Abstract
Background:
Spinal muscular atrophy (SMA) is a hereditary motor neuron disorder, characterized by the degeneration of motor neurons and progressive muscle weakness. There is a large variability of disease severity, reflected by the classification of SMA types 1–4.
Objective:
The aim of this cross-sectional study was to determine the nature of swallowing problems and underlying mechanisms in patients with SMA types 2 and 3, and the relationship between swallowing and mastication problems.
Methods:
We enrolled patients (aged 13–67 years) with self-reported swallowing and/or mastication problems. We used a questionnaire, the functional oral intake scale, clinical tests (dysphagia limit, and timed test swallowing, the test of mastication and swallowing solids), a videofluoroscopic swallowing study (VFSS), and muscle ultrasound of the bulbar muscles (i.e. digastric, geniohyoid and tongue muscles).
Results:
Non-ambulant patients (n = 24) had a reduced dysphagia limit (median 13 ml (3–45), and a swallowing rate at the limit of normal (median 10 ml/sec (range 4–25 ml). VFSS revealed piecemeal deglutition and pharyngeal residue. We found pharyngo-oral regurgitation in fourteen patients (58%), i.e. they transported the residue from the hypopharynx back into the oral cavity and re-swallowed it. Six patients (25%) demonstrated impaired swallowing safety (i.e. penetration aspiration scale > 3). Muscle ultrasound revealed an abnormal muscle structure of the submental and tongue muscles. Ambulant patients (n = 3), had a normal dysphagia limit and swallowing rate, but VFSS showed pharyngeal residue, and muscle ultrasound demonstrated an abnormal echogenicity of the tongue. Swallowing problems were associated with mastication problems (p = 0.001).
Keywords
GLOSSARY
Spinal muscular atrophy
Survival motor neuron
Medical Research and Ethics Committee
Dysphagia limit
Timed test swallowing
Videofluoroscopic swallowing study
International Dysphagia Diet Standardisation Initiative
Bolus residue scale
Penetration aspiration scale
Test of mastication and swallowing solids
Surface electromyografy
INTRODUCTION
Hereditary proximal spinal muscular atrophy (SMA) is caused by a loss of the function of Survival Motor Neuron 1 (SMN1) gene [1]. The disorder is characterized by alpha-motor neuron degeneration, abnormalities of neuromuscular junction anatomy and function, muscle atrophy and weakness. SMA is one of the most common hereditary neuromuscular disorders, with an incidence of approximately 1 in 10,000 live births [2]. The wide severity spectrum is reflected by the clinical classification of SMA that distinguishes types 1, 2, 3, and 4 around the time of diagnosis, based on age at onset and highest of two achieved motor milestones, i.e. independent sitting or walking [3–5]. Patients with SMA type 1 may acquire head control or learn to roll but will never learn to sit independently (i.e. ‘non-sitters’); patients with SMA type 2 achieve independent sitting, but not walking (i.e. ‘sitters’); patients with SMA type 3 learn to walk independently (i.e. ‘walkers’). SMA type 4 is characterized by adult onset and relative mild weakness. Since SMA is a progressive disorder, patients often lose acquired motor skills later in life. Therefore, the term ‘non-sitters’ may refer to both patients with SMA type 1 and patients with SMA type 2 who have lost the ability to sit independently, and ‘non-walkers’ to patients with SMA type 2 and those with type 3 who lost the ability towalk [5].
The most important feature of SMA is muscle weakness, which is more pronounced in legs than arms and also affects axial, intercostal and bulbar muscles (such as submental and tongue muscles). Patients with SMA frequently report issues such as impaired jaw function, fatigue when chewing, difficulty swallowing solid food and choking [6–11]. Swallowing problems are accompanied by an increased risk of respiratory complications and malnutrition, but studies investigating the nature of swallowing problems and underlying disturbed mechanisms in SMA, are sparse. Clinical observational studies reported reduced active maximal mouth opening (aMMO), caused by fatty degeneration of the bulbar muscles, especially the lateral pterygoid muscle (responsible for opening the jaw) [12–14]. Videofluoroscopic swallowing study (VFSS) revealed piecemeal deglutition (i.e. swallowing the bolus in small successive particles) and pharyngeal residue in children with SMA type 2 [15]. Until now, swallowing problems have not been studied in a larger number of patients with SMA, nor using a combination of techniques. In a previous study we examined the mastication function in patients with SMA type 2 and 3 [16]. In the present study, the swallowing function was investigated in the same patients. This study aimed to contribute to our understanding of the nature of swallowing problems and underlying mechanisms, in patients with SMA types 2 and 3. The second aim was to investigate the relationship between swallowing andmastication.
