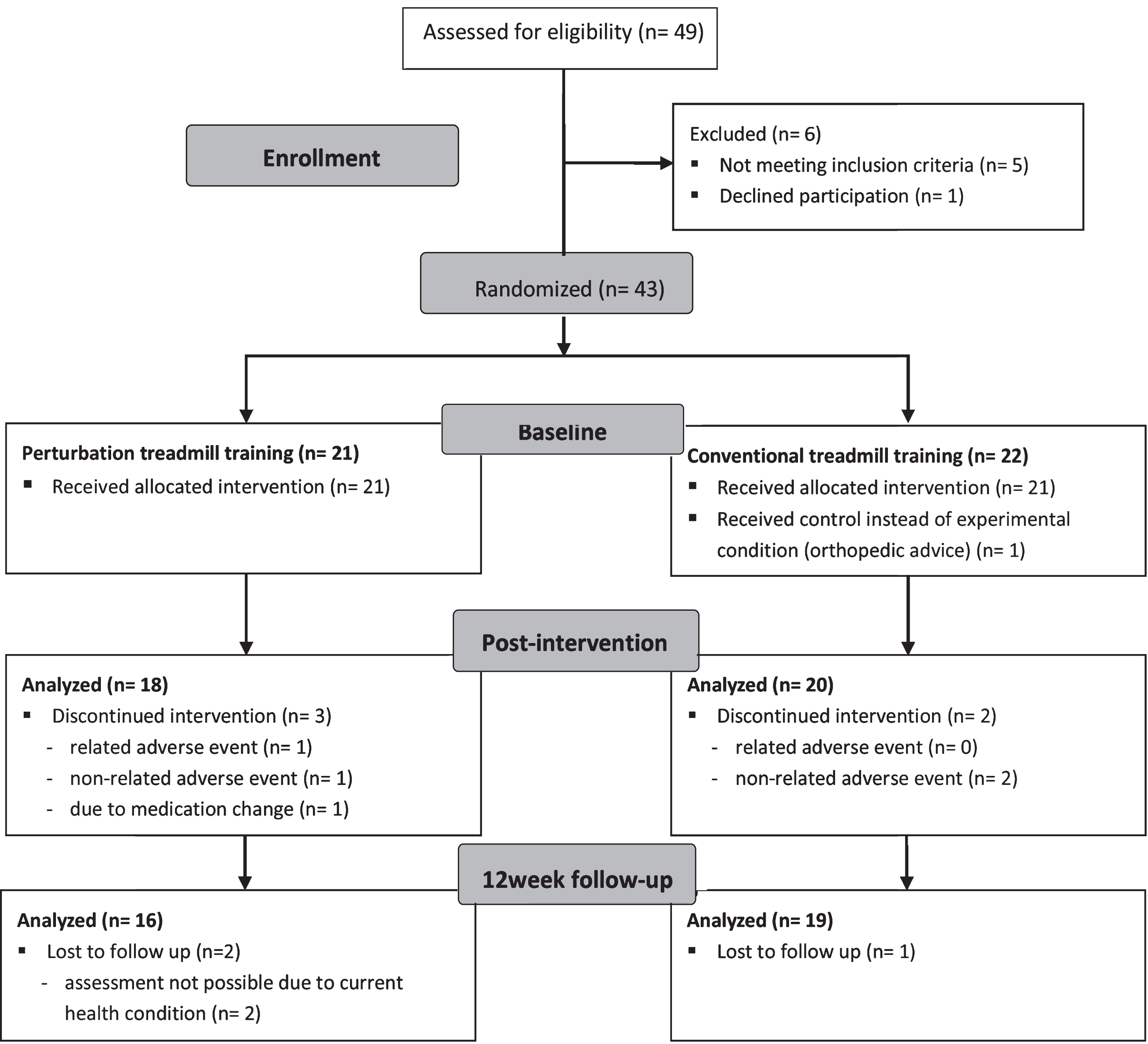
Flow chart.
Patient characteristics at baseline
†Chi Square Test; LEDD, Levodopa equivalent daily dose; H&Y, Hoehn and Yahr disease stage; UPDRS-III, Motor score of the Unified Parkinson’s disease rating scale; MoCA, Montreal Cognitive Assessment.
Clinical motor assessment
PD patients were clinically assessed at baseline, after 8 weeks of intervention, and at 12 weeks follow up. The assessments were performed by a movement disorders specialist blinded for group allocation using the UPDRS motor score (UPDRS-III) [24] including the ‘Postural Instability and Gait Difficulty’ subscore (PIGD) [9], and the single items ‘Gait’ and ‘Postural stability’. Participants performed assessments and intervention in stable ON medication without presence of clinically relevant motor fluctuations or dyskinesia. Time points of assessments did not differ between the three visits and regular therapy regimes and present medication were not altered during the intervention.
