Abstract
Background:
Spinal muscular atrophy is an autosomal recessive neuromuscular disease leading to ongoing degeneration of anterior horn cells in the spinal cord. Nusinersen is the first approved treatment for the condition, an intrathecally administered antisense oligonucleotide. It modulates pre-RNA splicing of the SMN2 gene and increases full-length SMN protein expression, thereby increasing SMN protein levels. The benefit of Nusinersen for patients with spinal muscular atrophy type 3 (SMA3) has recently been shown in several real-world cohorts.
Objective:
We aim to elucidate not only the effect of therapy with Nusinersen, but the development of the disease course after discontinuation of treatment. To our knowledge, there are so far no reports on the effects of Nusinersen discontinuation.
Methods:
We report on a 45-year-old female patient with genetically confirmed SMA3 and a disease duration of 40 years prior to treatment onset.
Results:
The patient was non-ambulantory, best motor function at treatment onset was holding arms with support, reflected in MRC of 3/5 in upper limbs. After having received Nusinersen for 11 months without complications, the patient showed improvement in motor functions, as measured by hand grip measurement (HGS), Hammersmith Functional Rating Scale Expanded (HFMSE), and Revised Upper Limb Module (RULM). Due to worsening of a pre-existing anxiety disorder, treatment was discontinued after six injections. Sixteen months later, progression of the disease became evident with worsening of HFMSE and RULM scores, while hand strength remained stable.
Conclusion:
Treatment with Nusinersen in SMA3 improves motor function in longstanding disease even in clinically advanced stages; however, after discontinuation of treatment, further progression mirroring the natural history of the disease is anticipated.
Keywords
INTRODUCTION
Spinal muscular atrophy is an autosomal recessively inherited neuromuscular disease leading to ongoing degeneration of anterior horn cells in the spinal cord. Main clinical symptoms are progressive muscle atrophy and muscle weakness, along with affection of the respiratory muscles [1]. In this context, the disease shows continuous progression during its natural course with permanent decline in motor functions [2]. The disease is divided into three main subtypes, which are distinguished based on age at symptom onset and the achievement of motor milestones [1].
In 96% of cases, the disease underlies a homozygous deletion of the SMN1 gene on chromosome 5q13.2. This results in a loss of SMN protein expression. The paralogous SMN2 gene differs from SMN1 only in a base substitution (C > T in exon 7), resulting in the production of a defective nonfunctional protein. The number of SMN2 genes is inter-individually variable, and more copies may have a positive effect on the course of SMA [3, 4].
Nusinersen is an intrathecally administered anti-sense oligonucleotide. It modulates pre-RNA splicing of the SMN2 gene and increases full-length SMN protein expression, thereby increasing SMN protein levels [1, 5]. The benefit of Nusinersen for adult SMA patients (sitters and walkers) has recently been demonstrated in several studies, resulting in a significant and additive effect over time regarding motor function improvements [5–7], as assessed by hand grip measurement (HGS), 6-minute walk test (6MWT), Hammersmith Functional Motor Scale Expanded for SMA (HFMSE) and Revised Upper Limb Module (RULM) [8–11]. Across all studies, there was good overall tolerability of Nusinersen, with the most common side effects being postpuncture headache and back pain at the puncture site [5, 6]. However, there is a per mill risk of developing hydrocephalus communicans after Nusinersen administration according to an official Biogen press release and an advice from the Federal Institute for Drugs and Medical Devices (Bfarm) [12, 13].
We aim to elucidate not only the effect of therapy with Nusinersen, but the development of the disease course after discontinuation of treatment. To our knowledge, there are so far no reports on the effects of Nusinersen discontinuation. In the light of upcoming therapies and longstanding treatments, experience with treatment discontinuation is highly relevant in daily practice and of increasing importance.