METHODS
Participants
Between August 2018 and 2019 patients of the Dutch SMA registry (i.e. patients enrolled in our ongoing population-based cohort study) with SMA type 2 and 3 were invited to participate [17, 18]. We included 27 patients aged 13 years and older. Those patients, who mentioned mastication and/or swallowing problems (i.e. fatigue or difficulty with chewing; feeling of food sticking in the throat; coughing when swallowing liquids or solid foods; mealtimes longer than 30 minutes; the need for diet modification; tube feeding required) were enrolled [8]. The study protocol consisted of a single hospital visit. Both the mastication and swallowing function were investigated. The results of the mastication function were published in a previous article [16]. At the time of enrollment, patients did not have access to SMN-augmenting therapies such as nusinersen or risdiplam. Six patients used pyridostigmine (i.e. a cholinesterase inhibitor that improves the strength of the muscles) [19, 20]. In order to prevent this drug from influencing the swallowing function, patients were asked not to take it on the day of the study.
The Medical Research and Ethics Committee (METC) of the University Medical Center Utrecht decided to only include patients with self-reported swallowing and/or mastication symptoms, and approved the study protocol according to Dutch legislation on clinical studies (METC 17–718). Informed consent was obtained from the patients, or in the case of patients younger than 16 years, from both patients and parents.
Study design
Swallowing was assessed, using a combination of a self-report questionnaire, dysphagia limit (DL), timed test swallowing (TTS), VFSS and muscle ultrasound of the digastric, geniohyoid and tongue muscles. Two experienced speech-language therapists (SLTs) (AMBH and LEH) performed the assessments.
Questionnaire and clinical scales
We used the questionnaire ‘Diagnostic List of Dysphagia and Dysarthria in (pediatric) patients with neuromuscular Diseases (DDD(p)NMD)’. The dichotomous questions of this questionnaire focus on masticatory, swallowing and jaw issues and consequences for mealtime duration, food adaptations, weight, and the occurrence and frequency of respiratory infections. This questionnaire was originally developed by a group of SLTs involved in the care of dysphagia in neuromuscular diseases with the aim of identifying feeding and swallowing problems [21]. The questionnaire overlaps with those used in other studies about feeding and swallowing problems in patients with SMA [6, 7]. It has been used previously in patients with SMA [8], and test-retest reliability was sufficient (n = 14, ICC = 0.94. (CI 0.80–0.98). We used the functional oral intake scale (FOIS). This is an ordinal scale, reflecting the functional oral intake of patients, with scores ranging from 1 ‘nothing by mouth’ to 7’ total oral diet with no restrictions’ [22].
Dysphagia limit
The aim of the DL test is to establish the maximum volume of water that can be swallowed in a single effort. It determines the presence and extent of piecemeal deglutition. This test has been validated with 75 healthy participants and 149 neurological patients with a swallowing disorder, and also 28 neurological patients without a swallowing disorder. No healthy participant had a dysphagia limit of <20 ml [23]. Reliability has not been explicitly investigated, but the observations are unambiguous due to the use of a syringe for the correct volume, and auscultation to determine the number of swallows. We offered patients 10 ml of tap water in a cup and asked them to swallow this in a single swallow. If the patient succeeded, we increased the bolus volume until the patient needed to swallow more than once. If the patient could not swallow 10 ml in one swallow, bolus volumes were gradually reduced. The largest volume swallowed in one effort was documented. We used cervical auscultation to assess the number of swallow sounds. The diagnostic value of 20 ml was used as the boundary between normal/abnormal volumes swallowed in a single effort [23].
Timed Test of Swallowing (TTS)
The aim of the TTS is to measure swallowing speed. Validation of this test with 150 ml water was performed, with 101 healthy participants, and 181 healthy participants by Nathadwarawala et al (1992) and Hughes et al (1996). No participant (aged <70 years) had a swallowing speed lower than 10 ml/sec. From 19 patients with a swallowing disorder, 14 patients had a swallowing speed <10 ml/sec. Intra and inter reliability of the test was sufficient.
We offered patients 100 ml of tap water. We instructed the patient to drink this as fast as possible, but safely’, and to say ‘yes’ when finished. The patient was videotaped laterally to determine the number of swallows and total time needed to complete swallowing 100 ml (time in seconds from the moment the first water passed the lower lip, until the patient indicated that he/she was finished). Not all patients were able to handle the cup independently (n = 12), in which case their results were excluded from analysis. We used the diagnostic value of 10 ml/sec as the boundary between normal and abnormal swallowing speed [24–26].
Videofluoroscopic Swallowing Study (VFSS)
VFFS was performed with a Philips Multi Diagnost Eleva C-arm, and VFSS images were collected in lateral view at 15 pulses per second, and pictures were stored in the picture archiving and communication system (PACS). Ambulant patients sat on a chair with a backrest adjustable for height to ensure an optimal upright position. Non-ambulant patients sat in their wheelchairs, meeting their individual needs. We used an iodine-containing nonionic contrast material. Three different consistencies were offered, and patients swallowed three boluses of each consistency. They started with IDDSI level 0, presented as a 10 ml sip in a cup. IDDSI level 3 which was thickened with a standardized amount of locust bean gum, checked on consistency, and presented on a dessert spoon (10 ml). The soft pieces bread (2×2 cm) of IDDSI level 7 were coated with contrast material and presented piece by piece [27].
