Abstract
Background:
Aberrant plasticity is closely linked to the development of levodopa-induced dyskinesia (LID) in Parkinson’s disease (PD).
Objective:
This study investigated whether dominant-side patients with PD exhibit a shorter time to LID development, based on the hypothesis that the dominant hemisphere may have greater plasticity than non-dominant-side patients.
Methods:
We analyzed data from 387 right-handed patients with PD who exhibited asymmetric motor deficits and received PD medications for ≥2 years (191 dominant-side and 196 non-dominant-side patients). The influence of side onset on time for LID development was assessed by Kaplan-Meier estimates and time-dependent Cox regression models based on the 5-year time point, after adjusting for age at PD onset, dopamine transporter activity in the posterior putamen, and daily levodopa dose.
Results:
LID developed in 46 (23.4%) patients with non-dominant-side PD and in 35 (18.1%) patients with dominant-side PD. The Kaplan-Meier analyses revealed that non-dominant-side patients developed LID earlier than dominant-side patients (p = 0.027). The time-dependent Cox regression models showed that the risk of LID within 5 years of treatment was significantly higher in non-dominant-side than in dominant-side patients (hazard ratio 1.954; p = 0.034), whereas the risk after 5 years was similar between groups (p = 0.528).
Conclusions:
The present study demonstrated that LID developed earlier in non-dominant-side than in dominant-side patients with PD. These results suggested a greater potential of synaptic plasticity in the dominant hemisphere that may exert a protective role for the development of LID compared to the non-dominant hemisphere.
INTRODUCTION
Levodopa-induce dyskinesia (LID) is an inevitable consequence of chronic dopaminergic therapy in most patients with Parkinson’s disease (PD) [1]; however, the timing of its development varies among patients. The mechanism underlying LID may be associated with altered synaptic plasticity within the denervated striatum in response to chronic pulsatile stimulations with exogenous dopamine replacement [2, 3]. Polymorphisms in brain-derived neurotrophic factor (BDNF) influence the time to develop LID [4], which supports the role of the innate capacity of neural plasticity in the development of LID.
Handedness, defined as hand preference or asymmetrical performance of motor tasks, is the most prominent human behavioral asymmetry [5]. In the brain, the dominant motor cortex shows a greater dispersion of elementary movement representations with more profuse horizontal connections compared with the non-dominant motor cortex [6]. Increased profuse interconnections in the dominant hemisphere may offer greater potential to form experience-dependent excitatory and inhibitory interactions between elementary movement representations [6, 7], which consequently, provide more efficient plasticity, compared with the non-dominant hemisphere. Accordingly, use-dependent facilitation of motor-evoked potentials (MEPs) is greater in the dominant than the non-dominant hemisphere [8, 9]. Furthermore, MEP facilitation, induced by repeated paired magnetic stimulation [10] or paired associative simulation [11], is greater in the dominant than the non-dominant hemisphere, suggesting that the potential for plastic synaptic change is greater in the dominant than non-dominant hemisphere.
The unilateral onset and persistent asymmetry of motor signs are unique features of PD [12]. In addition, LID first appears on the side of the body most affected by PD motor symptoms [13]. In a previous paper, we have shown that patients with dominant-side PD have better motor compensation than those with non-dominant-side PD [14], suggesting that side onset contributes to motor manifestations in PD. If LID is associated with plastic capacity of the brain, we hypothesize that dominant-side patients may develop LID earlier than non-dominant-side patients, because there is greater potential for plastic synaptic change in the dominant hemisphere, which may serve as a substrate for LID development. Task-specific hand dystonia, such as musician’s cramp, could be an additional example of aberrant motor plasticity [15]. Approximately 80% of musician’s cramp in keyboard players occurs in the dominant hand [16], which supports our assumption; however, the contribution of fine motor stress to a greater degree in the dominant compared with the non-dominant hand could not be excluded. We performed this study to investigate whether side onset of PD influenced the time to develop LID.
