Abstract
Keywords
INTRODUCTION
Camptocormia in Parkinson’s disease (PD) is the disturbance of posture involving involuntary truncal flexion induced by standing or sitting [1]. Camptocormia is a well-described clinical feature of PD and other parkinsonian syndromes; however, the mechanism of its pathogenesis remains unclear [2]. Moreover, effective treatment options for camptocormia are limited and frequently futile. Conservative therapies include physiotherapy, the injection of botulinum toxin, and the discontinuation of causative drugs [2–5]. Invasive therapeutic measures include local surgical approaches or deep brain stimulation (DBS). Previous reports have shown that DBS of the subthalamic nucleus (STN) or globus pallidus internus is effective in treating camptocormia [3, 6–9]. However, the efficacy of DBS for camptocormia varies and besides symptom duration there is currently no predictive marker for an improvement of the bending angle after DBS.
Recent studies including our own have concluded that camptocormia in PD is predominantly myopathic [10–13]. Therefore, we investigated the atrophy and degeneration of paraspinal muscles in patients with PD and concomitant camptocormia (PDCC) before DBS.
MATERIALS AND METHODS
Standard protocol approvals, registrations, and patient consents
All patients provided written informed consent in accordance with the approved protocol granted by the ethics committee of the Nagasaki Kawatana Medical Center (Nagasaki, Japan).
Patients and clinical evaluations
A diagnosis of PD was made according to the UK Brain Bank Criteria. We retrospectively reviewed the clinical course of 14 consecutive patients who underwent bilateral subthalamic nucleus deep brain stimulation (STN-DBS) and suffered from significant camptocormia between January 2009 and December 2013 [14]. The clinical characteristics of the 14 cases of PDCC are summarized in Table 1. We measured the thoraco-lumbar angle (TLA) and defined camptocormia as marked anterior flexion at least 30° of the thoracolumbar spine, with or without additional laterodeviation. The flexion needed to appear in the standing or walking position and disappear in the recumbent position. No signs of fixed kyphosis should be present (e.g., as in osteoporotic kyphosis). We excluded all patients with scoliosis. Using photographs, we evaluated camptocormia by measuring the thoracolumbar angle between the long axis of the femur and the upper thoracic plane preoperatively, 6 months after surgery, and at the time of the last follow-up in statuson-medication.
Clinical features of 14 patients with Parkinson’s disease and concomitant camptocormia
STN-DBS = subthalamic nucleus deep brain stimulation, UPDRS = Unified Parkinson’s Disease Rating Scale, pre = preoperative state, post = postoperative state, LEDD = levodopa equivalent daily dose, M = male, F = female.
We assessed age at onset, sex, PD duration, age at the time of operation, duration of camptocormia until surgery, and the levodopa equivalent daily dose (LEDD; mg/day). The following conversions were applied to calculate the LEDD: ropinirole X 20: pergolide, pramipexole, and cabergoline X 100; levodopa with decarboxylase inhibitor X 1; levodopa with decarboxylase and COMT inhibitor X 1.3 [15]. PD symptoms were assessed using the Unified Parkinson’s Disease Rating Scale (UPDRS). Axial symptoms were assessed using subscores of seven items from the UPDRS part III. An axial score was modified and calculated according to Bloch et al.’s method for combining items 18 (speech), 19 (facial expression), 22 (rigidity), 29 (gait), and 30 (postural instability) [16]. Preoperative levodopa responsiveness was determined by UPDRS part III including axial score when the patient was in the off-medication and on-medication states (the responding rate was calculated). The score after a drug-free period exceeding 12 h was defined as the practical medication-off state and the score at 1–2 h after the administration of the usual morning medications as the practical medication-on state. Assessments were performed two weeks before and 6 months after surgery by one independent observer from ourdepartment.
Bilateral STN-DBS
Prior written informed consent of the patients and their families was obtained. All patients underwent bilateral STN-DBS. We used a Leksell Stereotactic System (Elekta, Stockholm, Sweden) and the Frame Link planning system (Medtronic, Minneapolis, Minnesota, USA) [17]. The tentative STN target was 4 mm posterior, 4 mm inferior, and 12 mm lateral to the mid-commissural point on the preoperative magnetic resonance imaging (MRI) console. Microelectrode recording and test stimulation studies were performed. The trajectory that included more than four positive recording sites (4.0 mm) was chosen for placement of the DBS electrode (model 3389, Medtronic). The accuracy of electrode placement in the STN was confirmed by postoperative computed tomography. All patients underwent bilateral procedures in a single operative session. Implantable pulse generators (Soletra or Activa SC, Medtronic) were simultaneously implanted subcutaneously in the subclavian regions.