We duplicated recordings of each bolus to analyze images of the VFSS, blinded for patient characteristics, consistency offered, and order of bolus presentation. Two SLTs (AMBH and LEH) scored the recordings together, in random order, on the following aspects: (a) number of swallows per bolus (i.e. passage of a bolus through the oesophagus), (b) pharyngeal residue with the bolus residue scale (BRS), (c) pharyngo-oral regurgitation, and (d) penetration/aspiration with the penetration aspiration scale (PAS). Any difference in ratings were repeated and debated until consensus was achieved.
We determined the limit of a usual number of swallows for the standardized boluses, applying reference values from healthy participants. Comfortable sip volumes of thin liquid (IDDSI level 0) in healthy adults range from 11–14 ml. Twenty percent of healthy adults perform a ‘cleanup swallow’ (i.e. a second swallow) [28]. Therefore, for a bolus of 10 ml thin liquid, we considered three or more swallows as abnormal. The mean swallow volume of moderately thick liquid (IDDSI level 3) in healthy adults was 4.9 ml (95% CI 4.4–5.4) [28]. Therefore, two or three swallows (a cleanup swallow included) were considered normal, and four or more swallows as abnormal. For the pieces of bread (IDDSI level 7), no reference values were available; we therefore used the reference values of moderately thick consistencies [28]. Swallowing efficiency was evaluated with the BRS. The scale classifies pharyngeal residue in the valleculae, piriform sinuses and/or posterior pharyngeal wall, ranging from 1 = no residue to 6 = residue in valleculae, on the posterior pharyngeal wall and in pyriform sinuses. A higher BRS score is associated with a higher risk of aspiration [29]. We scored BRS regardless of pre-swallow pharyngeal residue. BRS was captured from the frame of ‘swallow rest’, where the piriform sinuses were at their lowest position after swallowing, as part of post swallow pharyngeal relaxation. In this study it was not possible to quantify the volume of residue because of the markedly dilated hypopharynx in several patients. In addition in some patients it was impossible to make fully lateral images due to severe scoliosis.
In some of the first few patients, we unexpectedly observed an exceptional phenomenon, best described as pharyngo-oral regurgitation’. Instead of moving the bolus into the oesophagus, they transported it from the hypopharynx back into the oral cavity and re-swallowed it. The phenomenon occurred when the bolus had been partially swallowed and pharyngeal residue was observed. This residue was transported from the pharynx back into the oral cavity and re-swallowed We documented whether this phenomenon was present in each subsequentpatient.
We used the PAS to determine by which extend material entered the airway, and by whether the material entering the airway was ejected. The 8-point scale ranges from 1 (no material entering the airway) to 8 (material enters the airway, passes the vocal folds, and no effort is made to eject) [30]. PAS scores >3 were considered as impaired swallowing safety. For PAS scores >2, we involved the radiologist. For these cases, the radiologist (RAJN) and a SLT (AMBH) independently scored the PAS. Differences in scores were resolved by consensus.
For interrater reliability, 30% of the images (70 out of 240 randomly chosen boluses) were rated independently by the raters. For interrater- reliability, the agreement of the number of swallows was analyzed with a two-way random effect intraclass correlation coefficient (ICC). ICC demonstrated excellent inter-reliability for the number of swallows (ICC r = 0.93, CI 0.88–0.95). Cohen’s kappa was almost perfect for pharyngo-oral regurgitation (κ= 0.81), substantial for pharyngeal residue (κ= 0.63) and moderate for penetration-aspiration (κ= 0.43).
Muscle ultrasound
We performed muscle ultrasound to visualize the structure of submental muscles (i.e. digastric, and geniohyoid muscles) and tongue muscles [31–33]. We used an Affiniti 70 Philips Ultrasound System (Philips, the Netherlands) with a 12-5 MHz transducer (Fig. 1). During examination head and neck of the patients were in a neutral position. A broadband linear intraoperative (so-called hockey stick) 14-5 MHz transducer was used for the tongue (Fig. 1). The patient opened his/her mouth, and the transducer was placed on the anterior part of the tongue. Data were stored as DICOM images in PACS. We scored images qualitatively, based on consensus (LEH and her colleague SLT, experienced in interpreting muscle ultrasound images) either as (1). ‘normal structure and echogenicity’, (2). ‘increased echogenicity with a moth-eaten pattern’ or (3). ‘increased echogenicity’ (Fig. 2) [31, 34]. Raters did not have access to patient characteristics or results of other assessments of the study.
