Abstract
Background:
Lewy body disorders, including Parkinson’s disease (PD), Parkinson’s disease dementia (PDD) and dementia with Lewy bodies (DLB), are characterized by profound central and peripheral monoaminergic dysfunction.
Objective:
To investigate whether these alterations depend on dementia status, we measured cerebrospinal fluid (CSF) and serum monoamine and metabolite levels across subgroups of the cognitive spectrum, and evaluated their marker potential afterwards.
Methods:
In total, 153 subjects were included, of which 43 healthy controls (HC), 28 PD patients with normal cognition (PD-NC), 26 patients with PD and mild cognitive impairment (PD-MCI), 18 PDD patients, and 38 DLB patients. The levels of monoamines and metabolites in paired CSF and serum samples were analyzed applying reversed-phase high-performance liquid chromatography with electrochemical detection.
Results:
Firstly, when comparing subgroups, CSF 3-methoxy-4-hydroxyphenylglycol (MHPG) levels were found lowest in HC and PD-NC groups and significantly higher in PDD/DLB patients. In addition, CSF 5-hydroxyindoleacetic acid (5-HIAA) levels differed significantly between HC and PD-MCI/PDD, and DLB patients (P≤0.001), but not between HC and PD-NC patients. Secondly, when performing logistic regression, it was shown that particularly CSF/serum MHPG levels and the serum MHPG to noradrenaline (NA) ratio effectively differentiated between HC and (non-)pooled PD subgroups (AUC = 0.914–0.956), and PDD and DLB patients (AUC = 0.822), respectively. Furthermore, CSF 5-HIAA was the most discriminative parameter to differentiate between PD-NC and PD-MCI (AUC = 0.808), and, PD-NC and PDD subgroups (AUC = 0.916).
Conclusions:
Our data revealed that especially alterations of the noradrenergic neurotransmitter system could distinguish between Lewy body disorder subtypes, pinpointing CSF/serum MHPG and NA as potential stage markers across the cognitive spectrum.
Keywords
INTRODUCTION
Dementia with Lewy bodies (DLB) is the second most common neurodegenerative disorder after Alzheimer’s disease (AD). Together with Parkinson’s disease (PD) and Parkinson’s disease dementia (PDD), DLB is considered to be part of the spectrum characterized by Lewy body pathology. Lewy body disorders share great overlap in clinical presentation, in both motor and non-motor symptoms, including neuropsychiatric disorders and cognitive impairment [1]. Although cognitive impairment is well known to be an important clinical hallmark of PDD and DLB, it can also already be seen in the early stages of PD. Research has shown that 24–36% of newly diagnosed PD patients show signs of mild cognitive impairment (PD-MCI) [2]. Presence of PD-MCI increases the risk of developing PDD [3]. In clinical practice, differentiating between PDD and DLB is based on clinical observation of the time of onset of motor or cognitive symptoms [4].
Studies investigating the underlying pathology of Lewy body disorders have been performed previously, but the exact mechanisms are still poorly understood. Generally, α-synuclein deposition in the substantia nigra is considered to be one of the core characteristics. However, Braak et al. [5] also showed the early involvement of the caudal raphe nuclei (RN) and locus coeruleus (LC)-subcoeruleus complex in the progression of PD pathology, the principle sites of serotonin (5-hydroxytryptamine; 5-HT) and noradrenaline (NA) synthesis, respectively. Monoaminergic dysfunction is, therefore, often found in PD [6–11], but its exact role in motor and non-motor disturbances remains unknown. More specifically, regarding the evolution in PD-related neuropathology, both brain stem nuclei are affected by Lewy bodies and neurites (stage 2 of PD-pathology) before the pars compacta of the substantia nigra is (stage 3 of PD-pathology). Generally, the neuropathological staging of Braak et al. [5] comprises a total of six stages, with lesions initially occurring in the dorsal motor nucleus of the glossopharyngeal and vagal nerves and anterior olfactory nucleus. The disease process in the brain stem pursues an ascending course with little interindividual variation. Eventually, it reaches the neocortex from stage 5 onwards.
Cerebrospinal fluid (CSF) and serum analyses can be of importance to get a better understanding of the pathological process of neurodegeneration and facilitate the identification of possible biomarkers to differentiate among dementia syndromes. In this regard, Herbert and colleagues [12] previously evidenced that the addition of CSF levels of the NA metabolite 3-methoxy-4-hydroxyphenylglycol (MHPG) to the traditional set of CSF AD biomarkers (Aβ1-42, T-tau, P-tau181P) improved the differentiation of DLB from AD, but not FTD. We also recently showed that MHPG levels were decreased across the brain in DLB compared to AD patients [13]. New recent findings from our group confirmed this assumption, since not only CSF MHPG, but also serum MHPG and/or NA levels significantly improved differential dementia diagnosis – such as between AD and DLB – if added to the CSF AD biomarker panel [14]. However, differentiating between PDD and DLB in a similar way is not yet possible. When solely focussing on Lewy body disorders, previous research shows changes in CSF monoamine levels, including decreased levels of the dopaminergic metabolites 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanillic acid (HVA), overall decreased or unchanged levels of NA and MHPG, and, contradicting results considering the 5-HT metabolite 5-hydroxyindoleacetic acid (5-HIAA) [15, 16]. Although these findings are of interest, they do not take different cognitive subgroups of Lewy body disorders into account, often resulting in a highly heterogeneous sample.
We, therefore, aimed to further investigate the discriminative potential of CSF and serum levels of monoaminergic neurotransmitters and metabolites (Fig. 1) across the cognitive spectrum of Lewy body disorders, to evaluate underlying pathology and identify possible subtype-specific biomarkers.