Population-based franconian PD patient cohort
From 2014 to 2016 the MDU Erlangen followed n = 406 PD patients and collected the data in a PD database defining a population-based cohort in Franconia (“Franconian PD cohort”) (mean±SD, age: 62.3±10.5 a; disease duration: 8.2±5.4 a; UPDRS-III: 19.3±13.5 points; H&Y: 2.3±0.9). From this cohort, both treadmill intervention groups (CTT, PTT) were recruited. In the clinical trial described in this study, we focused on the comparison between intervention effects of CTT and PTT. Since it is not feasible to choose a blinded non-intervention group for physical exercise, we selected an additional control from the Franconian PD cohort receiving best medical treatment (BMT). PD patients (n = 20) were matched by age, gender and disease duration to CTT and PTT. Thus, the BMT group did not differ in anthropometric or disease specific data at baseline (Supplementary Table 1), but were not able to participate in the clinical trial due to logistic reasons: distance to the hospital, lack of transport from home to hospital for two times per week. The BMT group completed the identical test battery as the intervention cohorts (PTT, CTT). Sustainability effects after 3 months of the treadmill interventions (CTT, PTT) were compared to this matched PD cohort that performed assessments at two consecutive visits within an average time frame of nine months.
Treadmill intervention
In this study, a standard medical treadmill (mercury, h/p/cosmos medical GmbH) mounted on a tiltable platform construction (zebris Medical GmbH) was used. This prototypic treadmill device and the training conditions have been previously described in detail [14, 16]. Shortly, three-dimensional tilting movements were induced to the treadmill surface. These perturbations result in constant adaptations of the patient’s postural control during walking. The CTT group received treadmill training on the identical treadmill device but without perturbations. All participants were secured with a safety harness on the upper body during treadmill walking. Prior to the training period, patients received one session of treadmill training for familiarization [14]. Structured treadmill training in both groups was realized in 16 sessions (two times per week over 8 weeks) lasting 30 minutes each. During the intervention period, treadmill speed was individually and progressively adapted according to each patients’ self-perceived exertion assessed by Borg scale (6–20) with a target range of 12–15 and by Likert scale (1–7) from 1 “not difficult” to 7 “extremely difficult” with a target range of ≤5 [16].
Statistical analysis
In this exploratory pilot study, a per-protocol analysis was performed. With regard to an intention-to-treat analysis, the small sample size and limited number of assessment time points may cause considerable bias to the data. Therefore, we decided to perform a complete-case analysis. Normality of data was tested by Shapiro–Wilk test and variance homogeneity by Levene test. In case of normally distributed and variance homogenous data, parametric tests were used to assess significant effects between groups. Otherwise, non-parametric measures were performed. Differences in baseline characteristics were tested using one-way ANOVA (interval data), Kruskal-Wallis test (non-parametric or not normally distributed parameters), and chi-square test (categorical data). Repeated-measures analysis (paired Wilcoxon test for dependent variables) was performed to detect significant within-group differences after the treadmill intervention (baseline vs. post-intervention; baseline vs. follow-up: post-intervention vs. follow-up), and to evaluate within-group changes in each group separately. Mann-Whitney U test was used to compare between-group differences as response to the intervention at post-intervention and follow-up. Kruskal-Wallis test was performed to evaluate between-group effects of both intervention groups and the BMT cohort (change between baseline and follow-up). Cohen’s d is presented as measure of effect size and was calculated by means and standard deviations (metric variables) or z-values (categorical variables). All statistical analyses were performed using SPSS software package version 21 (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0, Armonk, NY, USA: IBM Corp.). Post-hoc power calculations were performed using the software G*Power (Version 3.1.9.2).
RESULTS
In this study, PD patients were recruited from a large cohort of the MDU Erlangen and stratified into two intervention cohorts (PTT, CTT). After 8 weeks of treadmill interventions and at a three months follow-up visit, the effects on motor symptoms were compared between both groups. Patient characteristics at baseline did not significantly differ between both cohorts (Table 1) and medication remained constant in the CTT and in the PTT group during the intervention phase. In order to evaluate the effect of the intervention, we compared motor symptoms at the follow-up visit to a population-based PD cohort matched by age, gender and disease duration. This PD cohort received BMT and was assessed at two consecutive visits within an average time frame of nine months.
Change from baseline to posttest
After 8 weeks of intervention, both groups significantly improved motor symptoms rated by the UPDRS-III with an average improvement of –6.7 points (PTT) and –4.1 points (CTT) (p < 0.000). Between-group effects did not reach significance level (p = 0.285), however, effect sizes indicated that improvements were proportionally larger in the PTT group (d = –1.22; change from baseline: –38 %, p = 0.001) compared to the CTT group (d = –0.67; –20 %, p = 0.046). Importantly, post-hoc tests revealed that only the PTT group significantly improved in the PIGD score (d = –0.72; –34 %, p = 0.005), UPDRS items ‘Gait’ (d = –0.79; –50 %, p = 0.023) and ‘Postural stability’ respectively (d = –0.95; –40 %, p = 0.008) (Fig. 2, Table 2).
