Abstract
Background:
Polyneuropathy (pnp) is recognized as a clinical feature of Parkinson’s disease (PD). Whether pnp is a result of the alpha-synucleinopathy or related to treatment is debated. Previous studies support underlying disturbances in the methionine cycle mediated by L-dopa.
Objective:
Describe possible relationships between methionine cycle metabolism and the development of pnp in L-dopa treated PD. Furthermore, we aim to investigate possible genetic risk factors by genotyping specific SNPs in enzymes involved in the abovementioned pathways.
Methods:
In a cross-sectional study design, L-dopa treated PD patients (n = 33) and controls (n = 16) were evaluated with biochemical and genetic analyses. Subjects were assessed clinically and with regards to signs of pnp using established clinical neuropathy rating scales.
Results:
16/33 patients fulfilled a study diagnosis of pnp compared to 0 age-matched controls. Levels of homocysteine (Hcy) were significantly higher in patients with pnp (n = 16) compared to controls. A significant correlation between neuropathy scores and Hcy was seen in the whole patient group (n = 33). A significant difference in the genotype distribution of the COMT A158G polymorphism was demonstrated, favoring the low activity genotype in patients with pnp compared to both controls and patients without pnp.
Conclusions:
Pnp is a prevalent condition in L-dopa treated PD and an association may exist with elevated levels of Hcy, possibly reflecting an underlying impaired cellular methylation capacity. Furthermore, an association may exist between the low activity COMT genotype and pnp. These preliminary findings and the suggested pathophysiological mechanisms should be confirmed in future large-scale studies.
INTRODUCTION
Parkinson’s disease (PD) is diagnosed based on the presence of bradykinesia, rigidity and tremor [1]. Previous studies have shown an increased prevalence of polyneuropathy (pnp) among patients with PD. One study, based on 58 patients with PD, calculated the prevalence of pnp as high as 55% [2]. Accumulative exposure to L-dopa [2–4], serum levels of methylmalonic acid (MMA) [2, 5], vitamin B12 [4], B9 [3] and homocysteine (Hcy) [4–6] have in varying degrees been associated with the development of a symmetric sensory predominant axonal pnp in PD. Furthermore, some studies have proposed a pathophysiology mediated by the alpha-synucleinopathy itself, possibly presenting with a small-fiber neuropathy [7, 8]. A complete understanding of the pathogenesis of pnp in PD is yet to be determined.