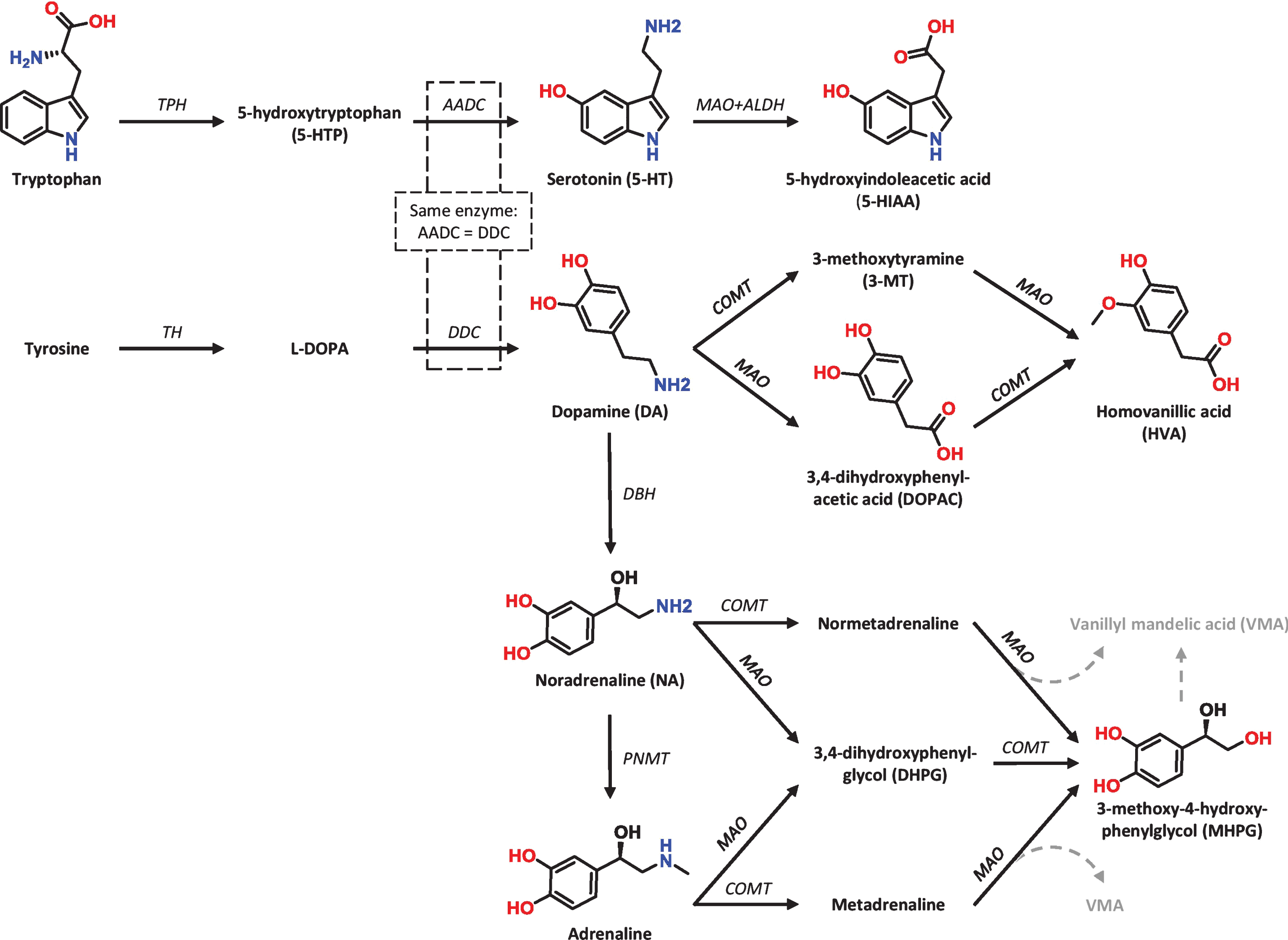
Schematic biosynthesis routes of monoamine neurotransmitters and their main metabolites. 5-HT is derived from the amino acid tryptophan, whereas DA (and thus NA and adrenaline) are derived from the amino acid tyrosine. The molecular structures are provided for the compounds that are quantified in this study by means of reversed-phase high-performance liquid chromatography (RP-HPLC); AADC, aromatic amino acid decarboxylase; ALDH, aldehyde dehydrogenase; COMT, catechol-o-methyltransferase; DBH, dopamine β-hydroxylase; DDC, DOPA decarboxylase; l-DOPA, l-3,4-dihydroxyphenylalanine (levodopa); MAO, monoamine oxidase; PNMT, phenylethanolamine n-methyltransferase; TH, tyrosine hydroxylase; TPH, tryptophan hydroxylase; Figure and Figure legend reprinted from Dekker et al. [37].
MATERIALS AND METHODS
Study population and inclusion protocols
The study included paired serum and CSF samples of 153 retrospectively included subjects, including 43 healthy controls (HC), 72 patients with a clinical diagnosis of idiopathic PD, and 38 patients with DLB. Samples were selected from the Biobank of the Institute Born-Bunge (University of Antwerp). All participants were recruited at the Memory Clinic of the Hospital Network Antwerp (ZNA)-Middelheim and -Hoge Beuken between 1991–2014. Of the PD patients, 28 were considered cognitively normal (PD-NC), 26 had mild cognitive impairment (PD-MCI) and 18 were diagnosed with PDD.
Subjects were included and diagnosed with PD according to protocol and criteria as described previously [17]. Of the patients with probable DLB, initially recruited for inclusion in a longitudinal, prospective study on neuropsychiatric symptoms in dementia [13], 9 had additional post-mortem confirmation of their diagnoses (i.e., definite DLB) following consented brain autopsy and neuropathological examination. Hospital records that matched date of sampling were available for all DLB and 52 PD patients, improving the certainty of the diagnosis. HC were sampled between 2001–2005 and had no neurological nor psychiatric antecedents or an organic disease involving the central nervous system. All HC were hospitalized during the time of lumbar puncture and blood collection, and mainly consisted of (i) subjects complaining of low back pain requiring a selective lumbar radiculography; (ii) patients with disorders of the peripheral nervous system (peripheral facial nerve palsy); and (iii) patients with subjective complaints in whom disorders of the central and peripheral nervous system were ruled out by means of an extensive clinical work-up [18]. Our study was conducted in compliance with the Helsinki Declaration and Ethics Approval for human sample collection of CSF and serum was granted by the Medical Ethical Committee of the Middelheim General Hospital (Antwerp, Belgium; approval numbers 2805 and 2806).
