Abstract
Background:
A qualified consensus suggests that a combination of levodopa with a peripherally acting dopa decarboxylase inhibitor continues to present the gold standard treatment of Parkinson’s disease (PD). However, as the disease progresses the therapeutic window of levodopa becomes narrowed. Pharmacological strategies for motor fluctuations are focused on providing less pulsatile and more continuous dopaminergic stimulation. Peripheral catechol-O-methyltransferase (COMT) inhibition improves the bioavailability of levodopa and results in a prolonged response.
Objective:
The primary aim of this study was to investigate the efficacy and safety of the two available COMT inhibitors; entacapone and tolcapone and the recently introduced opicapone.
Methods:
Electronic databases were systematically searched for original studies published within the last 37 years. In addition, lists of identified studies, reviews and their references were examined.
Results:
Twelve studies fulfilled the inclusion criteria. 3701 patients with PD were included in this systematic review.
Conclusions:
Adjuvant treatment of PD patients experiencing motor fluctuations with entacapone resulted in improvement of motor function and was well tolerated. Therefore, entacapone presented an acceptable benefit to risk ratio. Tolcapone appeared to result in a greater therapeutic effect. However, this was not consistent across all motor variables and studies, and thus would not support its use, given the current onerous monitoring that is required. Opicapone was not associated with adverse reactions in a phase III trial but did not present a greater efficacy than entacapone, and thus further studies are required in order to illustrate its cost effectiveness.
INTRODUCTION
Parkinson’s disease
Parkinson’s disease (PD) is the second most common neurodegenerative disorder after Alzheimer’s disease [1–5] affecting more than 10 million people worldwide [6–8]. It is recognised by the cardinal features of resting tremor, rigidity, bradykinesia, and impaired postural reflexes with an asymmetric onset [9–14]. The histopathological hallmark of PD is the preferential degenerative loss of dopaminergic neurons in the substantia nigra pars compacta and the presence of intraneuronal proteinaceous inclusions termed Lewy bodies [15–21]. This dopaminergic striatal depletion leads to a disrupted balance between the striatopallidal and pallidothalamic output pathways which is manifested by the characteristic parkinsonian motor features [14, 23]. Aging consists the most important risk factor in the development of PD, however the aetiology remains elusive, being attributed to a complex interplay of genetic susceptibility and environmental exposures [24–27]. Given that the population is aging, the number and proportion of older persons with PD is increasing [28–30] with far reaching economic, social, and health care implications [31, 32] as well as a renewed impetus to improve its management and pharmacotherapy [33].
Levodopa: A gold standard with fluctuating effectiveness
Forty years after its introduction, L-3,4-dihydroxyphenylalanine (levodopa; L-dopa)—a dopamine precursor that can cross the blood-brain barrier—still remains the gold standard in the symptomatic treatment of PD [34–39]. However, as the disease progresses the L-dopa’s therapeutic window becomes narrowed (wearing off) and patients oscillate between ‘on’ and ‘off’ states characterised by dyskinesia and involuntary movements [20, 40–43]. The prevalence of this wearing off phenomenon has been reported in approximately 50% of patients within 5 years of L-dopa treatment [44, 45]. The pathogenesis of these motor complications remains abstruse, hypotheses include; modifications in the presynaptic and postsynaptic signal transduction [46, 47] glutamate NMDA receptor dysregulation [48] and maladaptive striatal and serotonin axon plasticity [49, 50]. In clinical practice, therapeutic strategies for motor fluctuations are focused on providing less pulsatile and more continuous dopaminergic stimulation at postsynaptic dopamine receptors that mimics physiological dopamine levels [51–56].
Indeed, patients with response fluctuations present concentration-effect curves that are steeper and markedly shifted to the right when compared with patients whose symptoms are sufficiently controlled [57, 58]. L-dopa has a complex pharmacokinetic profile as it is evident by its low bioavailability, short plasma half-life, erratic gastric emptying, facilitated diffusion across the blood brain barrier and extensive peripheral O-methylation and decarboxylation [58, 59]. The latter occurs by aromatic amino acid decarboxylase (AAD) and constitutes the controlling step for the formation of dopamine in Parkinsonian patients [60, 61]. For this reason, current Parkinson’s disease treatment practice involves the co-administration of L-dopa with a peripheral dopa-decarboxylase inhibitor (DDCI, such as benserazide or carbidopa), which results in reduced peripheral metabolism and therefore amplified central effectiveness and decreased dopaminergic side-effects [62, 63] shown in Fig. 1.
