Abstract
Background:
Whether dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD) are distinct disorders or two subtypes of the same entity, is not yet fully understood. There is a dearth of knowledge on differences in longitudinal clinical outcomes between DLB and PDD.
Objective:
We aimed to compare longitudinal trend of cognitive decline, mortality, and their determinant factors between patients with DLB and PDD.
Methods:
At baseline, we recruited 1110 DLB, and 764 PDD patients registered in the Swedish dementia registry (SveDem) during 2007–2015. Cognitive status was assessed at baseline and each follow-up visit by mini-mental state examination (MMSE). At least one follow-up MMSE was available for 411 (37.0%) DLB and 229 (30.0%) PDD patients. Rate of cognitive decline and mortality risk were compared between the two groups.
Results:
After an average of 2-years, the rate of cognitive decline did not differ between DLB (1.1 MMSE unit/year) and PDD (1.2 MMSE unit/year) groups (p = 0.970). There was no significant difference in the median survival time between DLB (4.0 years) and PDD (4.1 years) groups (Log rank p = 0.614). Antipsychotic drug use in DLB and larger number of medications in PDD were the most important determinants of faster annual cognitive decline.
Conclusions:
Our findings from real-world clinical practice demonstrated that the rate of cognitive decline and mortality do not differ significantly between DLB and PDD at least over 2 years, yet, various factors might determine clinical outcome in these two groups. It seems that DLB and PDD are probably similar synucleinopathies, with phenotypical variations in the order of manifestations rather than course of progression and clinical outcome.
INTRODUCTION
Epidemiological studies show that dementia with Lewy bodies (DLB) is a common form of dementia in the elderly population with an estimated incidence rate of 112 per 100,000 person-years [1]. Likewise, the incidence of dementia in Parkinson’s disease (PDD) is approximately 100 per 100,000 patient-years [2] with a lifetime risk of as high as 80% in PD population [3]. Diagnostic criteria for DLB include progressive dementia and fluctuating cognition in association with visual hallucination and Parkinsonism [4]; while the diagnosis of PDD requires a period with pure motor symptoms and the beginning of cognitive impairment more than one year after the onset of motor disorder [5]. Recently, new diagnostic criteria have been released by the Movement Disorders Society (MDS) which do not include dementia at diagnosis as an exclusion criterion for PD diagnosis, thus further blurring the distinction between PDD and DLB [6].
Due to the substantial overlap in clinical and pathological characteristics, there is controversy whether DLB and PDD are one entity or two distinct conditions in a continuum between Alzheimer’s disease (AD) and Parkinson’s disease (PD). In a registry-based survey in Sweden, we previously showed that PDD patients referred to memory clinics were younger than DLB patients and required fewer diagnostic tests to establish dementia diagnosis [7]. Moreover, differences in sensitivity to neuroleptic drugs and response to anti-Parkinson and anti-dementia medications have been demonstrated between DLB and PDD [8]. Amyloid pathology is more common in DLB than PDD [9, 10], which may hypothetically result in different trends of cognitive decline between the two conditions. It has been shown that the pattern of cognitive dysfunction including attention, episodic memory, and executive functions, differ between patients with DLB, AD and PDD [11].
For a better understanding of the underlying pathology of neurodegeneration, further evidence is required whether clinical outcome differs significantly between DLB and PDD. Most of the previous studies are single-center investigations with small sample size which lack longitudinal follow-ups. In addition, the role of different demographic variables, medications, and other determinants on trend of cognitive decline and mortality in DLB and PDD have not yet been completely understood. Using data from the Swedish Dementia Registry (SveDem), we firstly aimed to compare the longitudinal trend of cognitive decline and mortality between large cohorts of individuals with DLB and PDD in a real-world clinical setting. Secondly, determinant factors of clinical outcome were investigated and compared in DLB and PDD.
METHODS
Participants recruitment
Data were obtained from the Swedish Dementia Registry (SveDem) (for details see http://www.svedem.se) with newly diagnosed dementia patients from the memory clinics and primary care units in Sweden. SveDem is a national web-based quality registry, established to improve the quality of diagnostic work-up, treatment and care of dementia across the country [12]. Up to the end of 2015, 58037 individuals with dementia were registered in SveDem, covering 35–40% of the entire expected yearly incident dementia cases [13]. In SveDem, dementia is generally diagnosed according to the ICD-10 criteria (WHO, 1993) [14] and registered in this incident-based survey since 2007. For the current study, we recruited data registered for all cases diagnosed with DLB (n = 1110) and PDD (764) in SveDem up until 30 December 2015. Diagnosis of DLB was based on the modified criteria recommended by the DLB Consortium [4] and PDD was diagnosed using the Emre et al. criteria [5]. These diagnoses were mostly made by specialists (i.e., geriatrician, neurologist or psychiatrist) in memory clinics (93%), or general physicians in primary care units or nursing homes (7%).
