Abstract
Background:
Subthalamic nucleus deep brain stimulation (STN-DBS) is an established treatment for selected Parkinson’s disease (PD) patients, but therapy is often limited by side effects. Previous studies indicate an inverse relationship of the therapeutic window (TW) to pulse width (PW) settings down to 60μs, but there is limited data available on the effect of shorter PWs.
Objective:
To define the TW of STN-DBS in PD at PW of 30μs (PW30) relative to standard PW settings at 60μs (PW60), and to compare speed of gait and speech intelligibility on the two PW conditions.
Methods:
Monopolar review data of 15 consecutive PD patients who had screening of contacts performed at PW60 and PW30 was used to calculate the TW at each contact. We compared the TWs of the most efficacious contact per STN, and a secondary analysis was performed comparing all contacts. Speed of gait with a timed 10 metre walk test, speech intelligibility, and perceptual characteristics of speech were also compared at the efficacy thresholds for PW60 and PW30.
Results:
The TW was significantly greater at PW30 [3.8±1.6mA] than at PW60 [1.7±1.1mA]. In the secondary analysis, 110 TWs could be calculated and these remained significantly higher at PW30. The timed 10 metre walk at PW30 was faster than at PW60, and perceptual rating scores of speech were significantly improved at PW30.
Conclusions:
STN-DBS in PD patients using a PW of 30μs significantly increases the TW compared to standard PW settings, and this effect is consistent across all contacts of an electrode. Speed of gait and perceptual speech scores are also improved at 30μs settings.
Keywords
INTRODUCTION
Deep brain stimulation (DBS) of the subthalamic nucleus (STN) is a well-established treatment option for patients with Parkinson’s disease (PD) with motor fluctuations and dyskinesias refractory to medical therapy [1–4]. However, the use of conventional programming parameters presents limitations in some patients due to the emergence of side effects at levels of stimulation required to control Parkinsonian motor symptoms [5–9].
The stimulation amplitude, represented by current or voltage, is the usual parameter titrated to adjust the level of stimulation. This has led to amplitude being used as the basis for defining the therapeutic window (TW), which is the magnitude of difference between the threshold for the required therapeutic effect and the threshold for side effects. Early studies examining the effect of pulse width (PW) between 60 and 450μs on therapeutic and adverse effects in PD patients noted wider therapeutic windows at lower PWs. The lowest available PW of 60μs on conventional systems was established as the standard value to be used in STN-DBS for optimal symptom control while minimising side effects in PD patients over the last two decades [10–13].
With technological advances in the last few years, it has become possible to utilise pulse width settings of less than 60μs with some DBS systems. Moreover, there are suggestions from modelling data that various PWs may selectively stimulate different neural elements [14, 15]. A wider TW enabled by using a pulse width shorter than 60μs may theoretically provide more scope to increase stimulation both initially and over time to the level required for optimal control of PD motor symptoms without the occurrence of adverse effects.
There has, however, been little published data looking at whether the effect of increasing the TW can be extrapolated using PWs below 60μs. Reich et al explored the effect of PWs between 10 and 120μs on the therapeutic window in four patients and reported an increase in TW of 182% at 30μs relative to 60μs. They also noted that despite using a higher stimulation current at lower PWs, the energy used in terms of charge per pulse was actually lower than at 60μs. However, efficacy thresholds could not be determined in many patients at PWs below 30μs due to the need for very high stimulation current [14].
Here, we report our findings on the effect of short PW on the therapeutic window from a larger series of patients in the early post-operative setting. Our retrospective analysis of monopolar review data aimed to quantitatively define the therapeutic window at a PW of 30μs relative to standard PW (60μs) in patients with STN stimulation for PD. In addition to this, we compare speed of gait, intelligibility of speech, and total electrical energy delivered (TEED) between standard (PW60) and short (PW30) pulse width settings at the thresholds for maximal rigidity control.
