Abstract
Background:
Caffeine intake has been associated with a lower risk of Parkinson’s disease (PD). This association is robust in men, but inconsistent in women due to a possible interaction with post-menopausal hormone (PMH) use.
Objective:
To (1) evaluate the association between caffeine intake and PD risk and (2) assess potential effect modification of the association by PMH use among women.
Methods:
We examined associations between caffeine intake and incident PD risk in the Nurses’ Health Study (NHS) (N = 121,701 women) and the Health Professionals Follow-up Study (HPFS) (N = 51,529 men). Dietary data on coffee and caffeine from other sources were collected every four years using a validated semi-quantitative food frequency questionnaire for both cohorts. Information on lifestyle and incident PD diagnosis was updated biennially and PD diagnoses were confirmed by medical record review. We estimated hazard ratios (HR) and 95% confidence intervals (CI) using Cox proportional hazards models.
Results:
We documented a total of 1,219 PD cases over the follow-up period. The multivariable-adjusted HR comparing the highest to lowest quintile of caffeine intake was 0.50 (95% CI: 0.37, 0.68; Ptrend<0.0001) in the HPFS. Among women, there was a suggestion of an interaction between coffee intake and PMH use (P = 0.08). In the pooled analyses combining men and women who have never used PMH, the risk of PD was lower as coffee intake increased (Ptrend<0.001).
Conclusions:
Our results support previous findings that increased caffeine intake may be associated with a decreased PD risk in men and women who have never used PMH.
Keywords
INTRODUCTION
A lower risk of Parkinson’s disease (PD) among coffee drinkers has been observed in numerous longitudinal studies [1–10]. This inverse associationwas also present for caffeine from non-coffee sources, such as cola beverages, chocolate, and non-herbal tea. However it was not present for decaffeinated coffee, suggesting that the inverse association is largely due to caffeine, rather than niacin or other biologically active compounds found in coffee [1, 11]. Caffeine serves as a non-selective adenosine receptor antagonist, and appears to have neuroprotective effects in animal models of PD by blocking the A2A subtype of adenosine receptor [12–15].
While a robust inverse correlation between caffeine intake and incident PD risk exists for men, a more complex association exists for women. Whereas a ‘beneficial’ association of caffeine was observed among women who do not use postmenopausal hormones (PMH) in most studies, conflicting results were observed among PMH users. However tests of interaction showed conflicting results, possibly due to low power [1–3, 16]. In the present study, we prospectively examined the associations between caffeine consumption and PD risk in the Nurses’ Health Study (NHS) and the Health Professional Follow-Up Study (HPFS). Results from the early follow-up of these cohorts have been published previously [2, 17] but we present here updated results after an additional 14 years of follow-up for the NHS and 16 years for the HPFS. In this study, we sought to elucidate the possible interaction between caffeine intake and PMH on PD risk. With 908 additional cases and increased power, we were able to investigate possible effects of caffeinated and decaffeinated beverages, use of different types of PMH among women, and updated PMH use over the follow-upperiod.
MATERIALS AND METHODS
Study population
The Nurses’ Health Study (NHS) enrolled 121,700 female registered nurses of ages 30–55 who returned mailed, self-administered questionnaires regarding medical histories, lifestyle and dietary factors in 1976. The Health Professionals Follow-up Study (HPFS) enrolled 51,529 male health professionals of ages 40–75 who returned similar questionnaires in 1986. The baseline for the analyses of this study for the NHS was 1980, while the baseline for the HPFS was 1986, as the first dietary assessments of caffeine consumption were collected then. For both cohorts, follow-up information on lifestyle factors wascollected every two years and dietary information was updated every four years. Participants with onset of PD or participants who died before the study baseline, participants who reported extreme caloric intakes (<800 or >4,200 kcal for men; <600 or >3,500 for women), and participants who lacked data on baseline coffee intake were excluded. Therefore, our analytic cohort included 90,254 women from the NHS and 47,474 men from the HPFS. This study was approved by the Human Research Committees at the Brigham and Women’s Hospital and the Harvard T.H. Chan School of Public Health.