Hospital records at the time of sampling comprised information regarding disease duration, neuropsychological performance and (psychotropic) medication use, all of which were reviewed retrospectively. The disease duration is referring to the moment the (probable) diagnosis was made by the medical specialist and the time between sampling. PD patients were classified as PD-NC (n = 28), PD-MCI (n = 26) or PDD (n = 4) based on the conclusion reports of extensive neuropsychological assessments from no more than three months before or after date of sampling. Due to variability in neuropsychological assessments and limited availability of individual test scores, only the Mini-Mental State Examination (MMSE) was included in the analysis. All DLB patients—and 14 out of 18 PDD patients—were clinically well-characterized, and underwent baseline/follow-up clinical, neuropsychological, behavioral and brain imaging assessments as part of their diagnostic work-up of dementia [13, 18].
CSF and serum sampling
A standard procedure was followed for CSF/serum sampling. CSF sampling was performed as described by Vermeiren et al. [19]. In short, lumbar puncture was performed at the L3/L4 or L4/L5 interspace between 08.00 and 10.00 a.m., after overnight fasting and having abstained from smoking for at least 12 hours. Morning medication was administered after lumbar puncture. A total of 16.5 mL was collected in 5 fractions (polypropylene vials (Nalgene; VWR, Leuven, Belgium): fraction C1 (4.5 mL), C2 (1.5 mL), C3 (1.5 mL), C4 (4.5 mL) and C5 (4.5 mL). Fractions C1-C4 were immediately frozen in liquid nitrogen, while the C5 fraction was centrifuged for 10 minutes at 3,000 rpm (Centrifuge 5702, rotor A-4-38; Eppendorf, Hamburg, Germany). For the current neurochemical analysis, the selection of the fraction was based on availability, absence of hemolysis (checked visually, to avoid potential oxidation effects), and on not having been thawed previously. This resulted in the use of either the C1 or C4 fraction.
For time-linked (paired) serum sampling, approximately 15 mL of total blood was collected following venous puncture in two serum gel tubes with clotting activator of the S-Monovette® 7.5 mL Z-gel subtype (Sarstedt, Nümbrecht, Germany) and centrifuged for 10 minutes at 3,000 rpm. Serum was then distributed to marked polypropylene vials and frozen in liquid nitrogen.
All samples were stored at –80°C in the Biobank facilities of the Institute Born-Bunge (Antwerp, Belgium) until analysis.
Neurochemical analysis
Concentrations of dopamine (DA), 5-HT, (N)A and their respective metabolites, i.e., DOPAC and HVA, 5-HIAA, and MHPG, were determined using a reversed-phase high-performance liquid chromatography (RP-HPLC) system with electrochemical detection (ECD) (ALEXYS™ Monoamine Analyzer, Antec Leyden, Zoeterwoude, Netherlands) [20].
Sample preparation prior to RP-HPLC-ECD was performed as described by Dekker et al. [21]. In short, precolumn separation was conducted using Amicon® Ultra 0.5 Centrifugal Filters (cutoff 3,000 Da; Millipore, Ireland), which were washed twice with 450 μL sample buffer beforehand by means of centrifugation (14,000×g, 25 min, 4°C). Next, serum and CSF samples were loaded onto the wetted filters and then centrifuged (14,000×g, 40 min, 4°C). The obtained filtrate was diluted into different fractions: the serum filtrate was diluted 4- and 15 times prior to RP-HPLC-ECD injection whereas CSF was diluted 2- and 7 times. Of all fractions, 5 μl was automatically injected onto an ALF-125 column (C18; 250 mm×1.0 mm, 3 μm particle size).
Statistical analysis
Nonparametric analyses were performed due to non-normal distribution of data. A Kruskal-Wallis test with post hoc analyses using the Mann-Whitney U tests and Bonferroni correction for multiple comparisons was used to compare continuous variables between groups (adjusted P < 0.005). The Chi-square test was used to test the association between dichotomous variables. In addition, we observed that a log-transformation of the of CSF/serum parameters (monoamines) led to an approximately normal distribution of these variables, enabling the use of parametric statistical analyses. These analyses allowed to select the most discriminative CSF and/or serum monoaminergic markers to predict disease status accounting for potential confounding effects of demographic and clinical variables. Subsequently, a multiple linear regression model (ANCOVA) was performed, including the log-transformed CSF/serum parameters as dependent variable, and, diagnosis, age and gender as independent variables. Due to multicollinearity effects between disease duration and medication use, the latter two variables could not be included in the model. In addition, a dot plot visualizing r–log10(P-values) of both the Kruskal-Wallis and ANCOVA tests was created, after which the most informative CSF/serum monoaminergic markers were selected to be included as predictor in stepwise forward conditional logistic regression analysis. Age was included in every regression model. The diagnostic performance of the fitted models was evaluated afterwards, by means of receiver operating characteristics (ROC) analyses (area under the curve (AUC) values). Finally, Spearman’s Rank-order correlations tests were calculated to evaluate the relationship between cognitive performance and monoaminergic CSF and serum levels. Missing values were excluded pairwise. All analyses were performed using SPSS 24.0 for Windows (IBM SPSS Software, Armonk, NY, IBM Corp) or R, version 3.4.0. for Windows.
RESULTS
Clinical and demographic characteristics
Demographic and clinical data of all subjects is summarized in Table 1. Significant differences in age at sampling, disease duration, MMSE rates, and, number of patients on dopaminergic medication compared to those free of such medication significantly differed between PD groups (P < 0.001). The same applied to psychotropic medication usage (P = 0.005).
Clinical and demographic characteristics
Data are presented as mean (SD); Only results out of individual group comparisons that remained significant after Bonferroni correction for multiple comparisons (i.e., 10 group comparisons for Mann-Whitney U tests; significant if P < 0.005) are mentioned below (depicted with superscript letters (P values) and in
CSF/serum concentrations of monoamines and metabolites
In CSF, significant MHPG, NA, HVA and 5-HIAA differences were found between groups. Results following Kruskal-Wallis analyses are summarized in Table 2. NA levels were significantly lower in DLB patients compared to all PD groups and HC (for all, P≤0.001). As for MHPG, levels were significantly higher in all the individual patient groups compared to HC (for all, P < 0.001). Furthermore, PD-NC subjects had lower MHPG levels than DLB patients (P = 0.001). MHPG levels seemed to gradually increase with increasing cognitive decline (Fig. 2B). Similarly, MHPG/NA ratios, indicative of the catabolic noradrenergic turnover, were significantly higher in DLB as opposed to all the other individual groups, including HC (for all, P≤0.001). Looking at HVA, DLB patients had significantly lower levels than HC and PD-NC patients. In addition, 5-HIAA concentrations were significantly lower in all groups compared to HC (for all, P≤0.001), except the PD-NC group. Moreover, 5-HIAA levels were also significantly decreased in PD-MCI, PDD and DLB patients compared to PD-NC (resp. P = 0.016, P = 0.006, P = 0.01) although significance was not maintained following Bonferroni correction (Fig. 2A). Finally, HVA/5-HIAA ratios, indicative of the serotonergic modulation of and interaction with the dopaminergic system, only differed significantly between HC and PD-MCI/PDD patients.