All patients were treated with unipolar stimulation using two contacts. The parameters were: frequency, 130–160 Hz; pulse width, 60–120 μsec, on both sides; stimulation amplitude,1.5–3.6 V.
The measurements of paraspinal muscles
MRIs of the thoracolumbar spine were performed three to six months prior to the operation to assess abnormalities of the paravertebral muscles and spine in that area. Axial T2 weighted image MRI scans were performed at four different spinal levels (L4 upper, L4 lower, intermediate L4/L5, and L5 upper). Measurements of the cross-sectional area (CSA) were determined by outlining the facial boundary of the bilateral paraspinal muscles (the psoas major, erector spinae [Es], multifidus [Mtx], and sum of Es and Mtx) and the vertebrae with Image J (National Institutes of Health, Bethesda, MD, USA) (Fig. 1a). The average of the right and left paraspinal muscles was computed at each aforementioned CSA. The ratio between the average CSA of each paraspinal muscle and the vertebrae of each level was expressed as the CSA ratio. Additionally, we measured the bilateral vertical width of Es and the minor axis of the vertebrae using Image J (Fig. 1b). The average vertical width of the right and left Es was computed. The distance ratio between the average width of Es and the vertebral minor axis was calculated.
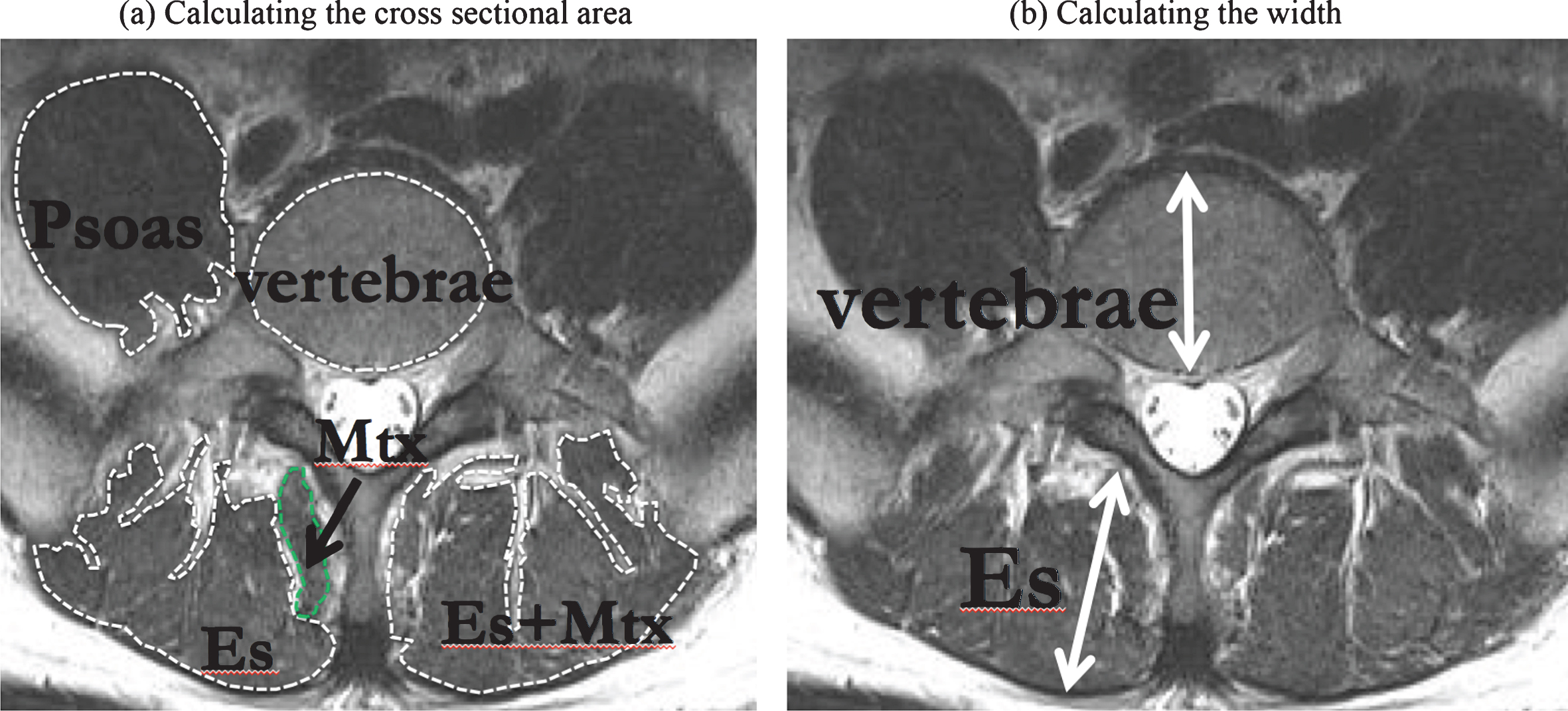
The measurements of paraspinal muscles. (a) Example trace used for calculating the cross-sectional area of the psoas major, erector spinae (ES, combined iliocostalis and longissimus), lumbar multifidus (Mtx) muscles, and vertebrae on an L4 axial magnetic resonance (MR) image. (b) Example of a region of interest (the straight-line is used for calculating the width of the vertebrae bone) showing the Es muscle from an L4 axial MR image.
Classification of the therapeutic response
Patients with PDCC were classified into three categories dependent on the response of their camptocormia to bilateral STN-DBS: a) effective (EF) = TLA became <30° and lasted six months after STN-DBS; b) partially effective (PE) = TLA decreased but persisted >30° with or without lasting six months after STN-DBS; and c) non-effective (NE) = no change in TLA after STN-DBS.