Dietary assessment of coffee consumption and other covariates
Nutritional information was ascertained via validated food frequency questionnaires (FFQs). The FFQs were self-administered and asked to report participants’ average intake pattern of a food or beverage item over the past 12 months. Nine possible multiple-choice responses were provided for intake frequency for each item, ranging from “never or less than once per month” to “6 or more times per day” for both cohorts. The 1980 FFQ included questions on servings of coffee with caffeine (cups), tea (cups), cola beverages (glasses), and chocolate (in 1-oz servings). From 1984 onwards, all FFQs additionally included items on decaffeinated coffee (cups), caffeinated and caffeine-free soda beverages (glasses). Information obtained from the U.S. Department of Agriculture food-composition sources was used to convert participants’ reported average intake of one serving of a caffeinated beverage or food over the preceding year into average daily intake of caffeine. The average caffeine content used for these calculations was estimated to be 137 mg caffeine per cup of coffee, 47 mg of caffeine per cup of non-herbal tea, 46 mg of caffeine per can or bottle of cola beverage, and 7 mg per serving of chocolate. The reproducibility and validity of the FFQs have been previously reported for the NHS [18] and the HPFS [19]. The validation studies on a subsample of the cohorts show a high correlation between self-reported intake of caffeinated beverages via the FFQ and four 1-week diet records. The Pearson correlation for coffee was 0.93 in the HPFS and 0.78 in the NHS, while the correlation for non-herbal tea was 0.77 in the HPFS and 0.93 in the NHS. Both cohorts had the same correlation for cola beverages (r = 0.84) [20, 21]. Information on other covariates regarding lifestyle characteristics was collected, including information on menopausal status and postmenopausal hormone therapy (PMH) use in the NHS.
Ascertainment of PD cases
Biennial self-report questionnaires were administered to ascertain new illness diagnoses. Lifetime occurrence of PD was first asked in the 1994 (NHS) and 1988 (HPFS) questionnaires and incident PD diagnoses were documented every two years thereafter via subsequent questionnaires. We asked self-reported PD cases for permission to contact their neurologist to request copies of their medical records to confirm the diagnosis. After obtaining their permission, we contacted patients’ neurologists and requested for them to either return a self-administered diagnostic questionnaire that asked to confirm the case or to send a copy of the patient’s medical records. In previous years, PD cases were considered confirmed if they fulfilled at least one of the following conditions: the treating neurologist rated the certainty of diagnosis as definite or probable; the final diagnosis of PD by a neurologist was included in the medical record; or the medical record indicated the presence of at least two out of three cardinal signs of PD (i.e. resting tremor, rigidity, bradykinesia) in the absence of features indicating other illness diagnoses. After 2003, a similar procedure was used to identify confirmed PD cases; however, medical records were requested from all PD cases, which were then reviewed by a neurologist specializing in movement disorders. If the diagnosis of the neurologist specializing in movement disorders conflicted with that of the original neurologist, the diagnosis of the movement disorder neurologist was used.
Statistical analysis
We conducted all analyses separately in each cohort and subsequently pooled cohort-specific estimates. Participants contributed person-years starting from age in months at baseline (1980 for NHS; 1986 for HPFS) until the age in months at the date of first PD symptoms, date of death, date of the latest completed questionnaire, or end of follow-up (June 2012 for NHS; January 2012 for HPFS), whichever occurred first. The analysis was stratified jointly by age in months at the start of follow-up and calendar year of the current questionnaire cycle in order to finely control for confounding by time.
We compared incident PD risk in cohort-specific quintiles of caffeinated and decaffeinated beverages. Within each cohort, cumulative averages of time-updated covariates, such as alcohol intake and physical activity, were categorized into quintiles. Nutrient intake (e.g., flavonoids, fructose, dairy protein, vitamin C, and vitamin E) was adjusted for total energy intake using the residual method to account for its correlation with nutrient intake [22]. We calculated age-adjusted and multivariable-adjusted hazard ratios and corresponding 95% confidence intervals using Cox proportional hazards model. The multivariable regression adjusted for the following time-updated covariates: pack years of smoking (never smoker, 1 to <5, 5 to <10, 10-<15, or ≥15), alcohol intake (in quintiles), and physical activity (in quintiles). Further analyses were conducted using baseline caffeine intake levels.