METHODS
Subjects
Study subjects were selected from the database of Yonsei Parkinson Center (consecutive patient sample from April 2009 to June 2015), who fulfilled the following selection criteria: (1) drug-naïve patients with PD with documented right-handedness; (2) patients who underwent dopamine transporter (DAT) imaging, using [18F] N-(3-Fluoropropyl)-2β-carbon ethoxy-3β-(4-iodophenyl) nortropane positron emission tomography (FP-CIT PET) scans; and (3) patients who received dopaminergic medications for ≥24 months or longer. PD was diagnosed according to the clinical diagnostic criteria of the UK Brain Bank [17], the presence of appropriate DAT defects on FP-CIT PET scans [18], and the presence of PD drug response during follow-up. Part III of the Unified Parkinson’s Disease Rating Scale (UPDRS-III) was used to assess PD severity in each patient at the time of FP-CIT PET acquisition. Patients had visited our outpatient clinic with 3- to 6-month intervals, and two movement disorder specialists (P.H.L. and Y.H.S.) assessed the presence of LID based on history and direct neurological examination at every visit. Survival duration was defined as the period from the initiation of dopaminergic medication to the onset of LID for patients with LID, or to the last time point of clinic visit for patients without LID. Parkinsonian medications prescribed at each visit were checked. The ethics committee of our hospital had reviewed and approved this study. This study was an analysis of pre-existing data; therefore, attainment of informed consent was exempted.
Assessment of asymmetry
The sum of the UPDRS-III items 20 to 26 (rest tremor, action or postural tremor, rigidity, finger taps, hand movements, rapid alternating movements of the hands, and leg agility) scores were calculated for each side of the body. The UPDRS asymmetry index was calculated using the following equation: (higher sum – lower sum)/(higher sum + lower sum). We adopted the definition of asymmetric PD as a UPDRS asymmetry index ≥0.2 or side difference of UPDRS-III score ≥4, which we have previously published [14]. From a total of 556 patients who fulfilled the selection criteria, 387 patients with asymmetric PD were included in the data analysis. Patients with greater UPDRS-III scores on the right side were defined as dominant-side PD patients, whereas those with greater UPDRS-III scores on the left side were defined as non-dominant-side patients.
Image acquisition and quantitative analysis of PET data
To assess striatal dopamine depletion, we obtained FP-CIT PET data using a GE Discovery STe PET-CT scanner (GE Healthcare, Milwaukee, WI). PET-CT image acquisition was performed according to previously described methodology [14, 19]. Quantitative analyses of FP-CIT PET data were performed as previously described [14, 18]. Image processing was performed using SPM8 (Wellcome Department of Imaging Neuroscience, Institute of Neurology, UCL, London, UK) with Matlab 2013a for Windows (Math Works, Natick, MA, USA). Quantitative analyses were based on volumes of interests (VOIs). Twelve VOIs of bilateral striatal sub-regions and 1 occipital VOI were drawn according to a previously described methodology [18]. DAT activity in each VOI was estimated using the specific/nonspecific binding ratio as a surrogate, which was defined as follows: (mean standardized uptake value of the striatal sub-region VOIs — mean standardized uptake value of the occipital VOI)/mean standardized uptake value of the occipital VOI. DAT activity in the posterior putamen was included in the data analysis, because DAT activity in this region is known to be closely associated with the severity of PD motor symptoms and the most consistent predictor for the development of LID [18, 20].