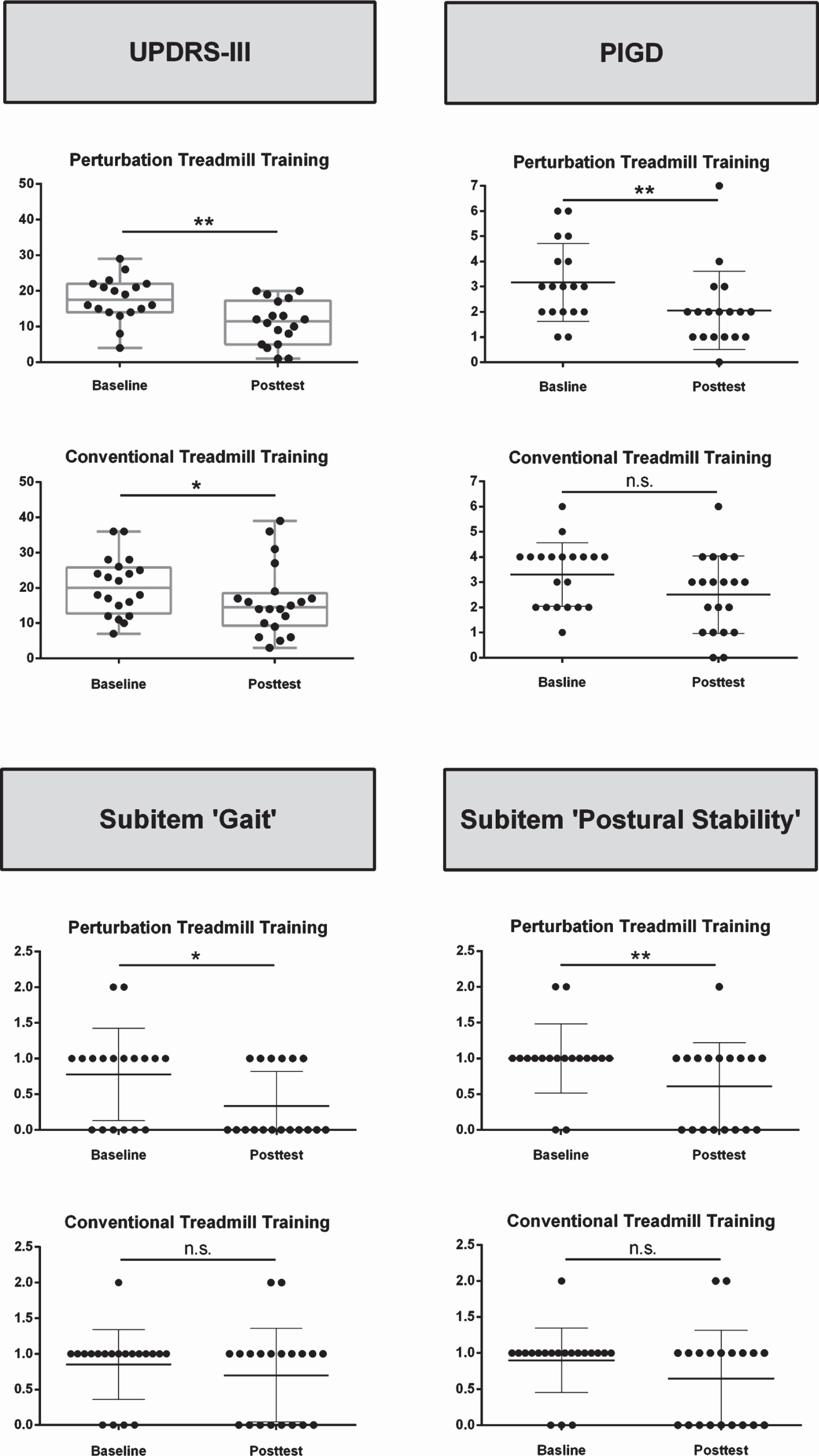
Improvements in the UPDRS-III (motor score), postural instability and gait difficulty score (PIGD), and UPDRS-III subitems ‘Gait’ and ‘Postural stability’ between baseline and posttest, separated in the conventional (CTT, at the bottom) and perturbation treadmill group (PTT, on top), *p < 0.05; **p < 0.01.
Changes of motor symptoms between baseline and post-intervention
*paired Wilcoxon-Test; **Mann-Whitney-U-Test; Effect size, Cohen’s d; LEDD, Levodopa Equivalent Daily Dose. Bold numbers indicate significance.