Ethical consideration
Patients and their relatives are informed orally and in writing about the register and can decline participation or withdraw consent. Ethical application for using longitudinal data registered in SveDem was approved by the regional ethics committee in Stockholm with the reference number 2015-1313-32.
Assessments
Baseline data consisting of age at dementia diagnosis, sex, living conditions, initial cognitive assessment by Mini-Mental State Examination (MMSE), medications and support from community (i.e., daycare, homecare) were obtained from the database. In SveDem, type of medication is recorded as anti-dementia treatment consisting of cholinesterase inhibitors and NMDA-antagonists, cardiovascular drugs, antidepressants, antipsychotics, anxiolytics, and hypnotics agents. The total number of regular daily medications was also calculated in each participant. Detailed data on anti-Parkinson medications, however, is not available in SveDem.
Outcome measurements
In SveDem, patients are usually followed-up every year. At each follow-up visit, cognitive status is re-assessed by MMSE. The latest date for follow-up data retrieval was 12 April 2016. One main outcome variable in this study was defined as the annual rate of cognitive decline measured by change in MMSE score divided by the follow-up time in year. By this calculation, adjusted for the follow-up time with valid cognitive assessment in each participant, we overcome the risk for survival bias since 44.3% of the participants died throughout the follow-up period. Secondly, we also defined mortality rate as another outcome measure. Death event and date are recorded in SveDem during the follow-up period if it occurs for any participant.
Statistical methods
Sample size calculation
To show a significant difference of at least 0.5-unit decline in MMSE scores between the DLB and PDD groups over the follow-up period, assuming standard deviation (SD) of 1 unit for the changes in MMSE scores and 0.05 chance for type I (α) error, at least 64 participants in each group were needed to achieve the statistical power of 80%. Our current sample size surpasses the minimum requirement and statistical power is fully met.
Univariate and multivariate analysis
Univariate between-group comparisons were performed using Chi2 or independent-samples t test where appropriate. Multivariate linear regression model was applied to compare rate of cognitive decline between the cohorts while adjusting for covariates, namely onset age, sex, and number of daily medications. We used multivariate ordinal probit regression to identify variables associated with faster cognitive decline within each cohort. In the ordinal probit models, quartile of the annual rate of cognitive decline was considered as the outcome and a comprehensive list of baseline, demographics and medication data were entered as independent variables. For each variable, corresponding regression coefficient (B) and the 95% confidence interval (CI) is reported.
Longitudinal data analysis
We used repeated measure ANOVA to compare the trajectory of cognitive decline recorded by MMSE scores throughout the consecutive follow-up visits. The time*group interaction effect was reported as the main statistic of interest.
Survival analysis
To compare mortality rate and the median survival time between the cohorts, Kaplan-Meier analysis and the Log rank test were used. We applied multivariate Cox regression models to compare the hazard for death between DLB, and PDD cohorts, while adjusting for the potential effect of onset age, sex, and number of daily medications. Moreover, Cox regression models were performed to identify factors relating to a faster or slower mortality rate over the follow-up period within each group. Hazard ratio (HR) and 95% CI was calculated for each determinant variable in each Cox model.
Statistical analyses were performed using IBM SPSS Statistics software (version 23.0). A two-tailed p-value of <0.05 was considered as the threshold for significant differences or associations in all analyses.
RESULTS
Demographics and baseline description
Baseline and demographic data were available for 1110 participants with DLB and 764 individuals with PDD (Table 1). In the entire study population, 1169 (62.4%) were male with no significant difference between DLB and PDD groups. The average age at the time of establishing the dementia diagnosis and thus baseline registration in SveDem was 1.5 years older in DLB patients (76.7 vs. 75.2 years, p < 0.001). However, at the time of dementia diagnosis, the PDD group had significantly lower MMSE score (20.7 vs. 21.4, p = 0.001). Regarding medications, after the diagnosis 84.1% of the DLB and 66.0% of the PDD cohort received treatment with anti-dementia medication (p < 0.001). However, PDD patients were receiving larger number of daily medications (6.3 vs. 4.7/day, p < 0.001).
