Abstract
Background:
A new extended release levodopa capsule (C/L ERC), Rytary®, has demonstrated improved “on” time in fluctuating Parkinson’s disease patients, compared to optimally dosed immediate release levodopa. The milligram dosing, however, differs markedly and no empiric ratio or formula for dose conversion currently exists.
Objective:
To determine the most effective conversion strategy from C/L to C/L ERC.
Methods:
We reviewed fluctuating PD patients with problematic “off” time who were converted to C/L ERC using a semi-structured dose titration schedule, and collected data regarding basic efficacy, tolerability, and dosing, in order to determine an empirically based dose conversion formula. We collected demographics, PD historic data, and other medication use.
Results:
Eighty fluctuating PD patients were given C/L ERC samples, 68 took at least one dose [46 male (67.6%), age 66.6±10.3 y], and 62 had adequate data for dose convergence calculations. At a mean follow-up of 119±101 days, [Range: 24–355 days], 43/68 (63.3%) remained on C/L ERC. CGI-I of “much improved” or “very much improved” was reported by 27/62 (43.5%) and dyskinesia scores from the Movement Disorder Society Unified Parkinson’s Disease Rating Scale item 4.1, (0–4 range)) tended to improve from 0.9±1.1 to 0.5±0.6, P = 0.08. The mean individual daily ratio was 2.0±0.6 : 1, [range 1.0–3.5]. A lower number of baseline daily L-dopa doses predicted a higher conversion ratio, but pre-conversion dyskinesia did not.
Conclusions:
This retrospective study found that C/L ERC was generally well tolerated and preferred by many patients. The mean total daily conversion ratio is 2 : 1.
INTRODUCTION
A novel carbidopa/levodopa extended release capsule (C/L ERC, Rytary®) consisting of tartaric acid, C/L-dopa powder and two different slow release beads, has been approved for use in Parkinson’s disease (PD). [1] In fluctuating patients, overall “on” time was superior to optimally dosed immediate release (IR) C/L and IR C/L with entacapone. [2, 3] The dosing is very different from conventional L-dopa and the official dosing recommendations are based on a priori calculated ranges from pharmacokinetic data that were used in the fluctuating clinical trials, not on empiric data. [2, 4] The majority of subjects in the studies did not conform to these doses, and needed to further increase their doses. A single post-hoc analysis based on several clinical trials reported a dose convergence ratio based on the data derived from those clinical trials, [5] but those trials had regimented dosing increments that did not allow for true dosing flexibility. No conversion ratio using data empirically derived from open and flexible dosing comparisons has been reported.
MATERIALS AND METHODS
We tabulated results in consecutive PD patients with fluctuations, defined by any wearing off, delayed on, incomplete on, or dose failures, who were switched from any other L-dopa preparation to C/L ERC. This was offered to patients who subjectively felt fluctuations were problematic enough to change their L-dopa dosing clinically. We recorded basic demographics (sex, age, duration PD, weight) from their medical record, and rated dyskinesia on Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) Question # 4.1 (0–4, based on percentage of awake day with dyskinesia). We calculated daily L-dopa dose equivalents ((L-dopa controlled release L-dopa*0.7)*1.1 if on a catechol-O-methyltransferase (COMT) inhibitor), number of L-dopa doses/day, and recorded concurrent PD medications. Fluctuating patients who were thought to be candidates for C/L ERC were then given adequate samples to identify an optimal dose, typically about 6–8 weeks, but this varied depending on starting dose and titration schedule. They were all switched immediately to C/L ERC and were given a C/L ERC titration schedule with identical doses taken throughout the day. In general, the initial individual dose was about 2x their initial morning dose of L-dopa equivalent. They were then prescribed at least 4 iterations of increasing doses, in 50 mg/dose increments, and asked to titrate to their optimal dose of C/L ERC, and to continue that dose. Patients were instructed to titrate upward to the next dose, as needed every 1–7 days. There was no set rule on number of doses/day but for the initial transition, 3–4 C/L doses converted to 3 doses of C/L ERC, 5–6 doses converted to 4, and 7 + doses converted to 5 doses for most patients. For example, a L-dopa dose of 100 mg 5x/day could switch to C/L ERC 4x/day starting at 195 mg, then 245 mg, then 290 mg, then 340 mg, then 390 mg. Patients were allowed to change this depending on results. We asked patients to continue other PD meds until the switch was complete and we identified an optimal dose, then allowed any changes. We followed patients until they stopped C/L ERC, or to the latest time point for up to one year from the switch if they continued on C/L ERC. We collected C/L ERC dose, percentage of time with dyskinesia before and after switch (MDS-UPDRS #4.1), clinical global impressions, and reasons for discontinuation at their next regularly scheduled visit, or via phone if needed. Descriptive statistics were performed. An analysis of covariance (ANCOVA) was used to assess the possible effects on the C/L ERC: C/L IR conversion ratio, employing five different variables: age, sex, weight, number of doses/day, and baseline dyskinesia scores. Data was collected from April 2015 to July 2016. The institutional review board waived full IRB consent as treatments were all standard of care.
