Abstract
Background:
Facial weakness is a key feature of facioscapulohumeral muscular dystrophy (FSHD) and may lead to altered facial expression and subsequent psychosocial impairment. There is no cure and supportive treatments focus on optimizing physical fitness and compensation of functional disabilities.
Objective:
We hypothesize that symptomatic treatment options and psychosocial interventions for other neurological diseases with altered facial expression could be applicable to FSHD. Therefore, the aim of this review is to collect symptomatic treatment approaches that target facial muscle function and psychosocial interventions in various neurological diseases with altered facial expression in order to discuss the applicability to FSHD.
Methods:
A systematic search was performed. Selected studies had to include FSHD, Bell’s palsy, Moebius syndrome, myotonic dystrophy type 1, or Parkinson’s disease and treatment options which target altered facial expression. Data was extracted for study and patients’ characteristics, outcome assessment tools, treatment, outcome of facial expression and or psychosocial functioning.
Results:
Forty studies met the inclusion criteria, of which only three studies included FSHD patients exclusively. Most, twenty-one, studies were performed in patients with Bell’s palsy. Studies included twelve different therapy categories and results were assessed with different outcomes measures.
Conclusions:
Five therapy categories were considered applicable to FSHD: training of (non-verbal) communication compensation strategies, speech training, physical therapy, conference attendance, and smile restoration surgery. Further research is needed to establish the effect of these therapies in FSHD. We recommend to include outcome measures in these studies that cover at least cosmetic, functional, communication, and quality of life domains.
Keywords
INTRODUCTION
Facioscapulohumeral muscular dystrophy (FSHD) is a slowly progressive neuromuscular disorder. Asymmetrical weakness of muscles in the face, shoulders, and upper arms are often the first symptoms. Later in the disease course, lower limb and trunk muscles become affected [1, 2]. It is the second most common inherited muscular dystrophy in adulthood, with an estimated prevalence of 1 per 8,000 to 1 per 20,000 [3, 4]. FSHD type 1 (95% of all patients [5]) is caused by contraction of D4Z4 microsatellite repeats on chromosome 4. This contraction leads to expression of a normally repressed DUX4 gene in skeletal muscles [6]. Expression of DUX4 induces the expression of other genes, leading to apoptosis, oxidative stress, and activation of the immune system. Altogether these factors lead to muscle dystrophy and weakness [7].
Facial weakness is an early symptom [8] and occurs in approximately 75% of FSHD patients [9]. Three facial muscles are mostly affected: 1) zygomaticus major muscle weakness causes difficulties in raising the corners of the mouth and can lead to an altered smile; 2) orbicularis oris muscle weakness can cause asymmetry of the mouth in resting position, which is more visible when blowing the cheeks; and 3) orbicularis oculi muscle weakness leads to difficulties in closing the eyelids [1, 2]. Taken together, this can contribute to an altered facial expression or even an expressionless face [1]. Figure 1 illustrates the reduced facial expressions in a young female with FSHD.

Facial expressions of a FSHD patient with facial weakness. These pictures are an example of facial expressions of a 27-year old FSHD patient with facial weakness in a neutral situation and during three primary emotions: happy, sadness and anger. Permission for usage of this pictures was granted by the patient.
Facial expressions are crucial in everyday communication, influencing first impressions [10], inferring emotions or personality characteristics [11], and raising empathy or involvement in others [12]. Facial expressions also elicit emotional mimicry [13], which serves as the base for smooth communication. Studies have shown that people in different cultures around the world can recognize emotions on the basis of emotional display in facial expressions [14]. Several studies have further shown that people with less or no facial expression are impaired in social interactions, because their emotions are less recognized and less mimicked. Patients with Bell’s palsy are known to experience more psychological distress [15] and have higher depression rates [16, 17], compared with people without facial weakness. Parkinson’s disease patients with facial masking are often judged by others as less attractive, supportive, social positive and more introverted, this is more pronounced in patients with more severe facial masking [18–22].
The initiative of this review arose from the observation of psychosocial distress in patients with FSHD with an altered facial expression in our recent studies [23–25]. In addition it is our clinical experience in the neuromuscular clinic that communication of FSHD patients is somewhat different compared to other patients. FSHD patients seem to be more direct, clear and redundant in their verbal communication, we hypothesized this to be the result of lack of supporting facial expression. Furthermore, an increasing number of FSHD patients at our expert centre make inquiries about available treatments. Currently, there are no curative treatments for FSHD, nor for the facial weakness it causes. Therefore, supportive treatment options will be important for reducing the negative consequences of facial weakness. We hypothesized that some supportive treatment options used in other neurological diseases with altered facial expression (Bell’s palsy, Moebius syndrome, myotonic dystrophy type 1, and Parkinson’s disease) could also be applicable to FSHD. Hence the aim of this systematic review is to provide an overview of available treatment options to improve facial expression and on therapies focussing on psychosocial consequences of having an altered facial expression in other neurological diseases with altered facial expression, and to evaluate their potential in FSHD patients.
METHODS
A systematic literature search was performed to provide an overview of literature on symptomatic treatment options in FSHD and four other neurological diseases with altered facial expression. Each disease is presented separately in the results section. Therapies were subsequently evaluated for applicability to FSHD. Therapy efficacy, pathophysiological targets, and generalisability to FSHD patients were taken into consideration during this evaluation.
Protocol
The methods of this systematic review were defined in advance and documented in a study protocol. The study protocol was registered at PROSPERO (CRD42020212524).
Type of study
Included studies were randomized controlled trials, quasi randomized controlled trials, non-randomized trials, case-control studies, and case reports. The last were included, because of the expected scarcity of published literature on this subject and to minimize the chance of missing potential treatment options for FSHD patients. Only prospective study designs were included. Conference abstracts were excluded. All studies had to be published in English or Dutch. Only studies from 1990 until June 24, 2022 were included, because we expected treatment options in older studies to be outdated for improving altered facial expression.
Participants
Studies had to include patients with one of the following neurological diseases: FSHD, Bell’s palsy, Moebius syndrome, myotonic dystrophy type 1, or Parkinson’s disease. Although the clinical features in these disorders are different, all have some degree of altered facial expression or will develop this in the course of their disease (see Fig. 2). We decided to exclude studies which included multiple causes of facial paresis. By doing so, influences of other disease mechanisms were diminished. In Moebius syndrome, patients have congenital uni- or bilateral facial and abducens nerve palsy due to cranial nerve impairments [26]. Hence, facial weakness is a distinct symptom in this disease. Furthermore, studies with patients with myotonic dystrophy type 1, also known as Steinert’s disease, were included. Studies with myotonic dystrophy type 2 were excluded, since facial weakness is not a pronounced characteristic in this subgroup [27]. Lastly, studies focussing on treatment of hypomimia in Parkinson’s disease were included. Parkinson’s disease patients with hypomimia have an altered facial expression and was therefore chosen as disease of interest.