Baseline and medication characteristics of the study groups
NS, not statistically significant; DLB, dementia with Lewy bodies; PDD, Parkinson’s disease dementia.
Cognitive decline
At least one follow-up data on cognitive assessment was available for 411 (37.0%) DLB and 229 (30.0%) PDD patients (Table 1). Rate of cognitive decline did not significantly differ between the two groups (1.1 MMSE unit/year in DLB and 1.2 MMSE unit/year in PDD) (p = 0.970). Results from repeated measures ANOVA also showed a similar trend in cognitive decline after three consecutive assessments, as illustrated in Fig. 1 (time*group p = 0.820). Adjusting for the effects of age, sex, and number of daily medications, DLB cohort had on average 0.62 (95% CI: –1.44 – +0.21) units less annual decline in MMSE score compared to the PDD group; however, the difference was not statistically significant (multivariate-adjusted p = 0.142).
We applied multivariate ordinal probit regression model to investigate factors associated with faster cognitive decline in each of the cohorts. As summarized in Table 2, antipsychotic drugs (B = 0.507, p = 0.039) in DLB and larger number of daily medications (B = 0.075, p = 0.056) in PDD were the most important determinant of faster annual decline in MMSE score.
Predictors of cognitive decline in dementia with Lewy bodies and Parkinson’s disease dementia (multivariate ordinal probit regression model to predict quartiles of rate of cognitive decline)
*Follow-up data on cognitive assessment was available in 411 DLB and 229 PDD patients (Table 1, Fig. 1); however, in the regression model some cases were excluded due to missing values in the list of covariates. NS, non-significant. Statistical significant regression coefficients are bolded.
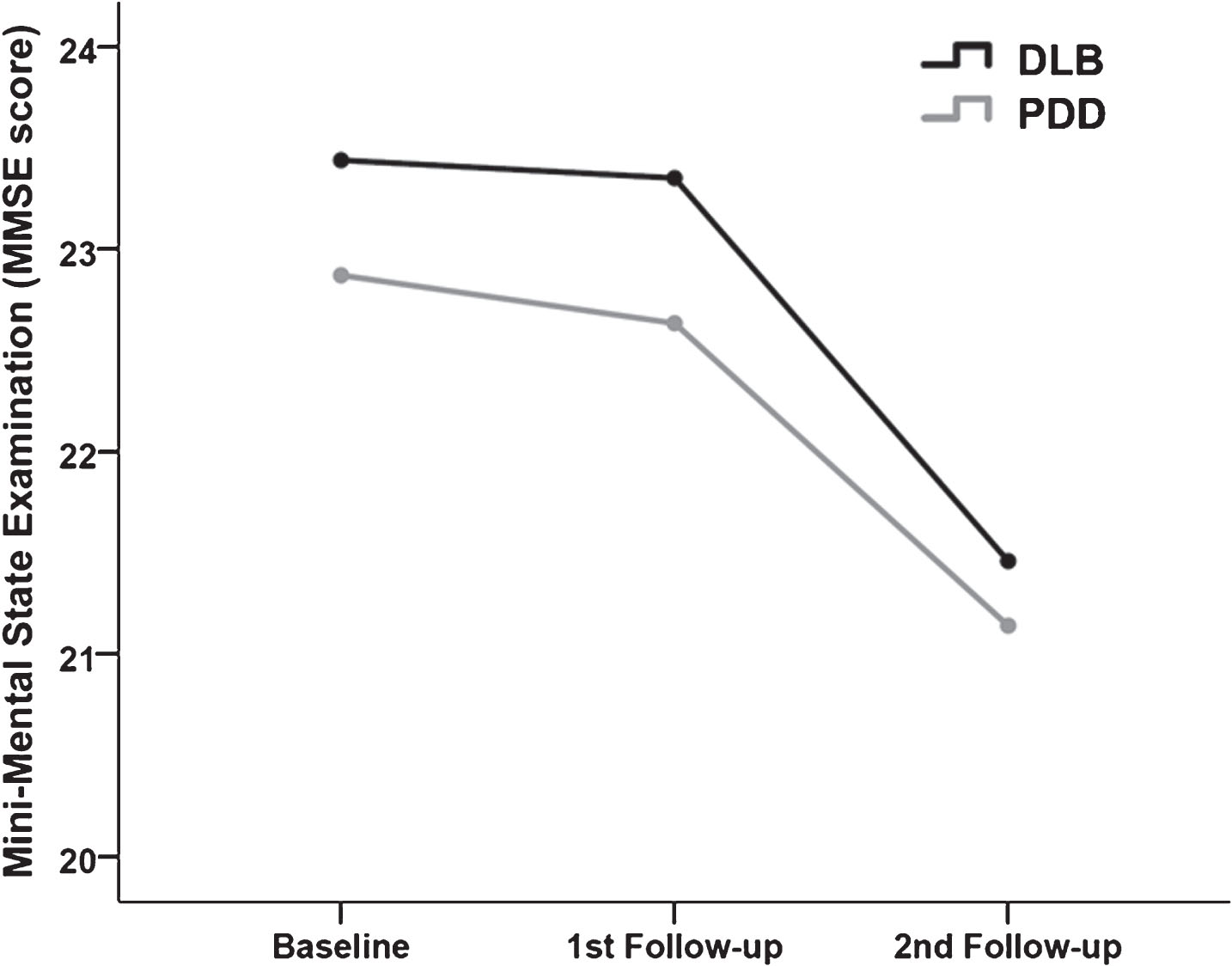
Trajectory of cognitive decline repeatedly measured by mini-mental state examination (MMSE) in patients with dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD) (repeated measurement ANOVA p-value = 0.820).
