Abstract
Abnormal sleep may associate with cognitive decline in Parkinson’s disease (PD). Furthermore, sleep dysfunction may associate with worse motor outcome. We hypothesised that PD patients with poor quality sleep would have greater progression in gait dysfunction, due to structural and functional overlap in networks subserving sleep and gait regulation. 12 PD patients and 12 age-matched controls completed longitudinal follow-up over 36 months. Poor sleep efficiency and greater sleep fragmentation correlated significantly with progression of step-width variability, a gait characteristic mediated by postural control, providing evidence that poor sleep in PD is associated with a more rapid deterioration in gait.
Sleep dysfunction is an integral component of Parkinson’s disease (PD), affecting 42–98% of patients [1, 2]. Rapid Eye Movement sleep behaviour disorder (RBD) is the prototypical sleep disorder associated with PD, but fragmented sleep in PD is multifactorial with a multitude of primary and secondary sleep disorders represented. Converging evidence from animal models of sleep deprivation and prospective human studies implicate poor sleep as a harbinger of cognitive decline [3]. Furthermore, changes in sleep architecture in early PD may predict progression to PD dementia [4]. The pathophysiological processes underpinning sleep dysfunction in PD are debated, but probably relate to a predilection of α-synuclein pathology for certain brainstem structures. The pedunculopontine nucleus (PPN), in particular, is pivotal in the regulation of the normal sleep-wake cycle; PPN cholinergic neurons display selective vulnerability in PD [5]. The PPN is also implicated in postural control and has attracted interest recently as a deep brain stimulation target for non-dopa-responsive axial symptoms in PD. Although numbers studied are small, it is notable that significant improvements in both daytime somnolence and nocturnal sleep stability have been described [6].
Additionally, data from large, prospective population studies demonstrate a link between objectively-recorded poor quality sleep and poor motor outcomes [7]. In normal older individuals, decreased gait velocity and increased gait variability, particularly in the context of dual-task walking conditions, are associated with poor sleep quality [8]. RBD, often a syndromic prelude to PD, is associated with subtle gait changes, particularly gait variability [9]. Secondary analysis of gait differences between our early PD cohort and age-matched controls [10] showed that step width variability was the most discriminatory gait measure in early PD (more so than gait speed and the variability of step length or time) [11].
We hypothesised that early sleep dysfunction in PD would predict more rapid deterioration in gait and sought to explore this in a well-characterised, incident PD cohort.
Participants were recruited from the Incidence of Cognitive Impairment in Cohorts with Longitudinal Evaluation- PD (ICICLE-PD) study; recruitment protocols and detailed methodology have been described previously [12]. The collaborative ICICLE-GAIT study recruited contemporaneously from within the same cohort. Briefly, newly diagnosed patients with idiopathic PD were recruited; those with significant memory impairment (Mini-Mental State Examination (MMSE) score <24), or meeting Movement Disorders Society (MDS) criteria for dementia, were excluded. Healthy, age-matched controls were recruited through community sources. The studies were approved by the local Research Ethics Committee; participants provided written informed consent. Participants who completed all elements of the study underwent ICICLE-PD assessments at baseline and 36-month follow-up visits.
This comprised medical assessment by a physician, recording demographics, educational exposure and medication use (including hypnotics). Motor phenotype was classified, and disease severity was rated by Hoehn & Yahr stage. Motor disability was assessed by the MDS Revised Unified Parkinson’s Disease Rating Scale (MDS-UPDRS). Levodopa-equivalent dose (LED) was also calculated. MMSE and Montreal Cognitive Assessment (MoCA) were used to assess global cognitive function [13]. Cognitive sub-domains were assessed by previously described methods [14]. Depression was rated with the Geriatric Depression Scale-15 (GDS-15).
Sleep was assessed at baseline, as described previously [15]. Patients participated in a semi-structured clinical interview with a specialist sleep physician, in addition to completing self-rated questionnaires (the Epworth Sleepiness Score and the Pittsburgh Sleep Quality Index). Patients and, where possible, bed partners completed an attenuated version of the Mayo Sleep Questionnaire, validated for probable RBD. Patients underwent one night of sleep respiratory home monitoring (Embletta) and three nights of lower limb actigraphy (CamNtech, Cambridge, UK) to measure periodic limb movements of sleep (PLMS). A cohort of participants then completed two weeks of wrist actigraphy using the same monitors, keeping contemporaneous sleep diaries.
Gait was assessed at baseline and 36 months, using single-task and dual-task paradigms, as described previously [10]. Briefly, assessments were performed using a 7-m-long x 0.6-m-wide instrumented walkway (GAITRite, CIR Systems, USA). Participants walked at their preferred pace for two minutes around a 25-m circuit incorporating the walkway. PD participants were tested approximately one hour after medication intake. For dual-task assessment, each participant completed the same circuit whilst repeating a string of numbers, the length of which was set at their seated maximum forward digit span. Gait characteristics were selected in keeping with a previously published model of gait validated in PD [16]. Variability characteristics were calculated using the within-person standard deviation (SD); asymmetry was calculated as the absolute difference between the mean of left and right steps.