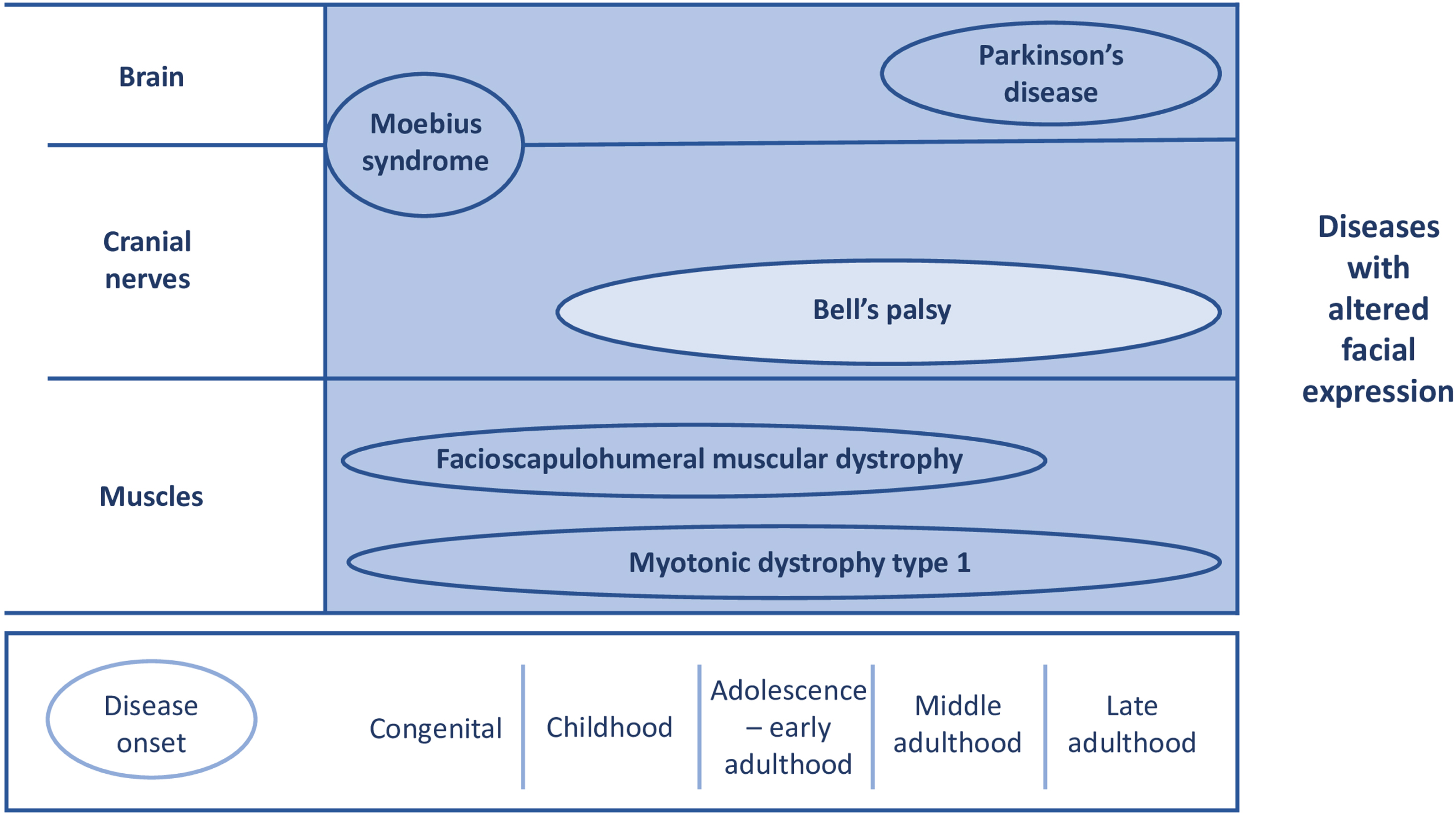
Overview for time of onset and origin of pathophysiology in the included diseases. X-axis: time of onset. Y-axis: anatomical localisation of pathophysiology.
Interventions
Studies had to focus on treatment options for altered facial expression or on improving psychosocial consequences of having an altered facial expression. Studies on treatments specifically addressing the underlying pathophysiology of other disorders than FSHD were not part of the scope of this review. For example, studies on antiviral medication or prednisolone for Bell’s palsy, specifically used in the acute stage of the disease, were excluded. Studies with these drugs could only be included when used as usual care and not as intervention. Studies on botulinum toxin treatment for synkinesis were not considered useful since they do not focus on facial weakness. Thirdly, studies with surgery or non-invasive treatments (laser therapy) involving only nerve transposition, reinnervation or nerve decompression were excluded, because these are not applicable to neuromuscular diseases in which the disease affects the muscle and not the nerve. Furthermore, studies with Parkinson’s disease medication or deep brain stimulation as intervention were excluded, because these treatment options are specific for Parkinson’s disease and not applicable to FSHD. Lastly, all studies focussing on complementary medicine (such as herbal medicine) were excluded. An exception was made for acupuncture, because this is already partly implemented in usual care around the world.
Outcomes measures
The main focus of outcome measures were the difference of facial expressions and quality of life before and after treatment of interest.
Search strategy
The following databases were searched: the Cochrane Library (1990 to June 24, 2020), Embase (1990 to June 24, 2020), and PubMed (1990 to June 24, 2020). The search was updated on June 24, 2022. The search strategy for PubMed is described in Table 1. The search strategies for the Cochrane Library and Embase are available upon request. In addition to the above-described search strategy, the references of included articles and articles citing one of the included articles, were also assessed for possible inclusion.
PubMed search strategy (1990 to June 24, 2022)
Study selection
The articles were screened independently for inclusion by two assessors (N.R. and W.G.) through consecutively title and abstract screening and full text screening. Disagreements about inclusion were discussed by the assessors and the principal investigator (N.V.) until consensus was reached. Covidence [28], a web-based screening and data extraction tool, was used for management of studies during the study selection procedure.
Quality assessment
Methodological quality was assessed using three critical appraisal tools of the Joanna Briggs Institute. To ensure that quality assessment tools were appropriate for every study, different tools were used for randomized controlled trials, quasi-experimental studies, and case reports [29, 30]. The tools consist of consecutively eight, nine and thirteen questions. Questions can be scored with “yes”, “no”, “unclear”, or “not applicable”. Every question is rewarded with one point, when the answer is “yes”. A higher total score is an indication for a lower risk of bias. Quality assessment was rated independently by two assessors (N.R. and W.G.). Disagreements about the quality assessment were discussed by the assessors and the principal investigator (N.V.) until consensus was reached. Covidence [28] was also used for management of studies during the quality assessment procedure.