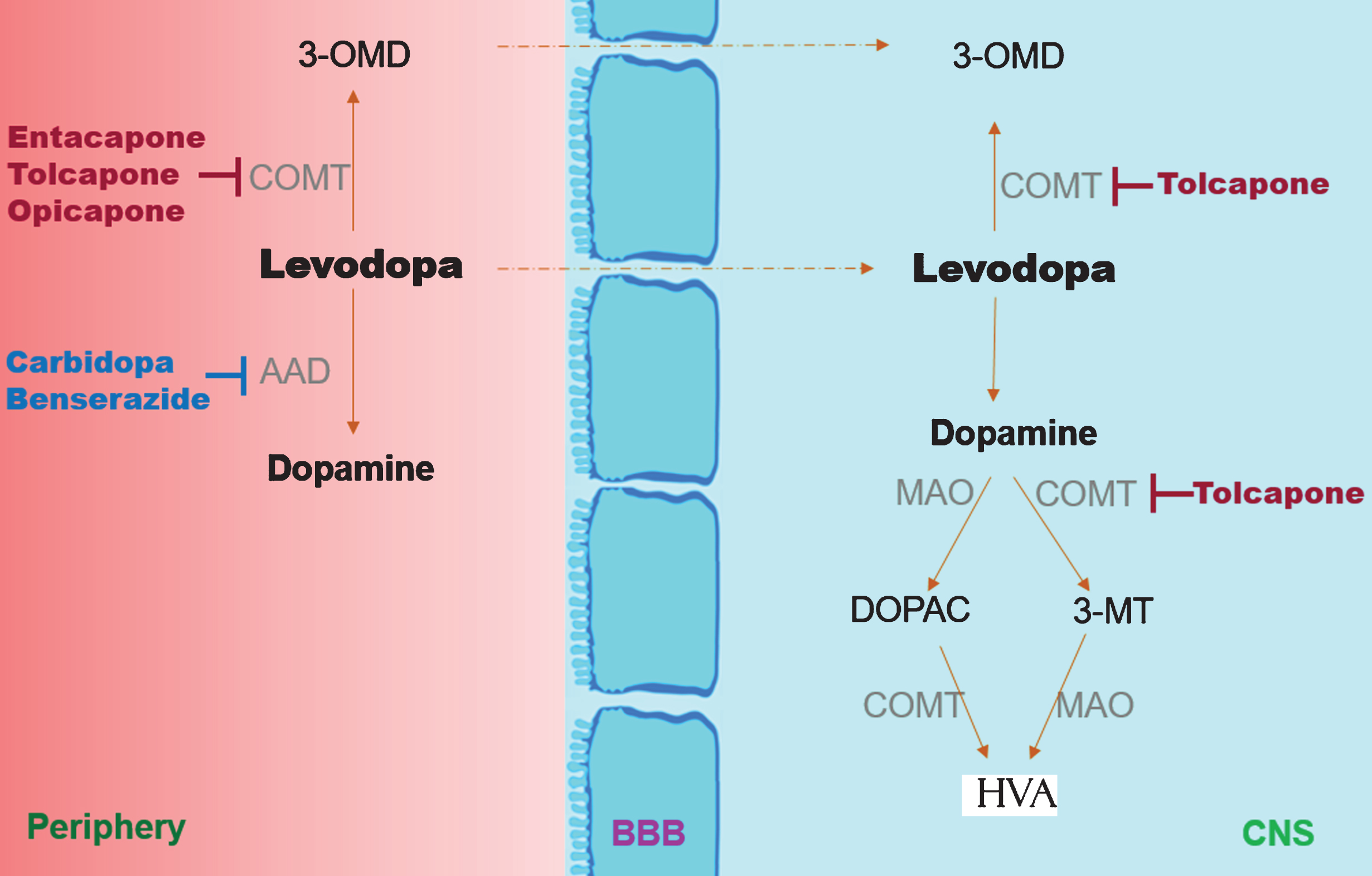
Simplified metabolic pathway of levodopa. The peripheral metabolism is inhibited by the aromatic L-amino acid decarboxylase (AAD) inhibitors (carbidopa, benserazide) and via the catechol-O-methyl transferase (COMT) inhibitors (entacapone, tolcapone, opicapone). PET scan studies and animal studies suggest that tolcapone may be able to also inhibit COMT in the CNS [64, 65].