PATIENTS AND METHODS
Fifteen consecutive PD patients (7 female, mean age 55±8 years, mean disease duration 14±4 years) who underwent DBS surgery with bilateral implantation of electrodes in the STN using Boston Scientific Vercise and Vercise PC systems in the 12 month period through to October 2017 were included. The mean preoperative UPDRS-III scores were 42±6 OFF and 15±6 ON medication. Patients underwent surgery under general anaesthesia without microrecordings using the Leksell frame and an MRI-guided and MRI-verified technique. Using this technique, we have previously reported a mean perpendicular error between planned target coordinates and electrode trajectory of 0.9±0.5 mm [16–19]. All patients in the series had verification of electrode placement following implantation and these were confirmed to be within 1.5 mm of the intended dorsolateral STN target. Figure 1 is a representative illustration of electrode placement in these patients.
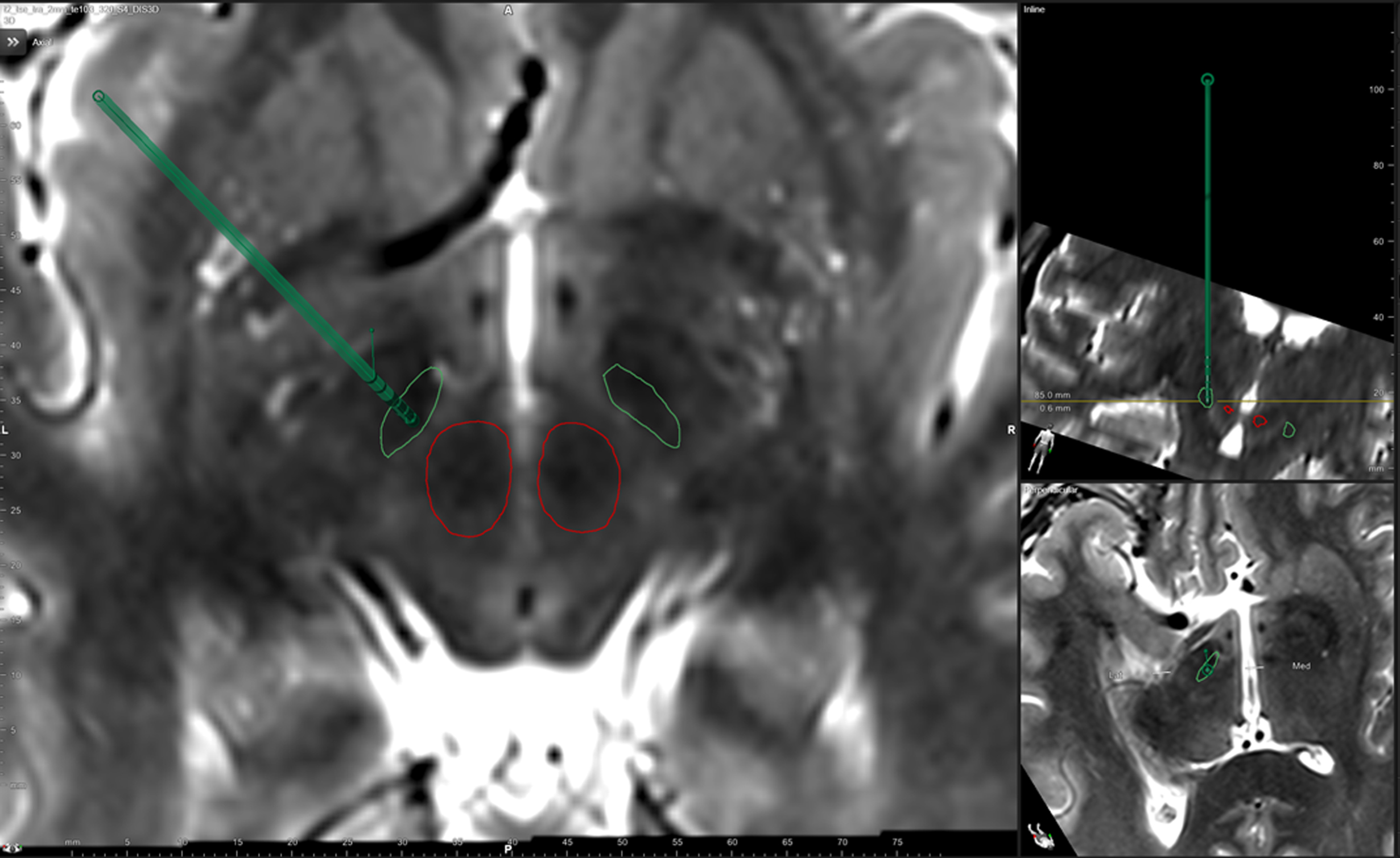
Representative illustration of electrode placement in the subthalamic nucleus target.