Statistical analysis
Commercially available statistics software (SigmaPlot®; SPSS, Inc., Chicago, IL, USA) was used to perform data analysis. Data were analyzed using a one-way analysis of variance (ANOVA) on ranks with three groups (EF group, PF group, and NF group). To quantify the relationship between the disease duration and thoracolumbar angle, imaging parameters (each CSA, and width of Es), and therapeutic response of camptocormia (i.e. EF, PF, and NE), the correlation analysis was used. Statistical significance was set to P < 0.05.
RESULTS
We consecutively enrolled 14 patients with PDCC (9 men and 5 women). The average age at onset was 51.9±9.7 years and the mean disease duration of PD was 13.1±4.9 years. Trunk flexion ranged from 30 to 80° with an average of 55.5±16.5°. Following STN-DBS, four (28.6%) patients were classified as EF, five (35.7%) PE, and five (35.7%) NE.
We found that the EF, PE, and NE groups did not differ in the age of PD onset, sex, disease duration, duration of camptocormia until surgery, age at STN-DBS, UPDRS III score, UPDRS III axial subscore, average TLA, or average LEDD (Table 2). Conversely, we found the statistical significance in the postoperative levodopa responsiveness based on UPDRS III score, UPDRS III axial subscore among 3 groups in postoperative state (Table 2), and there were significant differences in the average CSA and average width of the paraspinal muscle at almost all levels (Fig. 2). There was no correlation between the disease duration and thoracolumbar angle, imaging parameters, and therapeutic response of camptocormia in correlation analysis.
Demographic features of the three groups of PDCC
PDCC = Parkinson’s disease and concomitant camptocormia, UPDRS = Unified Parkinson’s Disease Rating Scale, pre = preoperative state, post = postoperative state, LEDD = levodopa equivalent daily dose. Asterisk means statistical significance. Statistical significance was set to P < 0.05.