Median CSF and serum monoamine and metabolite levels
Median (IQR), Kruskal-Wallis analyses (P < 0.05) with post-hoc Mann-Whitney U tests (significant if P < 0.005 after Bonferroni correction). Latter data that remained significant are depicted in
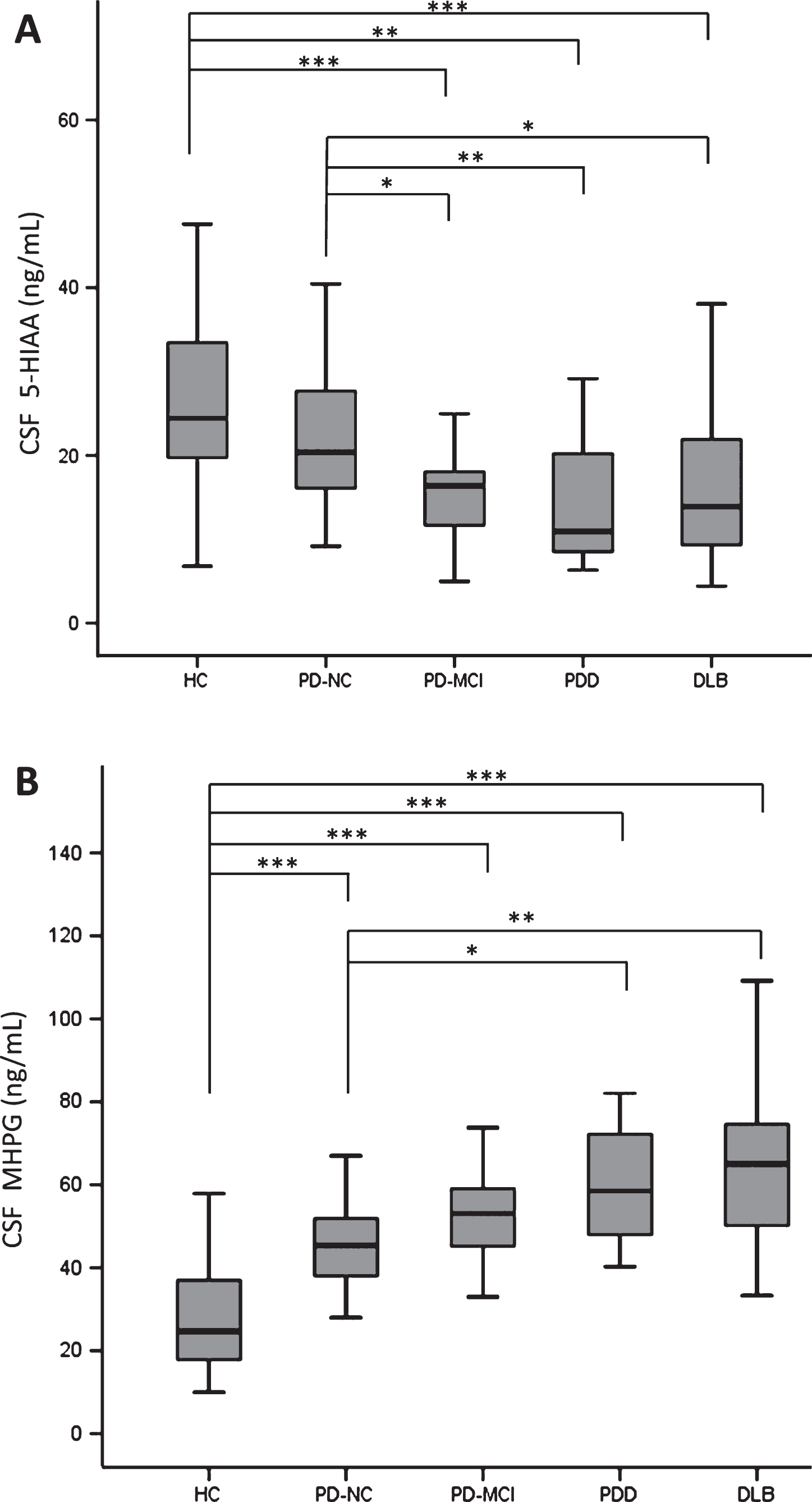
Boxplots showing median values and IQR (min-max ranges: whiskers) of (A) CSF 5-HIAA levels; (B) CSF MHPG levels in HC, PD-NC/MCI, PDD and DLB subjects; *P < 0.05, **P < 0.01, ***P < 0.001; 5-HIAA, 5-hydroxyindoleacetic acid; CSF, cerebrospinal fluid; DLB, dementia with Lewy bodies; HC, healthy controls; MHPG, 3-methoxy-4-hydroxyphenylglycol; PD, Parkinson’s disease; PD-NC, PD with normal cognition; PD-MCI, PD with mild cognitive impairment; PDD, PD dementia.
Entrapped air (oxygen) in the sampling tubes caused oxidation effects so that 5-HT was only measured in a very low number of CSF samples (Table 2).
The significant findings observed in serum were mostly confined to differences either between HC and PD(-NC/MCI) or DLB subjects, or, PD(-NC/MCI) and DLB subjects. No significant differences were found between PD-NC and PD-MCI subgroups (Table 2).
To allow for evaluation of potential confounding effects of clinical and demographic variables (e.g., age and gender) on monoaminergic data, ANCOVA analysis with post hoc Tukey correction tests was performed using the log-transformed CSF/serum monoamine data (residuals). Results (r–log10(P-values)) for all CSF and serum parameters following both Kruskal-Wallis and ANCOVA analyses were comparatively visualized by means of Fig. 3. The figure shows that most differences (dots) that survived Bonferroni correction (P < 0.0019; dashed line) were similarly significant, apart from CSF/serum MHPG, CSF/serum NA, serum HVA, and, the CSF MHPGA/NA ratio, which became more significant following ANCOVA. Results also indicated that age and gender were negligible as confounding factors. Moreover, post-hoc Tukey tests revealed results to be astonishingly similar to the post-hoc Mann-Whitney U tests for each CSF/serum parameter during individual group comparisons (data not shown).