For tests of trend, the mid-category scores (median caffeine intake value within each quintile) was modeled as a continuous variable to allow for possible nonlinear associations. Secondary analyses included incorporating a lag period by excluding the first years of follow-up (2, 4, and 6 years), evaluating decaffeinated beverages and total caffeine from non-coffee sources, such as non-herbal tea and other caffeinated beverages, and using cumulative averages of coffee intake in categories: less than one cup of coffee per day (“<1”), 1–3 cups per day (“1–3”), 3–5 cups per day (“3–5”), 5 or more cups per day (“≥5”), and the reference group of no coffee intake or less than 1 cup per month (“0”). We also conducted analyses additionally adjusting for BMI, dairy intake, dietary intake of antioxidants, and total energy intake. Similar analyses were performed among women in the NHS cohort, stratified by PMH use (ever/never). PMH status was updated over the study period. If a woman ever initiated PMH use, she would be considered an ever-user for all consequent follow-up questionnaires. To determine statistical significance of a possible interaction between coffee intake and PMH use, we conducted likelihood ratio tests by comparing the log-likelihood of a model including interaction terms to that of a model without the interaction terms on the multiplicative scale in each cohort. Results from the HPFS and NHS cohorts were pooled using random-effects modeling to account for the variation in the effect of caffeine on PD risk between the cohorts. All statistical analyses were conducted using SAS (SAS Institute, Cary, NC).
RESULTS
There were a total of 1,219 cases (590 in HPFS, 629 in NHS) observed over the 3,485,803 person-years of follow-up. Women had a higher baseline meancoffee intake compared to men (women: 2.22 cups/day, men: 1.34 cups/day). Participants with the highest caffeine intake tended to smoke more, were generally more likely to have higher alcohol intake, and be less physically active compared to participants with the lowest caffeine intake (Table 1).
Baseline characteristics according to quintile of total caffeine intake in the Health Professionals Follow-up Study (1986) and the Nurses’ Health Study (1980)
Abbreviation: SD, standard deviation. aValue from 1984 questionnaire.
Overall, higher caffeine intake was associated with a lower PD risk among men and women who have never used PMH (pooled multivariable-adjusted Ptrend = 0.05) (Table 2); the pooled multivariable-adjusted HR comparing the highest to lowest quintile of caffeine intake in men and women with never PMH use was 0.62 (95% CI: 0.39, 0.98; p = 0.04; Pheterogeneity = 0.09). Consistent with previous studies, higher total caffeine intake was associated with a lower risk of PD among men (multivariable-adjusted Ptrend < 0.0001). The risk of PD decreased monotonically with increasing quintile of caffeine intake; the HR comparing the highest quintile to the lowest quintile of caffeine consumption 0.50 (95% CI: 0.37, 0.68; P < 0.0001) in men. For women, neither caffeine nor coffee intake was associated with PD risk (Ptrend = 0.30, Ptrend = 0.39, respectively) (Supplementary Table 1).
Hazard ratios of developing Parkinson’s disease according to quintile of caffeine intake in the Health Professionals Follow-up Study (1986–2012) and the Nurses’ Health Study (1980–2012)
aAdjusted for age (years). bAdjusted for age, pack years of smoking, physical activity, and alcohol intake. cPooled analyses for men (HPFS) and women (NHS) who have never used PMH.
Among women who have never used PMH, there was a trend towards an inverse association with categories of coffee intake, though the results were not statistically significant (Ptrend = 0.16) (Table 3). However, when specific types of hormonal therapies were examined (e.g., progesterone, estrogen, combination therapy), we observed a borderline significant interaction between coffee intake and hormonal therapy use (P = 0.08). Furthermore, there was a significant interaction between coffee consumption and use of progestin only (Ratio of HR: 1.59; 95% CI: 1.11, 2.30; Pinteraction = 0.01). However, this result should be interpreted cautiously due to the low number of cases (n = 8). When we restricted our analyses to “definite” cases, an inverse association was observed specifically among women who have never used PMH (multivariable-adjusted Ptrend = 0.01) (Supplementary Table 2).
Hazard ratios of Parkinson’s disease by coffee intake and postmenopausal hormone use (PMH) in the Nurses’ Health Study (1980–2012)
aAdjusted for age (years). bAdjusted for age, pack years of smoking, physical activity, and alcohol intake. cPooled analyses for men (HPFS) and women (NHS) who have never used PMH.