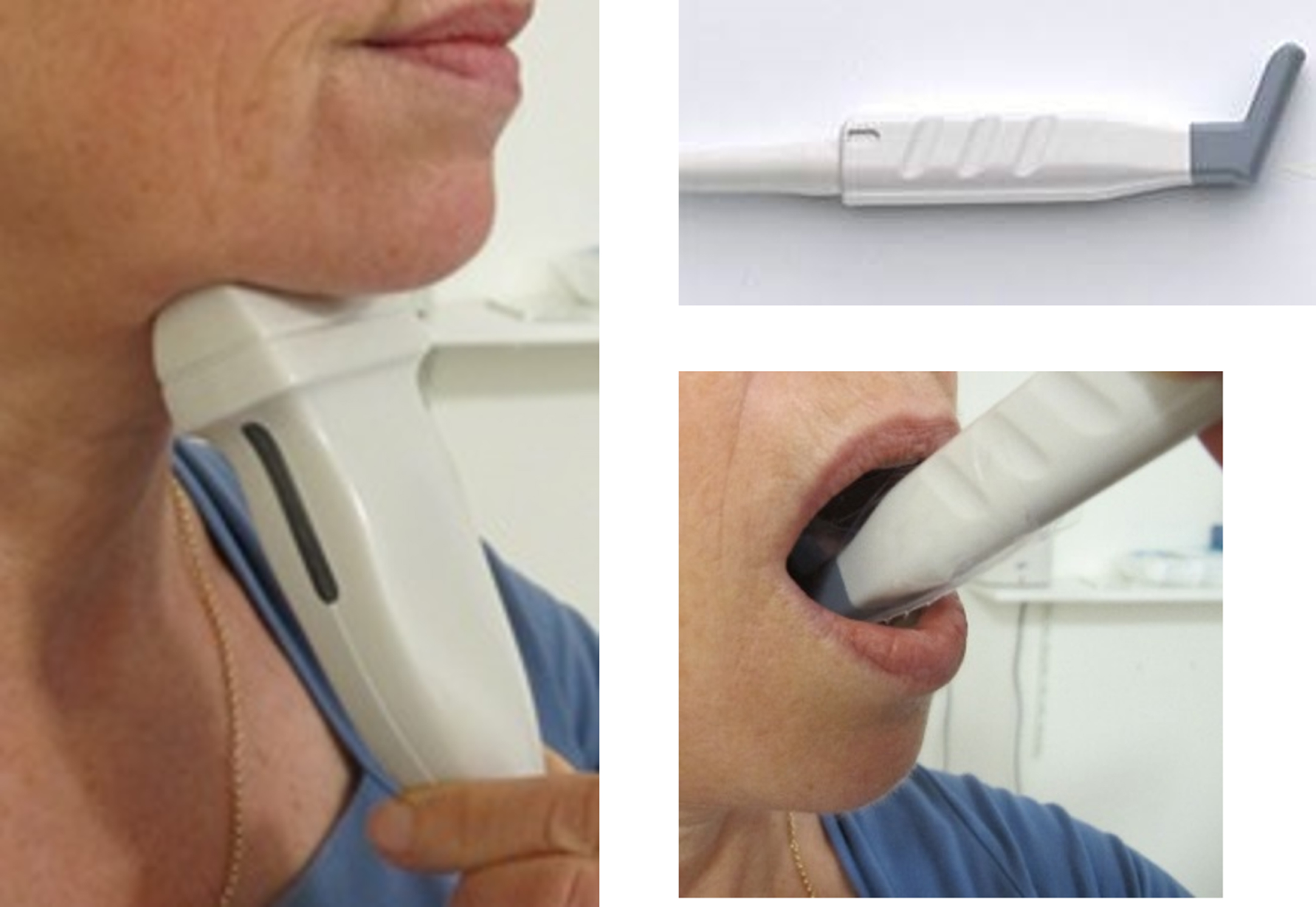
Muscle ultrasound of the submental and tongue muscles. A 12-5 MHz transducer was used for the submental muscles (i.e. digastric, and geniohyoid muscles) (left); a 14-5 MHz transducer (hockey stick) was used for the tongue muscles (right).
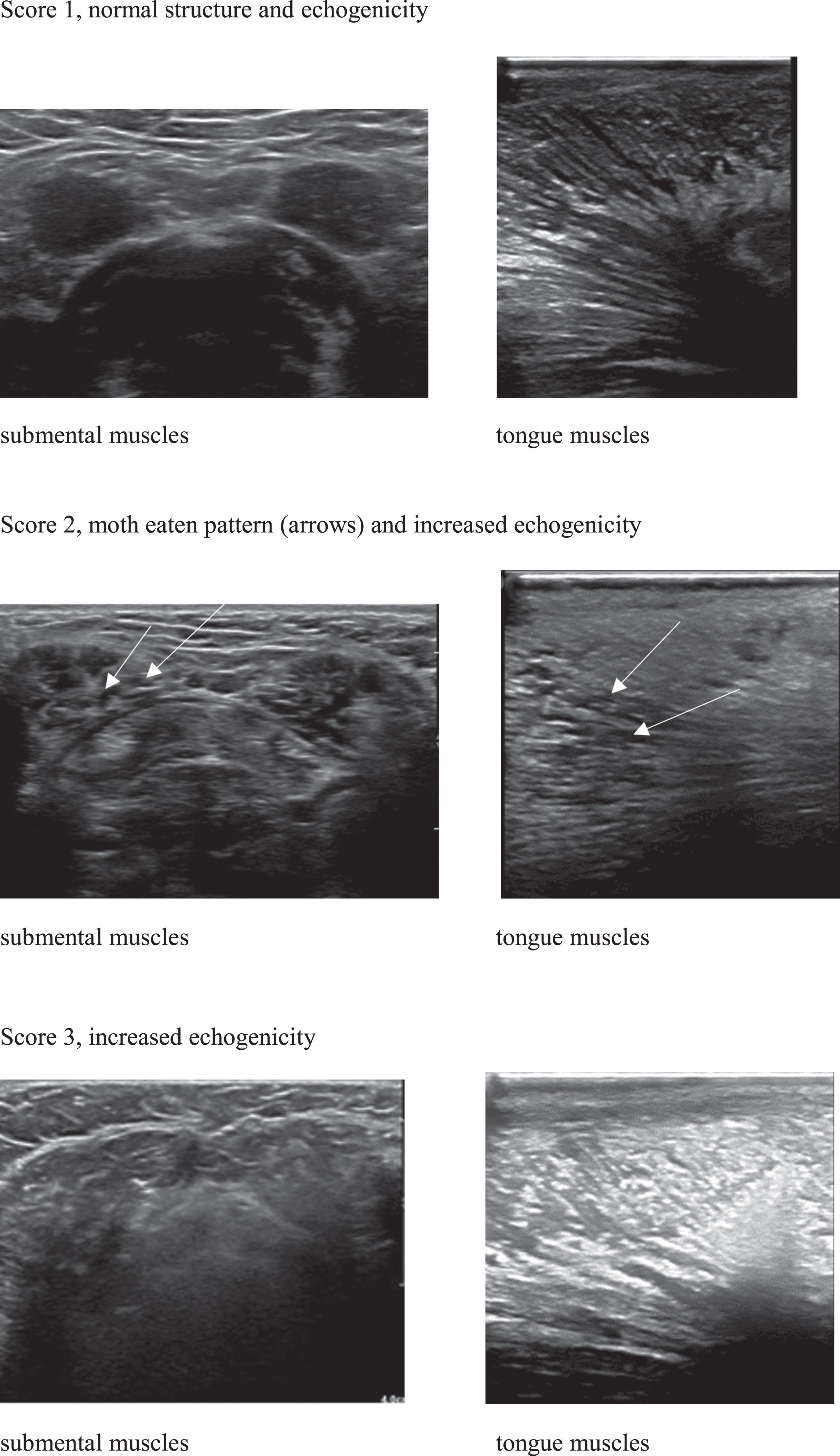
Muscle ultrasound images of the submental and tongue muscles, with normal echogenicity (score 1), increased echogenicity with an inhomogeneous, moth-eaten pattern (arrows) (score 2), and increased echogenicity, where muscle outline fade and disappear (score 3).
Relation between swallowing and mastication
To investigate the relation between swallowing and mastication problems, we used the DL test, and the test of mastication and swallowing solids (TOMASS) [35], described in a previous study [16]. For this test the patient eats a standardized cracker as fast as possible. We used the total time (TT) in seconds needed to finish the cracker as an outcome measure of efficiency of mastication.
Statistical analysis
We used descriptive statistics for patient characteristics, data of the questionnaire, DL, TTS, VFSS and muscle ultrasound (median (range), n (%), or median (IQR). The results of the questionnaire, clinical and instrumental tests were stratified according to the level of motor function: non-ambulant versus ambulant patients.
For analysis, we used the highest (i.e. worst) scores of the BRS, PAS and the number of swallows for each bolus for analysis. Associations between the PAS scores (dichotomized < /≥3) for IDDSI level 0 and questionnaire’s items were analyzed with a Fisher exact test for categorical data. For the relation between swallowing problems and mastication problems we used the DL (ml), and the TOMASS, (total time (sec) needed to finish the cracker), and calculated a Spearman’s correlation.
A p-value of <0.05 was considered as significant. Data were analyzed using SPSS 26 (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 26.0. Armonk, NY: IBM Corp).