Mortality
During follow-up, 502 (45.2%) DLB and 329 (43.1%) PDD patients died (Chi square = 0.857, p = 0.354). Baseline characteristics of the participants who died during follow-up are summarized in Table 1. At the time of dementia diagnosis, the median survival time was calculated as 4.0 years in DLB and 4.1 years in PDD using Kaplan-Meier statistic. As illustrated in Fig. 2, there is no significant difference in the mortality rate between DLB and PDD groups (Log rank test p = 0.614). Adjusting for age, sex, number of daily medications and baseline MMSE, results from a multivariate Cox regression model showed no significant difference in the hazard of death between the two groups (HR = 1.00, 95% CI: 0.86–1.16 for DLB vs. PDD, p = 0.975).
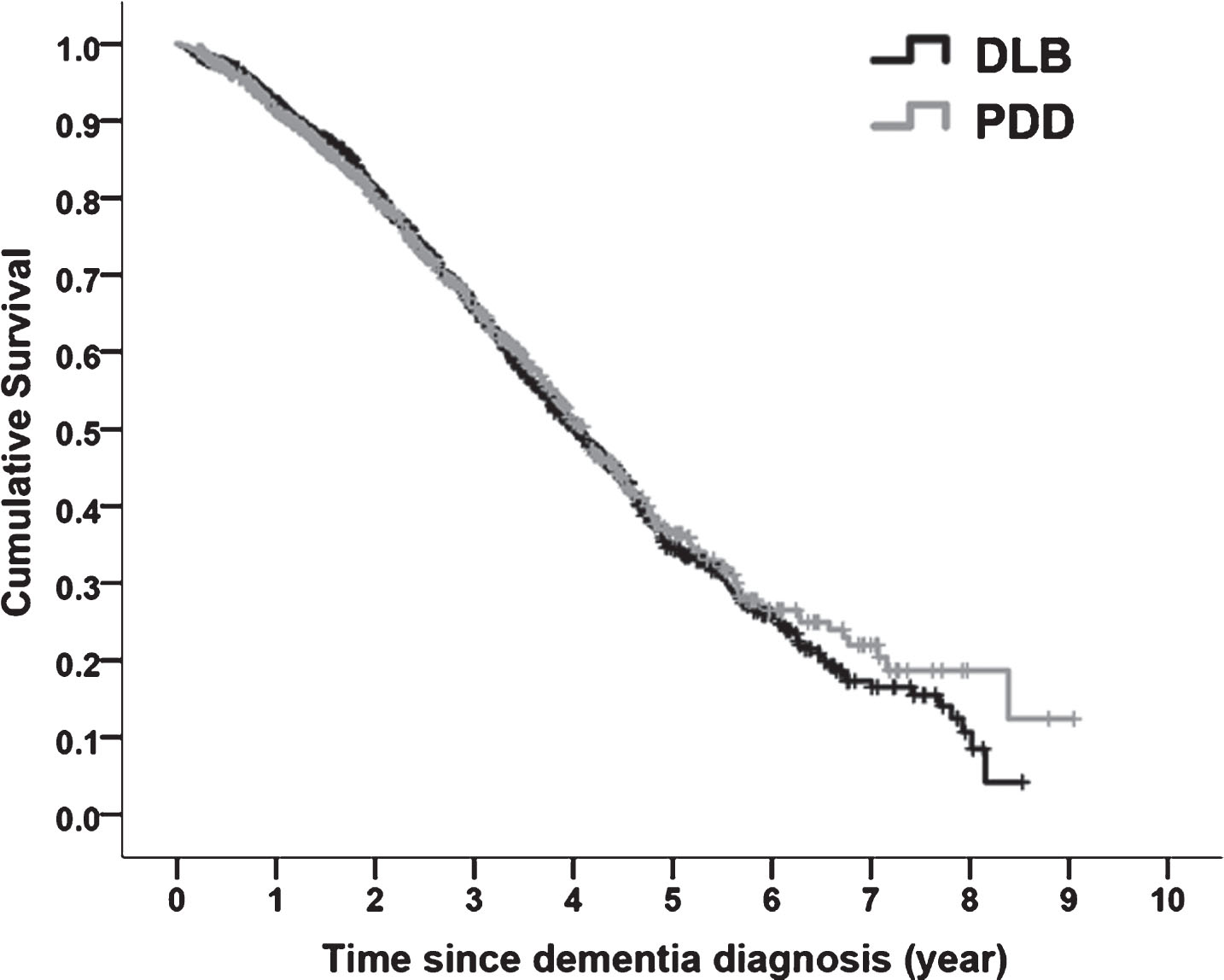
Kaplan Meier survival curve for mortality outcome in patients with dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD) (Log rank test p-value = 0.614).
We further assessed factors influencing mortality in each study cohort. As summarized in Table 3, male sex, older age at dementia diagnosis and lower BMI were common risk factors of higher mortality rate in both cohorts. In addition, larger number of medications increased hazard of death in DLB patients (HR = 1.04, 95% CI: 1.00–1.08). Only among the PDD population, cardiovascular (HR = 1.48, 95% CI: 1.04–2.09) and antidepressant medications (HR = 1.76, 95% CI: 1.28–2.42) were associated with a larger hazard of death, while receiving anti-dementia drugs reduced hazard of death by 29% (HR = 0.71, 95% CI: 0.51–0.97).
Predictors of mortality in dementia with Lewy bodies and Parkinson’s disease dementia (multivariate cox regression model to predict mortality)
*Data on mortality outcome was available for all participants showing that 502 DLB and 329 PDD patients died during the follow-up. HR, hazard ratio; NS, non-significant. Statistical significant regression coefficients are bolded.