Statistical analyses
Data were expressed as means±standard deviations. An unpaired t-test was performed to compare numeric variables and a χ2 test was used for non-parametric variables between dominant- and non-dominant-side patients. To illustrate the risk for LID, Kaplan-Meier estimates for LID-free survival were calculated for dominant- and non-dominant-side patients. The association between patient groups and the development of LID was evaluated using log-rank and Breslow tests. Cox proportional hazard models were used to assess the influence of side onset on the time to develop LID. In this analysis, potential confounding factors that were found in our previous paper [20], such as age of PD onset, daily levodopa dose, and DAT activity in the posterior putamen were included as covariates. We tested the proportional-hazards assumption by including an interaction term between patient groups and natural-log-transformed follow-up time with a statistical significance of p < 0.10. Additionally, the assumption of proportionality was checked by log-minus-log survival plots: the time-dependent covariate analysis results were statistically significant (p = 0.077) and log-minus-log plots of the two patient groups were crossed each other (Supplementary Figure 1), which suggested that the proportional-hazards assumption was not reasonable. Thus, we used the time-dependent Cox regression model to assess the effects of side onset on the development of LID based on a 5-year time point (i.e., follow-up period within 5 years vs. >5 years) because the gap between the two groups in log-minus-log plots became closer after this time point. Next, DAT activity in the posterior putamen of the dominant- and non-dominant-side was divided into tertiles based on their distributions. DAT activities in the dominant-side posterior putamen was analyzed in dominant-side patients, while the non-dominant-side posterior putamen was analyzed in non-dominant patients. Kaplan-Meier curves for LID-free survival were compared in patients with different tertile groups. The association between tertile groups and the development of LID was evaluated using pair-wised log-rank tests. All statistical analyses were performed using SPSS Statistics 23 (IBM Corporation, Armonk, NY). A p value < 0.05 (two-tailed) was considered statistically significant.
RESULTS
Demographic characteristics
Among 387 patients with asymmetric PD, 191 (49.4%) were dominant-side patients. Demographic and clinical variables as well as DAT activities in the posterior putamen were shown in (Table 1). Dominant-side patients exhibited lower UPDRS-III scores than non-dominant-side patients (p = 0.044), but other baseline variables as well as DAT activities in the posterior putamen were similar between the two groups. During a median follow-up period of 51 months (range, 24–104 months), 81 patients (20.9%) develop LID. At follow-up, daily levodopa doses and prevalence of LID were also comparable between two groups.
Clinical characteristics and dopamine transporter activity
Data are means±SDs or percentage. PD, Parkinson’s disease; MMSE, Mini-Mental Status Examination; UPDRS-III, Unified Parkinson’s Disease Rating Scale, part III; DAT, dopamine transporter; LID, levodopa-induced dyskinesia.
The effects of side onset on the development of LID
The Kaplan-Meier curves showed that LID-free survival progressively declined with the length of dopaminergic treatment in two groups. However, dominant-side patients showed a higher chance of survival compared to non-dominant-side patients; this difference appeared prominent within earlier years of treatment (PBreslow = 0.027), but waned after 4-5 years of dopaminergic treatment (PLog - rank = 0.170; Fig. 1). Next, we used the time-dependent Cox regression model based on the 5-year time point and adjusted for the effects of potential confounders, including age at PD onset, daily levodopa dose, and DAT activity in the posterior putamen. Accordingly, the risk of LID development within 5 years of treatment was significantly higher in non-dominant-side patients compared with dominant-side patients (hazard ratio [HR], 1.954; 95% confidence interval [CI], 1.053–3.625; p = 0.034), while the risk was similar between groups after 5 years of treatment (Table 2). In addition, age at PD onset (HR, 0.953; 95% CI, 0.929–0.978; p < 0.001), DAT activity in the posterior putamen (HR, 0.372; 95% CI, 0.216–0.642; p < 0.001), and daily levodopa dose (HR, 1.002; 95% CI, 1.001–1.003; p < 0.001) were also significant predictors of LID.