Data collection
Data were collected on study (year of publication, authors, country, type of treatment, and study design); participants characteristics (number of participants, age, disease of interest, and duration of symptoms); and on outcome measures (outcome assessment tool, outcomes for facial weakness or facial expression and, if available, psychosocial outcomes).
RESULTS
Results of the search
The primary database search, executed on June 24th, 2020 yielded 3,133 articles (The Cochrane Library: 202, Embase: 1,709, PubMed: 1,222). A total of 2,249 articles remained after removing duplicates. Title and abstract screening resulted in 138 eligible articles for full text screening. Thirty-two articles were included after full text review. Screening of reference lists of the included studies which were cited in one of the primary included studies resulted in another eight studies for possible inclusion. Seven of these eight studies were included, and one study was excluded during title and abstract screening. Update of the search, executed on June 24th, 2022, yielded one extra article. Altogether, 40 studies met inclusion criteria for this review (details in Fig. 3).
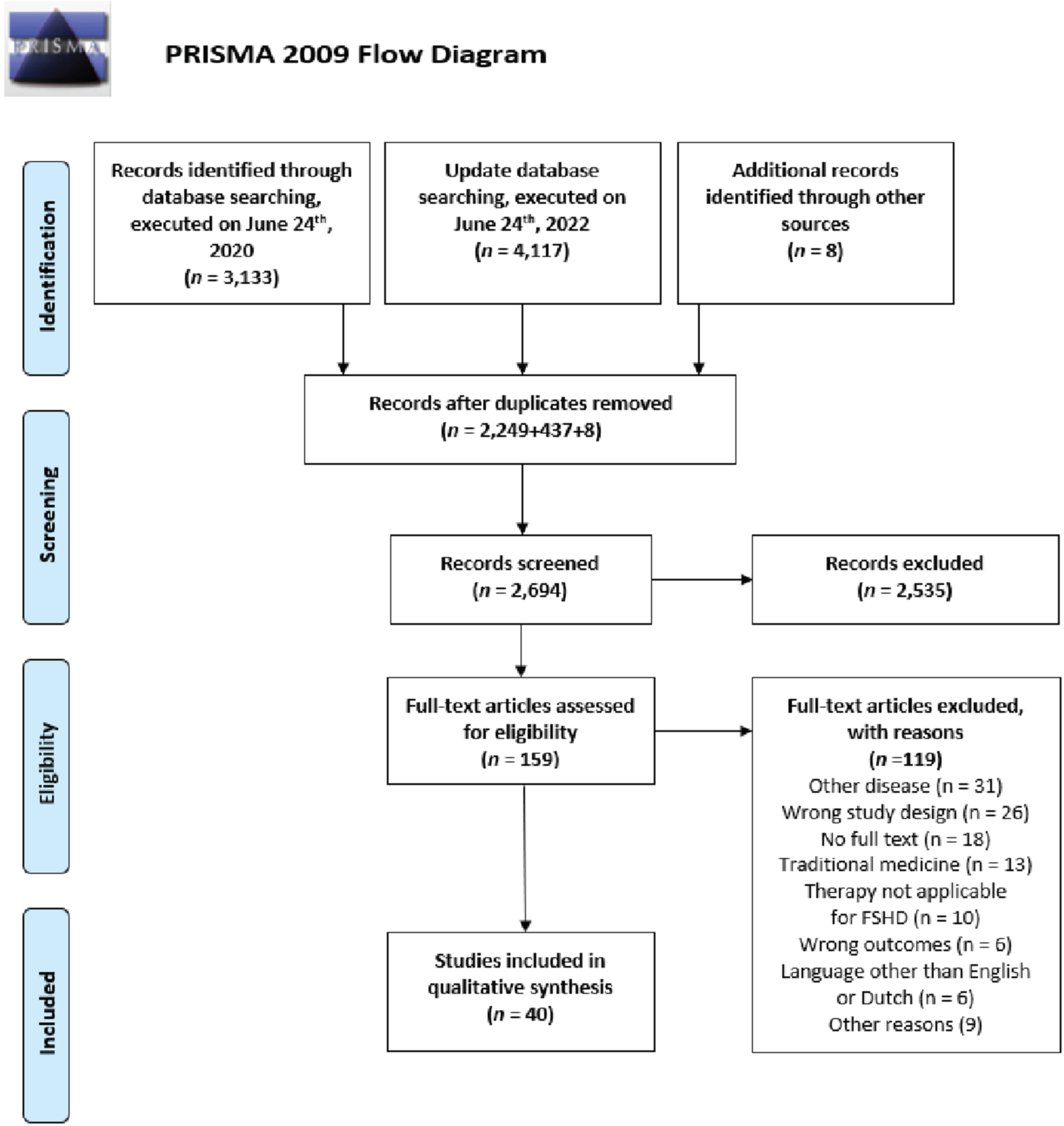
PRISMA flowchart for study inclusion. This flowchart presents the results of the literature search and the number of selected studies for each step of study inclusion.
Study characteristics
In the included studies, all five neurological diseases were addressed. More than half of the 40 included studies were studies with Bell’s palsy patients (21 studies) [31–51]. Second most included study population was patients with Moebius syndrome (nine studies) [52–60]. The other three diseases were present in five (Parkinson’s disease) [61–65], three (FSHD) [66–68], and two (myotonic dystrophy type 1) [69, 70] studies. Seventeen studies were set up as randomized controlled trial, twelve studies were case reports, others had a quasi-experimental study design. Most studies were performed in Asia and North America (33% each), the other studies in Europe (28%), Africa (5%), and Australia (3%). A variety of different types of therapies were present in the included studies, with physical therapy (n = 10), surgery (n = 7), and speech therapy (n = 7) as most included. Characteristics of the included studies are presented in Table 2.