An extended monopolar review was performed 2–10 days post-operatively (mean 4.3±2.5) as part of standard clinical assessment to screen for therapeutic and adverse effects of stimulation at each contact prior to initiating DBS therapy.
All patients were assessed in the OFF medication state, having withheld all dopaminergic medication for at least 12 hours prior to screening. Each of the four contacts on each lead was screened in ring mode to determine the efficacy threshold and the side effect threshold to the nearest 0.1 mA, according to our usual standard of care. We defined the efficacy threshold as the lowest current required to produce complete or near complete loss of contralateral rigidity. Rigidity was assessed at the wrist, initially with 0.5 mA increments of stimulation current at 60 second intervals until maximal loss of rigidity was achieved. The exact threshold was then determined with assessments at 0.1 mA increments.
The same procedure was followed to determine the side effect threshold, which was defined as the first clinically evident side effect reported by the patient or observed by the assessing clinician that either persisted for longer than two minutes or was not tolerated by the patient for this length of time.
Assessments were done at standard pulse width (60μs) and repeated at short pulse width (30μs), or vice versa with a one hour interval between sessions. The order of the PW condition tested was alternated with each subsequent patient for the screening, gait and speech assessments, so that 8 patients were tested on PW60 then PW30 and 7 in the reverse order. Patients did not have knowledge of PW settings at the time of assessments. The frequency setting was kept constant at 130 Hz for all patients, and the implantable pulse generator (IPG) was always programmed as anode. Stimulation was programmed in ring mode only for each of the four contact groups per STN lead. Supplementary Table 1 shows the most efficacious contact for each patient. The therapeutic window was calculated as the difference (in mA) between the side effect and efficacy thresholds for each of these contacts at standard and short PW. Speed of gait was assessed with a timed 10 metre walk at the efficacy threshold for each PW condition.
Speech assessment consisted of a 60-second monologue, a reading task, and the Assessment of Intelligibility for Dysarthric Speech computerised version (Speech Intelligibility Test; SIT) [20], assessed OFF-medication at the stimulation efficacy threshold for each PW condition. The SIT requires patients to read a list of sentences. A rater (TG) transcribed the sentences for each condition without knowledge of PW settings. The SIT score provided is the number of words transcribed correctly (SIT% ). Both the 60-second monologue (MON/42) and sentences read (SIT/42) were rated using the perceptual rating scale developed by Darley, Aronson and Brown (1969) [21]. The speech samples were rated on a number of speech parameters (out of 7), namely Articulation, Respiration, Resonance, Phonation, Prosody and Rate, giving a composite score out of 42.
TEED was calculated using the formula [(Voltage2×Frequency×Pulse width)/Impedance] after conversion of the stimulation current to voltage using individual contact impedances [22].
Statistics
R software version 3.4.1 was used for statistical analysis [23]. Normality of data and homogeneity of variances between the two conditions were verified using the Shapiro-Wilk and Bartlett tests respectively, with a significance level set to 0.05. Where data was not normally distributed, the non-parametric Wilcoxon signed rank test was used.
We performed two sets of analyses for the therapeutic window: First, we compared the most efficacious contact (i.e. the one with the lowest efficacy threshold at 60μs) in each STN electrode with repeated measures on the two PW conditions using a paired sample t-test. A further comparison was then made with paired samples for the entire set of contacts (four per STN lead) on the two PW conditions, to determine if any change in TW was consistent across alternative contacts to those used in the clinical setting.