The methionine cycle describes metabolic pathways involved in the cytosolic recycling of Hcy to methionine by means of remethylation (Fig. 1). As shown in Fig. 1, maintenance of the cycle is dependent on the presence of vitamin B12 and B9 [9]. Catechol-O-methyltransferase (COMT) is an enzyme which acts both within and outside of the central nervous system (CNS), catalyzing the methylation of L-dopa but also other substrates such as dopamine and other catecholamines as well as catechol estrogens. COMT is thus thought to play an important physiologic role [10, 11]. In order to function, COMT requires interaction with s-adenosyl-methionine (SAM) which serves as a methyl donor in various other physiological reactions [9, 11–13].
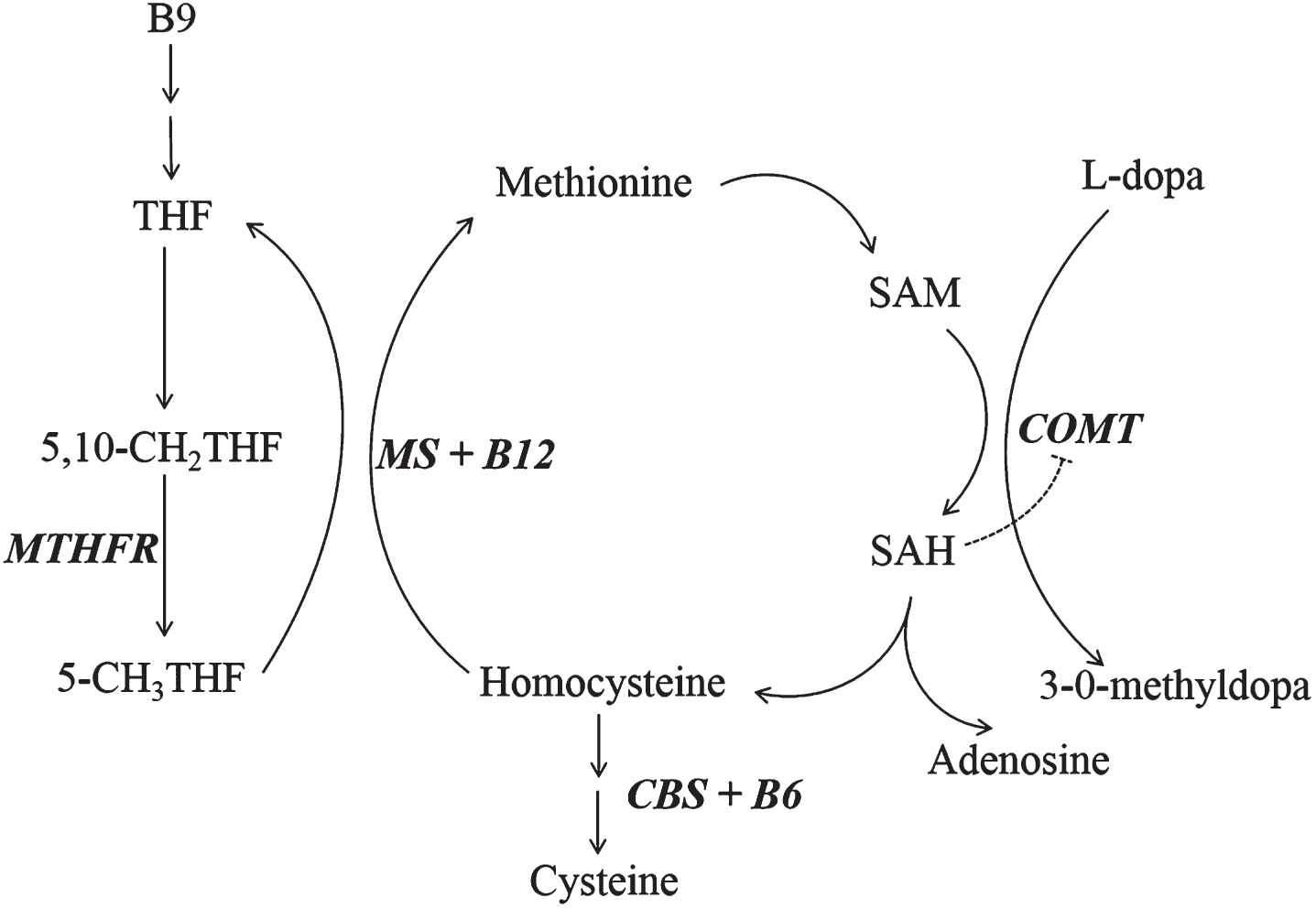
The methionine cycle. Continuous access to methionine maintains SAM as a methyl donor in various physiological methylation dependent reactions. After donating a methyl group, SAM is subsequently metabolized to homocysteine (Hcy). In order for Hcy to be remethylated by methionine synthase (MS) back to methionine, the presence of vitamin B12 and an intact folate cycle are required. In the figure we have, as an example, put in COMT as a receiver of methyl groups from SAM illustrating the potential effects on methionine cycle metabolism of L-dopa loading in a PD patient. The line from SAH to COMT illustrates the noncompetitive inhibitory effect of SAH on COMT activity. Abbreviations: THF – tetrahydrofolate; 5,10-CH2THF –5, 10-methylenetetrahydrofolate; 5-CH3THF - 5-methyltetrahydrofolate; MTHFR - methylenetetrahydrofolate reductase; MS – methionine synthase; SAM - s-adenosyl-methionine; SAH - s-adenosyl-homocysteine; COMT - catechol-O-methyltransferase; CBS – cystathionine beta synthase.
Single-nucleotide polymorphisms (SNPs) have been described in the enzyme methylenetetrahydrofolate reductase (MTHFR) which is involved in the methionine cycle (Fig. 1). Two SNPs, C677T rs1801133 and A1298C rs1801131, are well studied. It is known that homozygosity for the MTHFR 677 T/T variant is associated with increased levels of Hcy due to induced thermolability of the enzyme with resulting decreased activity. The A1298C SNP is also associated with decreased enzyme activity, although to a lesser extent [14, 15].
SNPs have also been described in COMT where A158G rs4680 is known to be an activity-regulating polymorphism. Valine homozygosity (G/G) in this position has been associated with high, and methionine homozygosity (A/A) with low, enzyme activity [16].
Homozygous and heterozygous mutations in the lysosomal enzyme glucocerebrosidase (GBA) are established genetic risk factors for the development of PD, to some extent supporting a role of lysosomal dysfunction in PD [17, 18]. Furthermore, vitamin B12 is known to be dependent on the lysosome for its intracellular metabolism [17, 20].