These findings were robust in additional analyses, including a lag analysis of 2, 4, 6, and 8 years, and further adjusting for BMI, total energy intake, vitamin C, vitamin E, dietary urate index, dairy protein, total dairy, fructose, and flavonoid intake, and the Alternate Health Eating Index (derived from intake of fruit, vegetables, nuts, soy, and other dietary components) [23]. Analyses considering servings of coffee linearly or total caffeine intake from non-caffeine sources did not change the results.
Furthermore, we investigated possible associations between decaffeinated coffee and caffeinated non-coffee beverages (e.g., soda) and PD risk among men and women who consumed less than 1 cup of coffee per day. Our results show that there was a significant inverse association between non-herbal tea consumption and PD risk among men across quintiles of cumulative averages (382 cases; multivariable-adjusted ptrend= 0.03) (Fig. 1a). The association between other caffeinated beverage intake from non-coffee sources and PD risk was borderline significant (385 cases; multivariable-adjusted Ptrend= 0.08) (Fig. 1b), while evidence for a potentially harmful effect was unexpectedly detected among men who drank decaffeinated coffee; per each increasing cup of decaffeinated coffee, there was a 13% higher risk of PD (HR = 1.13, 95% CI: 1.01, 1.25; P = 0.03). Finally, consistent with the direction of results for caffeine intake, marginally significant effects were found in the association between consumption of other caffeinated beverages from non-coffee sources and reduced risk of PD (72 cases; multivariable-adjusted P trend = 0.05) among women who have never used PMH. No statistically significant relation between tea and PD risk was found, possibly due to the low number of cases (n = 84) (Supplementary Figure 1).
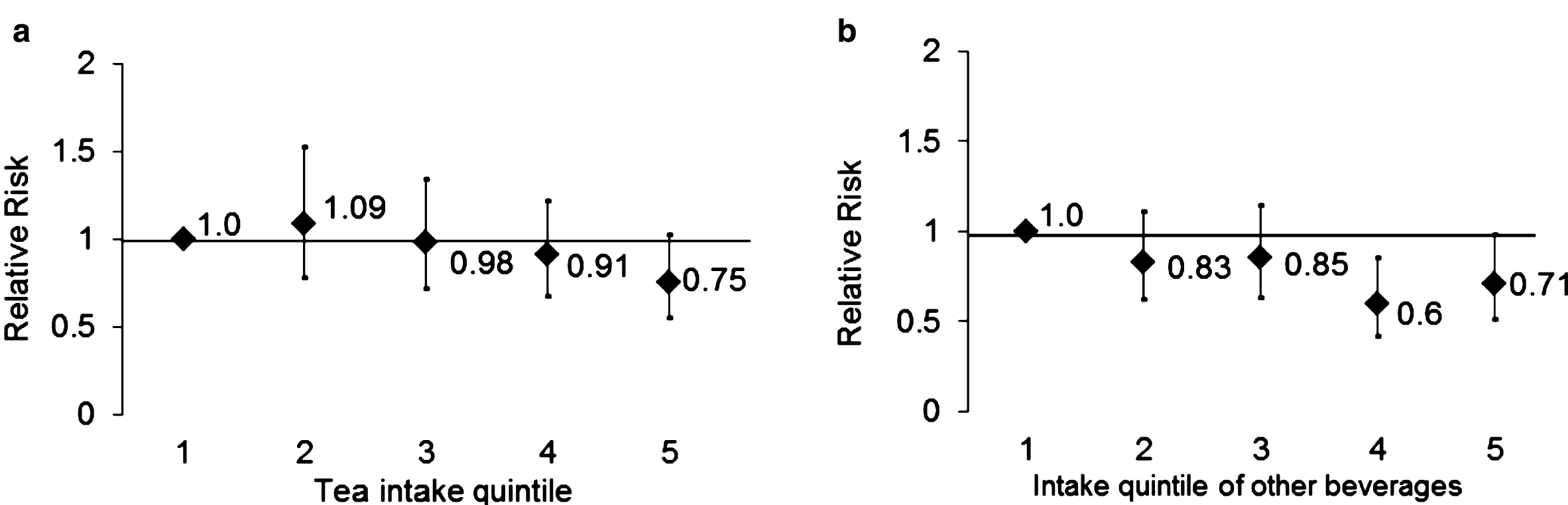
(a, b) Associations of (2a) tea (ptrend = 0.03) and other (2b) caffeinated beverages (Ptrend = 0.08) with PD using quintiles of cumulative average intake level, adjusting for pack years of smoking, physical activity, and alcohol intake among men who consume less than 1 cup of coffee per day.