RESULTS
The median post-operative UPDRS rigidity score OFF stimulation (UPDRS item 3.3) was 2 [range 0–3], with two patients not having any clinically detectable rigidity at the contralateral wrist (presumably resulting from the persisting stun effect of electrode implantation). Complete rigidity control with stimulation (score 0) could be achieved in all patients using the most efficacious contact. Persistent side effects at these contacts for PW60 and PW30 respectively included slurred speech (11; 9), facial (8; 9) or limb (5; 5) muscle contraction, sensory symptoms (4; 11), gaze deviation or diplopia (3; 2), autonomic symptoms (1; 3) and other (2; 2) which included head discomfort at PW60 and vertigo at PW30.
The efficacy threshold was significantly greater at PW30 (3.2±1.1 mA) than at PW60 (2.2±0.6 mA) as was the side effect threshold at PW30 (6.7±2.0 mA) compared to PW60 (3.8±1.1 mA). These are illustrated in Fig. 2.
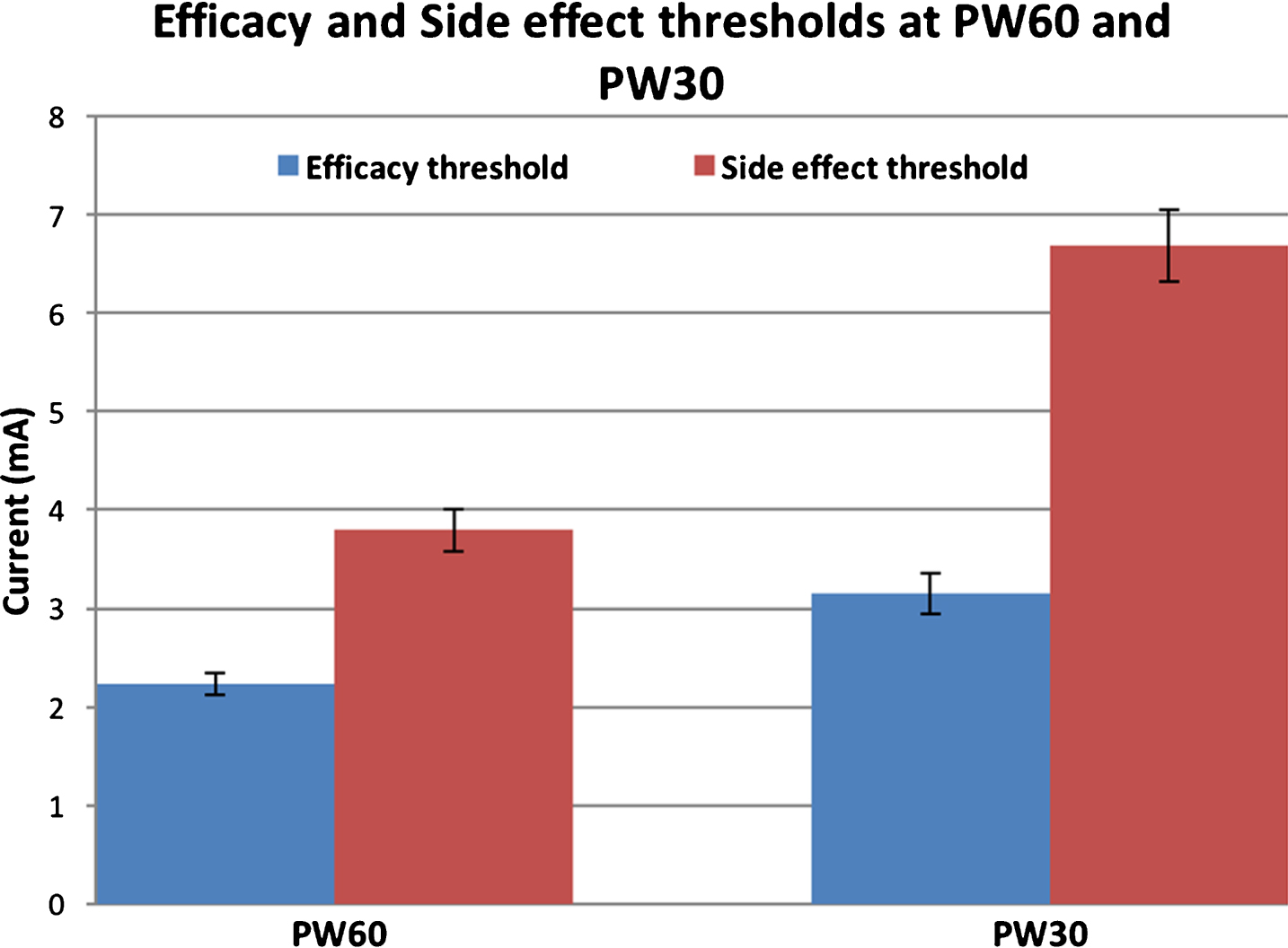
Mean efficacy and side effect thresholds at PW60 and PW30 for the most efficacious contact per STN lead [n = 28; error bars represent standard error of mean].
