Abstract
Background
In myasthenia gravis, impaired postsynaptic sensitivity to acetylcholine results in failure of neuromuscular transmission and fatiguing muscle weakness.
Objective
Develop an ex vivo muscle contraction assay to test cannabinoids and other substances that might act on the myasthenic neuromuscular junction to restore control of the muscle.
Methods
Tubocurarine was added to an ex vivo, mouse phrenic nerve-hemidiaphragm muscle preparation to reduce acetylcholine sensitivity. This produced a myasthenia-like decrement in twitch force during a train of 10 nerve impulses (3 / sec). Endplate potential (EPP) recordings were used to confirm and extend the findings.
Results
Surprisingly, addition to the bath of dimethylsulphoxide (DMSO), at concentrations as low as 0.1%(v/v), partially reversed the decrement in nerve-evoked force. Intracellular electrophysiology, conducted in the presence of tubocurarine, showed that DMSO increased the amplitudes of both the spontaneous miniature EPP (MEPP) and the (nerve-evoked) EPP. In the absence of tubocurarine (synaptic potentials at physiological levels), an adaptive fall in quantal content negated the DMSO-induced rise in EPP amplitude. The effects of cannabinoid receptor agonists (solubilized with DMSO) in the contraction assay do not support their further exploration as useful therapeutic agents for myasthenia gravis. CP 55,940 (a dual agonist for cannabinoid receptor types 1 and 2) reversed the beneficial effects of DMSO.
Conclusions:
We demonstrate a powerful effect of DMSO upon quantal amplitude that might mislead pharmacological studies of synaptic function wherever DMSO is used as a drug vehicle. Our results also show that compounds targeting impaired neuromuscular transmission should be tested under myasthenic-like conditions, so as to avoid confounding effects of synaptic homeostasis.
Keywords
INTRODUCTION
Myasthenia gravis is an autoimmune disease characterized by acute and severe fatiguing muscle weakness resulting from failure of neuromuscular transmission [1, 2]. Most cases are caused by autoantibody-mediated disruption of postsynaptic elements of the motor endplate [3–5]. Current treatment options for myasthenia gravis include steroids and other drugs that neutralise or suppress production of autoantibodies as well as drugs, such as acetylcholinesterase inhibitors, that act at the neuromuscular junction (NMJ) to restore synaptic function [6, 7]. Combinations of these therapies have been effective in reducing mortality for most myasthenic patients, but their prolonged use over time is associated with development of adverse side effects [8]. Moreover, approximately 10%of patients with myasthenia gravis are classified as ‘refractory’, failing to respond to available treatments, or responding inappropriately [9]. In particular, cholinesterase inhibitors can be ineffective or even harmful for some forms of myasthenia [7, 10]. New symptomatic drugs that target the NMJ are needed to increase the treatment options for myasthenia patients and their physicians.
The development of new drugs targeting the NMJ requires a quick and robust assay to screen lead compounds. Active immunization and passive IgG transfer rodent models of myasthenia gravis permit late-phase preclinical testing of candidate drugs [11–13], but they are not suitable for the earlier phase when multiple compounds must be tested. Ex vivo endplate potential (EPP) recordings can provide detailed information about the synaptic effects of a compound, but they are very labour-intensive and impractical for testing multiple drugs. Isolated phrenic nerve-diaphragm preparations have been used extensively in the past to test the effects of substances on muscle contraction [14–17]. The preparation has also been used to successfully elucidate mechanisms of neuromuscular diseases [18, 19] but this has usually involved tissue from animal models of disease, the maintenance of which often raise ethical concerns. These considerations point to the need for new, fast, accessible and reproducible assays to test drugs for their ability to restore failing neuromuscular transmission. Such assays should take account of the physiology and pathophysiology of neuromuscular transmission.
The safety factor for neuromuscular transmission is a measure of the degree to which the EPP is more than sufficient to trigger an action potential in the muscle fibre [20]. At the myasthenic NMJ autoantibodies suppress the postsynaptic response to each vesicle-load of acetylcholine (the quantal amplitude). During trains of nerve impulses, the number of vesicles of acetylcholine released in response to each impulse (the quantal content) declines naturally with each successive nerve impulse [21]. Since the EPP is the product of the quantal amplitude and quantal content, the safety factor becomes marginal, with the postsynaptic action potential often failing during the latter impulses of a train [22]. The reduced quantal amplitude triggers an adaptive (presynaptic) increase in quantal content [23, 24]. Recent work suggests that this involves a rapid homeostatic response: attempting to maintain the EPP amplitude and the safety factor [25–27]. By the time weakness becomes evident in myasthenic muscles, the adaptive increase in quantal content has already been challenged to the limits of its homeostatic capacity. Candidate therapeutics should therefore be tested in an assay where the parameters of quantal synaptic transmission are likewise modified. One way to mimic the reduced safety factor associated with myasthenia gravis is by partially blocking the activation of postsynaptic acetylcholine receptors using tubocurarine [28].
Recent work has suggested that synthetic cannabinoids might have potential as a novel therapeutic strategy for myasthenia gravis. Agonists of cannabinoid receptor 1 (CB1R; a G-protein coupled receptor) have been variously reported to modify either quantal content or quantal amplitude, or both (for a review see [29]). In a recent exploratory study, we reported increases in the amplitudes of both the MEPP and EPP after a synthetic cannabinoid receptor agonist, WIN 55,212-2, was added to the mouse phrenic nerve-hemidiaphragm muscle preparation. The increased quantal amplitude was explained by an increase in the diameter/volume of the synaptic vesicles [30]. Here we describe an ex vivo, contraction-based assay which can assess the potential of cannabinoids, and other substances, to restore the safety factor for neuromuscular transmission in a myasthenic context without the need to generate disease model animals. Cannabinoids are very hydrophobic, requiring substantial volumes of dimethylsulphoxide (DMSO), or organic solvents, such as ethanol, to dissolve in aqueous media. Using our contraction assay and follow-up electrophysiological recordings we report an unexpected, strong effect of the DMSO upon the safety factor for neuromuscular transmission, attributable to an increase in quantal amplitude. Our results reveal the potential confounding effects of solvents and synaptic homeostasis in studies of synaptic function.
MATERIALS AND METHODS
Mouse husbandry and ethics
All mouse experiments described in this paper were conducted with the approval of The University of Sydney Animal Ethics Committee (approval number 2017/1153) in compliance with the NSW Animal Research Act 1985 and the Australian Code of Practice for the Care and Use of Animals for Scientific purposes. Female C57BL/6 mice were obtained from the Animal Resources Centre (Western Australia), were group housed in individually ventilated cages and were fed ad libitum with continuous access to water. Mice (6-7 weeks old) were killed by intraperitoneal injection of pentobarbital (32.5 mg) to provide tissue on the day of the experiment. Both hemidiaphragms from a single mouse were used in order to simultaneously run electrophysiological and muscle contraction assays. No specific preference for left or right hemidiaphragms was taken for either experiment. A total of 90 mice were used in this study.