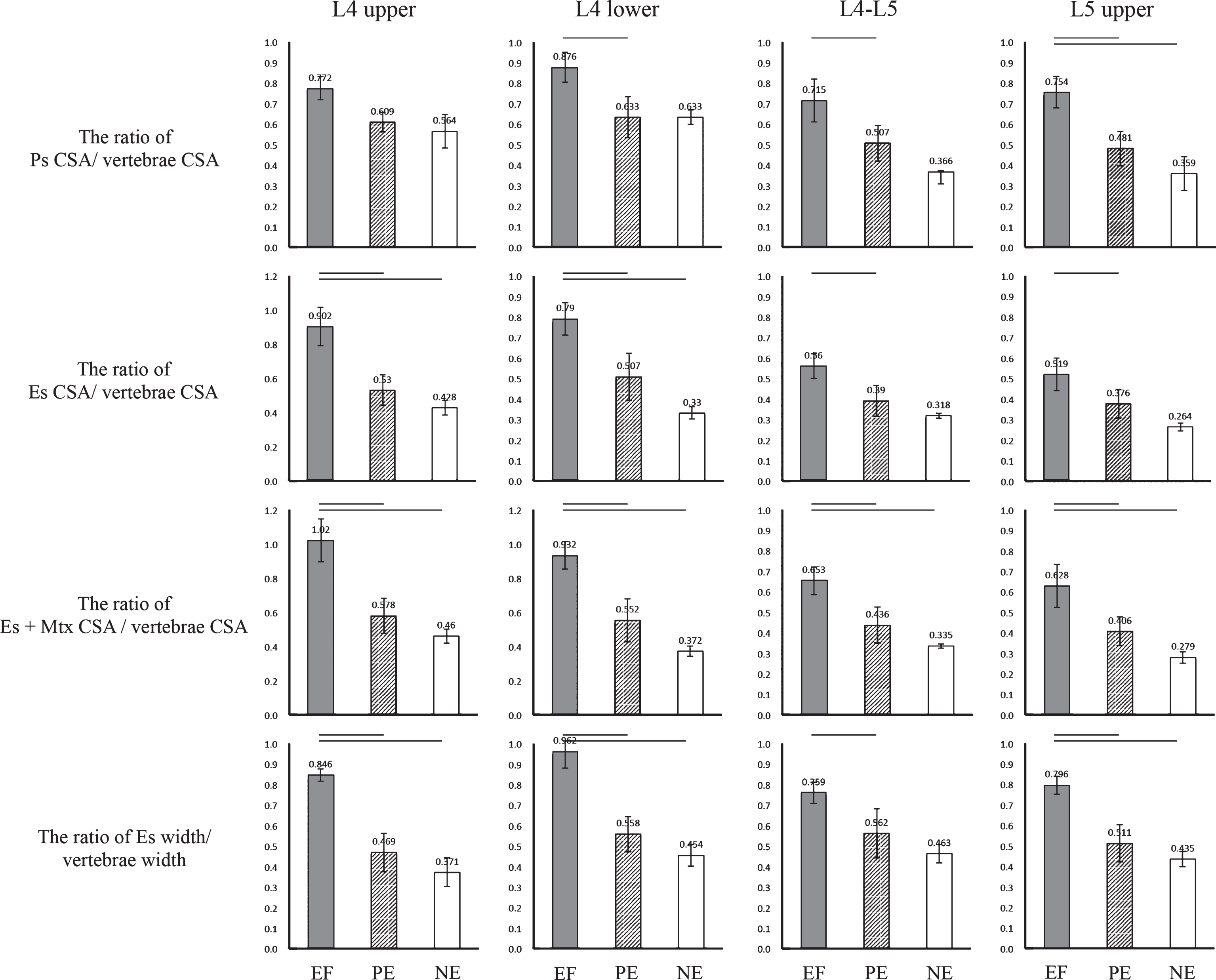
Results of the paraspinal cross-sectional area (CSA) and width. Comparison of the CSA ratio from calculating each paraspinal muscle (psoas major = Ps, erector spinae = Es, multifidus = Mtx) and the width of the Es of the four levels in the three groups [effective (EF) = black bar; partially effective (PE) = grey bar; and non-effective (NE) = white bar]. The bar denotes the level of statistical significance (P < 0.05).
DISCUSSION
The present results demonstrated that CSA was significantly larger in the EF group than in the PE and NE groups in most of the lumbar paraspinal muscles at the L4–5 levels. The width of the paraspinal muscle was markedly longer in the EF group than in the PE or NE groups. Consequently, both the truncal flexor and extensor muscles were atrophic in the NE group.