COMT: A second pathway of levodopa metabolism
Interestingly enough, administration of levodopa combined with a DCCI is thought to shift the peripheral metabolism of L-dopa towards the catechol-O-methyl transferase (COMT) pathway (Fig. 1) [66, 67]. COMT is a ubiquitously expressed catabolic enzyme with soluble and membrane bound isoforms [68] that can methylate (inactivate) catecholamines as well as their hydroxylated derivatives and catechol structured drugs such as L-dopa [69, 70]. Specifically, COMT catalyzes the magnesium dependent transfer of the methyl groups from the coenzyme S-adenosyl-l-methionine to the m-hydroxyl group of catechol compounds [71, 72], such as the conversion of L-dopa to 3-O-methyl dopa (3-OMD) [73–75]. The latter metabolite has no therapeutic value and a long half-life (∼15 h) [76] as opposed to the 60–90 minutes of L-dopa [77]. Its properties include a further reduction of L-dopa’s bioavailability in the brain by competing for active transport across the blood–brain barrier [78, 79] though the clinical significance of this finding has been contended in the relevant literature [80, 81].
COMT inhibition
COMT inhibitors (COMTIs) were examined initially as a therapeutic target based on the premise that they could lead to the following pharmacological benefits: (i) prolonged L-dopa half-life and increased area under the curve, (ii) reduced formation of 3-OMD, diminished L-dopa plasma level fluctuations, and (iii) extended clinical benefit of reduced L-dopa dose administration [67, 83]. Two nitrocatechol-type selective, reversible COMTIs were developed in the 90’s as adjuncts to L-dopa/DCC; entacapone and tolcapone, the clinical use of the latter is currently restricted due to potential hepatotoxicity and requires onerous monitoring [84–86].
Moreover, entacapone is thought to be acting in the periphery, while there is evidence that tolcapone may be acting centrally by crossing the blood brain barrier [65, 87]. They both exhibit linear pharmacokinetics at therapeutic doses, however, in healthy volunteers entacapone has a short half-life of elimination (t1/2β ∼1.3 h), which corresponds well to L-dopa, whereas tolcapone has a longer 4 h half-life and higher bioavailability (∼65% vs 35%) [84, 88–91]. Therefore, it is recommended that tolcapone is administered three times every day, whereas entacapone is administered with every scheduled dose as a separate medication [82, 93]. A single tablet containing L-dopa, carbidopa and entacapone with the brand name Stalevo offers a bioequivalent and potentially more convenient administration regime [94, 95]).
In clinical pharmacokinetic studies, both COMTIs have been shown to increase the AUC and half-life of levodopa, a finding that was highly correlated with improved motor complications in PD patients [67, 96–100, 67, 96–100]. Unfortunately, there is a paucity of studies comparing the efficacy of tolcapone and entacapone in PD, in fact there are no direct comparisons in randomised controlled trials [101, 102]. Lees (2008) suggested that a synopsis of literature supports a greater efficacy of tolcapone [90]. However, a closer inspection of the studies considered reveals important methodological differences, inclusion of open label [103] uncontrolled studies [104] and lack of statistical significance [105]. In a Cochrane review (2004), tolcapone appeared to result in a greater ‘on time’ compared to entacapone (1 h 1 min vs 1 h 55 min) [101]. Nonetheless, the aforementioned observations have been derived from indirect comparisons between trials, and therefore should be interpreted with caution [102].