Therapeutic windows could be calculated in 28 out of 30 STNs, (due to lack of detectable contralateral wrist rigidity in two). The TW for PW60 was 1.7±1.1 mA [median 1.8; range 5.0 mA] and for PW30 was 3.8±1.6 mA [median 3.5; range 6.3 mA], as shown in Fig. 3. The mean increase in TW at PW30 was 197% compared to PW60 [t (27) = 10.6, p≤0.001, r = 0.89].
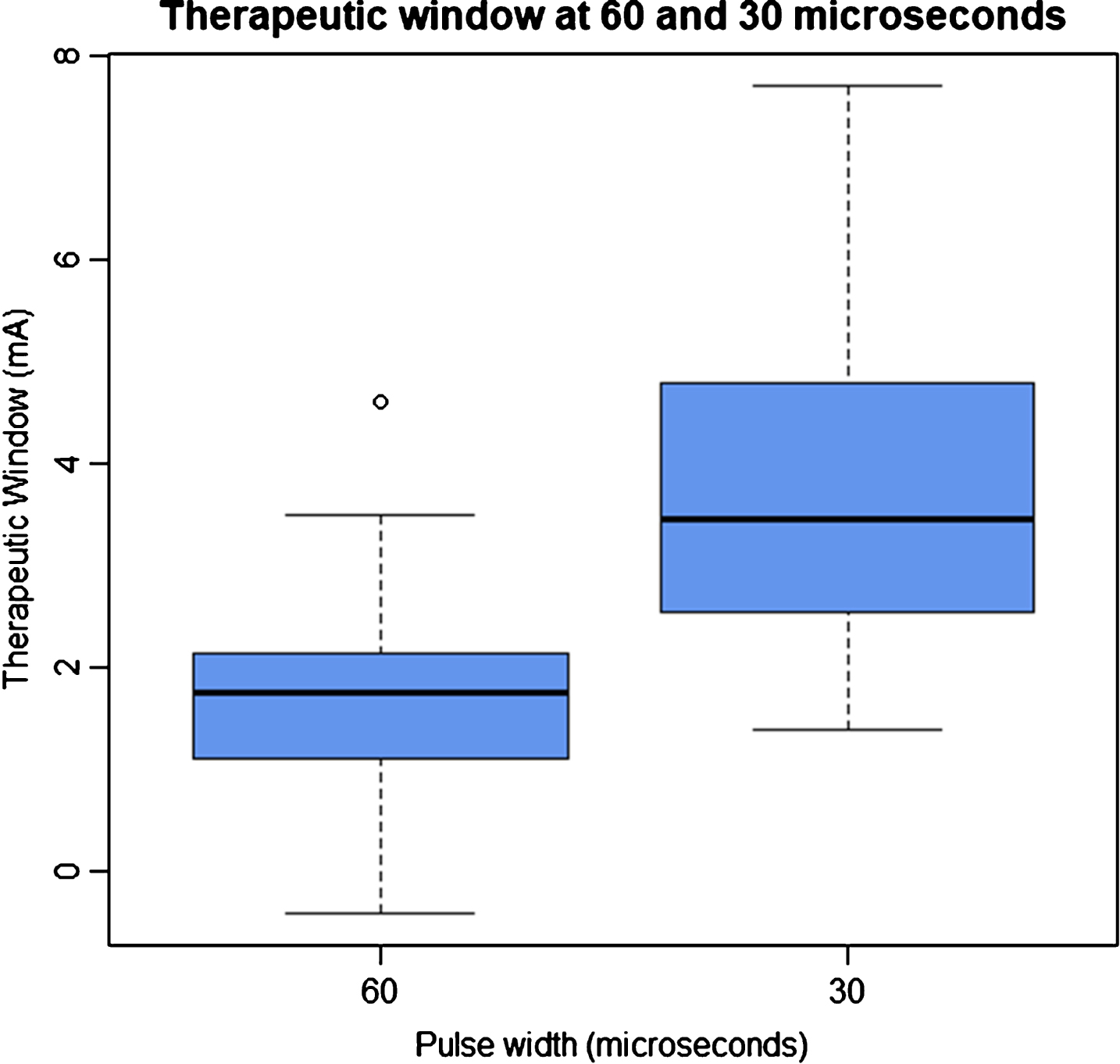
The therapeutic window (difference between side effect and efficacy thresholds) at pulse widths of 60 and 30 microseconds for the most efficacious contact per STN lead [n = 28].
The total electrical energy delivered (TEED) was not significantly different between PW60 (55±41 J/s) and PW30 (54±40 μJ/s) at the threshold for efficacy [t (27) = 0.31, p = 0.76], as shown in Fig. 4.
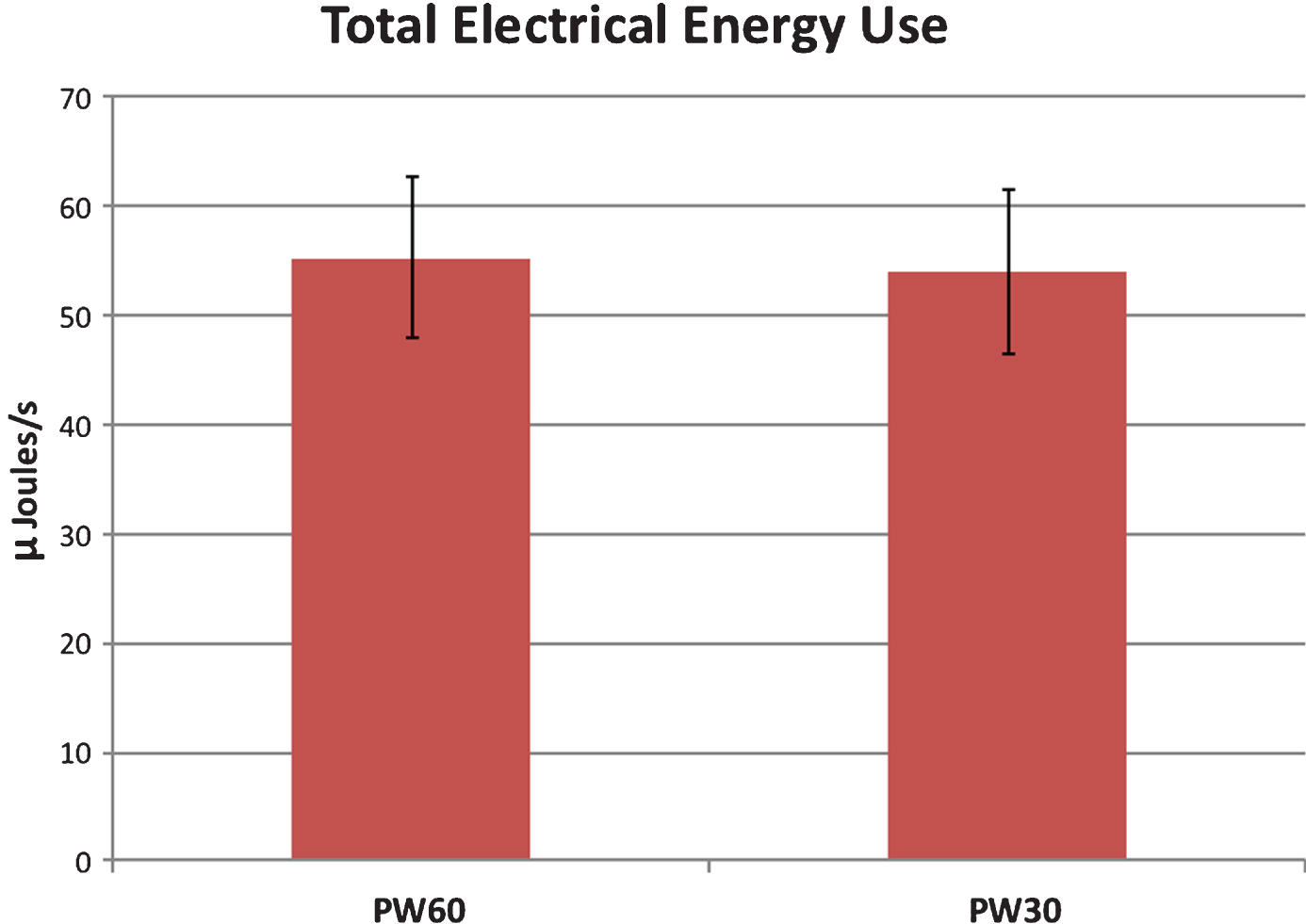
Mean total electrical energy use in micro-Joules per second for PW60 and PW30 settings at the efficacy thresholds, for the most efficacious contact per STN lead [n = 28; error bars represent standard error of mean].