Actigraphic data was computed by customised software (CamNtech, UK). For lower limbs, this produced a periodic limb movement index (PLMI); for wrist actigraphy this generated an estimate of total sleep time, sleep efficiency (SE) and a sleep fragmentation index (SFI). Pearson and Spearman correlations (and subsequent partial correlations) were performed using SPSS (v.22, IBM).
Twelve PD patients and twelve control participants completed baseline and 36 month assessments. Relevant clinical and demographic data are presented in Table 1. One PD participant took a hypnotic agent PRN during the two week period of wrist actigraphy. One PD participant had moderately severe obstructive sleep apnoea with an apnoea-hypopnoea index of 15.0/hr; five PD participants had PLMI >5.0/hr, of which four were >15.0/hr –two of these patients satisfied diagnostic criteria for Restless Legs Syndrome (RLS) in wakefulness. Three patients had probable RBD.
Participant characteristics and gait performance as baseline and change in gait
*Signifies difference between PD and control groups p < 0.05. MoCA, Montreal Cognitive Assessment; GDS, Geriatric Depression Scale; MDS-UPDRS, Unified Parkinson’s Disease Rating Scale; Results expressed as mean (SD).
There were no significant correlations at baseline between sleep parameters and gait characteristics. However, unfavourable baseline sleep indices correlated with greater change at 36 month follow-up in gait parameters associated with postural control (Table 1; Fig. 1). Poorer SE was associated with greater increase in step width in controls (Pearson’s r = 0.630; p < 0.05), and greater step width variability in PD (Pearson’s r = 0.723; p < 0.01). These associations were also evident under dual task conditions. Higher levels of sleep fragmentation were associated with an increased single-task step width over the 36 months in PD only (Pearson’s r = 0.588; p < 0.05) as well as increased dual-task step width variability in PD (Pearson’s r = 0.844; p = 0.001). Cohen’s d effect size was 0.95 for the group difference in change of step width variability over 36 months, for both single- and dual-task step width variability.
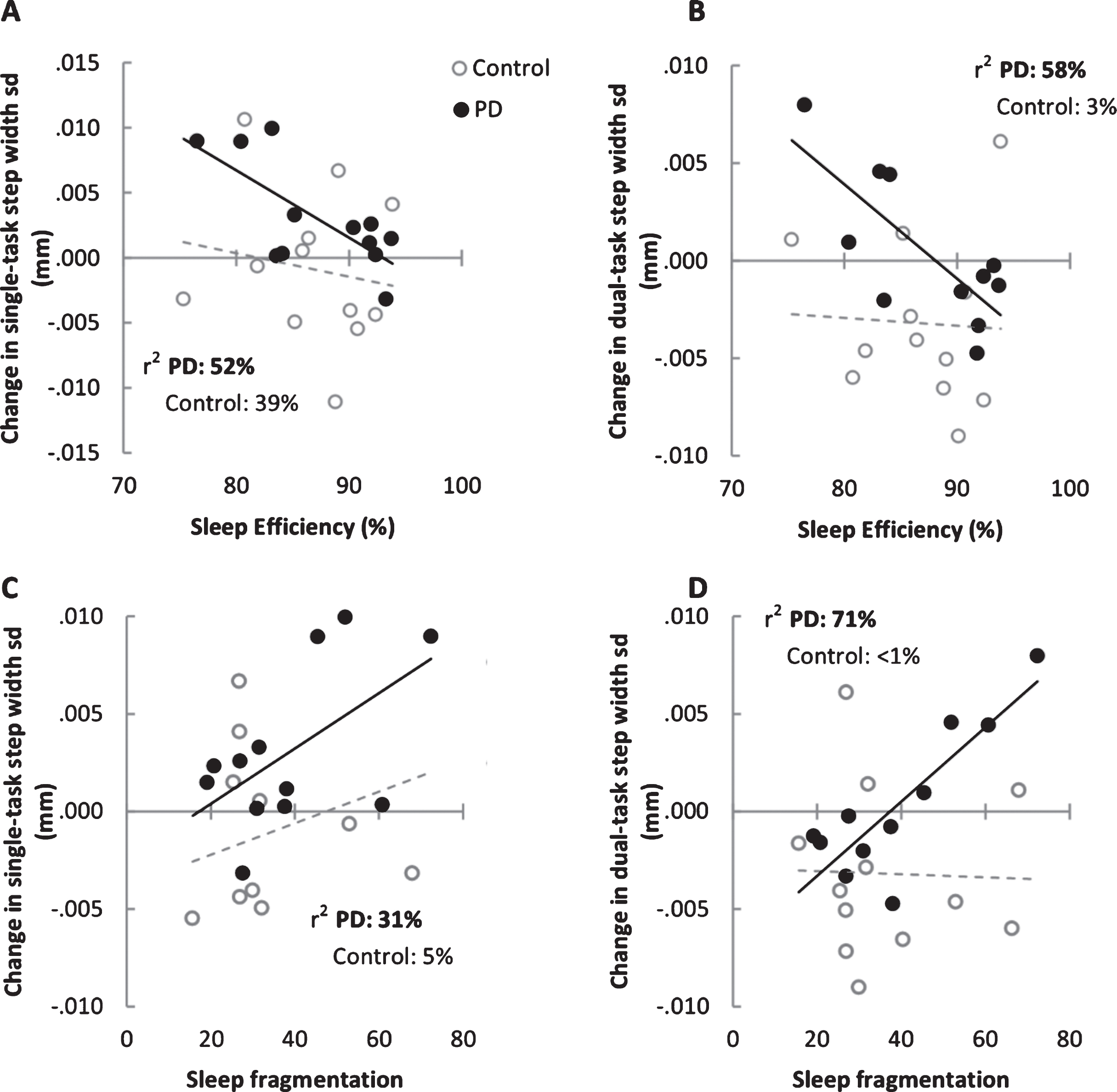
Scatterplots between baseline sleep characteristics and change in single-task (Panels A and C) and dual-task (Panels B and D) step width variability over 36 months (36 month assessment – baseline assessment). Lines of best fit are indicated by solid black lines for PD participants and the dashed lines for controls.