RESULTS
C/L ERC samples were offered to 80 fluctuating PD patients (Table 1). Three were lost to follow-up, 2 dosed incorrectly and were excluded, 6 decided not to start C/L ERC, and 1 died the day after starting from an unrelated cause. Sixty-eight subjects (46 male (67.6%), age 66.6±10.3 y, duration of PD 10.4±5.0 y) had any follow-up data. Of these, 34 were previously taking exclusively immediate release (IR) C/L, 12 took exclusively controlled release (CR) C/L, and 22 took a combination of IR and CR C/L. Six subjects discontinued after a single dose, prior to establishing any optimal clinical equivalent dose (4 due to immediate adverse event, and 2 due to reduced efficacy, but who only took the lowest initial dose). Sixty-two subjects had adequate data and 43 of those were continuing C/L ERC at their final time point, up to 1 year after initiation. The primarily reasons for discontinuing C/L ERC after starting it were: cost/lack of superiority (9), adverse event (5), worse effect (4), and surgical intervention (1). Patients with prior deep brain stimulation placement (DBS) usually discontinued (7/9). The mean f/u (N = 62) was 119±101 days, [Range: 24–355 days]. The final data point was obtained in clinic (35) or via phone interview (33).
Subject Flow
In subjects with adequate data (N = 62), the mean initial individual C/L ERC dose was 308±117 mg/dose, taken 3–5 times/day. The final dose after titration was 387±141 mg. The number of daily L-dopa doses decreased from 4.4±1.2 to 3.8±0.6. The total daily calculated L-dopa dose increased from 823±432 mg/day to 1,481±636 mg/day, corrected for 3 subjects who remained on entacapone (Table 2). The mean of all the individual C/L ERC to baseline calculated C/L daily ratios was 2.0±0.6 [range 1.0–3.5] to 1. Only the number of L-dopa doses per day significantly affected this ratio. A higher ratio was predicted by a lower number of L-dopa doses per day. (Table 3, Fig. 1) The ratio was not significantly different in subjects without any baseline dyskinesia 2.03±0.83 vs. those with baseline dyskinesia 1.87±0.56. The mean dyskinesia score (0–4) was similar after conversion 0.87±1.1 to 0.83±0.8, NS (N = 62).
Baseline Data and Demographics
Tests of Between-Subjects Effects
aR Squared = 0.198 (Adjusted R Squared = 0.111). bComputed using alpha = 0.05.
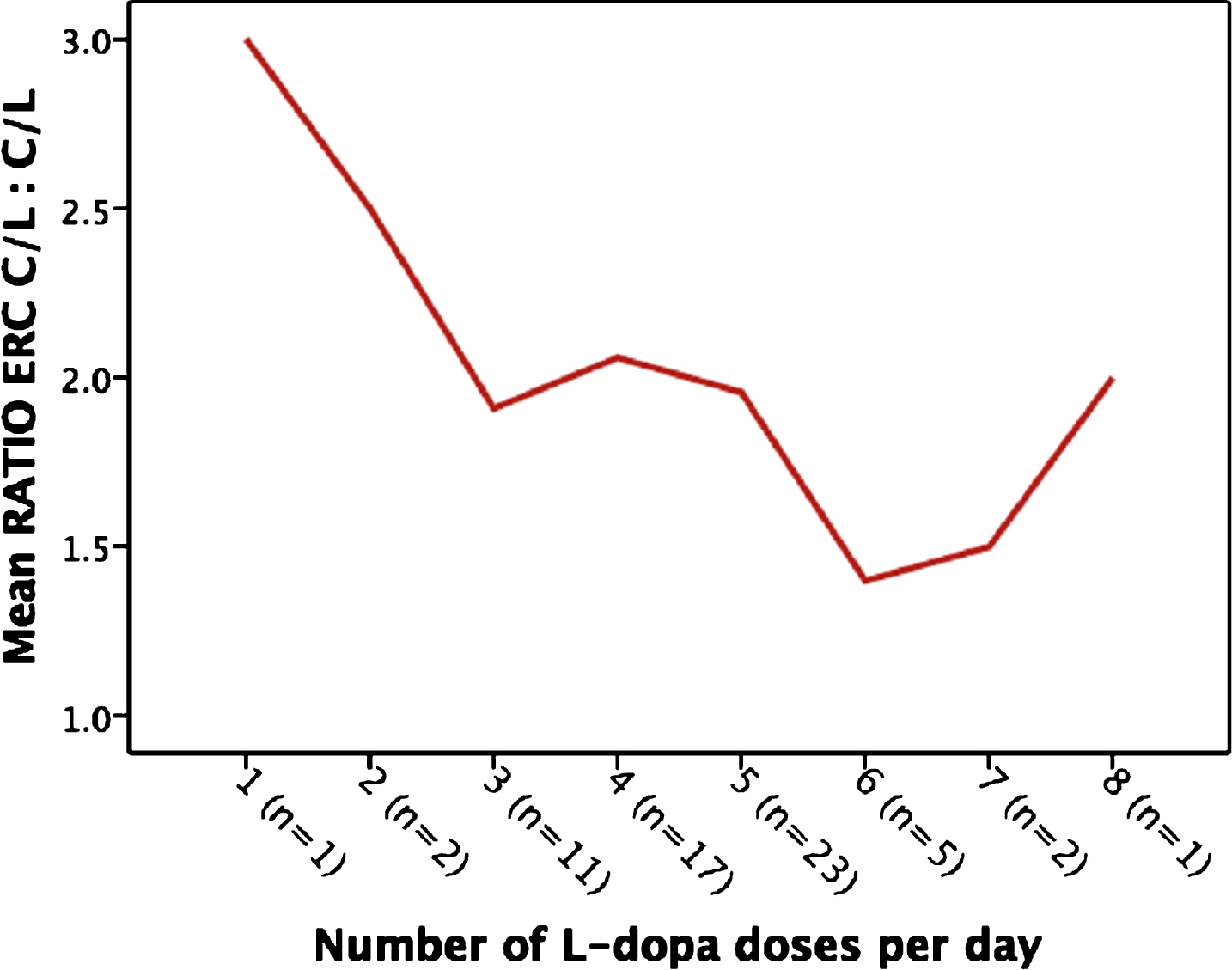