DISCUSSION
Main findings
Our study is one of the largest to compare clinical outcome between DLB and PDD cohorts over time. In general, most of the differences between these two cohorts were quite ignorable. Neither the rate of cognitive decline nor mortality risk were significantly different between DLB and PDD groups. Yet, some of the factors influencing clinical outcome may differ between the two groups. Higher use of antipsychotic medications probably reflecting burden of psychotic symptoms seemed to be an important predictor of faster cognitive decline in DLB. However, in patients with PDD, number of daily medications in general increased the risk of more rapid cognitive decline. In the PDD cohort, daily medications include a number of dopaminergic medications for parkinsonism; hence, this risk factor is likely to reflect severity of parkinsonism represented by more anti-Parkinson medications, which could also affect cognitive status in PDD patients. In line with our previous reports [15, 16], female sex and higher BMI were associated with decreased mortality rate in both cohorts. Specifically, in the PDD cohort, higher burden of cardiovascular diseases and depression (represented by cardiovascular medications and antidepressants) increased the hazard of death by 48% and 76%, respectively. While on the other hand, anti-dementia medication decreased mortality risk by 29% in PDD patients. This is in line with previous findings by our group reporting reduced mortality in AD patients treated with cholinesterase-inhibitors [17]. Nevertheless, one should also consider the potential confounding role of cardiovascular burden in the protective association between anti-dementia medications and mortality. Using data from the same cohort, SveDem, we have previously shown that patients with dementia tend not to be prescribed acetylcholinesterase inhibitors if they have higher cardiovascular co-morbidity burden (46% vs. 58%) [18].