Electrophysiological recordings
For EPP recordings, hemidiaphragm muscle with attached phrenic nerve were rapidly dissected and pinned onto Sylgard-coated dishes under Ringer’s solution (136 mM NaCl, 5 mM KCl, 1 mM NaH2PO4, 12.8 mM NaHCO3, 10 mM Glucose, 1 mM MgCl2 and 1 mM CaCl2 bubbled with 95%oxygen / 5%CO2). The concentration of CaCl2 (1 mM) used for both EPP and contraction recordings was lower than the 2 mM used in our previous studies (see Discussion). Muscle action potentials were blocked by adding 0.5–1μM μ-Conotoxin GIIIb (Peptide Institute, Japan) to the bath solution 1 hr before beginning the recordings. The phrenic nerve was sucked into a glass pipette attached to a DS2A Mk II isolated voltage stimulator (Digitimer, Welwyn Garden City, UK). Muscle fibres were impaled with sharp borosilicate glass electrodes filled with 3 M KCl. A Model 1600 Neuroprobe amplifier (A-M Systems, Elkhart IN, USA) and Powerlab 2/20 with LabChart7 software (AD Instruments, Bella Vista, NSW Australia; 1 kHz low-pass filter) were used to record postsynaptic potentials. Recordings from a fibre were only included in the analysis if: 1) resting membrane potential (RMP) was more negative than –60 mV, 2) fluctuations in the RMP remained within a 2 mV range over an initial 1-minute recording period and, 3) observable MEPPs had a rise time of < 3 ms. For each impalement, MEPPs were recorded for 1 minute, followed by EPPs evoked by supra-maximal nerve stimulation (45 pulses at 1 Hz), followed by 60 stimuli at 40 Hz. After completing pre-treatment recordings, the pinned hemi-diaphragms were rinsed and then reincubated for 2 hr in fresh oxygenated Ringer’s solution containing μ-Conotoxin GIIIb (as above) and the treatment of interest. Post-treatment recordings then repeated the pre-treatment protocol. For each hemidiaphragm muscle/experiment, recordings were sampled from 10–12 muscle fibres, pre- and post-treatment, and the average values obtained were compared.
Quantification of MEPPs and EPPs was performed using semi-automated methods in LabChart7. The ‘Cyclic Measurement’ function was used to identify and mark MEPP and EPP events in each trace with detection settings set to LabChart’s intrinsic ‘Sine shape’ option. The detection threshold was set and confirmed manually for each trace to account for differences in signal-to-noise ratios. LabChart’s data pad was then used to identify MEPP and EPP events using markers set by the ‘Cyclic Measurement’ function and voltage values were gathered at the base and peak of each event. The amplitudes of the MEPP or EPP were based upon these values. For each fibre MEPP and EPP amplitudes were normalised to a resting potential of –80 mV. Values were not corrected for non-linear summation, because the factors underlying Martin’s correction, f, have not been comprehensively determined in the context of our experimental preparation, and incorrect usage of Martin’s correction can lead to improper representation of EPP amplitude values [31–33]. Our decision to not correct should be kept in mind when comparing our results to those of others who did employ Martin’s correction. When used, the correction typically increases reported EPP values by approximately 20%at mouse neuromuscular junctions [30]. For each individual muscle fibre, quantal content was calculated by dividing the average MEPP amplitude by average EPP amplitude.
Muscle contraction assay
The method outlined in Zanetti et al. (2018) [15] was used to prepare the phrenic nerve and attached hemidiaphragm muscle for contraction recordings, with the following modifications. The dissected hemidiaphragm was attached to a force transducer (Aurora Scientific, Aurora ON, Canada) using a hook passed through the central tendon. The preparation was bathed in Ringer’s solution (composition as described above) at room temperature. The phrenic nerve was stimulated via two platinum wires, attached to an S88X dual output stimulator (Grass Instruments, Astro-Med, West Warwick RI, USA). The muscle was adjusted to the optimal length for twitch contractions. The nerve was then subjected to trains of supra-maximal stimuli (10 pulses at 3 / sec) at 90-second intervals. Contractions were recorded continually. Each experiment consisted of the following sequence: 1) pre-treatment contractions recorded for at least 1 hour; 2) tubocurarine chloride added to the bath (final concentration 700 nM; Sigma St Louis, MO, USA) with recordings continued for at least another 1.5 hours; 3) treatment of interest added into the bath with recordings continued for at least another 2.5 hours; 4) neostigmine added to the bath (final concentration 500 nM) with recordings continued for 30 minutes. The tubocurarine-induced failure of neuromuscular transmission during each train of 10 stimuli was estimated by measuring the decay in the twitch amplitude during each train of nerve stimuli (Fig. 1A). Twitch amplitude was quantified using LabChart’s ‘Cyclic Measurement’ function as outlined above for EPPs. To assess force decrement, the twitch force after each stimulus of the 10-pulse train was expressed as a fraction of the force from the first pulse of the same train. The fractional drop was averaged for the last three trains recorded at time points: T3, T4 or T5 (Fig. 2A). The effect of a treatment was expressed as the percentage restoration of force at Tn relative to T2 using the following formula.
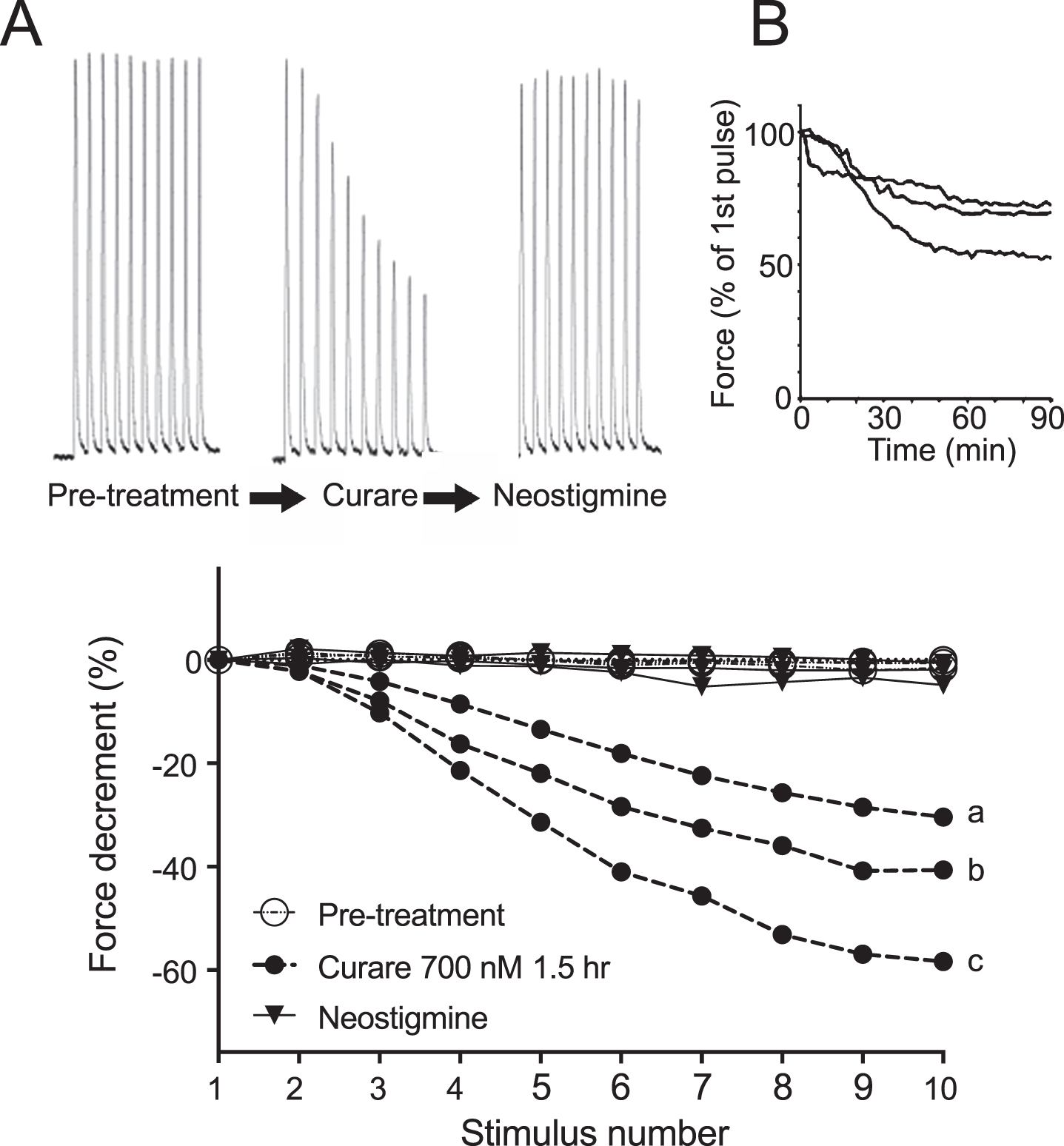
Decrement in nerve-evoked force after applying tubocurarine to the isolated phrenic nerve-hemidiaphragm muscle preparation. (A) Representative force transducer recordings show twitch contractions evoked by trains of 10 nerve stimuli (3 / sec), before (Pre-treatment) and after addition of tubocurarine (curare) and subsequently, neostigmine. (B) Onset of the force decrement with time after addition of curare. The force of the tenth twitch of each train is expressed as a percentage of the first twitch of the same train. Traces show results from three muscle preparations. (C) Curare-induced decrement of force during a train of 10 impulses. Symbols show steady pre-treatment twitch force (open circles), decrement 90 min after application of curare (filled circles showing 3 preparations: a, b and c) and force restoration after further addition of neostigmine (filled triangles).