Overview of included articles
1Data about differences between groups was missing. 2Data was only presented in a figure. Precise data was not presented. *=statistical significance. Abbreviations: ARTIC = articulation treatment, CR = case report, d = days, EMG = electromyography, ENoG = electroneurography, freq = frequency, GaAlAs = gallium-aluminum-arsenide, h = hours, HR = hazard ratio, Hz = hertz, LSVT = Lee Silverman Voice Treatment, max = maximum, m = mean, min = minutes, MSF = Moebius Syndrome Foundation, mth = months, NA = not applicable, NR = not reported, OR = odds ratio, PD = Parkinson’s disease, QE = Quasi experimental, RCT = randomized controlled trial, sec = seconds, wk = weeks, y = years. Outcome assessment tools used in any of the included studies: CAS = Chuang cortical adaptation stage: staging system for functional progress of transferred muscles. Stages range from: I (no movements) to V (spontaneous smile, without unvoluntary movements. CES = Communicative Effectiveness Survey: 8 item instrument for assessing communicative effectiveness. Scores range from 0 to 32. Higher scores are an indication for better communicative effectiveness. CPIB = Communicative Participation Item Bank: 10 questions for assessing communicative participation. Every question is score with a 4-point scale. Scores are converted into T-scores. Higher scores are more favourable. FaCE = facial clinimetric evaluation scale: 15 item instrument with six domains covering facial functioning. Total score ranges from 0–100. Higher scores are an indication for better facial function. FACEM = facial expression measurement: program with model of the face with 12 facial measures. FACS = Facial action coding system: anatomically based system for assessing facial expressions and movements. FDI = Facial disability index (FDI): consists of two domains, namely physical function, and social/well-being function. Scores are: 0–100, with 100 as best score. HADS = Hospital Anxiety and Depression Scale: 14 item instrument for assessing anxiety or depression. Scores range from 0–21 per subscale. Higher scores give a greater indication for anxiety, or depression. Happiness rating: Rating used for happiness indication. Scores range from 1 (not very happy) to 7 (extremely happy). Hay’s rating scale = Assessment tool for static position and active movements improvements after surgery. Scores range from 1 (perfect) to 9 (very marked imperfection). HBS = House-Brackmann scale. This is a facial grading system, ranging from I (normal function) to VI (total facial paralysis). HBS 2.0 = updated version of House-Brackmann scale. This facial grading system ranges from 4 (normal function) to 24 (total facial paralysis). Points are given per facial region: Eye, eyebrow, nasolabial fold, and oral region. IOPI = Iowa Oral Performance Instrument: device for measuring lip pressure in kilopascals. Paresis index: assessment tool for evaluating facial paresis in resting position and during voluntary movements. Scores range from 0–10, with higher scores as an indication for more facial paresis. PNSE = Positive and Negative Social Exchange: tool for assessing social support with 4 subscales: companionship, emotional, informational, and instrumental. Scores on 5-point scale, with higher scores as indication for more social support. PSQ = Perceived Stigma Questionnaire: 21 item instrument for assessing stigma by patients with visible differences. Every question has a 5-point scale, ranging from never to always. Higher scores are an indication for more perceived stigma. Satisfaction score = non-validated questionnaire for evaluating satisfaction after smile surgery. Scores range from 0–5, with 5 as maximum satisfaction score. SB = Sunnybrook facial grading system. This is a tool for assessing symmetry of facial movements and resting symmetry. Scores range from 0 (total facial paralysis) to 100 (normal symmetry). SES = Smile excursion score: scores the visibility of teeth during a wide smile. Scores range from 0 (no teeth visible) to 4 (teeth, or premolar tooth visible). SMILE = Scaled Measurement of Improvement in Lip Excursion: for assessing facial function by using iris diameter as scale of reference for facial features in a photo. SurgS = Surgeon’s score: non-validated score system performed by surgeons for assessing facial asymmetry (static and dynamic). Scores are: ‘poor’, ‘good’, or ‘excellent’. TA = Transcription accuracy: percentage of correct transcribed words during a listener’s transcription. It is used as an intelligibility measurement. UWDSE = University of Washington Disability Self-Efficacy Scale: 6 item instrument for assessing self-efficacy by patients with disabilities or chronic illness. Scores range from 6–30, higher scores are an indication for less self-efficacy. VHI-30 = Voice Handicap Index: 30 item instrument for assessing impact of voice disorders in daily life. Scores range from 0–120. Higher scores are an indication for more self-perceived impact of voice disorder.
Quality assessment
The randomized controlled trials scored on average eight out of thirteen points. Two questions about blinding were mainly not applicable to the included studies. The non-randomized controlled trials scored on average five out of eight points. Two questions were not applicable since these questions evaluated differences between included participants groups which was relevant in only three of the non-randomized controlled trials [31, 69]. The other nine trials had only a single participant group. The case reports scored on average seven out of eight points. The main limitation of these studies was a lack of described medical history of the included patients, as six out of eleven studies did not report this adequately. All details of the quality assessments are shown in Tables 3–5, respectively.
Quality assessments of included randomized controlled trials (Joanna Briggs Institute Critical Appraisal Checklist for Randomized Controlled Trials)
NA=not applicable, NR = not reported. Questions used in the Joanna Briggs Institute Critical Appraisal Checklist for Randomized Controlled Trials: 1: Was true randomization used for assignment of participants to treatment groups?. 2: Was allocation to treatment groups concealed?. 3: Were treatment groups similar at the baseline?. 4: Were participants blind to treatment assignment?. 5: Were those delivering treatment blind to treatment assignment?. 6: Were outcomes assessors blind to treatment assignment?. 7: Were treatment groups treated identically other than the intervention of interest?. 8: Was follow up complete and if not, were differences between groups in terms of their follow up adequately described and analyzed?. 9: Were participants analyzed in the groups to which they were randomized?. 10: Were outcomes measured in the same way for treatment groups?. 11: Were outcomes measured in a reliable way?. 12: Was appropriate statistical analysis used?. 13: Was the trial design appropriate, and any deviations from the standard RCT design (individual randomization, parallel groups) accounted for in the conduct and analysis of the trial?.
Quality assessments of included non-randomized controlled trials (Joanna Briggs Institute Critical Appraisal Checklist for Quasi-Experimental Studies)
NA=not applicable, NR = not reported. Questions used in the Joanna Briggs Institute Critical Appraisal Checklist for Quasi-Experimental Studies: 1: Is it clear in the study what is the ‘cause’ and what is the ‘effect’ (i.e. there is no confusion about which variable comes first)?. 2: Were the participants included in any comparisons similar?. 3: Were the participants included in any comparisons receiving similar treatment/care, other than the exposure or intervention of interest?. 4: Was there a control group?. 5: Were there multiple measurements of the outcome both pre and post the intervention/exposure?. 6: Was follow up complete and if not, were differences between groups in terms of their follow up adequately described and analyzed?. 7: Were the outcomes of participants included in any comparisons measured in the same way?. 8: Were outcomes measured in a reliable way?. 9: Was appropriate statistical analysis used?.
Quality assessments of included case reports (Joanna Briggs Institute Critical Appraisal Checklist for Case Reports)
NA=not applicable, NR = not reported. Questions used in the Joanna Briggs Institute Critical Appraisal Checklist for Case Reports: 1: Were patient’s demographic characteristics clearly described?. 2: Was the patient’s history clearly described and presented as a timeline?. 3: Was the current clinical condition of the patient on presentation clearly described?. 4: Were diagnostic tests or assessment methods and the results clearly described?. 5: Was the intervention(s) or treatment procedure(s) clearly described?. 6: Was the post-intervention clinical condition clearly described?. 7: Were adverse events (harms) or unanticipated events identified and described?. 8: Does the case report provide takeaway lessons?.
Study outcomes
Study outcomes are presented per neurological disease. An overview of all outcomes measures is shown in Table 2. We discuss the main findings below.