One possible way of further improving the efficacy of COMTIs has been hypothesised to be the development of long acting COMTIs that exhibit high affinity [91]. Based on this premise, opicapone—a novel, third generation COMTI with a once daily regimen—was introduced recently [106]. It was authorised by the European Medicines Agency (EMA) in June 2016 [107] and was launched in the UK in October 2016. It is licensed for adjuvant therapy to preparations of DDCI in adults with Parkinson’s disease who are experiencing end-of-dose motor fluctuations and cannot be stabilised on those combinations [108]. Opicapone is a hydrophilic 1,2,4-oxadiazole analogue with a pyridine N-oxide residue at position 3 resulting in high specificity and efficacy and in in vivo studies it did not cross the BBB. It has a very high binding affinity and thus a prolonged dissociation rate constant [109]. Indicatively, in human erythrocytes, the half-life of opicapone induced COMT inhibition is over 100 h [110]. In a pilot study, Ferreira et al. (2015) showed that once daily administration of opicapone (5–30 mg) in PD patients resulted in significant–dose dependent but not proportional- increase of levodopa bioavailability (assessed as AUC) and improvement of motor function [111]. The evidence supporting the use of opicapone, entacapone, and tolcapone as adjunctive therapy in PD patients will be explored in this systematic review.
Review aim
The primary aim of this review was to systematically compare the effectiveness and safety of COMTIs (entacapone, tolcapone, opicapone) versus placebo as an adjunct to levodopa in PD patients that had been already on established L-dopa treatment. The secondary aim was to investigate whether COMTIs would influence the progression of motor complications and whether their efficacy would depend on baseline motor function status. Thus this review aims to complement earlier reports by updating the available evidence and by extending the scope of the questions.
METHODS
Design of studies
Prospective double blind randomised placebo-controlled studies comparing one or more COMTIs versus placebo.
Participants
Patients with a clinical diagnosis of idiopathic PD that had been already on established L-dopa/DCC treatment for at least 1 month prior to randomisation. There was no age limit or baseline motor function restriction.
Types of interventions
Oral administration of levodopa/DCCI and one of the three COMTIs versus placebo: There was no restriction on the type of DCCI, and entacapone could be administered as a triple combination of levodopa/carbidopa/entacapone (Stalevo).
Types of outcomes measured:
Improvement of ‘on’ time Reduction of ‘off time’ Unified Parkinson’s disease rating scale Change of motor fluctuations compared to baseline Reversibility post-withdrawal Levodopa dose change from baseline Activities of daily living and any measures of well-being Adverse reactions with emphasis on liver toxicity Dose of COMTI Type of DCCI Incidence and causes of participant withdrawal
Search strategy
Cochrane library, Embase, Medline, NICE HDAS, Ovid, Scopus, PubMed and Web of Science were searched in January 2017. The reference list of the most relevant articles was also re-examined for verification purposes. The chronological range was 1980 – 2017; this time period was selected in order to include the discovery of the second generation COMTIs (entacapone, tolcapone). The search terms and further information on the exclusion criteria can be found in Supplementary Material A and B, the selection process is illustrated in the flow diagram below (Fig. 2).