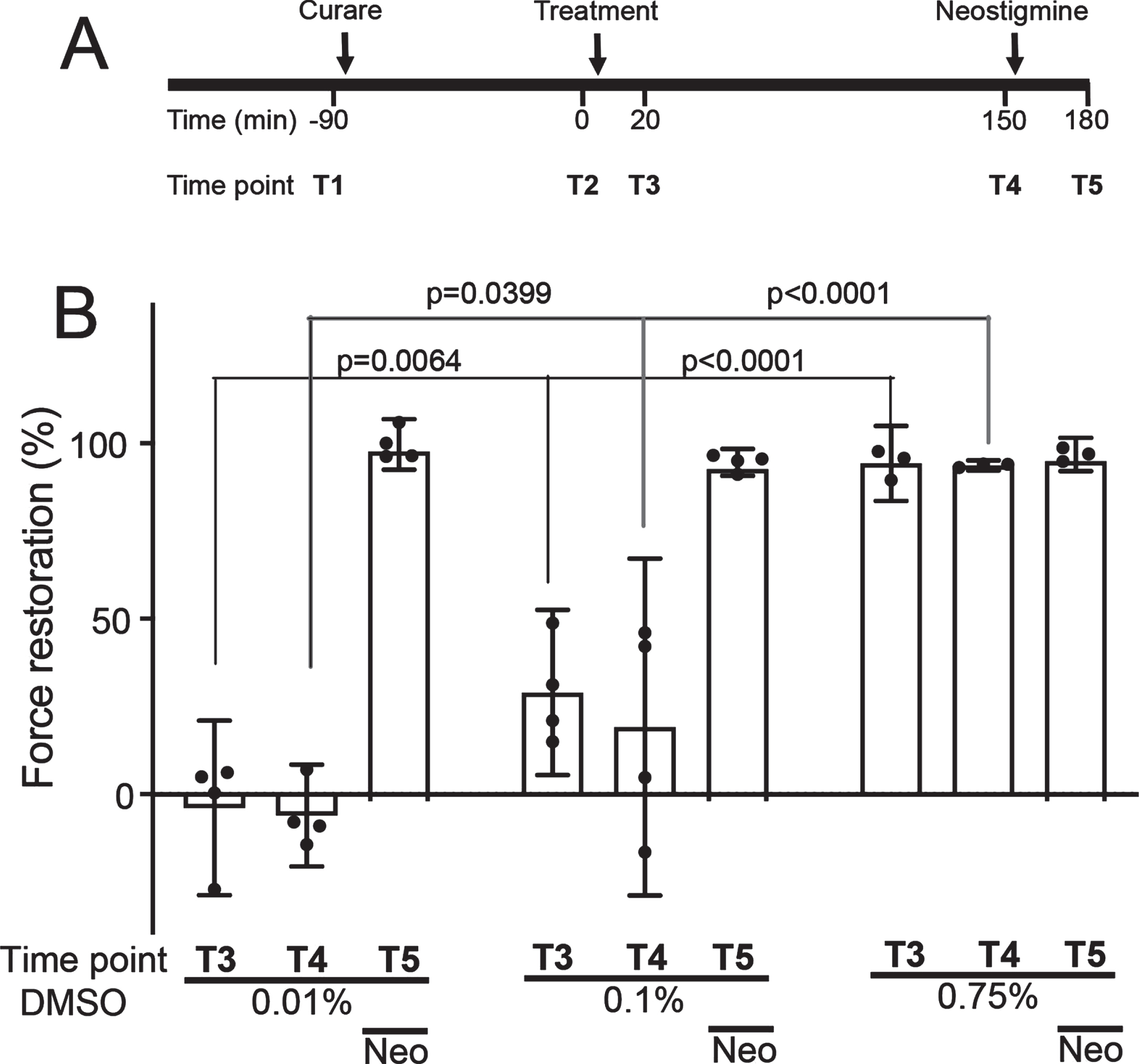
DMSO restores nerve-evoked contraction to curarized muscles. (A) Timeline for a standard ex vivo muscle contraction assay. Pre-treatment contraction trains were recorded at time point T1, immediately before adding 700 nM tubocurarine (curare) to the bath. The curare-induced decrement was quantified 90 min later (T2), immediately before addition of the experimental treatment (time zero). The effect of the treatment upon the curare-induced force decrement was quantified 20 min and 150 min later (T3 and T4). Neostigmine was then added and reversal of the force decrement was quantified at 180 min (T5). (B) Recovery from the force decrement after treatment with DMSO. Force restoration specifies the extent to which the force of the tenth twitch of the train was restored to the force of the first twitch of the same train (see Methods). Results are shown for the T3 and T4 time points (20 min and 150 min after addition of the indicated concentration of DMSO). The T5 time-point column demonstrates restoration of force after further addition of neostigmine to the preparation. Error bars show mean and 95%confidence interval for n = 4 muscle preparations/mice. Symbols show results for individual preparations. P-values are the result of mixed repeated measures two-way ANOVA with Tukey’s multiple comparisons post-test for DMSO concentration (simple effect).
Force restoration at time Tn (%) = ((Tn-T2)/(T1-T2))×100.
The absolute force of the first twitch of the train was not affected by tubocurarine. For example: 10.7±2.6 mN (mean±SD) before curare, 10.6±3.3 mN after curare (p = 0.81 paired student’s t-test, n = 3; also see Supplementary Table 1).
Drug preparation
WIN 55,212-2, CP 55,940, ACEA, HU-308 and AM-251 were obtained from Tocris Bioscience (Bristol UK) and were stored at –20 °C as 100 mM stocks in DMSO, with the exception of CP 55,940, which was stored in DMSO at 10 mM and AM-251, in DMSO at 50 mM. Cannabinoid/DMSO stocks were diluted into Ringer’s solution in a 10 ml polypropylene conical tube, which was vortexed vigorously before application to the experimental preparation. Cannabinoids dissolved at or above 9500 PPM in DMSO precipitated when diluted into Ringer’s solution, despite vortexing (Supplementary Figure 1). For this reason, all the cannabinoid stock solutions used in the experiments reported here contained less than 9500 PPM of the cannabinoid compound (concentrations are specified in the text).
Statistical analyses
Graphpad Prism 7 was used for statistical analysis and generation of graphs. Data was checked for normality using a Shapiro-Wilk test. Where data was normally distributed, a two-tailed, paired Student’s t-test was used to compare post-treatment and pre-treatment mean values, otherwise Wilcoxon matched pairs signed rank tests were used (as indicated). To compare more than two groups, where data was normally distributed, one-way ANOVA with Dunnett’s multiple-comparisons post-test was used. Otherwise, Friedman tests with Dunn’s post-test were used. Data involving two independent variables was analysed with two-way ANOVA and Tukey’s post-test for multiple comparisons. Significance was determined at p < 0.05. All error bars represent the mean ± 95%confidence interval. The sample size (n) represents the number of mice/muscles analysed. For each muscle, synaptic potentials were sampled from 10–12 fibres to estimate the mean for the muscle, before and after treatment. MEPP amplitudes were based on the average of all detectable MEPPs within a 1-minute timeframe per muscle fibre whereas MEPP half-times were based on the average of 10 MEPPs per muscle fibre.