RESULTS
Table 1 summarizes patient characteristics.
Patient characteristics
Abbreviations: SMA, spinal muscular atrophy; F, female; M, male; n, number of patients; aNon-invasive ventilation: ventilation support provided via a face mask. Patients used non-invasive ventilation during the night. b Invasive ventilation: positive air pressure support provided via a tracheostomy. The patient used invasive ventilation, with inspiratory and expiratory pressure during day and night; aMMO, active maximum mouth opening; n/a: not applicable. cPatients with SMA type 2 are non-ambulant; FOIS, functional oral intake scale.
Questionnaire
The most common swallowing problem of the non-ambulant patients (n = 24) was difficulty swallowing solid food (92%). The feeling of food sticking in the throat was reported by 18 of 24 patients (75%). None of the patients reported problems swallowing liquids exclusively. Impaired swallowing safety was captured by ‘choking more than once a day’, ‘coughing with solid foods’, and ‘coughing with liquids’. These complaints were reported by, respectively, 25% 29% and 29% of the patients. Twenty-one patients (88%) managed swallowing and/or mastication problems by dietary modification (i.e. pureeing solid food or cutting it into small pieces, or avoiding certain kinds of food). Half of the patients reported mealtimes longer than 30 minutes (54%).
Two of three ambulant patients reported the feeling of food sticking in the throat. These patients did not mention other swallowing problems, nor did they report modified diets or mealtimes >30 minutes.
Clinical swallowing tests
Seventeen non-ambulant patients (71%) had an abnormal DL (i.e. <20 ml). The median DL was 13 ml (3–45). The TTS of non-ambulant patients was at the lower limit of normal, with a median swallowing speed of 10 ml/sec (range 4–25 ml). Eight of 12 patients (67%) had a TTS of >10 ml, and four patients (33%) <10 ml.