FSHD
Only three case reports with FSHD patients were included. The studies did not use any of the universally used assessment tools or other systematic outcome measurements. In Dua et al. [66] a 57 year old woman received dermal fillers with cog threads on both sides of the cheek. After three weeks, the patient noted improvements of jawbone contour, nasolabial folds, and facial symmetry, but there was still a deflation at the most affected side. Acupuncture was the treatment of interest in Liu et al. [67], which was given in two groups of acupuncture points to a 62-year-old woman for two periods of five months. After ten months, improvements were observed by herself and her practitioner for several facial muscle functions, such as eye closure and top lip movements. The last included study on FSHD by Matsumoto et al. [68] performed surgery for patients with functional and aesthetic problems because of a drooping lower lip, due to facial weakness and muscle atrophy. The surgery procedure consisted of introducing auricular cartilage craft in the lower lip, to ensure lower lip elevation. All three included patients had a lower lip elevation of minimal four to seven millimetres. Only technical surgical outcomes were given as outcome measure.
Bell’s palsy
Twenty-one studies were included with Bell’s palsy patients with five different treatment options, most frequently physical therapy. Mean time since diagnosis varied from less than one day [31] to 26 years [46]. Most used outcome measurement scores were House-Brackmann scale (HBS) and Sunnybrook facial grading system (SB), with respectively eleven [31, 48–51] and eight [32, 51] studies. HBS is a scale for assessing facial paresis with scores range from I (normal function) to VI (complete facial paralysis). SB is a tool for assessing (a)symmetry of facial movements and synkinesis with the scores range from 0 (complete facial paralysis) to 100 (normal symmetry).
Outcomes of studies with physical therapy varied, regarding recovery rate and time to recovery, although most studies showed better outcomes for physical therapy, compared to usual care. Out of the nine studies with physical therapy, five contained Kabat physical therapy [32, 42]. This is a proprioceptive neuromuscular facilitation method originating from Kabat et al. [71]. Two studies of Monini [41, 42] showed that some patients who received Kabat physical therapy had more improvements on HBS and a faster recovery, compared with controls who received usual care. Patients with HBS IV or V, who received Kabat physical therapy, had better recovery rates than controls and other patients. Furthermore, patients in the therapy group had on average a faster recovery [41]. Similar results were seen in another study of Monini [42], were patients who received Kabat physical therapy had 17 times the odds for improving≥3 grades on HBS score (odds ratio = 17.73), compared with a control group. Barbara et al. [33], a study which looked also at Kabat physical therapy, showed significant better improvements in HBS for therapy group, at day 15 after start of treatment. Similar results were seen in Ghous et al. [38], where patients who received Kabat physical therapy had statistical improvements on the physical subscore of a facial disability assessment tool (FDI). Social subscores of FDI failed significance.
Manikandan [39] also showed significantly more improvement for the physical therapy group (facial neuromuscular re-education) compared with control group, although both groups improved over time. Cederwall et al. [37] performed a study with a quasi-experimental study design, showing that 26 to 42 weeks of physical therapy resulted in improvements in the SB scale. A control group was lacking and data were only presented graphically. In Martineau et al. [51] intervention method was mirror therapy. Patients did twice a day facial exercises in front of a mirror. There were significant better outcomes for the therapy group compared with the control group for HBS scale and FaCE (a facial clinimetric evaluation scale) outcomes during follow-up. There were no significant differences for SB scale outcomes.
In contrast to earlier mentioned studies, Nicastri et al. [43] found no differences in recovery rate and time to recovery between the physical therapy and control group. Only more severely affected patients (HBS V and VI) had faster recovery after physical therapy.
Two case reports showed improvements in facial symmetry after physical therapy. Aranha et al. [32] reported about a seven-year-old patient and showed improvements in the SB scale (from 21 to 75 points) after 14 days of Kabat physical therapy. Another case report by Brach et al. [35] about a 71-year-old, also reported an increase in SB scale (from 17 to 68 points) after physical therapy. In this case report the physical therapy consisted of facial neuromuscular retraining techniques and duration of follow up was 14 months.
Four studies investigated the effects of electrical stimulation on facial palsy. Three of these studies found no significant difference for recovery rates between the therapy and control group. Alakram et al. [31] found no statistical differences in recovery rates (37.6% vs 29.6%) in HBS scores between the group treated with transcutaneous electrical nerve stimulation and the control group. Marotta et al. [40] found no differences between neuromuscular electrical stimulation and control group as well. SB scores, respectively 47.2 and 37.3, did not statistically differ after treatment. However, they did find more symmetry in voluntary movements in the therapy group. Sandeep et al. [45] added heat by infrared to electrical stimulation. In this study, no statistical difference after treatment between therapy and control groups (mean difference in SB scores was respectively 43.1 and 43.7) was reported. In contrast to the other three studies, Tuncay et al. [49] did find significant more improvement in both facial grading (mean HBS: III to I) and facial disability (FDI) for the therapy group, compared to controls.
Outcomes after acupuncture as treatment for improving facial palsy differed. Three studies, of which two case reports, showed improvements after acupuncture, [34, 50] and two randomized controlled trials did not find statistically significant improvements [44, 48]. One of the randomized controlled trials (Öksüz et al. [44]) reviewed acupuncture treatment (19 selected points in a meridian) in patients with a disease duration of minimal three months. The other randomized controlled trial (Tong et al. [48]) included patients with a disease duration of maximum 12 days and used eight points in a meridian for acupuncture. Post-treatment there were no statistically differences between acupuncture and control groups for facial grading scores (HBS) in both randomized controlled trials [44, 48]. However Öksüz et al. [44] did find improvements in electromyography values favouring the acupuncture group. Two case reports showed improvements in facial grading (HBS) after acupuncture. Cayir et al. [36] reported an improvement of HBS IV to II after 20 sessions (15 selected points in a meridian), and Wong et al. [50] showed an improvement of HBS IV to III after 25 sessions (10 selected points per session). A Pakistani study by Bokhari et al. [34] also reported improvements after acupuncture (seven selected points in a meridian), although only descriptive results were shown and none of the widely used outcome assessment tools were used.
The following two treatment options were only included in one study each: blood injection and intramuscular injections with hyaluronic acid-based fillers. A case report by Seffer et al. [46] investigated the effects of subcutaneous and muscular injection of peripheral homologous blood at affected facial side. Improvements were reported in facial movements and facial symmetry, but an objective measurement was not used. Starmer et al. [47] used intramuscular injections with hyaluronic acid-based filler at affected sides. Lip pressure (Iowa Oral Performance Instrument) improved after treatment (2.2kPa to 5.3kPa) and all patients had improvements for articulation and less anterior spillage. Besides Bell’s palsy patients, one FSHD patient was also included. Lip pressure improved from 0kPa to 6kPa (right side of lip) and from 0kPa to 7kPa (left side of lip) in this patient.