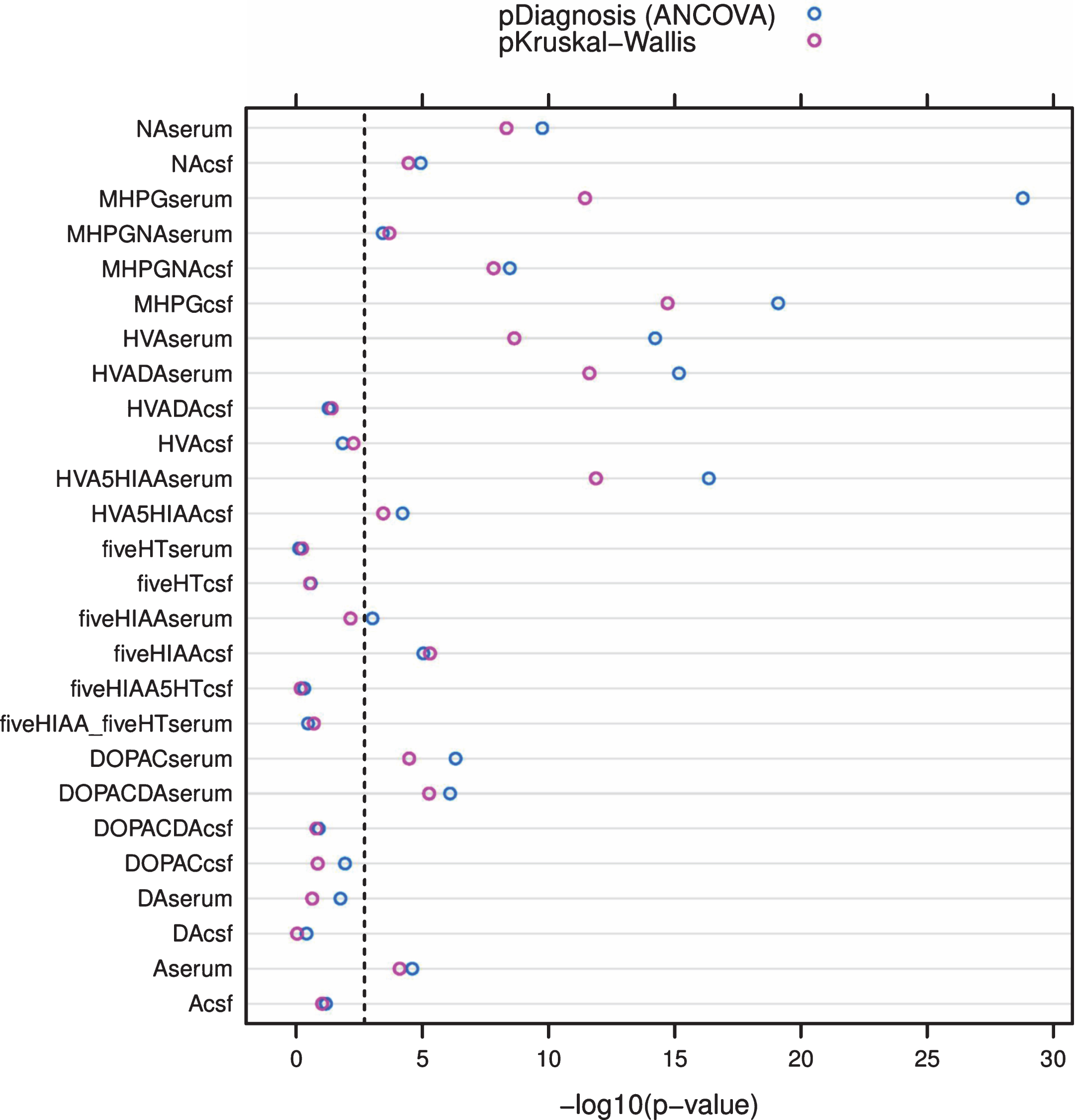
Dot plot visualizing and comparing (i) the obtained r–log10(P-values) of the CSF/serum monoamine data compared among groups following the original Kruskal-Wallis analyses (pinkish dots), as well as (ii) the r–log10(P-values) of diagnosis as independent variable for all log-transformed CSF/serum monoamines under study (dependent variable) following ANCOVA, with the inclusion of age and gender as independent variables (confounders) (blueish dots). The dashed line represents the Bonferroni-corrected threshold (P = 0.0019; 0.05 divided by 26 hypothetical tests); A, adrenaline; fiveHIAA or 5HIAA, 5-hydroxyindoleacetic acid; fiveHT, serotonin; CSF, cerebrospinal fluid; DOPAC, 3,4-dihydroxyphenylacetic acid; DA, dopamine; HVA, homovanillic acid; MHPG, 3-methoxy-4-hydroxyphenylglycol; NA, noradrenaline.
Finally, CSF/serum MHPG, CSF/serum NA, CSF/serum MHPG/NA ratio, CSF 5-HIAA, and, serum HVA were selected as the most informative markers of disease status, thereby ignoring significant markers which were potentially altered due to medication (please see below), i.e., serum DOPAC/DA and HVA/DA ratios (dopaminergic medication).
Diagnostic performance of the most significantly different CSF/serum monoamines and metabolites
A model only containing serum MHPG efficiently distinguished HC (n = 36) from the pooled PD-subgroups (n = 67; excluding DLB) (sensitivity (S): 83.3%; specificity (Sp): 98.5%; AUC: 0.927). Performance improved with the addition of serum NA (AUC: 0.984). In order to increase the sensitivity, extra addition of CSF MHPG/NA made the model nearly perfect (S: 94.4%; Sp: 96.4%; AUC: 0.989). The combination of serum MHPG and NA was sufficient to effectively discriminate between HC (n = 36) and pooled PD-NC+MCI subgroups (n = 50) (S: 88.9%; Sp: 95.1%; AUC: 0.981) (Fig. 4A). For the HC (n = 36) vs. PD-NC (n = 26) comparison, the combination of serum MHPG, HVA and age as independent variables in the model yielded high S/Sp values (S: 94.4%; Sp: 92.3%; AUC: 0.991). A model in which only serum MHPG/NA remained after regression analysis distinguished PDD (n = 17) from DLB (n = 35) patients with fair to good diagnostic power (S: 73.3%; Sp: 84.8%; AUC: 0.822) (Fig. 4B). Finally, the forward conditional method only kept CSF 5-HIAA and age in the model as the most optimal parameters to distinguish between PD-NC (n = 20) and PD-MCI (n = 26) (S: 65.0%; Sp: 88.5%; AUC: 0.808), and PD-NC and PDD subjects (S: 80.0%; Sp 81.3%; AUC: 0.916).