The ambulant patients had a median DL of 40 ml (range 20–45) and a TTS of 17 ml/sec (range 14–20).
Videofluoroscopic swallowing study
The results of VFSS are presented in Table 2.
Results of the VFSS of patients with SMA type 2 (n = 18) and 3 (n = 9): number of swallows (I), bolus residue scale (II) pharyngo-oral regurgitation (III), penetration-aspiration scale (IV) when swallowing a standardized bolus for IDDSI level 0 = thin liquid, IDDSI level 3 = moderately thick liquid, IDDSI level 6 = soft & bite-sized food
Values are expressed as median (Inter Quartile Range), or n (%). VFSS: videofluoroscopic swallowing study. IDDSI: International Dysphagia Diet Initiative. Number of swallows of a standardized bolus: the number of swallows needed to swallow 10 ml IDDSI level 0 (thin liquid), 10 ml IDDSI level 3 (moderately thick) and 2×2×0.5 cm IDDSI level 6 (soft and bite sized). Penetration-aspiration scale Rosenbek: scale to determine by which extend material enters the airway, and by whether the material entering the airway is ejected. Bolus residue scale: scale to classify pharyngeal residue in the valleculae, piriform sinuses and/or posterior pharyngeal wall. Pharyngo-oral regurgitation: the transport of the bolus from the hypo-pharynx back into the oral cavity again, before it is re-swallowed. Table cells with –refer to: no patients with this score. *1 missing. a1 ambulant patient, b2 ambulant patients, c3 ambulant patients d1 non-ambulant patient aspirated on pharyngeal residue of the previous bolus.
Piecemeal deglutition was observed in non-ambulant patients with thin liquid (79%), moderately thick liquid (33%) and soft solid food (33%). In most non-ambulant patients, there was pharyngeal residue following ingestion of thin liquid (79%), moderately thick liquid (88%) and soft solid food (22%). A striking finding was pharyngo-oral regurgitation (i.e. transporting the bolus from the hypopharynx back in the oral cavity and swallowing again) in 14 non-ambulant patients (58%), almost all on IDDSI level 7. Five of these patients had pharyngo-oral regurgitation with more than one consistency. Impaired swallowing safety (i.e. Pas >3) was observed in 6 non-ambulant patients (25%) with SMA type 2. Three of them had laryngeal penetration and three patients had aspiration. In five out of six patients, penetrated or aspirated material was not ejected from the airway. Two of these six non-ambulant patients aspirated also on pharyngeal residue of the previous boluses when swallowing soft solid food (Table 2).
Ambulant patients demonstrated no piecemeal deglutition but two of three patients had residue in the valleculae or pyriform sinus. There was no laryngeal penetration, aspiration or pharyngo-oral regurgitation in the ambulant patients (Table 2).
Muscle ultrasound
Eighteen non-ambulant patients (75%) had increased echogenicity of the submental muscles. The ultrasound had an inhomogeneous moth-eaten pattern. The echogenicity of the tongue was increased in seventeen patients (71%), and in nine of these, the ultrasound revealed a moth-eaten pattern. In seven patients, the echogenicity of the tongue could not be determined, as it was impossible to place the transducer on the tongue, due to a limited maximum mouth opening.
Ambulant patients had normal echogenicity of the submental muscles, and two of three patients had increased echogenicity of the tongue (one with a moth-eaten pattern).
Relation between VFSS and the questionnaire
Pas scores >3 were significantly associated with ‘daily use of antibiotics’ (p = 0.011) and ‘coughing with liquids (p = 0.001), but was not significantly associated with ‘choking’ (p = 0.588).
Relation between swallowing and mastication problems
The correlation between swallowing and mastication problems was significant (rs = –0.62, p = 0.001). An increase in the time needed to finish a standardized cracker was associated with a decrease of DL (Fig. 3).