RESULTS
Curarized phrenic nerve-hemidiaphragm muscle contraction assay
A simple ex vivo model of myasthenic muscle was established by recording trains of nerve-evoked contractions from a phrenic nerve-hemidiaphragm muscle preparation (10 stimuli at 3 / sec, applied every 90 seconds). Prior to treatment, each stimulus of the train produced a twitch of similar force (Fig. 1A, Pre-treatment). Addition to the bath solution of the acetylcholine receptor antagonist, 700 nM tubocurarine chloride (hereafter referred to as curare), resulted in a decremental response to the train of nerve impulses (Fig. 1A). This was reminiscent of the decrement in the compound muscle action potential recorded from myasthenic muscles [22]. The percentage decrement in force reached a stable plateau value 60–90 minutes after the addition of curare, at which point the effect of curare was quantified (Fig. 1B). Individual muscle preparations varied in the plateau decrement value reached after adding curare (Fig. 1C, filled circles: 43±14%; mean±SD). Addition of the cholinesterase inhibitor, neostigmine (500 nM), at the end of each experiment consistently restored twitch force to pre-treatment values (Fig. 1A&C, triangle symbols), confirming that the curare-induced force decrement reflected a reduced safety factor for neuromuscular transmission [20], rather than muscle fatigue.
The curarized muscle preparation was then used to assess the ability of other compounds to restore the safety factor for neuromuscular transmission. Figure 2A illustrates the timeline for such an experiment. The initial (baseline) force was recorded at time T1. Curare was then added to the bath and resulting decrement in force was assessed 90 minutes later (T2). The test compound was then added to the bath and its effect upon force was assessed after 20 minutes (T3, short-term effect) and 150 minutes (T4, longer-term effect). Finally, neostigmine was added to the bath, and the resulting restoration of force was assessed 30 minutes later (T5).
DMSO can restore nerve-evoked force to curarized diaphragm muscles
We first tested the effects of DMSO, a polar aprotic solvent widely used to solubilize compounds in aqueous media. A range of DMSO final concentrations were tested: 0.01%v/v (equivalent to 1.41 mM), 0.1%, (14.1 mM) and 0.75%(105.8 mM). Addition of DMSO to the bath solution produced a concentration-dependent restoration of nerve-evoked force in the curarized muscles. The lowest concentration (0.01%DMSO) had no effect, 0.1%DMSO produced a partial force restoration (∼20–30%) while 0.75%produced complete force restoration. The effects of adding DMSO were rapid and sustained. The force restoration reached a plateau value within 20 minutes of adding DMSO to the bath and this was sustained over at least 150 minutes (Fig. 2B T3 & T4). These results from our curarized muscle model of myasthenia show that DMSO can produce a rapid, concentration-dependent improvement in the safety factor for neuromuscular transmission.
DMSO increases quantal amplitude at motor endplates
To clarify the effects of DMSO on neuromuscular transmission, muscle fibres were impaled with capillary microelectrodes before, and 2 hours after, addition of DMSO to the bath. Compared to the pre-treatment values, addition of 0.01%DMSO to the bath solution did not produce any detectable changes in MEPP amplitude, EPP amplitude or quantal content (Fig. 3A-C 0.01%paired data sets, Supplementary Table 2). Higher concentrations of DMSO (0.1%and 0.75%) each produced an approximate 30%increase in MEPP amplitude (Fig. 3A). The half-width of the MEPP was not affected by 0.75%DMSO (3.11 ± 0.35 ms pre-treatment vs. 3.04±0.42 ms post-treatment; p = 0.46 paired Wilcoxon test, n = 8). The increased quantal amplitude was opposed by an approximate 20%reduction in quantal content (Fig. 3C), thereby precluding an increase in EPP amplitude, even in response to 0.75%DMSO (before 17.0±2.3 mV and 18.4±4.6 mV after addition, Supplementary Table 2). Whilst it is not possible to directly infer whether DMSO acted on pre- or post-synaptic processes (or both), these results are consistent with the idea that the DMSO-induced increase in quantal amplitude prompted a homeostatic-like reduction in quantal content (see Discussion).
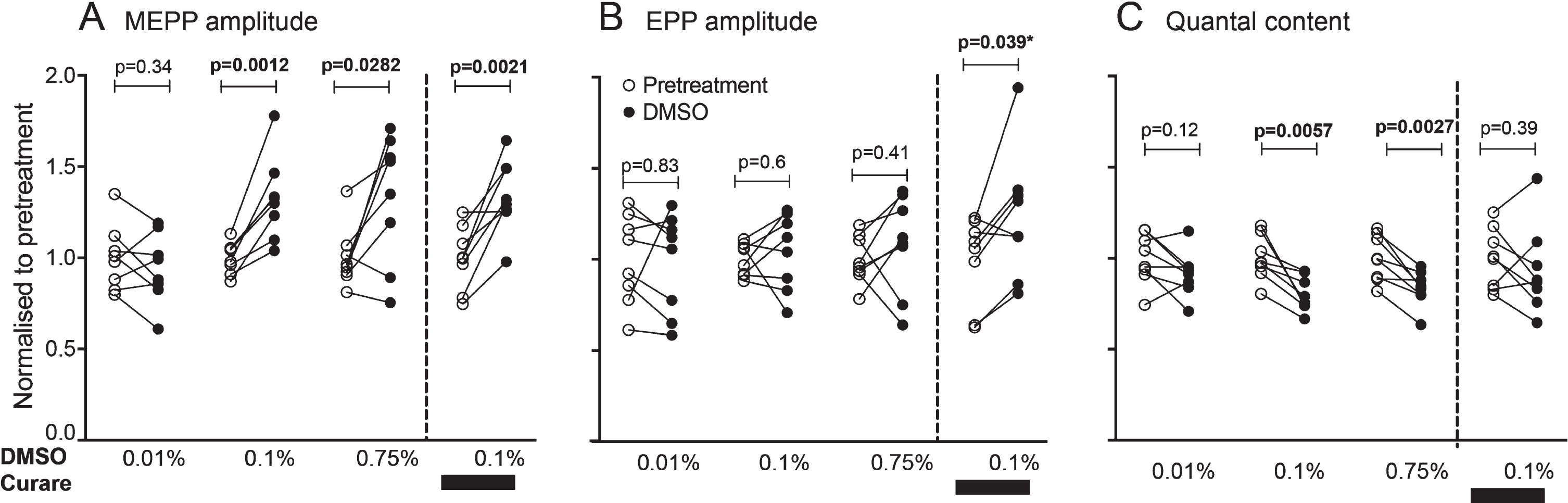
DMSO raises the quantal amplitude for neuromuscular synaptic transmission. (A) Paired comparison of miniature endplate potential (MEPP) amplitudes before (pre-treatment) and after addition of the indicated concentration of DMSO to the electrophysiology bath. Each muscle data point was normalised to the mean of the pre-treatment data (Supplementary Table 2 lists the absolute values recorded). (B) Paired comparison of endplate potential (EPP) amplitudes. (C) Paired comparison of quantal content values. For each parameter (panels A, B & C) the last paired comparison represents separate experiments in which the effect of DMSO were assessed in the continued presence of 500 nM tubocurarine chloride (curare). P-values are the results of paired Student’s t-tests except for the result indicated by an asterisk, where the post-treatment data did not pass the Shapiro-Wilk test for normality and a Wilcoxon ranked sign test was used instead (n = 8 mice/preparations in each comparison).