Sustainability of intervention mediated effects
The substantial improvement regarding UPDRS-III after the intervention period of 8 weeks persisted in both groups during the 3 months follow-up period (baseline vs. follow-up: PTT: –6.2 points, CTT –3.8 points, p < 0.000). No between-group effects were present, however larger effect sizes in the PTT group indicated stronger improvements (PTT: d = –1.25; change from baseline: –37%, p = 0.001, and CTT: d = –0.63; –19%, p = 0.064). With regard to PIGD as well as the subitems ‘Gait’ and ‘Postural stability’ significant improvements were detected from baseline to follow-up in the PTT group solely (PIGD: d = –0.84; –45%, p = 0.004, ‘Gait’: d = –1.03; –63%, p = 0.005, and ‘Postural stability’: d = –0.89; –67%, p = 0.013). In contrast, there were no significant changes in the CTT group (PIGD: d = –0.42; –18%, p = 0.138, ‘Gait’: d = –0.30; –25%, p = 0.366, and ‘Postural stability’: d = –0.37; –22%, p = 0.257) (Fig. 3, Table 3).
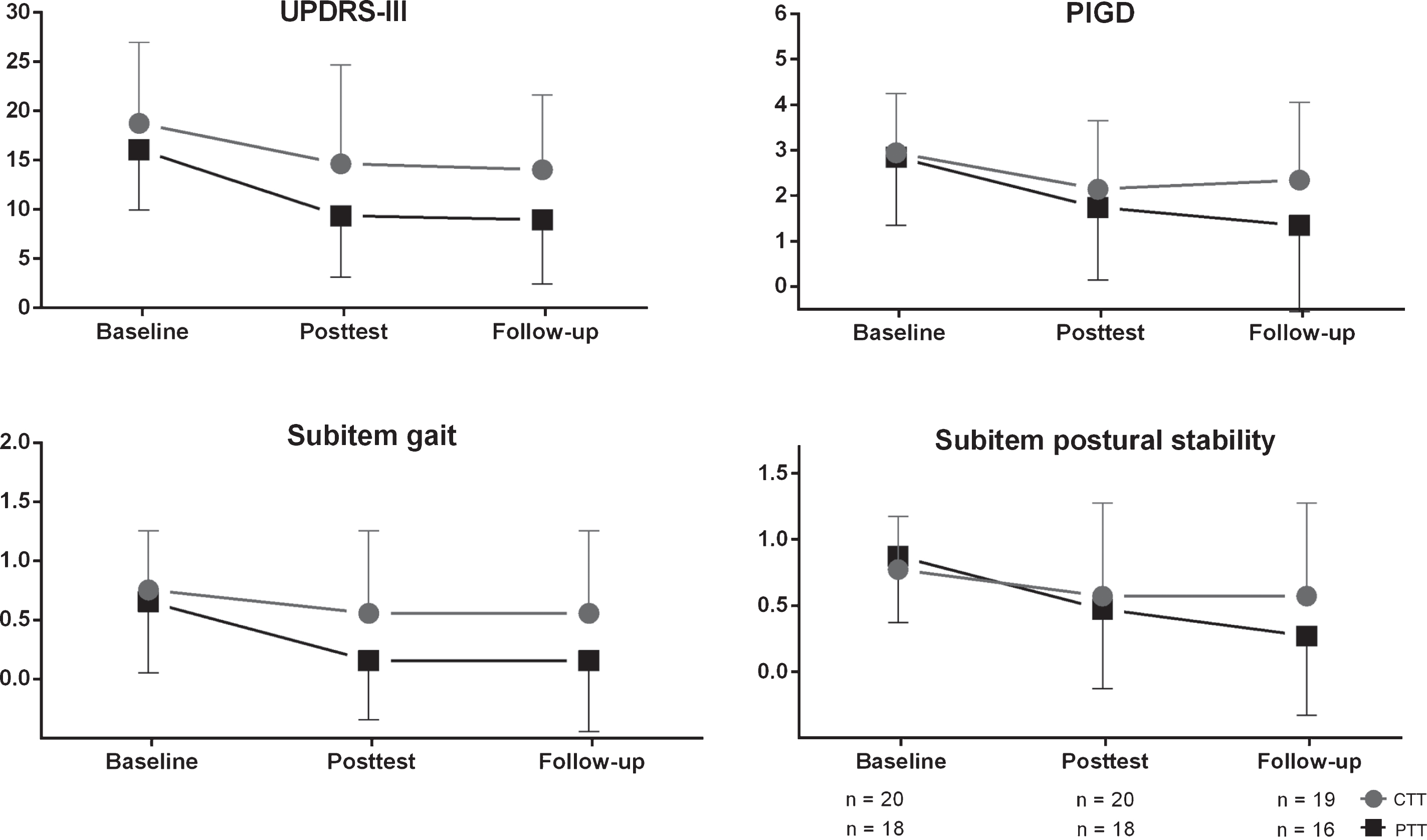
Sustainability effects of treadmill interventions at follow-up visit (5 months after baseline). CTT, Conventional Treadmill Training; PTT, Perturbation Treadmill Training.