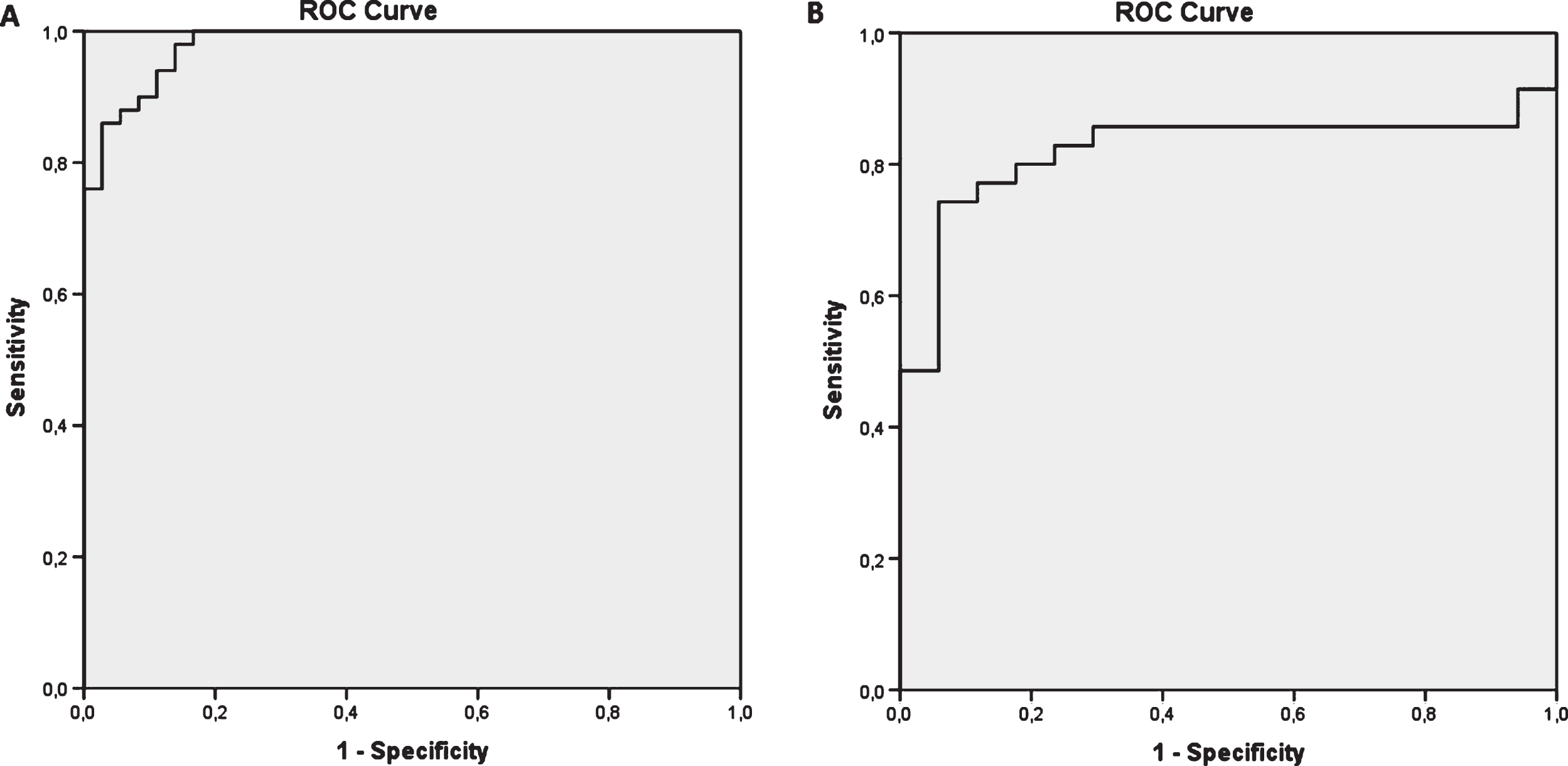
Diagnostic performance of newly composed models only comprising the most significantly different monoamines and metabolites (A) HC vs. pooled PD-NC/MCI subgroups (serum MHPG + NA; AUC = 0.981); (B) PDD vs. DLB patients (only serum MHPG/NA ratio; AUC = 0.822).
MMSE scores showed a small but significant positive correlation with CSF 5-HIAA concentrations in the pooled PD group, which comprised PD-NC, PD-MCI and PDD patients (rho = 0.418; P = 0.01; n = 39) (Fig. 5).