We repeated our EPP recordings with a second series of muscle preparations, this time after adding 500 nM curare to the bath solution, to simulate myasthenic conditions. As expected, the curare produced a marked reduction in MEPP amplitude, compared to untreated preparations (from 0.91±0.08 mV down to 0.28±0.03 mV). Consistent with previous studies [26], the curare-induced drop in quantal amplitude triggered an increase in quantal content (+66%). This adaptive rise in quantal content was not sufficient to prevent a reduction in EPP amplitude (–66%). In the presence of curare, addition of DMSO again caused an increase the MEPP amplitude (∼35%; Fig. 3A, fourth set of paired data). Under these myasthenic-like conditions, DMSO also produced an increase in EPP amplitude (∼25%), there being no opposing reduction in quantal content (Fig. 3A-C, Supplementary Table 3). These findings indicate that, under myasthenic-like conditions, DMSO can increase both the quantal amplitude and EPP amplitude, thereby explaining the increased safety factor for sustained contraction.
DMSO maintains synaptic strength during high frequency signalling
Since sustained contraction depends upon effective tetanic activation, we examined the EPP response during high-frequency trains of nerve impulses. In the absence of curare, a train of nerve stimuli at 40 / sec produced in an initial potentiation of EPP amplitude (up to ∼10%between the 1st and 3rd pulse) followed by synaptic depression (EPP amplitude declining to ∼80%of its initial value by the 30th pulse; Fig. 4A). The plateau EPP amplitude at the end of a high-frequency train is thought to reflect the rate of quantal release that the nerve terminal can sustain during ongoing high frequency neurotransmission [34]. The presence of curare increased the initial facilitation (to ∼20%; Fig. 4B&D, open circles). The subsequent synaptic depression reduced the EPP to about 70%of its peak value (compare Fig. 4B). With the curarized preparation, addition of 0.1%DMSO kept the EPP amplitude approximately 30%above the pre-treatment level (with curare but no DMSO) throughout the 40 Hz train (Fig. 4B). In summary, when applied to curarized muscle preparations that mimic the synaptic effects of myasthenia, DMSO sustainably raised the amplitudes of both the MEPP and EPP.
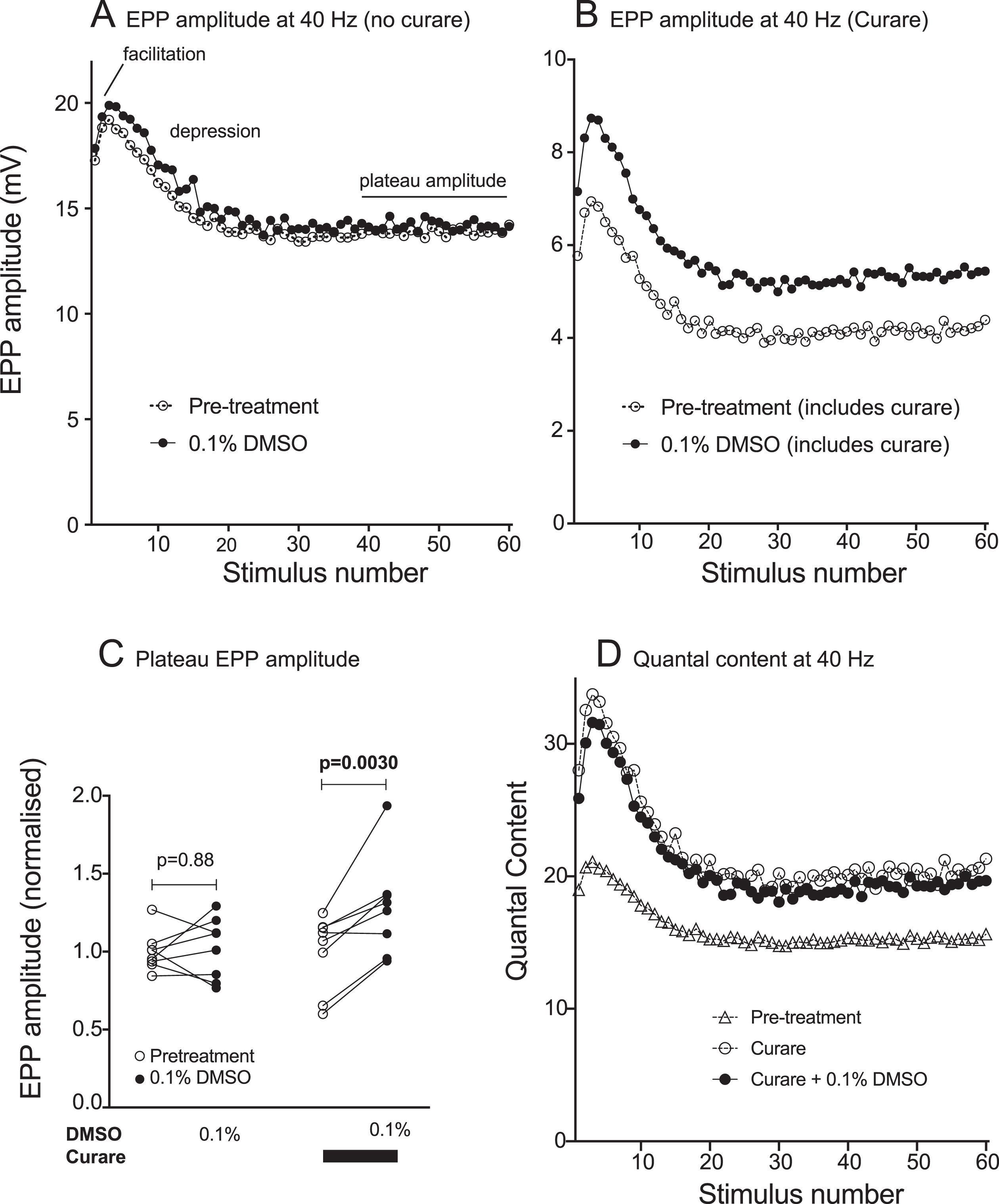
Effects of curare and DMSO upon high-frequency neuromuscular transmission. (A) EPP amplitude during a train of nerve impulses at 40 / sec. Initial facilitation was followed by synaptic depression and a plateau EPP amplitude, reached by 1 sec into the train. Symbols show pre-treatment results (open circles) and results after bath application of 0.1%DMSO (filled circles). Symbols in panels A, B & D represent mean values for n = 8 mice/preparations. (B) Comparable experiments conducted in the continued presence of 500 nM tubocurarine (curare) showing a sustained increase in EPP amplitude after addition of 0.1%DMSO (symbols show means for n = 8 mice/preparations). Note the difference in vertical scale values between A & B. (C) Paired comparisons showing changes in the plateau EPP amplitudes before and after addition of 0.1%DMSO. P-values are for ratio paired Student’s t-tests. (D) Quantal content during the 40 Hz stimulus train. Curare caused an adaptive increase in quantal content (open circles) when compared to no curare (open triangles). Subsequent addition of 0.1%DMSO had little further effect upon quantal content (filled circles).
Cannabinoid receptor agonists reverse the positive effects of DMSO on force restoration
Since cannabinoids are hydrophobic, they must be dissolved in DMSO or ethanol prior to dilution into aqueous solutions. Given the unexpected effect of DMSO per se upon the amplitude of synaptic potentials and upon the safety factor, we sought to minimise the volume of DMSO solvent. In the case of WIN 55,212-2 (a dual CB1R/CB2R agonist), a final aqueous concentration of 0.1%DMSO solvent was required to achieve a final concentration of 10μM WIN 55,212-2 in the bath solution. Higher aqueous concentrations of WIN 55,212-2 required more DMSO to prevent the cannabinoid precipitating when diluted into the bath solution (Supplementary Figure 1). Similar results were found for other cannabinoids studied (not shown). The following experiments specify the final aqueous concentrations of both the cannabinoid and the DMSO vehicle.