A deficiency of vitamin B12 and B9 could, in theory, lead to increased levels of Hcy, reflecting a reduced capacity for remethylation of Hcy to methionine. One could expect that treatment with L-dopa stimulates COMT activity, resulting in an increased demand for vitamin B12- and B9-dependent reactions to maintain low levels of Hcy [9, 22]. Studies investigating the role of COMT-inhibition on methionine cycle metabolism in L-dopa treated PD have reported conflicting results with some [12, 24] seeing decreased levels of Hcy together with increased levels of vitamin B9 and B12, while others [2, 25] seeing decreased levels of vitamin B12 and B9 or absence of effects on Hcy-levels. Moreover, one study gave support for a protective role of COMT-inhibition in the context of development of pnp in L-dopa treated PD [24].
In this study we aim to describe possible relationships between methionine cycle metabolism and the development of pnp in patients with L-dopa treated PD. Furthermore, we aim to investigate possible genetic risk factors by genotyping specific SNPs in enzymes involved in the methionine cycle and mutations in the lysosomal protein GBA.
MATERIALS AND METHODS
Subjects
The study was approved by the regional ethical board of Stockholm, Sweden (reference number 2015/2271-31/2) and the patient work was performed in accordance with the Helsinki Declaration. A written informed consent was obtained from patients and controls. All study subjects were assessed during the period 2016-01-01 – 2016-07-31.
Subjects were recruited from an existing research cohort with consecutively included patients and controls or from regular visits during the abovementioned period at the outpatient clinic of the Department of Neurology, Karolinska University Hospital, Stockholm. Participants, from both sources, were recruited by inquiring about general interest in participating in a study concerning PD, B vitamins and neuropathy. Participants were given the same consent forms to fill out.
A controlled study population was selected with the following criteria:
Inclusion criteria:
Age 55–70 years Possible or probable PD according to Gelb et al. criteria [26]. Ongoing treatment with L-dopa
Exclusion criteria:
Existing diagnosis of pnp Existing diagnosis of diabetes mellitus Overconsumption of alcohol, defined as an existing documentation in the participant’s patient journal of overconsumption or a diagnosis of alcohol related disorders.
After reviewing patient journals, subjects meeting the criteria were invited to participate in the study.
Clinical assessment
Subjects were assessed during one visit according to a specific protocol. General data regarding smoking habits, disease-specific questions, Hoehn and Yahr staging and current medication was obtained through oral interview and review of patient journals. Ongoing B vitamin substitution was documented. Non-prescription multivitamins were documented as “multivitamins”. The calculation of daily L-dopa dose was based solely on L-dopa containing drugs, in line with the study hypothesis of L-dopa mediated disturbance of the methionine cycle. Established conversion factors were used for L-dopa slow release formulations and levodopa-carbidopa intestinal gel (LCIG) [27]. Other dopaminergic medications were also documented. Cognitive functions were screened in all subjects with Montreal Cognitive Assessment (MoCA). Signs of pnp were evaluated with two clinical assessment scales: UENS (Utah Early Neuropathy Scale) and NIS(legs) (Neuropathy Impairment Scale (legs)) [28, 29]. The choice of scales was based on the aim of being able to detect an early sensory predominant length dependent pnp. Clinical diagnosis of pnp was predefined as > 4/42 p as measured with UENS. This cut-off value has been used in a previous study evaluating pnp in PD [30].
Biochemical analyses
1) Venous blood samples were collected from all subjects between 8:30 AM and 4:30 PM. With the exception of Hcy [31] and folate [32], to our knowledge no significant effects of circadian rhythmicity on the levels of the studied biochemical parameters have been reported. Biochemical analyses were made at the Clinical Chemistry Laboratory at Karolinska University Hospital. Hcy in plasma was measured with an enzyme cycling and photometric method (Cobas 8000 instrument; Roche Diagnostics; Switzerland). Vitamin B12 in serum was measured with an immunoassay employing a competitive test principle using intrinsic factor specific for vitamin B12 (Modular E instrument; Roche Diagnostics, Switzerland). MMA in serum was measured with an isotope dilution method followed by mass spectrometry (LC-MS/MS Quattro Premier and Xevo TQ MS instrument). Vitamin B9 in serum was measured with an immunoassay using a competitive test principle (Modular E instrument). In one patient, measurement of vitamin B12 and B9 failed due to hemolysis.
Genetic analyses
In 4 subjects DNA was extracted using the Qiagen AllPrep (Hilden, Germany) at the department of Clinical Genetics, Karolinska Hospital. In the remaining subjects, whole blood PBMC (peripheral blood mononuclear cells) were collected and frozen in –80°C, until DNA was extracted using standard methods (Qiagen AllPrep and PureLink Genomic DNA Mini Kit) according to manufacturer’s indications (Thermo Fisher Scientific; Sweden).