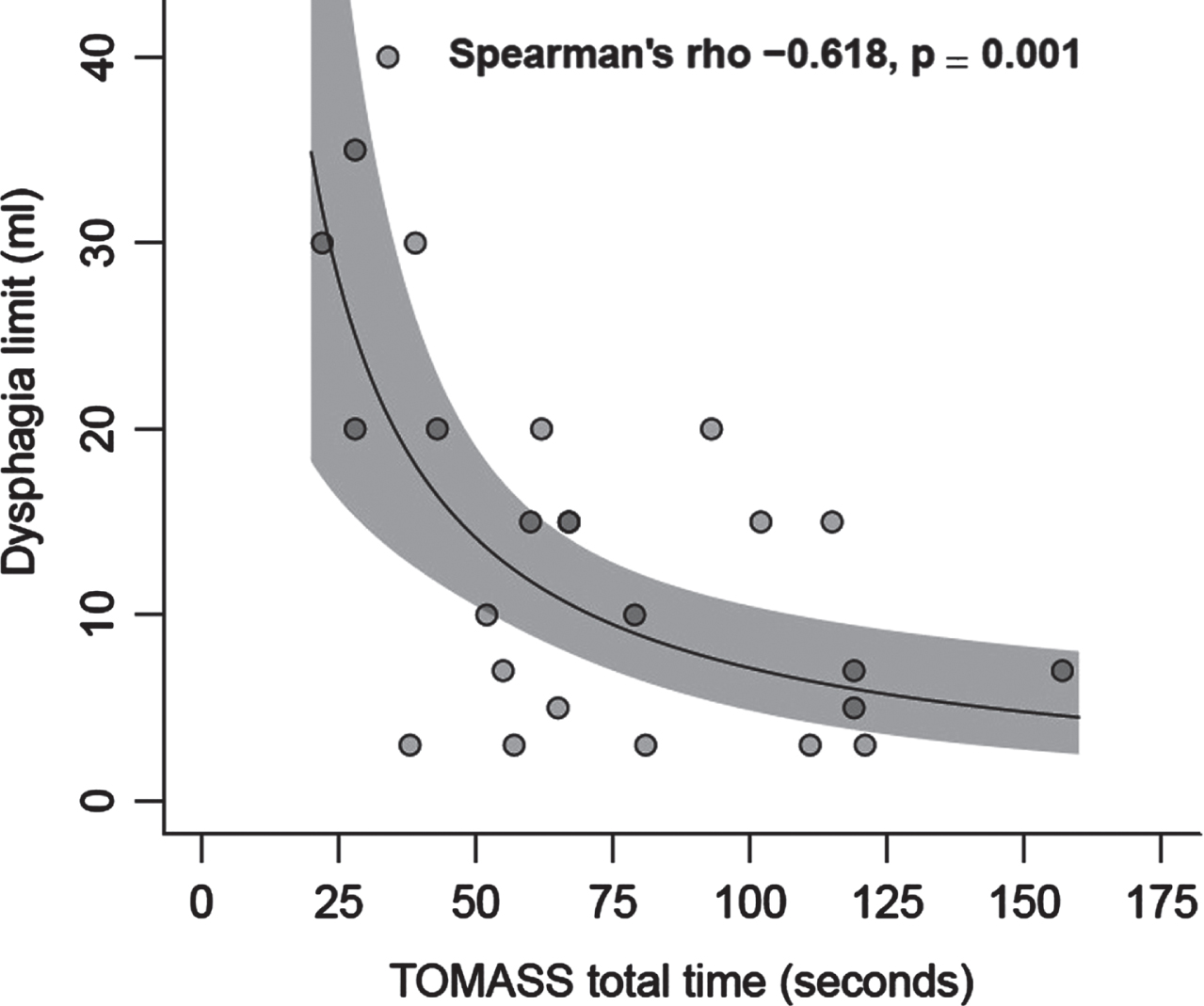
Correlation between swallowing and mastication problems, using DL (ml) and TOMASS (total time in sec).
DISCUSSION
This study provides insight into characteristics of swallowing problems in adolescent and adult patients with SMA types 2 and 3. Almost all non-ambulant patients swallowed inefficiently as reflected by piecemeal deglutition, pharyngeal residue, and pharyngo-oral regurgitation after stagnation in the hypopharynx. Only patients with SMA type 2 had impaired swallowing safety. Ambulant patients demonstrated no piecemeal deglutition or pharyngo-oral regurgitation, but did have pharyngeal residue after swallowing. We cannot exclude that patients with SMA type 3 will eventually develop significant symptoms of swallowing problems later in life. Investigation of more and older patients will probably clarify this.
Our results indicate that pharyngeal residue is common across the severity spectrum of SMA type 2 and even type 3, confirming the previous study in 6 pediatric patients with SMA type 2 [15]. Studies in healthy participants, found minimal pharyngeal residue and a significant association between pharyngeal residue and indirect aspiration on subsequent clearing swallows [28, 37]. Patients in our study could not clear post swallow residue, by performing a dry swallow (i.e. a voluntary swallow on request, unstimulated by a bolus) or by swallowing sips of water. Most patients were not aware of pharyngo-oral regurgitation and we observed no coughing. Pharyngo-oral regurgitation has not yet been described in patients with SMA, but a previous study by Hanayama et al, delineated this phenomenon in children and young adults with Duchenne muscular dystrophy [38]. They reported ‘difficulty passing the bolus through the cricopharyngeal muscle, combined with some pressure to regurgitate the bolus back into the oral cavity’. We hypothesize that this mechanism is a compensation strategy as a response to bulbar muscle weakness which is described in patients with SMA, [13, 39]. It affects the propulsion of the pharyngeal wall, elevation and forward movement of the hyoid, and opening of the cricopharyngeal muscle during swallowing. Pharyngo-oral regurgitation especially occurred in patients who demonstrated piecemeal deglutition, suggesting that dividing the bolus into small particles, was not a sufficient compensation mechanism.
Muscle ultrasound of bulbar muscles revealed an abnormal muscle structure with a characteristic moth-eaten pattern. Increased echogenicity on muscle ultrasound is caused by replacement of muscle tissue by fatty infiltration in bulbar muscles [14, 33]. The moth-eaten pattern consists of dark areas, described as viable motor units as reported by Wijntjes, and areas with increased echogenicity, reflecting permanent denervation andfibrosis [40].