Moebius syndrome
Smile reanimation surgery techniques were the intervention method in six out of the nine included studies. All these studies had a quasi-experimental design (n = 2) [55, 60] or a case report design (n = 4) [52, 59]. They all performed a free functional muscle transfer with reinnervation and a nerve graft. Used muscles varied: latissimus dorsi, gracilis, rectus femoris, or temporalis muscle. All six studies reported improvements, measured with variable outcome measurements. Four studies [55–57, 60] used only, or in most patients, the gracilis muscle for the free muscle transfer. Domantovsky et al. [55] had a long-term follow-up duration of 20 years, in which they assessed facial functioning with FaCE. A mean score of 62.3 out of 100 was found during follow-up. The lowest subscore was for facial movements (35.6 out of 100). All patients reported improvements in quality of life, communication, and self-confidence. However, surgery did not fulfil expectations in all patients; in an interview they noted that they still talked, looked and felt different than others. A Canadian study by Zuker et al. [60] found improvements for all patients: 70% of the participants had oral dysfunction (drooling and fluid loss while drinking) before surgery, which significantly improved after surgery. Six patients with speech difficulties all experienced improvements after surgery, and three of these group of patients were even symptomless. Lifchez et al. [56] and Lu et al. [57] both mostly used gracilis muscle for the free muscle transfer, Lifchez et al. [56] used the latissimus dorsi muscle once and Lu et al. [57] used the rectus femoris muscle once. Lifchez et al. [56] noted that two of the three patients could smile independently of jaw closure after surgery. The other case report (Lu et al. [57]) showed smile excursion (smile excursion score) improvements after surgery, and all patients could synchronously smile after the intervention. Two other case reports mostly used the latissimus dorsi muscle for the free muscle transfer. Amer et al. [52] reported a bilateral symmetrical smile 12 months after surgery. Woollard et al. [59] noted improvements for static and active movements (Hay’s rating scale) and good to excellent facial symmetry (surgeon’s impression).
Three studies were included with other treatment options for Moebius syndrome patients, of which two articles focused on a Moebius Syndrome Foundation conference in 2014. Bogart et al. (2016) [53] investigated the differences in social and emotional outcomes between attenders and non-attenders. Attenders tended to have more improvements for coping with stigma, social comfort, and perceived knowledge than non-attenders. There were no differences between attenders and non-attenders for companionship, anxiety, depression, and disability self-efficacy. Bogart et al. (2017) [54] looked at the reasons for attending or not attending the conference, using data from Bogart et al. (2016) [53]. Most frequently mentioned reasons for attending were: getting companionship, emotional support, and informational support. The most common reasons for not attending were financial reasons and energy limitations. Conference limitations were also assessed and most mentioned were, lack of age-appropriate activities and lack of relevant information.
The last included study with Moebius syndrome patients is a Danish study by Michael et al. [58], investigating the benefits of a social skill workshop for learning compensatory expressive behaviour. Both a group with Moebius syndrome and a group with controls (people without Moebius syndrome) were included and observed during social interactions. The social workshop consisted of group discussions, role-plays, group activities, and writings sessions. Comparisons between both groups before treatment, showed more facial expressivity, more fidgeting, more gesturing, and less variable speech rate in the control group. Both verbal and nonverbal communication improved after the workshop: more gesturing, more facial expressivity and more head movements were observed in both patients and controls after the workshop. Although both groups improved, the increase in gesture rates, facial expressivity, and fidgeting was statistically higher in patients. Moreover, speech rate variability increased, but speech rate was lower in both groups after the workshop. Rapport (‘the ability to connect with others in a way that creates a climate of thrust and understanding’) [72] was not increased when measured with self-reports, but observers did report a significant increase in both groups. Social anxiety and social competence assessments in the Moebius syndrome group did not differ after treatment.
Myotonic dystrophy type 1
The two included studies with myotonic dystrophy type 1 patients focused on respectively the influence of warming up on speech quality [69], and on an exercise program for improving lip muscle strength [70]. De Swart et al. [69] made a ten-minute speech protocol to review the effect of warming up on myotonia and on flaccid dysarthria (due to facial weakness), because of the aim of this review focus was only on outcomes referring to the flaccid dysarthria. Signs of flaccid dysarthria were still visible after warming up: maximal performance on sound prolongation tasks was poor, more variability on maximum repetition rate tasks, and maximum speech rate was low. However, warming up did not worsen the symptoms of flaccid dysarthria. Sjögreen et al. [70] found that a 16-week exercise program with an oral screen led to lip-strength improvements in 88% of the patients (n = 7). There was no statistical difference with patients who did not receive treatment.
Parkinson’s disease
The studies on Parkinson’s disease used voice treatment or orofacial physical therapy. The outcomes after treatment were similar in these studies, as they all reported some improvement. Every study used different outcome assessment methods.
Lee Silverman voice treatment (LSVT) was used in three studies [61, 65]. This is a communication intervention for improving vocal intensity in Parkinson’s disease patients, by exercises varying from saying words to conversational speech [61]. Bryans et al. [61] reported a significantly improvement of vocal intensity after LSVT. Besides, patients noted improvement in effectiveness of their speech in daily conversations (Communicative Effectiveness Survey). Dumer et al. [62] reviewed not only LSVT, but also ARTIC, wherein orofacial movements are trained to improve articulation. Assessments for facial expression movements (FACS), showed only improvements for frequency and variability in the LSVT group. After treatment, there were no longer statistical differences for frequency and variability for facial expressions between controls and LSVT group. This is in contrast with before the LSVT sessions, as frequency and variability for facial expressions were lower in Parkinson’s disease patients. A study by Sapir et al. [65] investigated the influence of LSVT on formant frequency and vowel goodness ratings (vowels produced in a sentence). They reported improvements in some formants and in vowel goodness ratings.
The only included study from Australia by Katsikitis et al. [63], noted improvements for mouth opening, after orofacial physical therapy, which contained exercises and brushing of muscles. This improvement was not seen in the control group. Lastly, Levy et al. [64] compared voice treatment with articulation treatment and results were presented with intelligibility assessment (TA). There was only a significant increasement after treatment in the voice treatment group, as the percentage of correct transcribed words by listeners increased from 53.6% to 85.1% .
DISCUSSION
The aim of this systematic review was to provide an overview of research on available treatments for improving facial muscle function and on therapies focussing on psychosocial consequences of having an altered facial expression in other neurological diseases to evaluate their potential in FSHD patients. We encountered only limited overlap for therapies between the different neurological diseases. A total of forty studies were included with twelve different treatment categories, of which acupuncture, physical therapy, speech treatment, and surgery were the intervention of interest in at least two different neurological diseases. The studies on FSHD were not performed in a systematic manner. Based on the results in the other neurological diseases, we consider several interventions of interest for FSHD: teaching (non-verbal) communication compensation strategies, speech training, physical therapy, conference attendance, and smile restoration surgery. The approaches call for systematic prospective research. The main findings are discussed below.