The COMT and MTHFR SNPs were genotyped with commercially available TaqMan assays; C_25746809_50 for rs4680, C_850486_20 for rs1801131 and C_1202883_20 for rs1801133 respectively according to the TaqMan® SNP Genotyping Assays Guide (ThermoFisher Scientific). Allele discrimination was performed with the software supplied with the instrument (CFX96 Touch™ Real-Time PCR Detection System, Bio-Rad, Sweden).
For the N370S mutation in the GBA gene, we used a custom-designed assay (forward primer: 5′ GTG ACC CTT ACC TAC ACT CTC T, reverse primer: 5′ GGT TCA GGG CAA GGT TCC), the reverse primer being specific for the GBA gene, thus excluding the pseudogene GBAP1. Allelic discrimination was run with 2 5′ fluorescence-labeled allele-specific probes: C_2286559_30 and C_2268560_20, genotyping master mix (Applied Biosystems), and 20 ng of genomic DNA. We used the default PCR fast cycling conditions, 10 min at 95°C, 15 seconds at 92°C and 1 minute at 60°C, for 40 cycles. Analysis was performed with software supplied with the instrument (QuantStudio 6 and 7, Flex Real-Time PCR System Software v 1,0) [33].
L444P gene mutation in the GBA gene was detected and analyzed with PyroMark Q96 apparatus [33]. PCR was performed using the Pyromark PCR Kit with 60°C as annealing temperature. Pyrosequencing reactions were conducted using Qiagen solutions and PyroMark Gold Q96 according to manufacturer’s instructions. Eighty degrees was set as temperature for annealing sequencing primer to the biotinylated PCR product immobilized to streptavidin-coated beads (GE Healthcare).
Statistical analysis
Statistical tests were performed with Statistica version 13 with p-values < 0,05 considered to be statistically significant. Nonparametric tests were used to calculate correlations (Spearman Rank Correlation) and group differences (Mann-Whitney U-test and Pearson chi-square test).
RESULTS
33 PD patients and 16 controls were included in the study population (Table 1). The mean age was very similar between the control and patient groups. A significant overrepresentation of males was apparent in the patient group. Comparing patients with regards to sex, no significant differences between levels of Hcy, MMA, MoCA, UENS, NIS(legs) or age were seen. Neither in the control group did differences between the same parameters with regards to sex reach significance.
Demographic characteristics of the study population. Daily L-dopa dose was calculated solely based on L-dopa containing medications with conversion factors used for slow release formulations and LCIG (levodopa-carbidopa intestinal gel)
acomparing patients with pnp and non-pnp patients. bcomparing patients with pnp and controls. ccomparing non-pnp patients and controls. 1Mann-Whitney U-test. 2Pearson’s chi-square test.
In the patient group, 16/33 (48%) fulfilled the study criterion for a diagnosis of pnp (Table 2). In contrast, in the control group no subject fulfilled this criterion. When comparing mean scores on UENS and NIS(legs), significantly higher scores were seen in the patient group with a significant correlation between scores in relation to Hcy-levels (Fig. 2A-B). Hcy-levels were significantly higher in patients with pnp compared to controls (Table 2, Fig. 3). No differences between serum levels of MMA were found between patients and controls (Table 2).
Clinical, biochemical and genetic characteristics of the study population. Significant difference in the distribution of the COMT A158G genotype is seen, favoring the A/A genotype, in patients with a study diagnosis of pnp compared to patients without pnp and controls
acomparing patients with pnp and non-pnp patients. bcomparing patients with pnp and controls. ccomparing non-pnp patients and controls. 1Mann-Whitney U-test. 2Pearson’s chi-square test, A158G: χ2a = 6,9, χ2b = 6,21.