Existing evidence
Even though many studies have investigated cognitive profile in DLB and PDD patients, there is a dearth of information on natural longitudinal history of cognitive decline and mortality in DLB and PDD from population-representative cohorts. A systematic review on 18 studies showed that people with DLB, on average, experience 3.4 units annual decline in MMSE score [19]. In our study, however, most of the participants were possibly in the early-stage of disease and therefore, the annual rate of cognitive decline was slower (1.1 in DLB and 1.2 in PDD). Another study has compared cognitive profile between 10 DLB, 17 PDD and 76 AD patients, which demonstrated significant differences. Frontal executive function was more impaired in DLB and PDD groups, while episodic verbal memory was more preserved in PDD patients compared to AD and DLB [11]. However, this was a cross-sectional study with rather small sample size in each group. Our findings are also in line with those reported by Kramberger et al. in a prospective multicenter study on 715 DLB and 280 PDD patients with two years of follow-up [20]. Similarly, they found neither statistical nor clinical significant difference in the mean annual decline of the MMSE score between DLB and PDD. Recently, in a population-based study, researchers have assessed mortality rate in individuals with a synucleinopathy diagnosis, among which 81 with DLB and 55 with PDD were included [21]. Compared with the general population, both DLB and PDD groups demonstrated a higher risk of death, yet with a similar HR (3.94 in DLB and 3.86 in PDD) [21]. In another study from a Norwegian population, the median survival time among individuals with DLB/PDD was estimated 4.4 years [22]. These reports are in line with our findings where no significant difference was observed in the mortality rate between DLB and PDD cohorts and the median of survival time at the time of dementia diagnosis was approximately 4 years in both cohorts. Although the DLB cohort had slightly higher MMSE score at diagnosis (21.4 vs. 20.7), still no significant difference was found in the mortality outcome between the two cohorts after adjusting the Cox regression model for baseline MMSE as a covariate.
Limitations and strength
We acknowledge some limitations of our study. Using data from a naturalistic registry-based data, we had no access to core clinical features of DLB and PDD (e.g., hallucination, motor symptoms), comprehensive cognitive assessments on various functional domains and detailed neuropsychiatric features. Only MMSE was available in SveDem as a global cognitive measure. Secondly, in SveDem as a large dementia registry, possible diagnosis of DLB and PDD is based on the latest guidelines; yet, no postmortem validation data is available to confirm diagnostic accuracy. Third, the average follow-up time in our study was nearly two years (maximum 8 years) which is still a short period to evaluate trajectories of cognitive status. In the future, updated results are required with a longer follow-up assessment. The rate of participants with follow-up cognitive assessment was slightly higher in DLB group. This probably reflect the fact that PDD patients are more likely to be visited by neurologists in Movement Disorder Clinics instead of Memory Clinics. In both groups, participants with no follow-up data were significantly older and had more severe dementia at baseline (data not shown). Nevertheless, there was no statistical interaction between group membership (DLB vs. PDD) and the association between baseline age and cognitive status with being followed-up. Therefore, the risk for attrition bias was low and the difference in attrition rates between the two groups was non-differential.
The current study has several strengths. First, it was performed using data from one of the largest cohorts of DLB and PDD that have been longitudinally followed-up. Our study had a very high statistical power to infer lack of significant difference between these two groups. Lastly, SveDem is a naturalistic cohort reflecting real-world clinical practice, which increases the generalizability of our findings.
Conclusion
Patients with PDD and DLB are consistent in several neuropsychological features such as frontal/executive dysfunction and prominent perceptual and visuospatial deficits [23]. Even though recent animal and human post-mortem studies have suggested some differences in spreading patterns of α-synuclein pathology in DLB and PDD [24], still their clinical outcome is more similar when compared to other dementia types. Considering DLB and PDD as distinct, but related disorders seems to be a more conceptual argument based on definition criteria rather than real differences in clinical outcome [25]. Our findings from real-world clinical practice demonstrated that rate of cognitive decline and mortality do not differ significantly between DLB and PDD, yet, various factors may contribute in clinical outcome in these two groups.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This study was financially supported by the grant received from the Research Foundation of the Swedish Parkinson’s Disease Association and we would like to appreciate their generosity. The authors are grateful to all colleagues and co-workers in SveDem for providing data as well as many thanks to all participants in SveDem including patients, caregivers, and staff. Data collection in SveDem is supported by the the Swedish Association of Local Authorities and Regions. ME was supported by the Swedish research council (grant 2016-02317) - and by a grant from the order of St John.