Using our curarized muscle contraction assay we first tested the effects of the dual CB1R/CB2R agonist, WIN 55,212-2 upon the safety factor for neuromuscular transmission. Application of 10μM WIN 55,212-2 / 0.1%DMSO to curarized diaphragms led to a partial (∼35%) restoration in force within 20 minutes, similar to effect of 0.1%DMSO alone. Unlike the effect of DMSO alone, the improvement in the safety factor after adding 10μM WIN 55,212-2 / 0.1%DMSO was transient, disappearing by 150 minutes (Fig. 5A, compare T3 and T4 time points). We next tested the effects of CP 55,940, a dual CB1R/CB2R agonist of greater specificity and potency compared to WIN 55,212-2. Addition of 1μM CP 55,940 / 0.01%DMSO to the bath produced no significant force restoration at either time-point (Fig. 5B). Addition of 10μM CP 55,940 / 0.1%DMSO produced a transient (∼25%) restoration of safety factor (Fig. 5C compare T3 and T4), similar to the effect of 10μM WIN 55,212-2/ 0.1%DMSO. Addition of CP 55,940 (75μM) / 0.75%DMSO produced a greater transient restoration of force (Fig. 5D). Thus, while DMSO alone (≥0.1%) acted rapidly to produce a sustained increase the safety factor for neuromuscular transmission (Fig. 2B), cannabinoid receptor agonists (co-applied with DMSO) appeared to act slowly to reverse the positive effect of the DMSO (Fig. 5A, C & D).
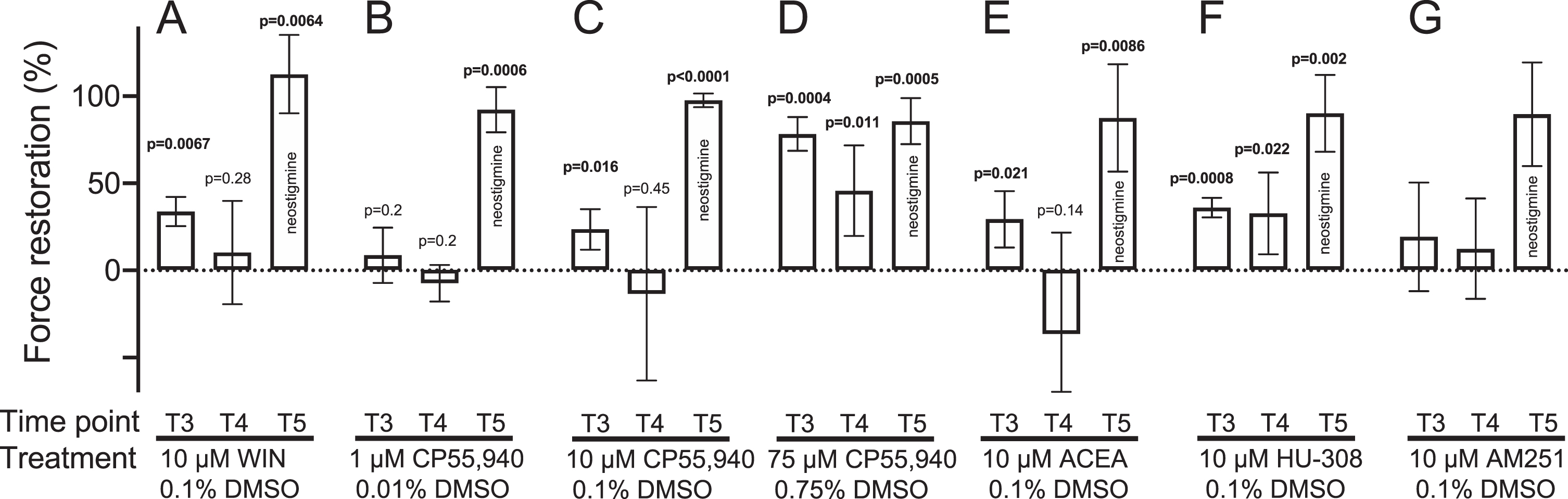
Cannabinoids reduce the safety factor for neuromuscular transmission. The curarized muscle contraction assay was treated with indicated combination of cannabinoid and DMSO (solubilizing agent). Histogram bars show the degree of recovery from curare-induced force decrement (force restoration) at the T3 (20 min) and T4 (150 min) time points after adding the indicated treatment to the bath, and at T5 (after addition of neostigmine). Panels show mean±95%confidence intervals for treatment with: 10μM WIN 55,212 / 0.1%DMSO (A; n = 3 mice/preparations); 1μM CP55,940 / 0.01%DMSO (B; n = 4); 10μM CP55,940 / 0.1%DMSO (C; n = 4); 75μM CP55,940 / 0.75%DMSO (D; n = 4); 10μM ACEA / 0.1%DMSO (E; n = 4); 10μM HU-308 / 0.1%DMSO (F; n = 4) and 10μM AM251 / 0.1%DMSO (G; n = 4). Each set of experiments (A-G) was separately subjected to repeated measures one-way ANOVA followed by Holm-Sidak’s multiple comparisons post-test comparing time points T3, T4 and T5 to the initial curarized state (T2, see Methods).
Agonists specific for either CB1R or CB2R were next tested in the curarized contraction assay. The combination of CB1R-specific agonist ACEA with DMSO (10μM ACEA / 0.1%DMSO) produced an effect similar to that with WIN 55,212-2 / DMSO and CP 55,940 / DMSO: a transient restoration of force that was reversed by 150 minutes (Fig. 5E compare T4 to T3). By contrast, the combination of CB2R-selective agonist HU-308 with DMSO (10μM HU-308 / 0.1%DMSO), produced a sustained increase in safety factor (Fig. 5F). The CB1R antagonist AM-251, in combination with DMSO (10μM AM-251 / 0.1%DMSO) did not produce any significant force restoration (Fig. 5G). The implications of the latter result remain unclear. The cannabinoids had little or no effect upon the amplitude of the first twitch contraction of the train (Supplementary Table 4). Overall, our contraction findings suggest that exogenous cannabinoids can act slowly, via CB1R (not CB2R), to reduce the safety factor for neuromuscular transmission.
Effects of cannabinoid/DMSO combination upon synaptic potentials
Our initial EPP results with cannabinoids became ambiguous as evidence emerged of the confounding effects of both DMSO solvent and the homeostatic response. We therefore focused upon trying to explain the combined effects of 10μM CP 55,940 / 0.1%DMSO upon the safety factor (Fig. 5C). Curare (500 nM) was included in the bath solution throughout the experiment to produce myasthenic conditions comparable to the contraction assay. Under these conditions, application of 10μM CP 55,940 / 0.1%DMSO to the bath caused an increase in MEPP amplitude and a reduction in quantal content, with no significant change in EPP amplitude (Fig. 6A-C). While 0.1%DMSO alone produced a sustained increase in EPP amplitude during 40 Hz nerve stimulation, the 10μM CP 55,940 / 0.1%DMSO combination did not (Fig. 6D-F). It is important to note that our EPP recordings began 2 hr after adding the treatment (DMSO±cannabinoid) to the bath solution, similar to the 150-minute time point (T4) in the contraction assay (Fig. 5). The presence of 10μM CP 55,940 produced a steady reduction in quantal content during the 40 Hz train of impulses (Fig. 6G-I). In summary, the negative effects of CB1R agonists upon the safety factor for neuromuscular transmission (Fig. 5A, C, D&E), are likely explained by a reduction in quantal content.