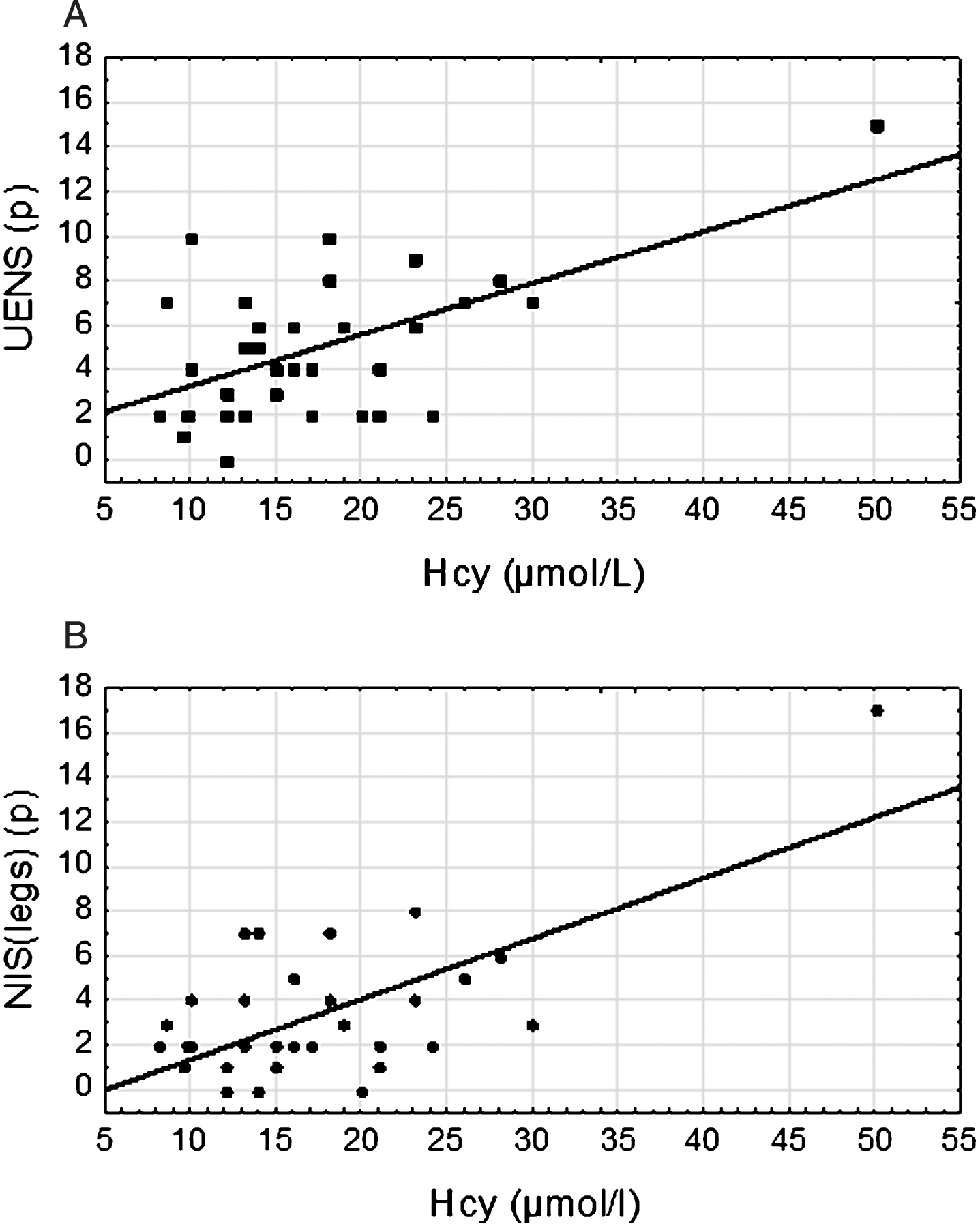
A. The graph shows significant correlation between UENS-score and Hcy-levels in plasma in patients with L-dopa treated PD. The y-axis represents the neuropathy score as measured with UENS and the x-axis represents the plasma levels of Hcy. Spearman rank correlation; r = 0,40, p = 0,022. B. The graph shows significant correlation between NIS(legs)-score and Hcy-levels in plasma in patients with L-dopa treated PD. The y-axis represents the neuropathy score as measured with NIS(legs) and the x-axis represents the plasma levels of Hcy. Spearman rank correlation; r = 0,37, p = 0,034.