Margraf et al. recently concluded the cause of camptocormia in PD to be a focal myopathy, because their data suggested myopathic changes from an electromyogram, MRI of the paravertebral muscles, and a histopathological analysis [10, 13]. Furthermore, they demonstrated that camptocormia in PD presents with a consistent lesion pattern composed of myopathic changes with type-1 fiber hypertrophy, loss of type-2 fibers, loss of oxidative enzyme activity, and acid phosphatase reactivity among lesions [12]. In the previous study, we observed significantly more abnormal findings in muscle MRI among patients with PDCC than in those without [14]. We confirmed atrophy and/or fatty degeneration in the MRI of the paravertebral muscle, which suggests chronic and degenerative changes. Therefore, myopathy could be a pathological core feature in the mechanisms of camptocormia in PD.
Regarding the cause of myopathic changes in PDCC, Schulz-Schaeffer et al. hypothesized that camptocormia led to impaired axial tone by proprioceptive dysregulation and myopathy occurred secondarily [18, 19]. Margraf et al. suggested that camptocormia in PD has acute and chronic stages, which can be distinguished by examining MRI of the paraspinal muscle [10, 13]. They also suggested that fat degeneration in the paraspinal muscles could result from a degenerative process caused by a deregulation of proprioceptive feedback and movement. Bloch et al. suggested that camptocormia is axial dystonia caused by the degeneration of the basal ganglia [15]. Postural control is a complex system involving central mechanism, especially the proprioceptive sensory feedback [3, 20]. Proprioceptive disintegration affects axial motor control, and the abnormal proprioception can occur in axial musculature in PD [12, 20]. We hypothesize that camptocormia develops with proprioceptive dysregulation as its first step, then changes in edematous and/or swelling in the paraspinal muscle develop in the second step, and fatty degeneration occurs in the final and chronic stage of the condition [3]. The duration of each stage likely differs by individual, which might explain why the efficacy of STN-DBS for camptocormia varies among patients with PDCC. STN-DBS probably improves bending symptoms only when proprioception is restored. In cases where declining paraspinal muscle volume is the result of secondary changes, STN-DBS would no longer be the best therapeutic choice for lacking muscle strength to maintain posture.
Although Margraf et al. did not find a significant correlation between the duration of PD and radiological variables of the paraspinal muscle on MRI, they found a strong positive correlation between fatty degeneration of the paraspinal muscle and the duration of camptocormia [13]. And several groups indicated that the prognostic factor for a beneficial DBS effect on camptocormia is its short duration [18, 21]. However, in the present study, there was no correlation between CSA of paraspinal muscles, therapeutic response of camptocormia and disease duration. Particularly, we found no statistical difference in the duration of camptocormia among 3 groups. This may be because the duration of the proprioceptive dysregulation stage varied among patients. Further, this may explain why we sometimes find that STN-DBS dramatically improves severe bending symptoms even among those who have been suffering with camptocormia for more than two decades. The measurement of size of paraspinal muscles in camptocormia may address muscle degeneration more precisely than the duration of camptocormia. Moreover, our findings suggested that the improvement of PDCC treated with STN-DBS paralleled postoperative levodopa responsiveness and motor symptoms.
Some limitations existed in this study. The limitations of this study were the retrospective design, the small number of subjects, and simple comparison analysis of 3 groups, i.e. the groups of EF, PE, and NE. A prospective, multi-center, and clinical observation is necessary to confirm the relationships between the severity of PD, camptocormia and the imaging parameters. The important limitation exists in the present study. We were supposed to perform the quantitative assessment of fatty degeneration, swelling, and edema in paravertebral muscle MRI. However, we could not evaluate different variables of body composition (e.g. skeletal muscle to adipose tissue ratio) in detail, which might be a confounder. Therefore, there is the possibility EF, PE, and NE group status reflected the impact of muscle degeneration on muscle mass.
To our knowledge, this is the first report that takes particular note of the relationship between myopathic changes on MRI and the efficacy of DBS for camptocormia in patients with PD. We conclude that the measurement of paraspinal muscle CSA and width is an easy way to predict the efficacy of STN-DBS therapy for camptocormia among patients with PD.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
DISCLOSURE OF SOURCES OF SUPPORT
This work was supported by a grant from a collaborative research initiative at the National Hospital Organization Network, Japan.
All patients provided written informed consent in accordance with the approved protocol granted by the ethics committee of the Nagasaki Kawatana Medical Center (Nagasaki, Japan).
Footnotes
ACKNOWLEDGMENTS
This work was supported by a grant from the collaborative research in the National Hospital Organization Network, Japan.