Research gap and recommendations
This systematic review was set up because of observations of psychosocial distress in FSHD patients with altered facial expression and lack of symptomatic treatment options. This scarcity was reflected by the low number of studies in FSHD in this review (n = 3). All three studies had a case-report design and lacked systematic outcome measures. Hence, level of evidence is low.
Compensation strategies. We consider teaching compensation strategies a potentially beneficial treatment option for FSHD patients. Patients with altered facial expression develop compensation strategies over time [73–75]. Compensation strategies can also be successfully taught, as Michael et al. [58] reported improvements in both verbal and non-verbal communication for Moebius syndrome patients. In a recent questionnaire study and qualitative study, we showed that younger FSHD patients experience more psychosocial distress because of facial weakness than older patients [23, 24]. Therefore, young FSHD patients could particularly benefit from learning compensation strategies. In addition to learning compensation strategies during a social skill workshop, other training modes could be considered: information provided by online modules, 3D model interactions, applications, or through peer-support. Learning how to deal with the consequences of facial weakness and learning how to optimally inform others, could also be implemented in a social skill workshop. Acceptance and Commitment Therapy (ACT) [76], a behaviour therapy focussing on improving psychosocial flexibility, might help in this learning process. Further research could investigate if FSHD patients benefit from learning compensation strategies and if combining this with ACT gives additional improvements on psychosocial outcomes.
Communication. Communication difficulties are common in FSHD patients (35%) [77, 78]. Nevertheless, treatment options aiming to improve communication have not been studied for FSHD patients. This in contrast to Parkinson’s disease and myotonic dystrophy [61, 69]. The impairments in communication in FSHD patients could be due to speech difficulties, but the influence of social interaction due to lack of facial expression could also interfere in this impairment. Therefore, it could be beneficial to improve both aspects, for instance by implementing speech training and learning compensation strategies in one training.
Patient conferences. Studies with Moebius syndrome patients reported about benefits and identified reasons for (not) attending Moebius syndrome patient conferences [53, 54]. Similar studies in FSHD have not been performed. These could give insight in the perspective of FSHD patients on attending in a conference. Conferences are important for sharing patients’ perspectives of their disease, for informing patients about latest insights in research progression and it is a place to introduce treatment options. It is also a place to facilitate peer support, which is support (emotional, informational, or appraisal) by another patient with the same disease or another disease with similar consequences by sharing experiences, provide suggestions, to enhance quality of life [79]. Peer support as part of shared medical appointment in patients with neurological diseases (FSHD as second largest patient category) has shown that it can improve self-reported quality of life [80]. Furthermore, it could be beneficial to supply information on facial weakness and its psychosocial consequences to a wider audience. That is especially important as patients with altered facial expression are perceived as more negative [19, 82] and when people are aware of the consequences of having facial weakness, prejudgements could potentially be reduced.
Physical therapy. Physical therapy has been incorporated in treatments of Bell’s palsy and Parkinson’s disease, but applicability to FSHD is uncertain. Most studies on Bell’s palsy patients showed tendency to improved recovery rates after physical therapy [33, 51]. Previously, two reviews have performed a literature search on physical therapy in Bell’s palsy patients [83, 84]. Both reviews concluded that there was neither benefit nor harm associated with physical therapy. However, these conclusions were based on searches completed in 2011 [83] and 2012 [84]. Subsequent to these reviews, six new studies were conducted. Among these, four studies reported more improvements after physical therapy [41–43, 51]. The remaining two studies were either of low quality [38], or a case report [32]. Consequently, since the searches of Baugh et al. [84] and Texeira et al. [83], the newer performed studies on this subject suggest more favourable outcomes for the groups undergoing physical therapy. Besides Bell’s palsy, one study reported improved mouth opening after orofacial physical therapy in Parkinson’s disease [63]. Because of the different pathophysiology (see Fig. 2), prediction of the effect of physical therapy in FSHD patients is difficult. It is currently unknown to what extent dystrophic muscles are capable of adaptation and strengthening by training. Skeletal muscle strength training in FSHD tends to be safe with limited improvements [85]. Aerobic training is also a safe intervention resulting in reduced fatigue [86] and increased walking speed [87]. Applicability of physical therapy for facial muscles has never been investigated in FSHD. Research could clarify if physical therapy can help in improving facial expressions.
Plastic surgery. Surgery as a treatment for facial weakness has not been studied systematically. Only one case series reported the effect of surgery on facial weakness in FSHD patients [68]. All three patients had a drooping lower lip with either functional or aesthetic problems. There is another case report on facial surgery in a FSHD patient, however facial weakness was not the indication and this study was therefore not included in the review. In this article a patient with early onset FSHD had surgery to reduce macroglossia and a frontal open bite through an osteotomy of the mandible in combination with a tongue reduction. No studies were performed on smile-restoration surgery in FSHD. This technique is often used in Moebius syndrome patients, with reported improvements in all included studies [52, 60]. Smile restoration surgery could be effective in FSHD patients who are severely affected by facial weakness, since the pathophysiology of FSHD is limited to muscles and does not affect nerve function. One major limitation in smile restoration in FSHD patients is the slowly but progressive nature of the muscle dystrophy and therefore the risk of transferring a potentially affected muscle. The Labbé technique (a lengthening temporalis myoplasty) where a tendon of the temporalis muscle is replaced from the coronoid process to the lips, has been applied in patients with peripheral facial paresis and might also be useful in FSHD patients [88, 89]. For the Labbé technique to be successful, it must first be proven that the temporalis muscle is spared in disease progression. Muscle ultrasonography could be used to investigate this, as disease severity correlates with muscle ultrasound findings in FSHD patients [90].
Cosmetic interventions. FSHD patients at our expert centre have frequently asked for the applicability of botulinum toxins and dermal fillers for improving facial symmetry. Although we specifically added botulinum toxins in the search strategy, we did not find any study assessing this. Dermal fillers were used in one included case report. This case report by Dua et al. [66] describes the usage of dermal fillers and cog threads and results showed partial improvements. However, these results should be interpreted with caution, because of study design, lack of systematic outcome measurements, and limited follow-up duration (3 weeks). Botulinum toxins are used for improving synkinesis in facial palsy patients [91, 92]. But outcomes of studies focussing on synkinesis were not part of this review, because it is not an equivalent of facial weakness and therefore no recommendations on applicability to facial weakness can be made. Thus, not enough evidence is available to conclude whether botulinum toxins and dermal fillers are useful options in FSHD.
Future studies. Future studies on treatment options to improve altered facial expression should preferably address different categories of outcomes measures: cosmetic, facial functioning, communication, and quality of life (see Fig. 4). Not one of the included studies addressed all these categories. Psychosocial outcome measures were especially under-represented. In addition, to allow comparison between different studies, it would be recommended to use similar study designs and outcome measures.