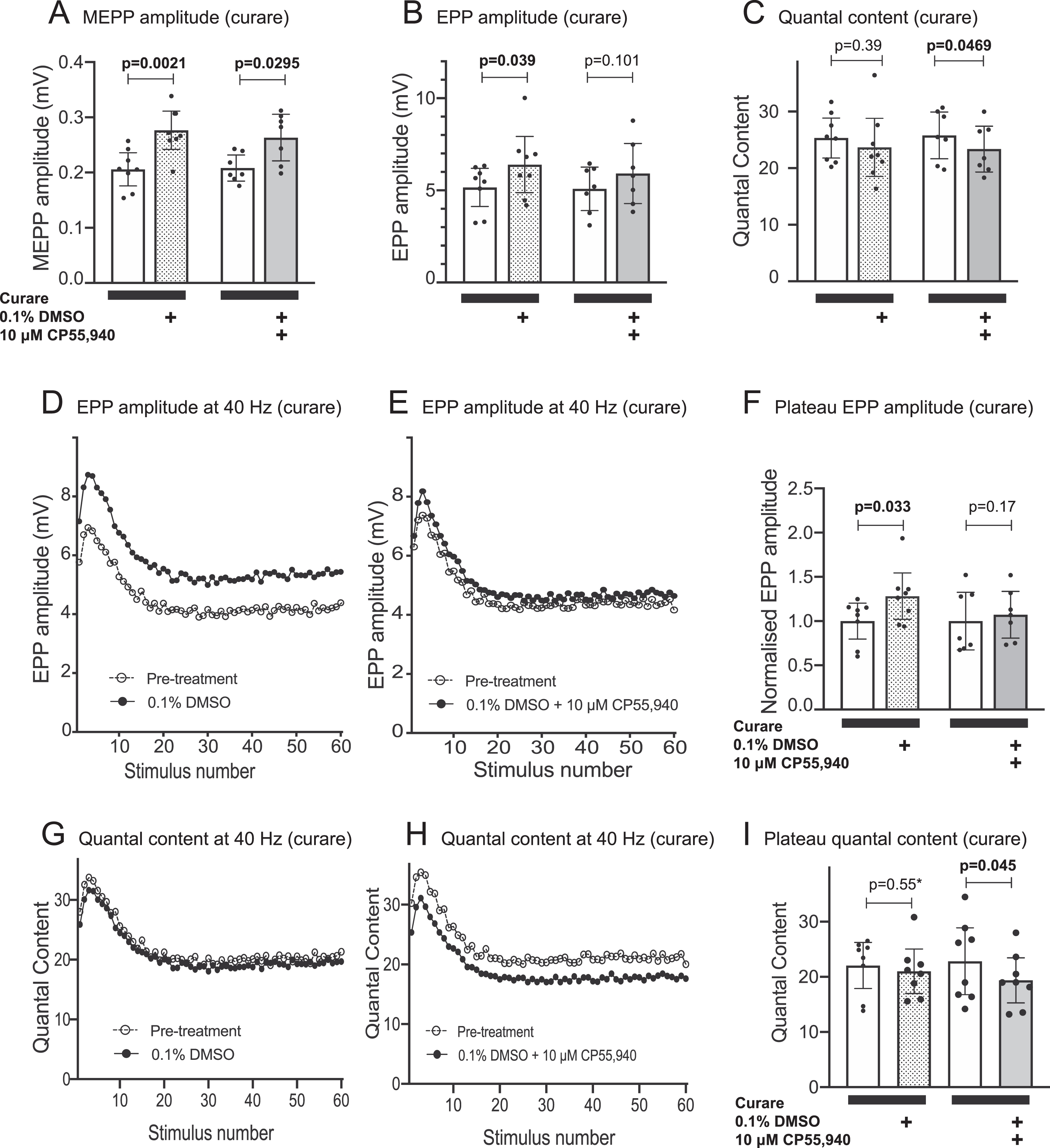
Combined effects of CP55,940 with DMSO upon quantal synaptic transmission at the myasthenic NMJ. The effects of CP55,940 (10μM, solubilized with 0.1%DMSO) upon synaptic potentials was assessed in the continuing presence of 500 nM tubocurarine chloride (curare). Data for the effects of 0.1%DMSO alone (Figs 4) has been replotted here for comparison. (A) Application of 10μM CP55,940 / 0.1%DMSO caused an increase in MEPP amplitude (paired comparison to pre-treatment recordings). (B) While 0.1%DMSO alone produced a significant increase in EPP amplitude, 10μM CP55,940 / 0.1%DMSO did not. (C) Significant reduction in quantal content after adding 10μM CP55,940 / 0.1%DMSO. (D-F) EPP amplitudes during repetitive nerve stimulation at 40 / sec. While 0.1%DMSO alone produced a sustained increase in EPP amplitude (D), 10μM CP55,940 / 0.1%DMSO did not (E). Panel F quantifies the plateau EPP amplitude (averaging the last 20 stimuli of the train). Treatment data has been normalised to the pre-treatment mean. (G-I) Quantal content during repetitive nerve stimulation at 40 / sec. Preparations treated with 10μM CP55,940 / 0.1%DMSO revealed a consistently lower quantal content compared to paired pre-treatment data (H), whereas treatment with 0.1%DMSO alone had no such effect (G). Panel I shows the plateau value of quantal content. Changes in parameters before and after addition of the treatment were compared by ratio paired Student’s t-tests except for the result indicated by an asterisk, where the post-treatment data did not pass the Shapiro-Wilk test for normality, and a Wilcoxon ranked sign test was used instead (n = 8 mice/preparations in each comparison for no curare experiment and n = 7 mice/preparation in each comparison for curare experiments).
DISCUSSION
In this study we present a robust, accessible ex vivo assay to screen compounds for their capacity to restore failing neuromuscular transmission. In the electrophysiology clinic, the primary test for myasthenia is a decrement in the compound muscle action potential amplitude during repetitive nerve stimulation [22, 35]. The presence of curare in our contraction assay caused a consistent decrement in force during successive trains of 10 nerve impulses, reflecting a reduced safety factor for neuromuscular transmission. Using this assay, we show that concentrations of DMSO as low as 0.1%partially restored the safety factor. It did so by increasing the amplitudes of both the MEPP and EPP. In contrast, CB1R agonists, solubilized with DMSO, cause a delayed negative influence upon quantal content, EPP amplitude and safety factor. These unexpected results highlight the need to carefully control for effects of widely used solvents, and to take account of synaptic homeostasis when studying the effects of drugs that target synaptic function.
Our contraction assay employed curare to mimic the myasthenic condition in an isolated nerve-muscle preparation. Application of 500 nM curare to hemi-diaphragms reduced the MEPP amplitude to approximately one quarter of its healthy value. This is comparable to the reduction in MEPP amplitude found in muscle biopsies from myasthenia gravis patients, compared to those from healthy individuals [36]. Synaptic depression, during a train of nerve impulses, is an innate feature of the NMJ. In myasthenic muscles, where the quantal amplitude is reduced, synaptic depression leads to decrements in the compound muscle action potential and twitch force [22]. Both our electrophysiology and contraction recordings were conducted in a lower concentration of calcium than we and others have used previously (1 mM vs 2 mM). We recorded slightly lower EPP amplitudes from untreated muscles, compared to EPP values we reported previously for untreated muscles in 2 mM Ca2 + (17 mV vs 21 mV [37]). However, this difference may be explained by our decision not to correct for non-linear summation. While a bath concentration of 1 mM Ca2 + may not be saturating to quantal release machinery, the contraction recordings show that EPPs remained well above threshold, prior to the addition of curare. Total plasma calcium in C57BL6J mice was reported to be approximately 2.4 mM but the level of free ionic calcium was 1.1–1.2 mM [38]. Thus the 1 mM Ca2 + that we added to the Ringer’s solution in the present study was comparable to plasma free calcium levels. In our contraction assay DMSO produced a rapid, concentration-dependent reversal of the myasthenic force decrement, reflecting improvement in the safety factor for neuromuscular transmission. Like neostigmine, a well-established therapeutic for myasthenia gravis, 0.75%DMSO fully restored contraction force.
We previously reported that cannabinoid receptor agonists, WIN 55,212-2 and anandamide, could increase EPP amplitude [30]. Using the new contraction assay we were able to dismiss this class of compounds from further consideration much sooner than would be possible using EPP recordings alone, and without resort to in vivo animal models of myasthenia gravis. The curarized ex vivo contraction assay might provide a translational bridge: facilitating the rapid development of lead compounds towards a candidate drug, prior to testing in animal models.