To detect impaired swallowing safety, the questionnaire proved to be helpful. The items ‘do you cough when swallowing liquids’ and ‘do you use antibiotics’ corresponded best with unsafe swallowing results of the VFSS (i.e. PAS >3). Affirmative replies to both questions are a clear indication for a VFSS. We think that VFSS should be used not only in case of coughing while drinking, but also when patients experience difficulty swallowing certain kinds of food, cough while eating solids, or when they have the feeling of food sticking in the throat’. We also advocate the use of clinical swallowing tests and muscle ultrasound in case of reported swallowing problems. It is necessary to prepare the patient for the possible outcomes of the assessments, for example, the occurrence of aspiration, or that eating solid food is too demanding for the patient and might bring the possibility of pharyngeal residue. Through shared decision making, the professional and patient discuss adaptations with the aim to establish and sustain the feasibility of oral feeding [41].
In this patient group, both swallowing and mastication function were investigated. Mastication problems were characterized by abnormal masticatory efficiency, reduced endurance and fatigue [16]. Not surprisingly, mastication problems occurred relatively often - in 60% of the patients –in combination with swallowing problems, and may, therefore, be an indication for a swallowing assessment and vice versa.
Our findings have practical implications for patients. In this study, more than 2/3 of the patients had residue in the valleculae, which is associated with impaired swallowing safety on the subsequent clearing swallow [37]. Since we do not know how long pharyngeal residue remains in the hypopharynx after swallowing, nor which method is best for clearing pharyngeal residue, it is recommended not to use coughing techniques (for instance a coughing machine) after mealtime, as these techniques involve deep inhalation and may cause aspiration of residue. A previous study in patients with SMA (using VFSS and sEMG), revealed more muscle strength when swallowing, and less post swallow residue with the head in a slightly bowed position [15]. It is probably best to correct a retracted head position during swallowing, if necessary with a headband, by moving the chin slightly downward and forward, in order to minimize a biomechanical contribution to swallowing difficulties.
In patients with neuromuscular diseases, we should be cautious about thickening liquids because it requires relatively more strength to swallow thick liquids [42]. VFSS can be used to determine the correct level of thickness for safe swallowing.
This study has some limitations. A small number of ambulant patients participated in the study. This can be explained by the inclusion criterion of having bulbar problems. Another limitation is that during VFSS, we used for technical reasons a pulse rate of 15 instead of 30 per second. It is possible that we missed milder forms of laryngeal penetration or aspiration. Finally, we did not rate the volume of pharyngeal residue. However, it has been found that BRS was significantly correlated to objective impedance manometry, and that a high BRS score was associated with a higher risk of aspiration [29, 44].
Future research should aim to investigate the results of pharyngo-oral regurgitation in a larger group of patients. Further studies should also involve the effect of a corrected head position on the amount of post swallow residue. We realize that the current assessment protocol did not capture muscle fatigability related to swallowing. Nearly half of the patients in this study had mealtimes longer than 30 minutes. It is not inconceivable that fatigability negatively affects the performance of swallowing during the course of a meal. Therefore, the relation between muscle fatigability and swallowing is a subject for future research.
CONCLUSION
In patients with SMA, who report swallowing and/or mastication problems, swallowing problems are characterized by inefficient and sometimes impaired swallowing safety, caused by widespread abnormalities of bulbar muscle structure. Ambulant patients clearly have a better bulbar function compared to non-ambulant patients, but they also show functional and structural abnormalities. These results help to understand the nature of swallowing disturbances in SMA. In this study almost all patients with SMA, who reported problems with eating or drinking, demonstrated disordered swallowing. This is important information for patients who experience swallowing problems, as they can seek help and become properly advised.
FUNDING
This study was supported by research grants from the Prinses Beatrix Spierfonds, WOB 14–25, by Stichting Spieren voor Spieren and Vriendenloterij.
ROLE OF THE FUNDING SOURCE
The funding source that provided support for the conduct of the research had no role in study design; in the collection, design, and interpretation of the data; in the writing of the report; and in the decision to submit the article for publication.
Footnotes
ACKNOWLEDGMENTS
The authors thank all patients who participated in this study.
AUTHOR CONTRIBUTIONS
AMBH, LEH, WLP designed the study AMBH, LEH and WLP contributed to the drafting of the manuscript AMBH, LEH, FA participated in the data collection AMBH and RPAE performed the statistical analysis AMBH, LEH, WLP, RAJN, RPAE, FA, CEE, IC, AJNB, and EG contributed to the interpretation of the data. All authors critically reviewed and revised the manuscript
DECLARATIONS
CONFLICT OF INTERESTS
AMBH, RAJN, RPAE, FA, CEE, IC, AJNB, EG, LEH declare no financial or other conflicts of interest WLP receives research support from the Prinses Beatrix Spierfonds, Stichting Spieren voor Spieren and Vriendenloterij. His employer receives fees for ad hoc consultancy services to Biogen, Avexis (member scientific advisory board), and Novartis (member data-monitoring committee).