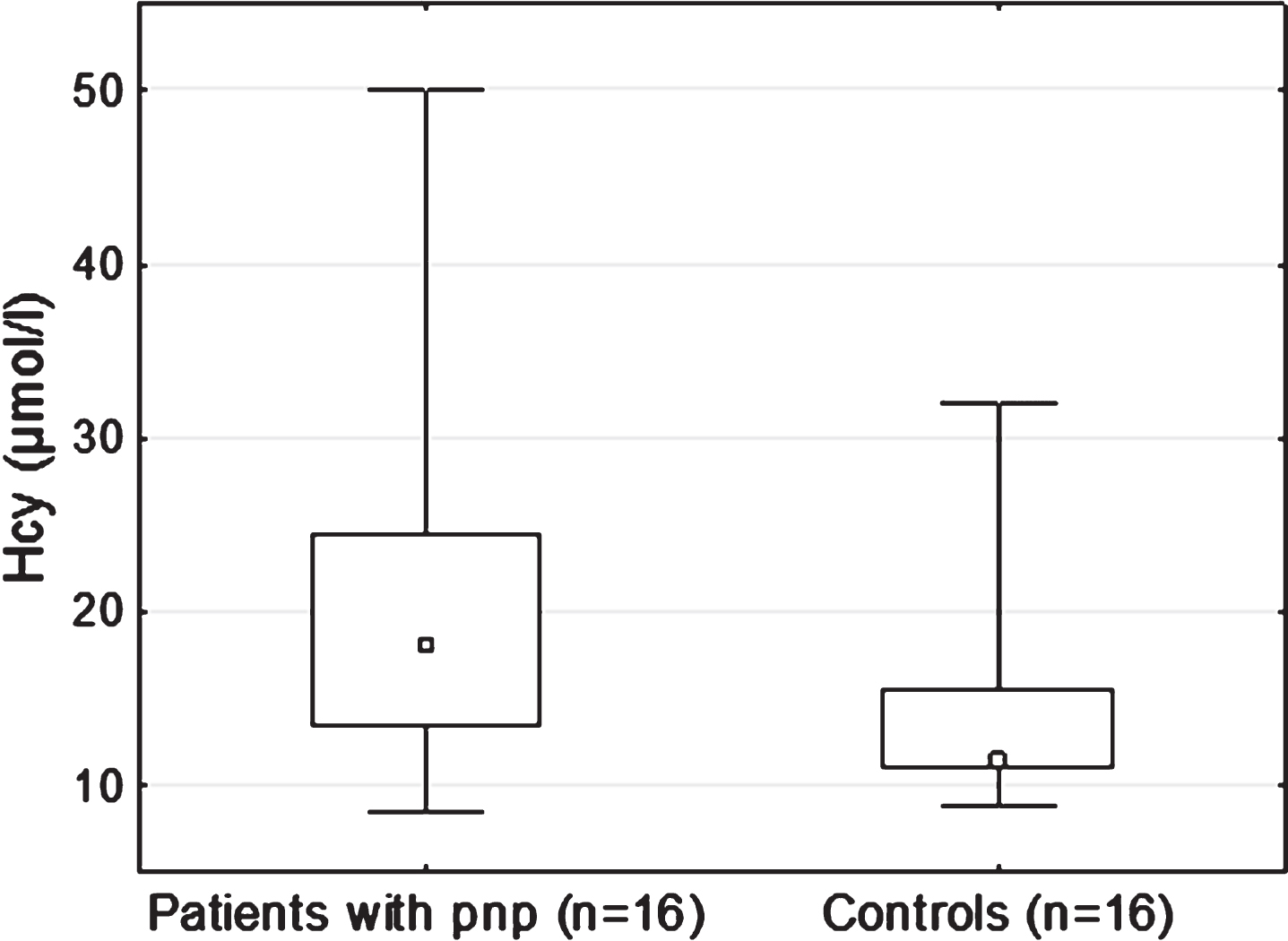
The boxplot shows significant difference in the Hcy-levels between PD patients with pnp compared to controls. The y-axis represents Hcy-levels in plasma and the x-axis categorizes subjects as controls and patients with pnp. Mann-Whitney U-test; p = 0,020.
Studying the entire patient group, no significant correlations were seen using Spearman rank correlation between age1, disease duration2 or daily L-dopa dose3 in relation to UENS (r1 = 0,27; p1 = 0,13, r2 = 0,23; p2 = 0,20, r3 = 0,17; p3 = 0,34), NIS(legs) (r1 = 0,29; p1 = 0,11, r2 = 0,25; p2 = 0,17, r3 = 0,27; p3 = 0,13), MoCA (r1 = 0,029; p1 = 0,87, r2 = –0,051; p2 = 0,78, r3 = –0,13; p3 = 0,48), Hcy- (r1 = 0,12; p1 = 0,49, r2 = 0,10; p2 = 0,56, r3 = 0,30; p3 = 0,086) or MMA-levels (r1 = –0,060; p1 = 0,74, r2 = 0,063; p2 = 0,73, r3 = 0,26; p3 = 0,14). No significant differences with regards to disease duration or daily L-dopa dose were demonstrated when comparing patients with and without pnp (Table 1). Furthermore, the total duration time of L-dopa therapy was calculated for 30/33 patients. No significant (p = 0,15) difference was found between patients without pnp (79,0±65 months) and patients with pnp (110±64 months).