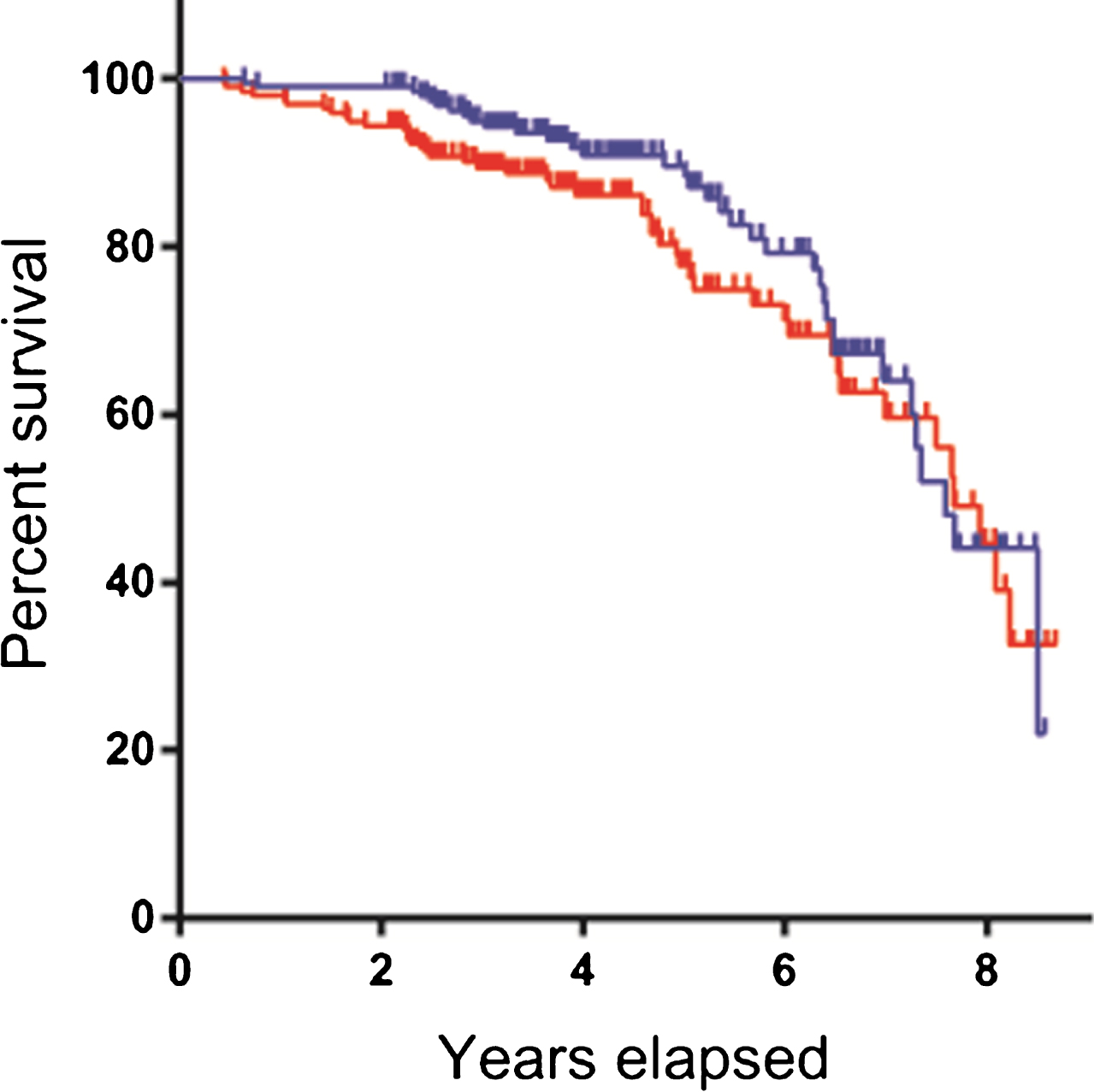
Levodopa-induced dyskinesia-free survival curves. Dominant-side patients (blue line) show a higher chance of survival compared with non-dominant-side patients (red line). This difference appeared prominent within the earlier years of treatment. The crosses in the graphs indicate censored data.
Multivariate Cox hazard models for prediction of the development of LID
LID, levodopa-induced dyskinesia; PD, Parkinson’s disease; DAT, dopamine transporter; CI, confidence interval.