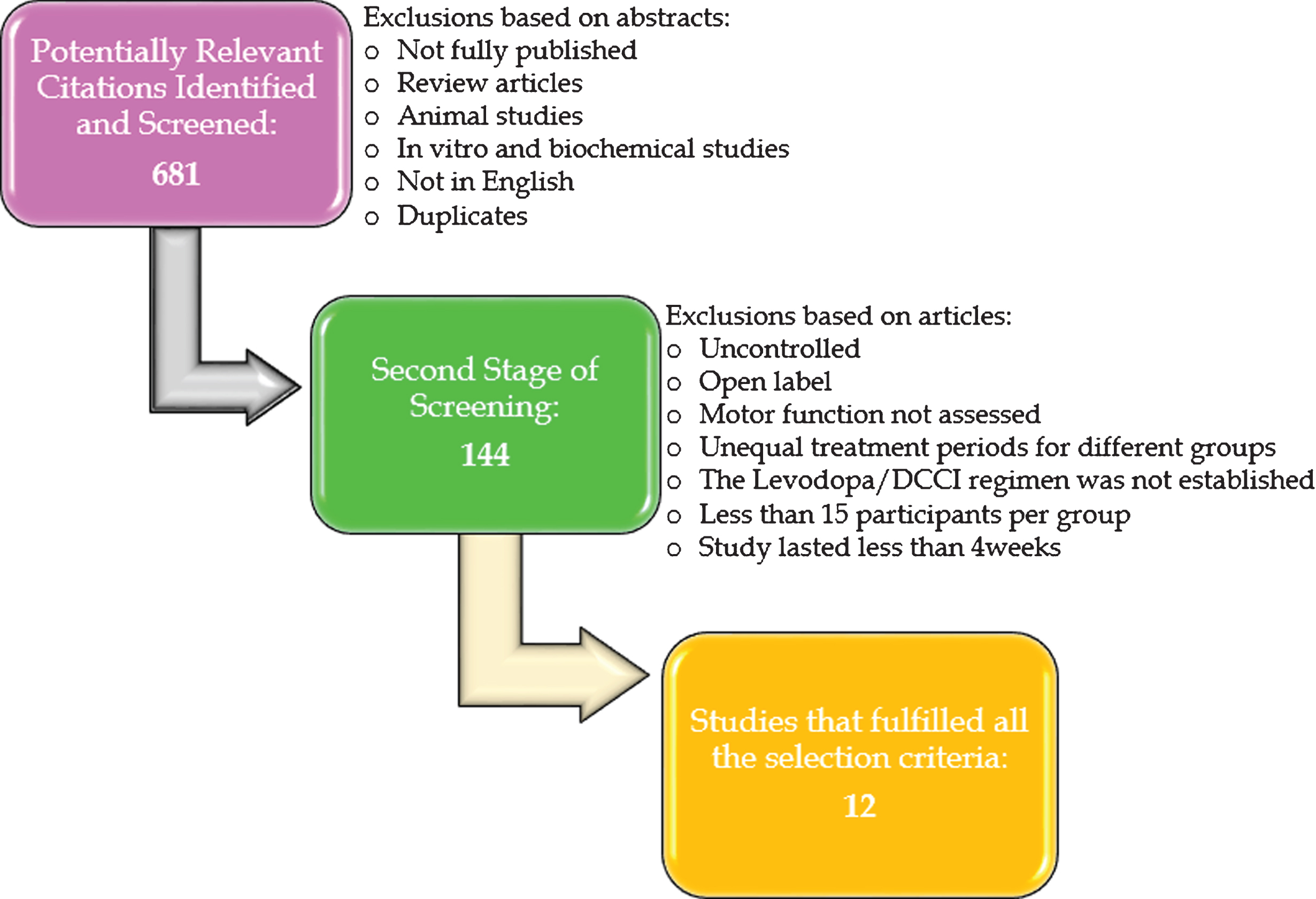
Study selection process for inclusion in review. The flow diagram illustrates the protocol that was followed in order to select the studies that fulfilled all the criteria that were set a priori.
RESULTS
Twelve RCTs with a total of 3701 participants were included in this systematic review [112–123]. Only one study compared two different COMTIs (opicapone and entacapone) versus placebo [123]. All RCTs reported an improvement in motor function following COMTI administration in at least one out of the four main dependent variables. At baseline, the pooled mean age (SD) of PD participants across the three treatment groups was 64 (±13) years and the male to female ratio was 2 : 1 with no significant differences overall. There was only one study which included more females than males in a 40 : 60 ratio [119].
The pooled mean baseline dose of L-dopa did not differ significantly for any of the groups; it was 691(±189)mg, 569(±136)mg, and 659(±21)mg for tolcapone, entacapone and opicapone respectively. However, the treatment with tolcapone resulted in a significantly greater reduction of pooled L-dopa baseline dose compared to entacapone and opicapone (p > 0.001). The pooled dose reduction was as follows: 155 (±720) mg for tolcapone, 48 (±37)mg for entacapone, and 22(±6)mg for opicapone. The dose reduction was not significant between entacapone and opicapone, though it should be noted that the power of such calculations is limited as only one RCT has investigated the effects of opicapone. Thus, the L-dopa dose reduction could be represented as follows; tolcapone>entacapone=opicapone. The same hierarchy was reflected in the ‘on’ and ‘off time’ variables, but the differences were not as robust. Specifically, tolcapone was associated with the greatest increase of ‘on time’ (differences n.s.) and highest reduction of ‘off-time’; 2.2(±0.6) h vs 1.6 (±0.6) h for entacapone and 1.6 (±0.2 h) for opicapone, though these time differences were borderline significant (p = 0.07).