While robust and convenient, the contraction assay has some limitations. The diaphragm muscle is composed of a mixture of fibres that differ in contractile force capacity (type 1, 2A, 2X and 2B fibres). Type 2B/2X fibres possess a higher safety factor for neuromuscular transmission than type 1/2A fibres [39]. Thus, force decrement measurements may dis-proportionately represent the type 1/2A fibre populations, rather than providing a linear scale of the number of affected fibres. An aspect of the assay requiring consideration is the substantial muscle-to-muscle variability in the percentage decrement (Fig. 1B, C). Our assay attempts to mimic the myasthenic condition by using curare to reduce the safety factor. Under such conditions, nerve-evoked contraction becomes very sensitive to small fluctuations in synaptic efficacy within an individual. Likewise, a small difference between individuals may be reflected in much larger difference in muscle function. The assay might be refined by comparing nerve-evoked force to force produced by direct electrical stimulation of the muscle.
Dimethylsulphoxide is widely used as a stock solvent both in clinical medicine and preclinical research, so its effects upon synaptic transmission were surprising. Experiments that use DMSO to dissolve an ‘active’ compound generally include DMSO in their control treatment, but such studies rarely report the effects of DMSO per se. DMSO easily penetrates lipid bilayers, and multiple unintended effects upon cells and tissues have been reported, including altered patterns of gene expression [40]. In our curarized contraction assay DMSO caused a rapid restoration of the safety factor for neuromuscular transmission (within 20 minutes). The effect of DMSO on the force decrement was concentration-dependent: partial restoration with 0.1%DMSO and full restoration with 0.75%DMSO. This was explained by an increase in the EPP amplitude, which in turn was explained by an increase in the quantal amplitude (MEPP). Published studies of the effect of DMSO upon neuromuscular transmission are limited. One early report described complex effects (both positive and negative) upon nerve-evoked contraction force when DMSO was applied to the bath at high concentrations (1–20%). The enzymic activity of acetylcholinesterase was partially inhibited by 5%, DMSO [41] but inhibition was nominal in DMSO concentrations tested below 0.7%[42]. McLarnon and colleagues used voltage-clamp to study the effects of DMSO upon neuromuscular transmission in the mouse diaphragm muscle [41]. At concentrations between 1%and 6%, DMSO caused a progressive slowing of the decay time for the miniature endplate current, consistent with cholinesterase inhibition. Paradoxically, the same concentration range of DMSO produced an opposing reduction in the amplitude of the miniature endplate current, which they attributed to a curare-like effect of DMSO on the acetylcholine receptor [43]. It is important to note that at 1%DMSO, both of these ‘postsynaptic’ effects were small in magnitude. Geron and Meiri applied 0.5%DMSO to the frog NMJ and found a 22%increase in MEPP amplitude [44]. After irreversibly inactivating acetylcholinesterase with phospholinium iodide, addition of 0.5%DMSO produced no further increase in MEPP amplitude [44]. The significance of the latter finding remains uncertain. Inactivation of cholinesterase increased the MEPP amplitude greatly (230%) possibly saturating postsynaptic acetylcholine receptors in the vicinity of the transmitter release site with acetylcholine. If so, an additional 22%DMSO-induced increase in the amount of acetylcholine per quantum might not have been detected. In our previous work, pyridostigmine (a reversible cholinesterase inhibitor) produced a 57%increase in MEPP amplitude, associated with an approximate 63%increase in MEPP half-width [30]. In the present work, 0.75%DMSO increased MEPP amplitude by ∼30%with no increase in half-width. Thus, the increased MEPP and EPP amplitudes we recorded after addition of DMSO cannot easily be explained by inhibition of acetylcholinesterase. DMSO can also act on the presynapse to increase the amount of transmitter released, presumably by modifying membrane properties [43, 44]. For example, 0.5%DMSO doubled the MEPP frequency [44]. A recent amperometry study with adrenal chromaffin cells provided evidence that DMSO can increase the amount of catecholamine released per quantum [45]. Millimolar concentrations of other common organic solvents, including ethanol and propylene glycol, have been reported to cause changes in EPP and MEPP amplitudes [46]. Thus, substances that can permeate and modify lipid bilayers may have multiple effects upon synaptic transmission that are relatively non-specific. Irrespective of their mechanism of action, our findings with DMSO highlight the problems of using solubilizing agents such as DMSO in studies of cell signalling. Final concentrations≥0.1%DMSO might distort the very signalling systems that we are trying to study. The effects of an ‘active’ solute might be qualitatively different if it is delivered together with DMSO. This should certainly be a concern for ex vivo and in vitro experiments studying the effects of cannabinoids upon synaptic transmission.
Our current results suggest that CB1R agonists (when co-delivered with DMSO), can negatively influence the safety factor for neuromuscular transmission. Ex vivo tissue studies such as ours often require a higher concentration of cannabinoids (e.g. 10μM) compared to concentrations that are sufficient to activate CB1R on monolayer cell cultures. This may be due to problems with diffusion and non-specific absorption of the hydrophobic cannabinoids in the bulk tissue. In any event, we needed to use DMSO at a final concentration of at least 0.1%to solubilize cannabinoid compounds to a final concentration of 10μM (see Methods). Thus, our results for cannabinoids represent the combined effects of the DMSO and the cannabinoid upon the nerve-muscle preparation. In our contraction assay, the mixture of 0.1%DMSO with CB1R/CB2R dual agonist (WIN 55,212-2 or CP 55,942) produced a rapid initial restoration of force within 20 minutes (attributable to the DMSO). This improvement was short-lived, with return of the force decrement by 150 minutes (Fig. 5A&C). The synthetic CB1R-specific agonist, ACEA, replicated this effect of WIN 55,212-2 and CP 55,942, whereas the CB2R-specific agonist, HU-308 did not (Fig. 5E&F). EPP recordings in the presence of curare, 2hr after addition of 10μM CP 55,942 / 0.1%DMSO showed an increase in the MEPP amplitude (due to the DMSO) but with an opposing reduction in quantal content (attributable to the CP 55,942). Consequently, 10μM CP 55,942 / 0.1%DMSO produced no significant increase EPP amplitude (Fig. 6). In the CNS, endocannabinoids often suppress transmitter release via cAMP/PKA-mediated modulation of calcium channels and synaptic vesicle-associated proteins [47–49]. Whether such mechanisms apply to the NMJ remains uncertain, with multiple conflicting reports (reviewed by [29]). In summary, our results show that DMSO increases the safety factor for neuromuscular transmission, whereas the combination of CB1R agonist with DMSO reverses this positive effect. CP 55,942, at least, caused a reduction in quantal content.
The effects of DMSO described here provide a novel insight into the synaptic homeostatic system at the NMJ. It has long been known that interventions that cause a reduction in the MEPP amplitude can initiate an adaptive increase in quantal content [23, 50]. This form of presynaptic potentiation is now thought to reflect a form of retrograde synaptic homeostasis which can manifest over short and/or long timescales [23, 25–27]. Our findings with DMSO demonstrate the converse: that an increase in quantal amplitude above the physiological level can prompt a reduction in quantal content. Gaviño and colleagues used a transgene to constitutively overexpress glutamate transporter in motor nerve terminals of Drosophila larvae [51]. The resulting chronic elevation of quantal amplitude was accompanied by lower quantal content (relative to wild-type flies). This was attributed to a homeostatic reduction in presynaptic calcium channels. Our experiments at the mammalian NMJ now show that an acute increase in quantal amplitude (above the physiological level) is sufficient to prompt an immediate reduction in quantal content. In the presence of curare (MEPP amplitudes well below the physiological level), DMSO again caused an increase in quantal amplitude but, no significant reduction in quantal content occurred (Fig. 2). Our results, combined with those of others [24, 26], support the existence of a rapid, bi-directional homeostatic system that makes adaptive changes to quantal content whenever there is a shift in the MEPP amplitude below, or above, a physiological ‘set point’.
Footnotes
ACKNOWLEDGMENTS AND FUNDING
This work was funded by the Lambert Initiative for Cannabinoid Therapeutics. We thank Drs Samuel Bannister, Jonathon Arnold and Iain McGregor of the Lambert Initiative for their advice and support.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