Changes of motor symptoms between baseline and follow-up visit after 3 months
*paired Wilcoxon-Test; **Mann-Whitney-U-Test; Effect size, Cohen’s d; LEDD, Levodopa Equivalent Daily Dose. Bold numbers indicate significance.
The comparison between posttest and follow-up assessment revealed no significant differences in UPDRS-III (p = 0.900), PIGD score (p = 0.776) as well as subitems ‘Gait’ (p = 0.976) and ‘Postural stability’ (p = 0.819) indicating that positive effects sustained after three months. In separate comparisons, paired Wilcoxon-test revealed that both groups (PTT and CTT) showed stable motor symptoms from post-intervention to follow-up, respectively. In the PTT group, effects on UPDRS-III (d = 0.09; change from posttest: +6%), PIGD score (d = –0.05; –5.6%), ‘Gait’ (d = 0.01; 0%), and ‘Postural stability’ (d = –0.22; –20%), persisted between posttest and follow-up. In the CTT group, there was no change from posttest as well (UPDRS-III: d = 0.01; 0%; PIGD: d = 0.10; +8%; ‘Gait’: d = 0.01; 0%; ‘Postural stability’: d = 0.08; 0%) (Table 4). Significant between-group effects were not detected.
Changes of motor symptoms between post-intervention and follow-up visit after 3 months
*paired Wilcoxon-Test; **Mann-Whitney-U-Test; Effect size, Cohen’s d; LEDD, Levodopa Equivalent Daily Dose. Bold numbers indicate significance.
Comparison to a matched population-based franconian PD patient cohort
Matched PD patients selected from a larger cohort that regularly visited the MDU and received BMT but no treadmill training were compared to both intervention groups at follow-up. The CTT and PTT group had a follow-up visit five months after baseline and the BMT group visited the MDU after an average time of 9±4 months after baseline. In the BMT group, clinical motor symptoms remained nearly constant over the nine months period between their routinely scheduled initial and follow-up visit. Within-group comparison in the BMT cohort revealed no significant changes in UPDRS-III (d = 0.08; change from baseline to follow-up: +4%), PIGD (d = –0.04; –3%), ‘Gait’ (d = 0.13; +14%), and ‘Postural stability’ (d = –0.13; –12%). Between-group effects evaluated clinically relevant differences between BMT, CTT and PTT (change from baseline to follow-up) and revealed significant effects for UPDRS-III (BMT: +0.8±8.1 points; CTT: –3.8±8.1; PTT: –6.2±4.7; p = 0.008), PIGD score (BMT: –0.1±1.5 points; CTT: –0.6±1.8; PTT: –1.4±1.5; p = 0.020), and ‘Gait’ (BMT: +0.1±0.6 points; CTT: –0.2±0.8; PTT: –0.5±0.5; p = 0.014) but not for ‘Postural stability’ (BMT: –0.1±0.6 points; CTT: –0.2±0.6; PTT: –0.6±0.7; p = 0.075) (Fig. 4, Table 5). Single comparisons revealed significant between-group differences for PTT vs. BMT solely (UPDRS-III: p = 0.001; PIGD score: p = 0.004; ‘Gait’: p = 0.003; ‘Postural stability’: p = 0.035). Between-group comparisons between CTT and PTT as well as between CTT and BMT did not reach significance level. L-Dopa equivalent daily dose was constant in each of the three groups between baseline and follow-up (Table 5). Beneficial effects of PTT compared to BMT were not only present in PIGD symptoms but also in the remaining items of the UPDRS-III (Supplementary Figure 1).