All treatment regimens were overall well tolerated, adverse events were encountered within the first five weeks and there were no deaths associated with any of the treatments. Entacapone was associated the least number of side effects compared to placebo, and tolcapone was associated with the most. Gastrointestinal adverse effects (nausea, vomiting, and diarrhoea) were overall the most frequent reason leading to discontinuation in all three groups, followed by aggravation of parkinsonism. A synopsis of the main findings is presented in Table 1.
Randomised double blind placebo controlled studies investigating the efficacy and safety of COMTIs in PD patients
DISCUSSION
COMTIs have been shown to prolong the therapeutic effect of L-dopa in different study designs, but due to methodological differences their clinical benefit in PD patients is not well defined [102]. The primary aim of this review was to compare the efficacy and safety of tolcapone, entacapone and opicapone in prospective randomised double blind placebo controlled trials.
Efficacy
The present review confirms that COMTIs reduced ‘off time’ and/or prolonged ‘on time’ by at least 1 h when assessed on patient diaries. The beneficial effects appeared to be reversible after the treatment was withdrawn [115–118]. The ‘off time’ variable represents the time period that the medication has worn off [124] and was the most consistent primary efficacy finding in this review. Indeed, only one study (entacapone vs placebo) did not report a significant improvement, however, it deviated from the standard patient characteristics [122]. Specifically, it was the only study conducted in Asia and it had the highest proportion of female participants, and a very low baseline dose [122] that may have led to a plausible floor effect [121].
In terms of relative efficacy, for the ‘off time ‘measurement, tolcapone was associated with the greatest reduction (overall an additional 48 min compared to other COMTIs at 200 mg). This finding is in agreement with previous reports comparing the effects of tolcapone and entacapone in PD patients [101, 125] and can be attributed to tolcapone’s pharmacokinetic and pharmacodynamics profile and its presence in the CNS as well as the periphery [88, 127].
Interestingly, however, one would have expected opicapone to present a greater efficacy than entacapone, based on preclinical pharmacological results [110] yet the two COMTIs did not differ in any of the primary variables [123]. This could be because the highest opicapone dose in that study (50 mg) was lower than the chosen entacapone dose (200 mg) [123]. The effect of a higher dose cannot be extrapolated due to potential toxicity and due to the fact that none of the considered COMTIs exhibited a linear dose-response curve. In fact, in the studies comparing different COMTI doses, a higher concentration was mostly associated with higher incidence of adverse effects and marginal clinical benefit or even a lower effect [112–115, 123]. The reason for this result has not been explored, it has been hypothesised that at higher dose of COMTIs may delay L-dopa’s Cmax and Tmax [82].
Furthermore, there was a lack of correlation between the assessment of motor function in patient diaries and the UPDRS assessment regarding total and motor subscale (III). Specifically, UPDRS (III) improved moderately in 3 out of 7 entacapone studies, and 3 out 5 tolcapone studies, while there was no evidence for opicapone. However, these differences can be attributed to the fact that UPDRS assessment offers ‘a snapshot’ of the patient’s condition [129]. Also, the timing of the assessment may be of critical importance as the effect of COMTIs versus placebo is more prominent as the day progresses [117], but assessments typically occurred in the morning [121].