Next, we grouped dominant- and non-dominant-side patients into tertiles based on the distribution of DAT activity in the posterior putamen of the dominant- and non-dominant-side, respectively. Non-dominant-side patients, those with severe DAT reduction (the lowest tertile group) had a higher risk for the development of LID compared with mild reduction (the highest tertile group; p = 0.004). By contrast, no difference was observed between tertiles in dominant-side patients (p = 0.629; Fig. 2).
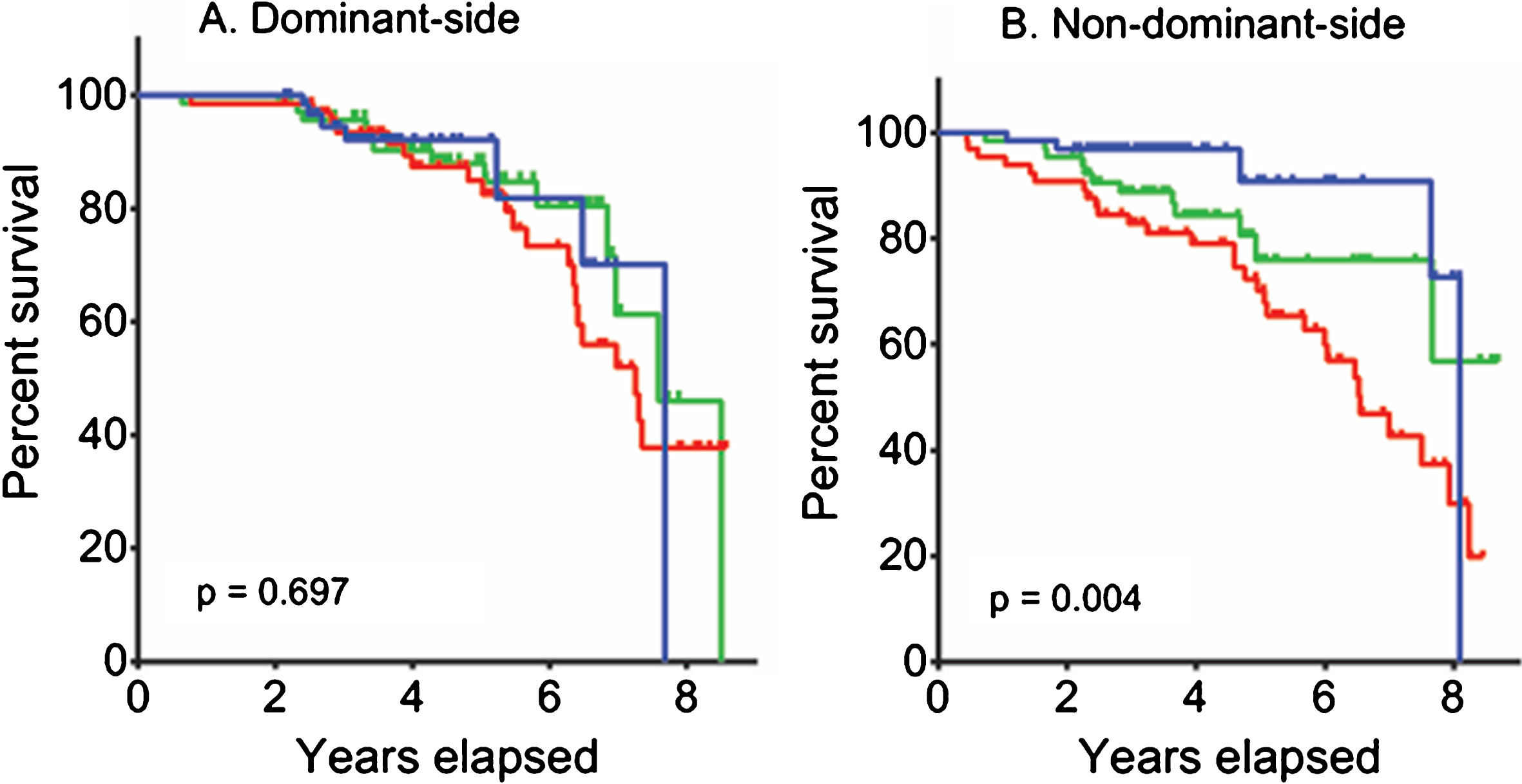
Risk of levodopa-induced dyskinesia according to dopamine transporter activity in the posterior putamen. Levodopa-induced dyskinesia-free survival curves are illustrated using Kaplan-Meier methods for different tertile groups based on dopamine transporter activity in the posterior putamen of the dominant-side in dominant-side patients (A), and the non-dominant-side in non-dominant-side patients (B). In each section, curves for the highest tertile group (i.e., the group showing a mile reduction in dopamine transporter activity; blue line) is adopted as the reference group, then compared with the middle (green line) and lowest tertile (red line) groups. In each section, log-rank comparison p values between the highest and lowest tertile groups are reported.