A subanalysis within the patient group was performed, comparing patients with (n = 11) and without (n = 22) ongoing B vitamin supplementation. Significant increases in UENS-score (p = 0,035), Hcy- (p = 0,0025) and MMA-levels (p = 0,0011) were seen in the non-supplementation subgroup. However, the increase in NIS(legs)-score did not reach significance (Supplementary material).
No significant difference was seen between non-pnp patients and controls regarding the distribution of the 3 analyzed SNPs in COMT and MTHFR when performing the Pearson chi-square test (Table 2). However, patients with pnp demonstrated a significant difference compared to both controls and non-pnp patients in the distribution of the COMT genotype, favoring the A/A genotype (Table 2).
Genetic analysis did not demonstrate any carriers of the GBA N370S mutation in the study population.
8 patients and 3 controls exhibited either deficiency or values in the gray zone with regards to serum levels of vitamin B12. 2 patients and 1 control exhibited vitamin B9 deficiency. No significant differences were seen in the levels of vitamin B12 (p = 0,43) and B9 (p = 0,96) between patients and controls.
DISCUSSION
The present study demonstrates a 48% prevalence of pnp (defined as UENS > 4p) in a L-dopa treated PD patient population followed at an outpatient clinic at a University Hospital. The pnp prevalence in the PD patients is markedly higher compared to the age-matched controls. The result is in line with previous studies that have shown a prevalence of pnp in PD of 38% [30] and 55% [2]. The UENS-score was significantly higher in the patient group compared to controls and correlated significantly with Hcy-levels for patients. Furthermore, Hcy-levels were significantly higher in patients with pnp compared to controls. These findings are in line with earlier studies, supporting a possible relationship between the development of pnp and a disturbed methionine cycle metabolism in PD [2–6].
Different hypotheses have been proposed to the pathogenesis of pnp in PD. Previous studies have, among other mechanisms, discussed underlying neurotoxic effects of L-dopa, MMA and Hcy as well as an underlying L-dopa-induced vitamin B12- or B9-deficiency [2, 34–36]. The explorative findings in this study, of higher Hcy-levels in patients with pnp compared to controls and significant correlation between UENS-score and Hcy-levels in the patient group, might give some support for Hcy as a possible marker for pnp in L-dopa treated PD. However, the study sample is small and these findings have to be confirmed in larger studies.
Reviewing the methionine cycle, elevated Hcy-levels can be regarded as a reflection of an impaired capacity to recycle Hcy back to methionine (Fig. 1). Methionine is needed for the production of SAM which is essential for important cellular methylation reactions. L-dopa exposure is expected to increase the demand for SAM, because of increased COMT-mediated methylation, and thus subsequently increase the levels of Hcy. In this context, supplementation with vitamin B9 and B12 could serve to uphold methylation capacity through SAM, by ensuring a continuous recycling of Hcy back to methionine. By which mechanisms impaired methylation capacity potentially could contribute to the development of pnp is yet to be better understood.
The COMT low activity genotype (A/A) has previously, in a Finnish prospective study, been associated with an increased risk of acute coronary events, possibly by means of interaction with elevated Hcy [37]. In the present study, we could see a significant difference in the distribution of the COMT genotype in pnp patients compared to both non-pnp patients and controls. Indeed, 50% of the pnp patients exhibited the A/A genotype compared to 18% of the non-pnp patients. Even though the study sample is small, this was still a surprising finding considering the conflicting data regarding COMT-inhibition in the context of pnp and methionine cycle metabolism in PD. There are possible roles of COMT in this context. S-adenosyl-homocysteine (SAH) is an intermediate in the methionine cycle and also acts as a noncompetitive inhibitor of COMT (Fig. 1). Consequently, high levels of Hcy can lead to an increased inhibition of COMT through the effects of SAH. COMT has a physiological function by means of COMT-mediated methylation metabolism of endogenous catechols, and impaired function has been thought to result in cytotoxic effects [11]. Entacapone mainly acts outside the blood-brain-barrier, whereas the COMT genotype affects cellular COMT function throughout the body. Thus, given a setting with high Hcy-levels, one could expect that the SAH-mediated reduction of COMT activity would be relatively more pronounced in patients carrying the low activity genotype since these patients have a constitutionally lower enzyme activity from start. In light of these thoughts, we speculate on whether patients with the low activity genotype could be more vulnerable to an elevation of Hcy and consequently, be at higher risk of developing an impaired COMT-mediated methylation in peripheral neurons that could contribute to pnp. These are speculations, driven by the results from this small study that must be confirmed and further explored in larger studies. Of note, we could not demonstrate a protective effect of entacapone in this study.