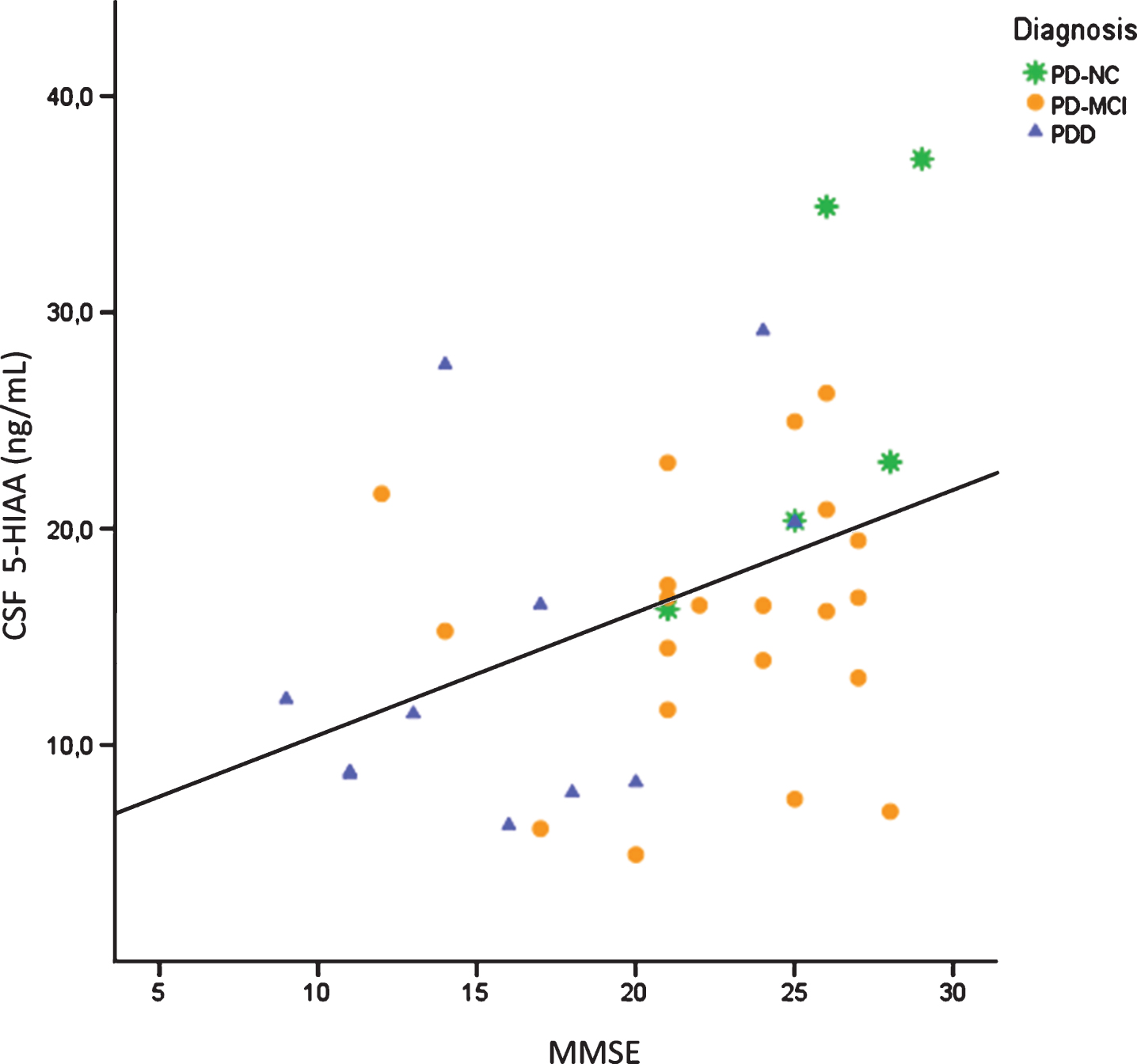
Significant Spearman correlation between MMSE scores and CSF 5-HIAA concentrations in the pooled PD group (r = 0.418, P = 0.01, n = 39); 5-HIAA, 5-hydroxyindoleacetic acid; CSF, cerebrospinal fluid; MMSE, Mini-Mental State Examination; PD, Parkinson’s disease; PD-NC, PD with normal cognition; PD-MCI, PD with mild cognitive impairment; PDD, PD dementia.
Confounding CSF/serum monoamine alterations related to medication
Serum DA levels were higher in PD-NC and DLB patients who were on dopaminergic medication compared to patients free of such medication (P = 0.041 and 0.003). In the DLB group, serum HVA/DA and DOPAC/DA ratios, both indicative of the catabolic dopaminergic turnover, were also higher (P = 0.007 and 0.002) in addition to decreased serum 5-HIAA concentrations (P = 0.034). As for the PD-MCI subgroup on dopaminergic medication, only CSF HVA/5-HIAA ratios were significantly higher (P = 0.023).
In HC, PD-MCI and DLB subjects on antidepressants, particularly 5-HT serum levels were much lower in comparison to their antidepressant-free counterparts (P = 0.001, 0.026 and <0.001, respectively), accompanied with increased serum 5-HIAA/5-HT ratios, indicative of the catabolic serotonergic turnover, in HC and DLB subjects (P = 0.002 and <0.001). DLB patients also had lower CSF 5-HIAA levels (P = 0.007). Furthermore, serum MHPG/NA ratios were much higher, and, CSF MHPG levels much lower, in HC subjects (P = 0.017) and PD-MCI patients (P = 0.012), respectively, who were administered antidepressants.
DISCUSSION
We analyzed and compared monoaminergic neurotransmitter and metabolite levels in paired serum and CSF samples of Lewy body disorders. Results revealed that MHPG differentiated between HC and the (non-)pooled PD groups, and between PDD and DLB patients (serum MHPG/NA ratio). Additionally, CSF 5-HIAA was associated with cognitive impairment.
CSF MHPG levels were higher in the cognitively more impaired subgroups. Therefore, degeneration of LC noradrenergic neurons accompanied by possible compensatory upregulation of monoamine turnover might be thought of [22, 23], particularly in PDD/DLB patients [6]. Comparably to the RN, the LC becomes lesioned in neuropathological stage 2 of PD [24] and is characterized by profound autonomic—meaning sympathetic—and more central noradrenergic neurotransmitter deficits, given its widespread connections with the rest of the cerebrum/cerebellum. Interestingly, the LC is the sole provider of NA via its efferent projections to the hippocampus, amygdala and prefrontal cortex, and has neuroprotective properties [25], so that compensatory changes, such as upregulated receptors or increased catabolic conversion of NA to MHPG to some degree, may occur in case of subtle to moderate LC neuronal loss [26]. This hypothesis might explain why our data shows higher CSF and serum levels of MHPG in PD-NC and PD-MCI patients compared to healthy controls, which has been previously assumed by Mann et al. [8] in 17 drug-free PD patients, showing a negative correlation between CSF MHPG and scores on the digit span, a cognitive task measuring working memory. Szot et al. [27] previously found consisting evidence of a compensatory mechanism in the LC by sprouting dendrites into the peri-LC dendritic zone, as determined by α2-adrenoreceptors, and NA transporter binding sites, as well as sprouting of axonal projections to the hippocampus not only in AD but also DLB subjects.