In the secondary analysis comparing data from all contacts of each electrode, 110 TWs could be calculated and these remained significantly higher at PW30 (3.5±1.7 mA) than at PW60 (1.4±1.0 mA); t (109) = 16.3, p≤0.001, r = 0.84.
The timed 10-metre walk using settings at the defined efficacy threshold at PW30 (8.7±2.3 s) was faster than at PW60 (9.2±2.7 s); p < 0.001.
The perceptual speech score for the reading task (SIT/42) was improved at PW30 (36±5) compared to PW60 (33±5); p < 0.01, as was the monologue task (MON/42; 35±5 for PW30 vs. 32±5 at PW60; p = 0.02). The SIT% scores were not significantly different for PW30 (96.5±4.1 % ) and PW60 (94.7±8.7 % ); p > 0.05.
DISCUSSION
Our results show that the therapeutic window (TW) of stimulation using a short pulse width setting of 30μs is significantly greater than that at standard PW settings. The magnitude of this increase is greater than two-fold and is consistent across all contacts of a given electrode. The change in TW was characterised by a relatively greater increase in the side effect threshold than the efficacy threshold at short PW settings compared to those at standard PW. Our findings are consistent with those reported in a smaller series previously [14] and is the largest cohort to date confirming the relative increase in the TW at PWs shorter that 60μs.
Furthermore, we found that energy consumption using short PW settings at the threshold for rigidity control was not significantly different to that at standard settings. This finding is somewhat unexpected with the assumption of requiring a proportional increase in stimulation amplitude, as a change in the voltage has a relatively greater impact on energy used per unit time than PW. However, our data demonstrates that while PW is halved with 30μs settings, the stimulation amplitude that defined the efficacy threshold was only 41% higher than at standard PW, resulting in no significant net difference in energy consumption.