Partial correlations confirmed that increased step width variability in PD participants with impaired sleep (poorer SE, r2 = 0.576 and higher SFI, r2 = 0.712) was independent of global cognition, as assessed by MOCA (see supplementary data); it was also independent of measures of attention. There was no relationship between either baseline sleep efficiency or sleep fragmentation index with evolution in LED or motor UPDRS scores (36 month mean +/–SD, 598.4 +/–275.5; 40.6 +/–10.0, respectively). There was no correlation between subjective sleep quality scores (ESS, PSQI) and gait characteristics; data derived from patient diaries did not reveal a relationship between extent of daytime napping and progression in gait disturbance.
Sleep quality, previously a “Cinderella” topic in neurodegenerative research, is attracting increasing attention as a window to the underlying pathophysiological cascade. The mechanisms regulating sleep quality are complex, with numerous brain regions implicated. However, the close anatomical and functional proximity between sleep and gait control centres makes the hypothesis that dysfunction in one domain will lead to abnormalities in the other an attractive one.
Our study highlights an association in an incident PD cohort between poor sleep and more rapid deterioration of gait-related postural control. We found a relationship between both poor sleep efficiency and higher indices of sleep fragmentation and greater step width variability in PD participants. People with early PD can present with a less variable step width, possibly reflecting an inability to adapt to dynamic locomotor challenges [10]. However, excessive step width variability may manifest with disease progression and has been associated with balance impairment [17] and higher risk of falls [18] in elderly community-dwelling individuals, and a trend toward greater fall risk in PD [19]. Whilst aberrant sleep patterns may be associated with broadscale effects over time, in that some adverse associations were also seen in our control group, it is conceivable that the deleterious impact of poor quality sleep is magnified in the setting of a disease affecting gait, particularly aspects mediated by postural control, hence the more compelling results in the PD group. It may be that patients with PD exhibit greater vulnerability to the ageing process. Furthermore, the association continues to hold after controlling for cognition, raising the tantalising prospect that the observation in this small, exploratory study may unveil a hitherto under-recognised link between sleep and gait in PD. Recent data from another group interrogating the functional neuroanatomy of pedunculopontine nucleus networks [20] have also suggested a link between disturbed sleep and postural control impairments in PD.
We acknowledge the small participant number involved as a limitation of this study. This reflects the proportion of subjects from the larger ICICLE and ICICLE-GAIT studies who completed all components of each assessment over time, and as such represents a small, but deeply phenotyped nested cohort. The small sample size precludes use of more definitive statistical analysis; nonetheless the association between sleep and gait characteristics related to postural control was robust. Furthermore, the consistency of the association across single- and dual-task protocols makes the possibility of Type I error less likely. Employing wrist actigraphy rather than full polysomnography may be considered a limitation, but wrist actigraphy is an established marker of sleep fragmentation in PD; furthermore in our study, sleep-disordered breathing and PLMS were sought and quantified.
Sleep disturbance in PD has multiple causes including sleep-disordered breathing, RBD, RLS and asymptomatic PLMS. To our knowledge, this is the first study to address a relationship between sleep indices and gait outcomes in an incident PD cohort. The suggestion that poor sleep, which is frequently remediable, may be associated with unfavourable motor progression warrants greater exploration in larger prospective studies.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was funded by grants from Parkinson’s UK (J-0802) and Lockhart Parkinson’s Disease Research Fund. ICICLE-GAIT is supported by the National Institute for Health Research (NIHR) Newcastle Biomedical Research Unit based at Newcastle upon Tyne Hospitals NHS Foundation Trust and Newcastle University.
The authors have no other financial disclosures or conflicts of interest relating directly to this work.