DISCUSSION
In these analyses of two large prospective cohorts, greater intakes of coffee and total caffeine, but not intake of decaffeinated coffee, were associated with a lower risk of Parkinson’s disease. These results were consistent in pooled analyses including men and women who have never used postmenopausal hormone therapy, confirming our previous findings [1–3, 16]. In addition, the lack of protective association between consumption of decaffeinated coffee and PD risk suggests that caffeine, rather than other biological components of coffee, is the presumptive protective factor. The strengths of our study include a large number of cases for increased power, high follow-up rates in all cohorts, and repeated assessment of dietary intake and lifestyle factors using a prospective study design, which minimizes the potential for recall bias and reverse causation.
Our study has several limitations. First, during the early stages of disease onset, participants’ dietary habits can change due to an altered sense of taste and smell, possibly influencing coffee-drinking behavior. Because of the prolonged prodromal period of PD, we cannot exclude the possibility of bias from reverse causation. However, this potential source of bias does not appear to be substantial, as we consistently found a strong inverse association between coffee consumption and the onset of first PD symptoms in lag-time analyses as well as using baseline intake levels. In addition, the association remained robust in analyses using baseline caffeine levels for both men and women who have never used PMH. Second, there may have been measurement error of the exposure because the FFQ, as with any other method of measuring dietary data, may not have captured the exact coffee intake. However, the FFQ has been validated in both of our cohorts; in addition, any measurement error is expected to be non-differential with respect to PD, which would likely bias our results towards the null. In addition, because the study cohorts were restricted to health professionals, our results may have limited generalizability to other populations. However, because our subgroup of the population has a higher consumption of caffeinated beverages compared to the general population, we were able to determine long-term PD risk with respect to a wide range of total caffeine intake. It should be noted that due to the observational nature of the study, the possibility of residual confounding and/or unmeasured confounding should be considered. However, despite several sensitivity analyses performed to adjust for potential confounders, estimates remained largely unchanged, indicating that unmeasured confounding is likely to be modest.
While there is conflicting evidence regarding the statistical presence of a caffeine-PMH interaction, most studies have reported that a significant protective effect of caffeine on PD is limited to women who have never used PMH [1–3, 16]. Our pooled results indicated that an inverse association between coffee intake and PD risk was present in men and women who never used PMH. Because post-menopausal hormone therapies frequently include a combination of estrogens and progestin, we further explored possible effect modification by different types of hormonal therapy. Progestin, as well as estrogens, has been shown to confer neuroprotection in animal studies [24–28]. Interestingly, our findings suggest a significant interaction between coffee intake and PD risk among women who were only treated with progestin compared to women who were not treated with any PMH, regardless of type. However, the number of cases in this subtype was low, partially because progestin-only therapy was rare; therefore, confirmation of these results are warranted. While we did not observe protective effects that were statistically significant of coffee intake among never users of PMH, we observed a marginally significant interaction between coffee intake and PMH use in women when we considered the different types of PMH therapies. It is also important to note that potential effects of PMH may have been attenuated over time, as the prevalence of PMH use peaked in the 1980’s and early 1990’s and declined over the recentyears [29].
Caffeine is believed to be protective against excitotoxicity and dopaminergic neuron injury by antagonizing brain adenosine A2A receptors [12, 30]. In animal studies, rodents that were administered caffeine were protected against loss of nigrostriatal dopaminergic neurons, induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) or its active toxin metabolite [12, 30–32]. Furthermore, studies have demonstrated that caffeine down-regulates neuroinflammatory responses and nitric oxide (NO) production, which may delay neuronal cell death [33].
Our epidemiological results support experimental evidence for effect modification by PMH for the association between caffeine and PD risk in animal models. For instance, in ovariectomized aged (retired breeder) female mice the neurotoxic effect of MPTP, as reflected by striatal dopamine depletion, was reduced by caffeine [34]. Similarly, in young male mice caffeine treatment attenuated MPTP-induced striatal dopamine loss; however, in male mice that were pretreated with estrogen caffeine did not have a neuroprotective effect.
In summary, the results of this study with two large longitudinal cohorts support our previous reports that total caffeine intake is inversely associated with PD risk in men and in women who have never used post-menopausal hormones.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