Following these compensatory mechanisms, one might expect gradually declining CSF MHPG levels in PDD and DLB as opposed to PD-NC/PD-MCI patients, as well as HC, when LC neuronal loss has become more severe. Remarkably, we could not detect such a decrease for CSF nor serum MHPG. On the contrary, CSF and serum NA levels tended to follow this pattern, with increased levels in the PD-NC subgroup, followed by a steep decrease in the DLB (but not PDD) subgroup (Table 2). Noteworthy, it has previously been evidenced that MHPG is distributed across the entire length of the spinal cord, which also may be involved in clearance of this metabolite, possibly making CSF MHPG a less certain indicator of central or LC-related NA metabolism. Conversely, peripheral (i.e., serum) MHPG changes may well reflect the extensive autonomic dysfunction that accompanies the disease, mainly since MHPG crosses both the CSF-blood and blood-brain barrier [28]. Latter event might explain why we were unable to detect lower CSF (or serum) MHPG levels in DLB/PDD, given the mixed concentration effects of central and peripheral MHPG.
Moreover, our results indicated that a denervated serotonergic system, i.e., the RN, might be related to cognitive decline in PD. Together with age, CSF 5-HIAA levels clearly differentiated between PD-NC and PDD and correlated with MMSE scores within the overall PD group. Although preclinical studies suggest a role of 5-HT in cognition [29], limited clinical data is available, especially in PD patients. For instance, in the recent study of Olivola et al. [30], PD patients had significantly reduced CSF 5-HIAA and 5-HT levels compared to either control subjects or AD patients. However, the authors did not find an association with motor or non-motor aspects, and no significant correlation with MMSE scores. On the contrary, in neuropathologically confirmed AD patients, we previously found a similar correlation between MMSE rates and 5-HIAA levels, albeit analyses were performed in postmortem frozen brain tissue. We theorized that 5-HT and also acetylcholine projections converge at several key target structures, such as the hippocampus, and that 5-HT receptors are important pharmacotherapeutic targets to enhance cognitive functioning [31]. In this last study, the dysfunctional serotonergic neurotransmission was also related to dementia severity, as measured by MMSE scores, more specifically in the temporal cortex. This is in agreement with the current study since CSF 5-HIAA levels were the lowest in PDD and DLB patients – preceding those of PD-NC and then PD-MCI patients.
One major study limitation is the use of dopaminergic/psychotropic medication during CSF and serum sampling, even though patients had ceased all medication one day earlier. On the whole, largest effects were seen for serum DA levels, and serum HVA/DA and DOPAC/DA ratios regarding dopaminergic medication, combined with alterations of serum 5-HT levels, serum 5-HIAA/5-HT rations, and CSF HVA/5-HIAA ratios for those who were on antidepressants. These results were, therefore, left out of the interpretation and discussion. The only alterations with regard to CSF 5-HIAA and MHPG levels were observed in DLB (P = 0.007) and PD-MCI patients (P = 0.012), respectively, on antidepressants (lower levels for both). Surprisingly, antidepressants created false negative data to some degree because statistical reanalysis in small subgroups of patients free of antidepressants revealed significantly higher CSF MHPG levels in PD-MCI as opposed to PD-NC subjects indeed (11 vs. 10; P = 0.003), which confirms our assumption of CSF MHPG as a potential marker. Neurochemical CSF analyses of C1 or C4 fractions, depending on availability, may also have introduced less accurate estimates of CSF HVA and CSF 5-HIAA levels since a marked rostrocaudal CSF concentration gradient exists for both compounds, independent of the body position, with an approximate 30% increase in concentration for each successively tapped 4 mL [32]. For CSF MHPG, there is only a slight 8% increase over 20r–25 mL of CSF drawn. The absence of such a gradient for MHPG suggests that a major part of lumbar CSF MHPG originates from the spinal cord. Another limitation of this study involves its retrospective character. Due to the retrospective analysis of clinical data, not all information was fully available. Important information on motor and non-motor symptoms could, therefore, not fully be taken into account. Furthermore, groups could only be partially matched for age and disease duration, but were fully gender-matched. In addition, medication data was lacking for some PD patients, even though the HC were well-documented with availability of all medication. Unfortunately, HC did not undergo similar neuropsychological testing. Finally, no CSF AD biomarker (Aβ1-42, T-tau, P-tau181P) [33] or CSF α-synuclein levels [34] were determined to look at the possible added discriminative value of all neurochemical markers, including monoamines, combined.
To conclude, we found that mainly noradrenergic neurotransmitter dysfunction seems to be a determining pathophysiological feature in Lewy body disorders. More specifically, serum MHPG, NA, MHPG/NA ratio, and CSF MHPG seem promising to differentiate between HC, PD(D) and DLB patients. Furthermore, similar as with metaiodobenzylguanidine (MIBG) scintigraphy showing sympathetic denervation of the heart in early PD [35] and DLB [36] for instance, measuring serum MHPG and NA levels may provide an affordable, swift and non-invasive way to give a first and accurate indication of dementia status during routine PD diagnostic work-up, or to simply exclude PD as potential diagnosis. Larger prospective follow-up studies, however, are essential in order to substantiate our findings, preferentially with inclusion of multiple cognitive data, CSF AD biomarker or CSF α-synuclein analyses, and repeated sampling to monitor progression. Similarly, various methodological issues that may influence monoamine levels, such as sample handling (freeze-thaw cycles, type of recipient), dietary and environmental (temperature, light) effects, require a thorough investigation beforehand. CSF sampling should be standardized as well, always analyzing the same first fraction (0r–2 mL).
COMPETING INTERESTS/CONFLICTS OF INTEREST
The authors declare that there is no conflict of interest.
Footnotes
ACKNOWLEDGMENTS
This research was supported by the Research Foundation Flanders (FWO), Interuniversity Poles of Attraction (IAP Network P7/16) of the Belgian Federal Science Policy Office, Alzheimer Research Foundation Belgium (SAO-FRA; P#16003), Methusalem excellence grant of the Flemish Government, agreement between Institute Born-Bunge and University of Antwerp, the Medical Research Foundation Antwerp, the Thomas Riellaerts research fund, Neurosearch Antwerp, and the Alzheimer Research Center of the University Medical Center Groningen (ARCG-UMCG).
The authors gratefully acknowledge the contribution and support of all patients, control subjects, relatives, caregivers, nursing and administrative personnel, and clinical staff involved.