An important research question that needs to be addressed is whether PD patients can benefit from COMTIs in combination with L-dopa if they are not experiencing a ‘wearing off’ phenomenon. Only three such RCTs have been completed, one investigated potential benefits of tolcapone [116] and two of entacapone [120, 121]. These studies lasted between 24 and 26 weeks with efficacy being based on UPDRS scores rather than patient diaries. They demonstrated a reduction in the L-dopa requirements which was well tolerated. Only Waters et al. (1997) reported a significant improvement in all sections of UPDRS [116]. Furthermore, Brooks and Sager (2003) reported a significant improvement only in part II of UPDRS (ADL) [120], while Olanow and colleagues (2004) showed a beneficial effect only in sections of other quality-of life measures (PDQ-39, SF-36,PSI) [121]. The potential effect of long term treatment in non-fluctuating PD patients has been controversial in other study designs as well. In a 39-week study, entacapone provided symptomatic benefits and did not increase motor complications [130]. By contrast, a 134-week study worryingly found that entacapone was associated with an accelerated onset of dyskinesia and a higher rate of MI, prostate and skin cancer [131]. Despite, methodological limitations these findings warrant further exploration.
Safety
All COMTIs considered were well tolerated and not associated with reported serious adverse experiences. In addition, there was no evidence of hepatotoxicity, though a total of 16 patients in the tolcapone group presented raised LFTs [112, 116], 6 of them withdrew and fully recovered, and the remaining spontaneously recovered. These results are in accordance with suggestions that the monitoring standards on tolcapone are disproportionately strict [132]. Although the European Medicines Agency (EMA) lifted the suspension of tolcapone for patients who fail to respond to other COMTIs, it entails strict liver function monitoring [133]. According to the EMA and NICE guidelines, tolcapone administration requires liver function tests prior to commencing treatment and then every 14 days for the first year of therapy [134, 135]. However, we suggest that these strict monitoring guidelines reflect historic case reports and the results of in vitro and animal studies rather than evidence derived from RCTS.
Furthermore, the entacapone treatment did not present any changes in haematological and laboratory tests, while there was one case of elevated LFTs in the opicapone group (5 mg). Notably, the incidence rate did not differ from the placebo group [123]. Dyskinesia, diarrhoea and nausea were the most commonly reported adverse effects with diarrhoea presenting overall the most common cause of discontinuation in the entacapone group [118, 120]. In addition, entacapone, as well as tolcapone, resulted in innocuous urine discoloration which may have led to the unblinding of some participants [113] and is the reason why studies proceeded with the addition of riboflavin in the placebo [112]. Lastly, even though entacapone is a chelator [136], there were no increased cases of anaemia compared to placebo.
Limitations and future studies
The length of the studies was short and therefore it is difficult to have these findings extrapolated into long-term treatment regimes. In addition, the available number of studies was small and all of them were conducted several years ago. The only exception was the study of Ferreira et al. (2016), it was conducted recently and investigated the effect of two different COMTIs [123]. Thus, further randomised controlled studies are required in order to delineate the relative safety and efficacy of different COMTIs. In addition, all studies except for one [114] permitted the concomitant use of dopamine agonists, this however may have resulted in interaction effects and may have further complicated the disentanglement of COMTIs’s independent effects.
Another limitation is that the studies considered in this review have utilised different protocols in the collection and analysis of diary data, which is likely to have influenced the magnitude of the calculated effects. Therefore, future studies should employ a standardised diary and data extrapolation system [124]. In addition, there are concerns about the recall bias and compliance of diary recording [137] and, hence, future studies could utilise electronic diaries [138] or kinetographs [139]. Interestingly enough, there has been no consideration of the potential confounding that may have resulted from the use of different DCC and L-dopa formulations as these may have altered the pharmacokinetic profile of L-dopa [140]. Certainly the aforementioned limitations constitute new avenues for further research.
CONCLUSION
In randomised double-blind placebo controlled studies, adjuvant treatment of PD patients experiencing motor fluctuations with entacapone was well tolerated and resulted in motor function improvement. Therefore, entacapone presented an acceptable benefit to risk ratio. Tolcapone appeared to result in a greater therapeutic effect, however, this was not consistent across all motor variables and studies, and thus would not support its use, given the current onerous monitoring that is required. The third COMTI considered, opicapone was not associated with adverse reactions in a phase III trial; nonetheless it did not present greater efficacy when compared with entacapone, and therefore further studies are necessary in order to illustrate its cost effectiveness. Indeed, several avenues for future research remain to be explored, including RCTs directly comparing the efficacy and safety of the three COMTIs, and revealing potential interaction effects of COMTIs with different dopamine decarboxylase inhibitors.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