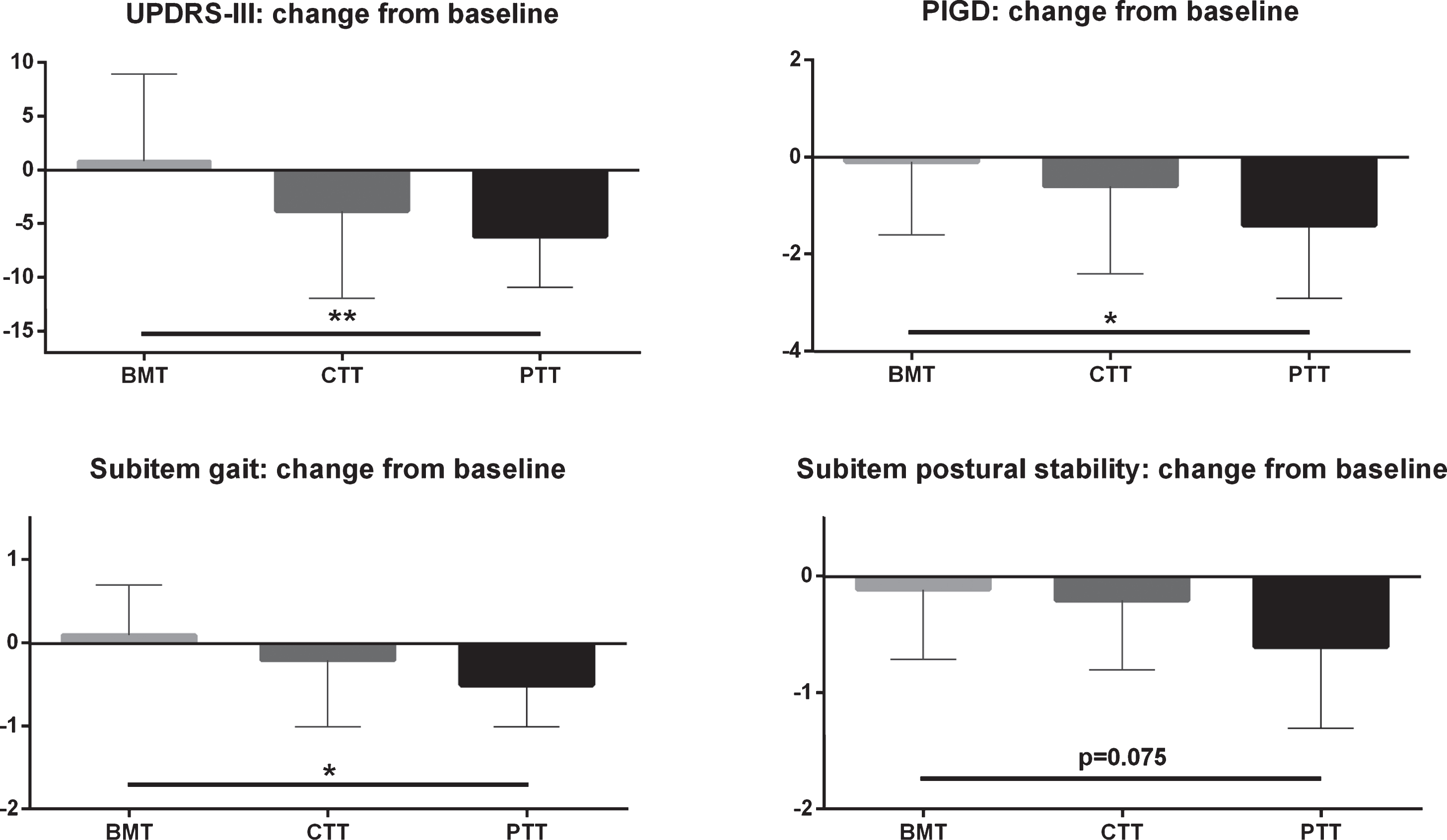
Change of clinical scores from baseline to follow-up. CTT, Conventional Treadmill Training; PTT, Perturbation Treadmill Training; BMT, Best Medical Treatment.
PD patients receiving Best Medical Treatment (BMT): Changes of motor symptoms between baseline and follow-up (∼9 months) and comparison to intervention groups at follow-up (∼5 months after baseline)
*paired Wilcoxon-Test; Δ Kruskal-Wallis Test; □Mann-Whitney-U-Test; Effect size, Cohen’s d; CTT, Conventional Treadmill Training; PTT, Perturbation Treadmill Training; PIGD, Postural Instability and Gait Difficulty Score; LEDD, Levodopa Equivalent Daily Dose. Bold numbers indicate significance.
DISCUSSION
In this study, we investigated whether eight weeks of treadmill training with additional postural perturbations (PTT) is superior to standard treadmill training (CTT) in improving global motor symptoms (assessed by UPDRS-III), as well as PIGD symptoms, and single items ‘Gait’ and ‘Postural stability’ in early to mid-stage PD patients. Both, PTT and CTT substantially improved global motor symptoms from pre-to post-intervention. Although we did not observe significant between-group differences for UPDRS-III, within-group effect sizes were considerably larger in the PTT group compared to CTT (PTT = –1.22; CTT = –0.67) and improvements sustained after 3 months follow-up. In particular, only the PTT group significantly improved gait and balance related motor symptoms (PIGD subscore, items ‘Gait’ and ‘Postural stability’). This finding indicates the clinical relevance of PTT improving specifically gait and balance aspects of motor impairment in PD.