DISCUSSION
The present study demonstrated the influence of side onset on development of LID in PD. The overall prevalence of LID was similar between dominant- and non-dominant-side patients; however, survival curves and Cox hazard analysis revealed that dominant-side patients had a greater chance of LID-free survival than non-dominant-side patients within 5 years of dopaminergic treatment, even after controlling for potential confounding factors. LID increases with the duration of levodopa treatment, and eventually occurs in most patients with PD. A systematic review has shown that approximately 90% patients with PD develop LID after 9–15 years of levodopa treatment [1]. Thus, the effect of risk factors linked to the development of LID may weaken as treatment duration increases, as shown in the present study. The effect of DAT depletion on the development of LID was observed in non-dominant patients; those with severe depletion exhibited a reduced chance of LID-free survival compared with those with mild depletion. However, this effect was absent in dominant-side patients, suggesting that the dominant hemisphere has a greater capacity to cope with the development of aberrant plasticity, i.e., LID, compared with the non-dominant hemisphere.
In contrast with our initial hypothesis, we have demonstrated that non-dominant-side patients showed an earlier development of LID compared with dominant-side patients. This suggests that more efficient intrinsic connections, i.e., greater potential for plastic synaptic changes, in the dominant hemisphere may enable a greater resistance to the development of aberrant corticostriatal plasticity. Our previous study has shown that dominant-side patients exhibit a greater ability to cope with PD-related pathological changes compared with non-dominant-side patients [14].
The BDNF val66met polymorphism is related to diminished synaptic plasticity in the cortex and striatum [21, 22]; however, it is linked to earlier development of LID [4], providing evidence to support a protective role for plastic capacity in the development of LID. The present study also showed that the degree of DAT depletion in the non-dominant putamen was closely associated with the time to develop LID; however, this relationship was not observed in the dominant putamen. This suggests that the dominant putamen has greater resistance to the development of LID resulting from dopamine depletion. Unilateral dopamine depletion in rats produced greater up-regulation of D2-like receptors in the right compared with the left striatum [23]. Replicating this finding in humans with PD would support the greater vulnerability of LID in the non-dominant compared with the dominant hemisphere.
Enrollment of a relatively large number of de novo patients is advantageous for the prognostic evaluation based on initial presentations. However, this study does have some notable limitations. First, this is a retrospective-designed study; therefore, we cannot draw definitive conclusions. Second, the prevalence of LID in this study (20.9% during a median follow-up of 51 months) was lower than has been previously reported (approximately 30% with 5-year follow-up) [24]. This may be due to our determination of the presence of LID via medical records, which may not detect mild LID. Further prospective studies with more quantitative and objective methodology would be needed. Third, hand dominance in this study was determined using patient’s self-reporting rather than a structured inventory; therefore, we were unable to analyze the influence of the degree of handedness. Finally, enrollment of only right-handed patients obscured whether the present finding also existed in the left-handed patients.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