In the present study, a subgroup analysis was performed examining the possible impact of B vitamin supplementation in the context of developing pnp. Patients not receiving supplementation had significantly higher UENS-scores than supplemented patients. However, the study sample in this subgroup analysis was small and the definition of supplementation encompassed varying combinations of vitamin B12 and B9 why no certain conclusions can be made (Supplementary material).
No significant differences in the distribution of the two analyzed MTHFR SNPs were seen between patients with pnp and non-pnp patients. Many patients received B vitamin supplementation, which together with the small study sample could contribute to the absence of a possibly different genotype distribution in the patients with pnp. Furthermore, this study could not demonstrate an association between the two studied GBA mutations and pnp in PD.
The main limitation of this study is the study definition of pnp. Only using clinical scales could both over- and underestimate the true prevalence of pnp. UENS was originally established to specifically detect early sensory predominant pnp, including signs of small-fiber neuropathy [28]. Based on previous phenotypical descriptions of polyneuropathy in PD, we deemed this scale suitable since we expect to see a length dependent sensory predominant pnp in PD. Furthermore, some evidence have pointed to the existence of a small-fiber neuropathy in PD [7]. UENS might therefore be even more sensitive than electrophysiology, which solely measures the function of large fibers. Moreover, we used the same predetermined cut-off value in UENS as used in a previous study by Rajabally and co-workers, also based on a PD population [30]. In this study, the 14 PD patients that had a positive UENS-score also underwent electrophysiological evaluation. Of these 14 patients, 9 had electrophysiological evidence of an axonal process and 5 had normal electrophysiology. The calculated prevalence in our study (48%), is in between the 38% prevalence reported by the Rajabally study [30] and another study by Toth and co-workers [2] that has also used electrophysiology besides clinical assessment for defining polyneuropathy and reported a 55% prevalence.
In the present study, we employed an age interval (55–70 years) as inclusion criteria. This was mainly used in order to exclude confounding factors such as age-related neuropathic findings. Furthermore, we expected a small sample size why a smaller age interval could possibly reduce the effects of outliers with regards to age. Subjects with diabetes mellitus and/or documented overconsumption of alcohol were excluded from this study. However, a systematic laboratory protocol to exclude other causes of pnp was not carried out in this study. Thus, it cannot be ruled out that other causes of pnp are prevalent in the patient group.
This study only examined patients with ongoing L-dopa treatment. Since accumulative L-dopa exposure is thought to be a possible risk factor for the development of pnp, it would have been of value to include a second patient group only treated with dopamine agonists or MAO-inhibitors. A study of this kind could potentially elucidate a contribution of the alpha-synucleinopathy itself to the development of pnp.
Furthermore, this study could not accurately provide an optimal measurement of accumulative L-dopa exposure. This was mainly due to lack of necessary dosage information in the journals from the patients’ entire disease duration. Many patients had been treated at different hospitals and clinics at the time of diagnosis why a confident estimation of previous therapies, including dosages, could not be carried out. We cannot rule out that a better estimate of the total accumulated L-dopa exposure would have been able to show significant correlations with regards to the development of pnp.
The study population was matched to age, but not to sex. Since it is known Hcy-levels are higher among men, it cannot be ruled out that this together with a smaller control group could have caused potentially false correlations and differences between the study groups. However, no significant differences within the patient or control group were demonstrated between levels of MMA, Hcy, MoCA, UENS and NIS(legs) with regards to sex.
In conclusion, this study proposes elevated levels of Hcy to be associated with pnp in L-dopa treated PD, possibly reflecting an underlying impaired cellular methylation capacity which may be implicated in the development of pnp. L-dopa is proposed as exerting a strain on the methionine cycle, resulting in an increased demand for vitamin B9 and B12 in order to maintain methylation capacity through SAM. However, the study population is small and adjustments for multiple confounders have not been performed why the results should be seen as preliminary with need of confirmation in larger studies.
Furthermore, we provide some novel evidence on the role of COMT and whether the low activity genotype could predispose the development of pnp in L-dopa treated PD through interaction with Hcy. Adequate supplementation therapy with vitamin B9 and B12 in L-dopa treated PD patients with elevated Hcy might play a role in preventing the development of pnp, however larger prospective treatment studies are needed to provide further support for this notion.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank the study subjects of this project. Funding was obtained from the ALF program at the Stockholm City Council. PS is a Wallenberg Clinical Scholar.