Effects of treadmill training on motor symptoms
In the PTT group, we observed a decrease of global motor symptoms rated by the UPDRS-III (–38%), and improvements in the PIGD subscore (–34%) including the subitems ‘Gait’ (–50%) and ‘Postural stability’ (–40%) which were not present in the CTT group after intervention. These positive within-group effects persisted until the follow-up visit in the PTT group. Beneficial effects of treadmill training in PD, in particular on motor symptoms and gait are in line with previous findings. In a review of 18 trials, improvements in gait speed and stride length have been reported [11]. A recent randomized controlled trial with 128 de-novo PD patients observed positive effects of 6 months high-intensive treadmill training on the UPDRS motor score (no progression) in comparison to standard care (+4 points). Other studies reported beneficial effects of treadmill training on clinical motor symptoms as well, showing improvements of the UPDRS-III between 10 and 32% [17–21], mostly <30%. This is in line with the improvements of the CTT group in our study which decreased UPDRS-III by 20% after intervention. PTT results in a superior improvement of motor symptoms (–38%) which might be related to mechanisms of additional sensory input. There is evidence that continuous aerobic exercise combined with deficit-targeted, intensive training, which is part of the PTT intervention, have the potential to improve neuroplasticity [25, 26]. In particular, this multisensory exercise paradigm is discussed to strengthen and improve striatal-thalamic-cortical-motor circuitry by mechanisms that include enhanced synaptic strength resulting from an increase of neurotransmitters in the basal ganglia, receptor density and dendritic spine formation [25]. We observed sustainable positive effects of PTT that were stable until follow-up. Importantly, this beneficial effect of PTT and not CTT over 5 months was larger than the best medical care effects in a comparable group of Franconian PD patients (BMT). Sustainable improvements in gait and postural stability items might be related to motor-learning effects caused by neuronal adaptations to external postural perturbations. Motor adaptations have been reported to be fundamental in neurorehabilitation since they may support to recalibrate the flexibility in the human motor control and are likely to result in long-term improvements of movement patterns [27, 28]. As a result of treadmill training studies including ours, findings indicate improvements of motor symptoms in PD. Perturbative stimulations appear to have additional positive effects on gait and balance. Future work will have to further explore the clinical long-term relevance and dose-response relationship of PTT.
Treadmill intervention compared to a population-based franconian PD patient cohort
Since the effects of physical exercise in both randomized groups were quite robust, we included an additional group of patients receiving BMT. The selected group was representative for our Franconian PD reference cohort regularly visiting the MDU Erlangen and matched to the clinical characteristics of the PTT and CTT cohorts. Here, the positive effects on general motor symptoms as well as gait and postural stability observed in the PTT group persisted after 3 months and are beneficial in comparison to standard medical care (BMT: UPDRS-III at nine months follow-up: +0.8 points). Interestingly, also the remaining items of the UPDRS-III score (without PIGD) significantly improved in the PTT group and sustained until follow-up indicating that positive effects of PTT do not only address gait and balance deficits. Improvements of the CTT group persisted as well from post-intervention to follow-up, however, these effects were not superior to standard medical care. Importantly, the PTT and CTT groups received best medical standard care comparable to the BMT group. This underlines the translational relevance of treadmill intervention and physical exercise leading to additive effects complementing pharmacological treatment. These results indicate that PTT may serve as an effective, sustainable, non-pharmacological therapy for the treatment of gait and balance deficits as well as motor symptoms in PD patients.
Relevance of physical therapy on motor impairment compared to L-Dopa effects
In this study, we aimed to evaluate in more depth the effect size of PTT for motor symptoms (PTT vs. CTT) but also the effect size of exercise training to pharmacological effects on motor symptoms as reported in clinical trials. In our study, we observed substantial improvements of UPDRS-III scores in both treadmill intervention groups (PTT: –6.7 points, CTT: –4.1 points) underlining the clinical importance of treadmill training as a complementary treatment option. In particular, the substantial improvement for UPDRS-III in our PTT group is supported by large effect sizes (d = –1.22). The effects on motor improvements in CTT and PTT were comparable to the established pharmacologically L-Dopa treatment in PD even at moderate or high L-Dopa dosage escalation. In a large clinical study with de-novo patients, 150 mg/d L-Dopa (moderate dose) resulted in 3.8 points reduction of UPDRS-III, and 600 mg/d L-Dopa (high dose) in 6.5 points respectively after 9 weeks of treatment (15, 16). Vice versa, PD patients that were not treated with antiparkinsonian medication (patients in the placebo arms of treatment studies) reach an annual rate of disease progression of 5 to 9 points in the UPDRS-III during the initial course of the disease (5 years) [29–34]. Since all PD patients in our study (CTT, PTT, BMT) received best medical treatment, these findings suggest that treadmill training, in particular PTT appeared to considerably reduce progression of global motor symptoms beyond L-Dopa response. These results indicate that treadmill training may serve as effective and additive therapy for motor symptoms in PD. Especially PTT substantially improves motor symptoms on top of L-Dopa treatment.
Practicability of the intervention, acceptance, and adverse events
In this proof-of-concept study, the intervention showed good practicability, was in general well accepted, and the compliance was very high in both groups as reported in the primary publication of this trial [16]. Most participants were able to walk completely without handrail support (CTT: n = 11, PTT: n = 11) or for more than 50 % of the sessions (CTT: n = 5, PTT: n = 3). Adverse events registered in this trial were overwhelmingly unrelated to the intervention (n = 4). In one potentially related case (n = 1 in the PTT group), there was a strong link to orthopedic comorbidity that already existed prior to participation in this study. For future studies, the level of training intensity with regard to the perturbation component should be individually adapted for each patient. In addition, a combined cardial-orthopedic pre-assessment is required prior treadmill intervention in order to assess comorbidities.