The timed 10-metre walk showed a small but significant improvement at short PW settings. However, given the magnitude of the difference in times between the two PW conditions, more detailed analysis of gait with sensitive objective outcome measures as well as long term subjective patient feedback need to be examined to substantiate any clinically significant benefit. Results of perceptual speech intelligibility showed both the monologue (MON/42) and reading tasks (SIT/42) were significantly better at PW30. In particular, articulation and rate subscores were noted to be sensitive to change in PW settings. However, there was no significant difference in speech intelligibility as measured by the SIT% score; this may be due our patients exhibiting only mild speech deficits at baseline and also due to assessments being performed early in the post-operative period, as speech disturbances following chronic STN DBS may appear several months following surgery. While the cohort of patients examined was not selected on the basis of speech impairment, this finding merits further scrutiny in patients with more profound stimulation induced dysarthria, who may potentially benefit more from short PW settings.
While therapeutic and adverse effects produced by stimulation delivered through a contact are inextricably linked to the precise location of the contact in the target area, global patterns emerging from systematic study of changes in parameter settings are useful despite the confounding factors of natural anatomical variation, variations in surgical technique and precision of targeting. Short PW settings may therefore have beneficial clinical applications in PD patients who have STN DBS. While it may not be relevant to those on standard settings who have an adequate TW, many patients require an increasing level of stimulation over time to control progressive PD symptoms, which may exceed the side effect threshold. Indeed, some patients are found to have a very narrow or even negative TW early in the course of DBS therapy. In these cases, short PW settings may provide an alternative that results in adequate control of motor symptoms while avoiding side effects such as speech and gait dysfunction.
The exact mechanism by which short PW stimulation results in different thresholds for efficacy and adverse effects in STN DBS remains to be fully elucidated, but work done in patients with essential tremor suggests reducing pulse duration may more selectively affect particular fibre pathways than others in close vicinity due to differences in chronaxies (measure of excitability of neural elements) [15]. Reich et al used model derived strength-duration curves for STN stimulation in PD patients to demonstrate a widening difference in the action potential initiation thresholds between small diameter fibres close to the electrode and larger diameter fibres further away. While they focussed on pyramidal tract activation as the adverse effect and postulated that corticosubthalamic fibres (the hyperdirect pathway) may be instrumental in rigidity control, the precise nature of responsible neural elements in the STN and how they are modulated with DBS in humans is unclear at this stage [14].
Limitations of our study include non-blinded clinical assessment of rigidity and side effects, assessment early in the post-operative course, and the lack of comprehensive motor assessment and long term follow up on the two conditions. While assessments on the two PW conditions were performed in alternating order for each subsequent patient and patients were not aware of PW settings during screening, speech and gait assessments, this was not a randomised trial and there is a possibility of inadvertent bias being introduced due to its inherent design and non-blinding of the assessor to PW settings. In particular, the reported improvement in speech and speed of gait based on our data should be interpreted with caution until these findings are further qualified with randomised blinded trials. It is also unclear at this stage if the increase in TW using PW30 in the acute setting will translate into a longer term clinical benefit.
We opted to use assessment of rigidity to define the efficacy threshold rather than bradykinesia or tremor as we have found it the most reliable and objective of these signs during DBS screening assessments. We also only compared PW at 30μs, and intermediate PW settings of 40μs and 50μs need to be explored as well, as they may have a role in cases where 30μs settings may not be well-tolerated or as efficacious for any reason. Given the possible underlying mechanistic notion of various PWs resulting in selective stimulation of different neural elements, it may indeed be that the optimal PW lies somewhere in this range, perhaps depending on the clinical variable being optimised.
While a significant increase in the TW of DBS therapy is a welcome finding, randomised double blinded trials with longer term follow up in patients who suffer from stimulation-induced side effects are now needed to confirm the sustained efficacy, tolerability, and thereby clinical application and incorporation into DBS programming paradigms, of short pulse width neurostimulation in PD patients.
CONFLICT OF INTEREST
No financial or material support was received for this work.
PL has received honoraria from Boston Scientific and Medtronic. MH and LZ have received honoraria and travel expenses from Boston Scientific and Medtronic for speaking at meetings. TF has received honoraria from Profile Pharma, BIAL, AbbVie, Genus, Medtronic, and St Jude Medical. All other authors declare no conflicts of interest.