Ratio of C/L ERC to C/L, as a function of baseline C/L doses per day.
After switch, 27/68 subjects reported their overall PD to be “much” or “very much” improved. (Table 4) An adverse event was reported by 15/68 subjects: hallucination (4), increased dyskinesia (3), hypotension (3), increased dreams (3), nausea (3), palpitation (1), edema (1), “felt funny” (1).
Clinical Global Impressions
DISCUSSION
The majority of subjects switched from other C/L preparations to C/L ERC continued on C/L ERC at last follow-up. The daily levodopa dose conversion of 2.0 : 1.0 is similar to that reported by Nausieda et al of 2.1 : 1. In contrast to one consensus paper, the presence of dyskinesia did not significantly mitigate this ratio, although subjects with pre-existing dyskinesia did tend to have a lower conversion ratio. [6] A new or worsening adverse event was reported by 15/68 patients, but dyskinesia did not increase, suggesting that improvement in subjective motor status was not at the expense of more dyskinesia.
This is an open label real world trial reporting on all patients who tried C/L ERC. The main advantage is that it includes a broader scope of patients than in clinical trials, representational of an actual clinic population. There were no formal inclusion/exclusion criteria, but no one was excluded based on age, other medical problems, other medications/surgeries etc. The main disadvantage is that additional variables are added, including aspects such as drug cost and travel logistics. For example, we did not feel we could segregate withdrawal due to cost/efficacy as a common response was paraphrased to “same or a little better, but not worth the extra cost”.
Nevertheless, we feel this report represents the best empiric data currently available to dictate conversion strategies to C/L ERC, and contradicts the official conversion instructions.
CONFLICTS OF INTEREST
There are no conflicts of interest of any author directly related to this report.
Competing interests not related to report:
William Ondo MD:
Grant support: Lundbeck, Parkinson’s Study Group, Sunovian, Luitpold, Restless legs Syndrome Foundation,
Speaker: TEVA, ACADIA, UCBPharma, Lundbeck, Neurocrine, USWorldMeds
Pablo Coss MD: none
Melissa Christie MD: none
Belen Pascual PhD: none
Footnotes
ACKNOWLEDGMENTS
There was no funding for this report and no other acknowledgments.