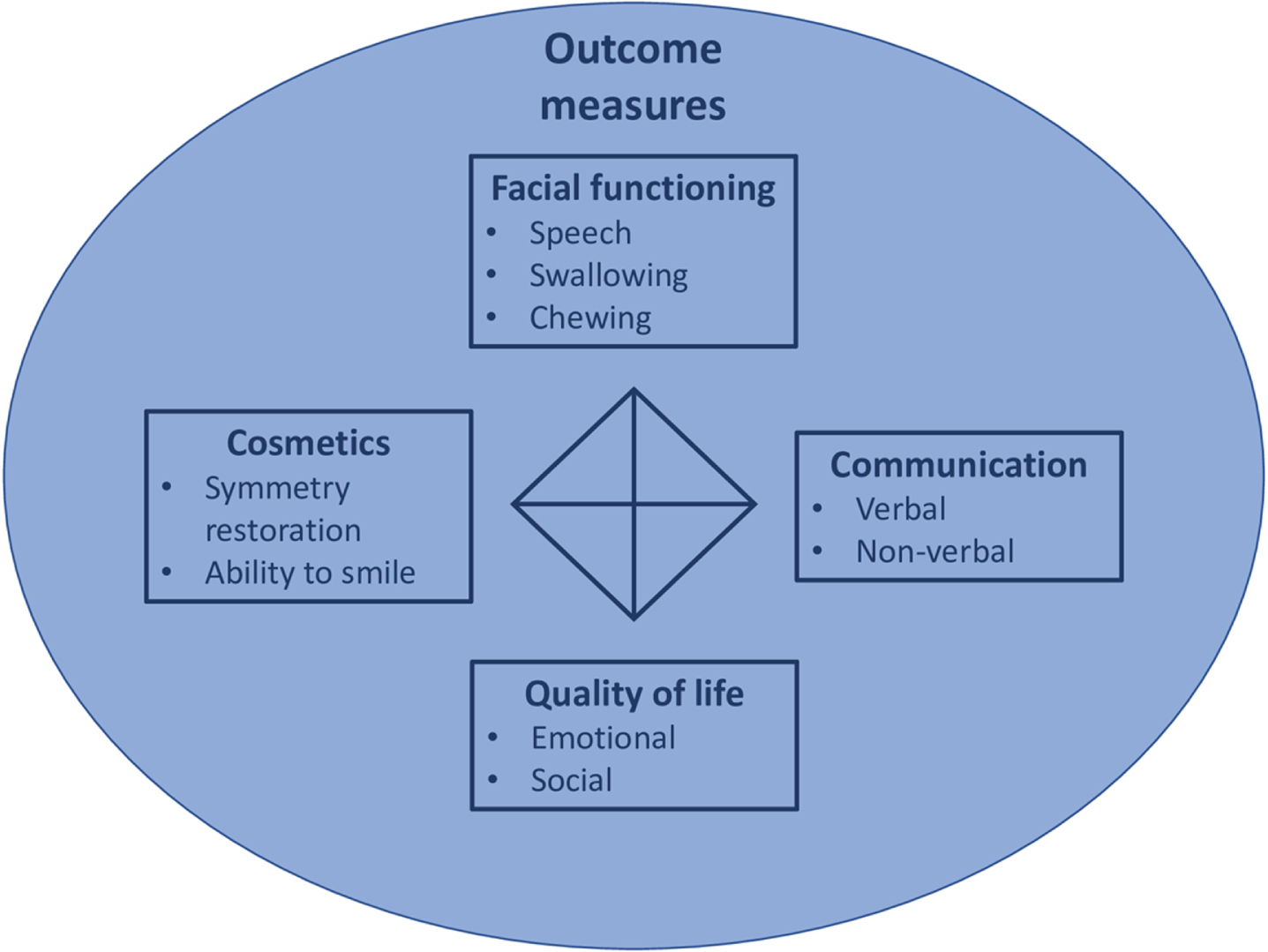
Different outcome assessment categories for assessing symptomatic treatments for improving altered facial expression. Four different outcome assessment categories: 1. facial functioning: consisting of speech, swallowing and chewing; 2. communication: consisting of verbal and non-verbal communication; 3. quality of life: consisting of emotional and social functioning; 4. cosmetics: consisting of symmetry restoration and ability to smile.
Limitations
Multiple neurological diseases were included, because of the expected scarcity of literature about treatment options for improving facial weakness in FSHD patients and of the expected wide applicability of certain symptomatic treatments. However, the anatomical location of the defect and the pathophysiology is different in these diseases (Fig. 2), therefore some recommended treatments are not applicable to FSHD.
When comparing the included diseases on altered facial expression, especially Parkinson’s disease has a different underlying mechanism. As bradykinesia causes the altered facial expression, whereas in the other included disease this is caused by facial weakness or facial paresis. Additionally, medication and associated on-off phenomenon can effect the degree of bradykinesia, potentially affecting the outcomes of symptomatic treatments. However, the included studies did provide valuable insights into current symptomatic therapies, which could be discussed for their potential applicability in FSHD.
Bell’s palsy patients have a high spontaneous recovery rate, as about seventy percent has a spontaneous recovery within three to six months [93]. Therefore, in studies with Bell’s palsy patients a high number of patients recovering after treatment, would also recover without treatment. Treatment effectivity could therefore be overestimated in this disease, this applies especially for studies performed in the acute phase of Bell’s palsy.
Of the included studies, 12 consisted of case reports. Level of evidence is low, and conclusions must be taken with caution. Nevertheless, these studies were included because they could point out treatment options that would otherwise not be identified and might be interesting for future research.
Studies performed before 1990 were excluded, because of the expected outdated treatment usage before 1990, especially for surgery techniques. Despite we could have missed important references.
CONCLUSION
This systematic review showed that literature on treatments aiming to improve facial weakness in FSHD is scarce and has low level of evidence. Several potentially applicable treatment options for improving altered facial expression were identified after evaluation of the included therapies. These were: teaching (non-verbal) communication compensation strategies, speech training, physical therapy, and conference attendance. Smile restoration surgery could be an option for severely affected patients, but it is necessary to first identify facial muscles which are not or only mildly affected. Further research is necessary for all these potential treatment options. We recommend use of functional clinical outcome measures in various categories: cosmetic, facial functioning, communication, and quality of life. Ideally these outcome measures become standardized, to ensure future comparison between studies.
In short, this systematic review identified opportunities for future research, to achieve the goal in providing symptomatic treatment options for improving the consequences of facial weakness in FSHD.
Footnotes
ACKNOWLEDGMENTS
Several authors of this publication are members of the Radboudumc Center of Expertise for neuromuscular disorders (Radboud-NMD), Netherlands Neuromuscular Center (NL-NMD) and the European Reference Network for rare neuromuscular diseases (EURO-NMD). We are grateful for the contributions of Spierziekten Nederland and the FSHD Stichting.
FUNDING
No funding received.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article.