Limitations
Although this study revealed promising results, between-group effects for clinical scores did not reach significance level. Our sample size was comparable to the majority of treadmill therapy studies in PD, yet statistical power was low as indicated by post-hoc power analysis. This analysis revealed a power of ≤36% meaning that statistical power was low due to the restricted sample size. At least 70 patients per group are necessary to detect statistically relevant between-group effects for clinical rating scales. A-priori power calculations were performed for the primary outcomes (change of walking speed, change of Mini-BESTest) and not for the UPDRS-III rating (secondary outcome). Future work must evaluate the effects of PTT on clinical motor symptoms in larger multi-center studies. Furthermore, the BMT cohort was retrospectively matched to the intervention cohorts (PTT, CTT). Since the time period to the follow-up visit was different between groups, the comparisons between groups should be interpreted carefully.
Conclusions
In conclusion, our pilot study revealed that eight weeks of treadmill training (CTT, PTT) improved motor symptoms in PD assessed by the UPDRS-III. Interestingly, beneficial effects on the PIGD score and clinical motor symptoms ‘Gait’ and ‘Postural stability’ were observed in the PTT group only. However, significant between-group effects were not detected. The positive effects of PTT persisted after 3 months and are of additive nature in comparison to standard medical care. In summary, PTT may be an effective and sustainable additive therapy for the improvement of motor symptoms, in particular gait and balance deficits in PD. Future studies should focus on long-term effects of PTT (≥12 months) in larger cohorts.
CONFLICT OF INTEREST STATEMENT
WA declares no conflict of interest. KP, SK, and SS have received institutional research grants from Deutsche Stiftung Neurologie (German Foundation Neurology) and together with BE, CP, HG, JK and JW from the Emerging Field Initiative of the Friedrich-Alexander University Erlangen-Nürnberg (EFI Moves, 2 Med 03). BE holds ownerships of Portabiles HealthCare Technologies GmbH and Portabiles GmbH, received compensation and honoraria from serving on scientific advisory boards for Abbvie GmbH, adidas GmbH, Bosch Sensortec GmbH, and ST Sportservice GmbH. KP has received institutional research grants from the Federal Ministry of Education and Research (BMBF), the German Statutory Pension Insurance, the Bavarian Virtual University German Foundation Neurology as well as compensation or honorary for serving on scientific advisory boards, lecturing or reviewing from Abbvie Gmbh, German Statutory Pension Insurance, BZgA, MS-Franken, Landkreis ERH and ICF. JW reports personal fees outside of the submitted work from Teva GmbH, Desitin Arzneimittel GmbH, Abbvie GmbH & Co. KG, and Biogen GmbH. JK reports institutional research grants from Bavarian Research Foundation; Emerging Field Initiative, FAU; EIT-Health; EIT-Digital; EU (H2020), German Research Foundation (DFG); BMBF. Industry sponsored institutional IITs and grants from Teva GmbH; Licher MT GmbH; Astrum IT GmbH; Alpha-Telemed AG. He holds shares of Portabiles HealthCare Technologies GmbH, Portabiles GmbH, Alpha-Telemed AG, and received compensation and honoraria from serving on scientific advisory boards for LicherMT GmbH, Abbvie GmbH, UCB Pharma GmbH, Athenion GmbH, and Thomashilfen GmbH; as well as lecturing from UCB Pharma GmbH, TEVA Pharma GmbH, Licher MT GmbH, Desitin GmbH, Abbvie GmbH, Solvay Pharmaceuticals, and Ever Neuro Pharma GmbH; Dr. Klucken has a patent related to gait assessments pending.
Footnotes
ACKNOWLEDGMENTS
We thank Dr. Z. Kohl, Dr. F. Marxreiter, Dr. J. Schlachetzki and Dr. M. Regensburger supporting patient recruitment, and Dr. S. Krinner (orthopedic assessment). We also thank Kathrin Weinmann, Susanne Seifert, and Julius Hannink supporting data acquisition. Furthermore, we thank the physiotherapists Marie-Kristin Dunker, Lyusyena Novokreshchenova, Surendar Devan, and Hasan Tariq. We especially thank our PD patients and families for participating in this study.
The clinical trial was supported by the Emerging Fields Initiative of the Friedrich-Alexander University Erlangen-Nürnberg, Germany (EFI Moves, 2 Med 03), and by the German Foundation Neurology (Deutsche Stiftung Neurologie). Further, the Bavarian State Ministry for Education, Science and the Arts, Munich, Germany (MotionLab@Home, E|Home Center), the Bavarian Ministry of Economic Affairs and Media, Energy and Technology (Medical Valley Award 2016, Risk-e-Gait) and MoveIT, an EIT Health innovation project, supported this work. Study sponsors were not involved in the study design, the collection, analysis, and interpretation of data, in the writing of the manuscript, or decision to submit the manuscript for publication.