Case presentation
We report on a 46-year-old female patient with genetically confirmed spinal muscular atrophy type 3 (deletion exon 7 and 8 of the SMN1 gene, 4 copies of the SMN2 gene). The patient presented for the first time at the age of 35 years at our neuromuscular outpatient clinic. At that time, she stated that birth and early childhood development had been completely normal. Family history revealed no additional affected family members. First symptoms appeared at the age of 3.5 years in form of proximal muscle weakness along with muscle atrophy. At 14 years of age, her ability to walk became limited. However, the diagnosis of SMA was only confirmed at the age of 23 in Hungary. At her first presentation at our unit at age 35, walking was only possible for a few steps with assistance.
Clinical examination then revealed proximal weakness of the upper (MRC 3-4/5) and the lower extremities (MRC 2-3/5). At that time, the only therapy option was best supportive care, in the form of physiotherapy and occupational therapy.
With ongoing disease, walking and standing were no longer possible at the age of 37. The patient developed a chronic pain syndrome, mainly due to the incorrect posture, problems with concentration, chronic fatigue and a worsening of her lung function with forced vital capacity of 1,74l (50% of predicted).
At the age of 41, the patient was not able to continue her work as a CAD (“computer-aided design”) specialist in a construction company. Due to ongoing worsening of her motor functions and fine motor skills she was not able to operate a computer mouse and keyboard sufficiently anymore; however, muscle strength was largely stable with intensive physio-therapy.
Following approval of Nusinersen, treatment was started in November 2017 at the age of 43.
Prior to start of treatment, lifting the arms up to horizontal level was only possible with support of another person; muscle strength had declined to MRC 3/5 in the proximal upper extremities and to MRC 2/5 in the lower extremities. Pulmo-nary function was largely unchanged.
The first intrathecal administration of Nusinersen was performed easily and without complications, although the patient had a mild right-convex scoliosis. No local anaesthetics, premedication or imaging was needed. At the next visit in December 2017 (day 14), the patient reported postpuncture headache starting two days after the first administration, lasting for four days, which she treated symptomatically with Metamizole. The second administration was equally easy to perform; during the third administration, the patient reported about pain at the puncture site and edema of the ankles and feet, and associated both symptoms with the Nusinersen therapy. However, these symptoms were not verified by a physician; at the next presentation in our unit, no edema was seen. No additional diseases were known, the etiology of the patient-reported edemas could not be finally elucidated.
Improvement of muscle weakness was first seen with the fourth and fifth administrations –both without any complications - after 64 and 173 days, respectively. Thus, an improvement in the degree of strength of upper arm abduction from 3/5 to 4/5 was noted bilaterally. In addition, she was now able to grasp and hold a glass with one hand again, and she reported that she could now push the wheelchair independently.
The sixth administration (day 299) at age 44 in September 2018 was again performed without complications, but the patient subsequently reported on severe persistent back pain at night with consecutive sleep disturbances. Due to the back pain and nocturnal restlessness, the patient fell out of bed resulting in a fracture of a lumbar vertebra (LV5). In addition, the patient suffered from frequent headache.
Following a Biogen press release on development of a communicating hydrocephalus not related to meningitis or bleeding during treatment with Nusinersen [12] in July 2018, the patient developed increasing psychological distress and anxiety about the CSF puncture and possible treatment complications, mainly communicating (nonobstructive) hydrocephalus. She therefore discontinued therapy with Nusinersen, now after six administrations and 299 days after treatment initiation. Treatment with an antidepressant for anxiolytics was briefly discussed with the patient, but the patient decided against psychopharmacologic therapy.
Until then, physiotherapeutic assessments had shown continuous improvement of scores over time. The patient achieved 21 points on the left upper extremity and 20 points on the right side in the RULM prior to treatment. An increase in the score up to 23 points on both sides was documented as early as the fourth administration. With the sixth administration, there was an increase to even 26 points on the left and 25 points on the right side. HFMSE showed an increase in the score from 6 to 11 points as well at the fourth administration. In the course, a decrease to 8 points on both sides was seen in the fifth and sixth doses, which however remained stable. The best progress was demonstrated in the HGS (Jamar handgrip). Initially, the strength achieved was 0 kg on both sides and could already be increased to 4 kg on the right and 5 kg on the left side with the fourth administration (see Table 1, Fig. 1).
Overview of physiotherapeutic assessments before, during and after treatment
*After discontinuation of Nusinersen.
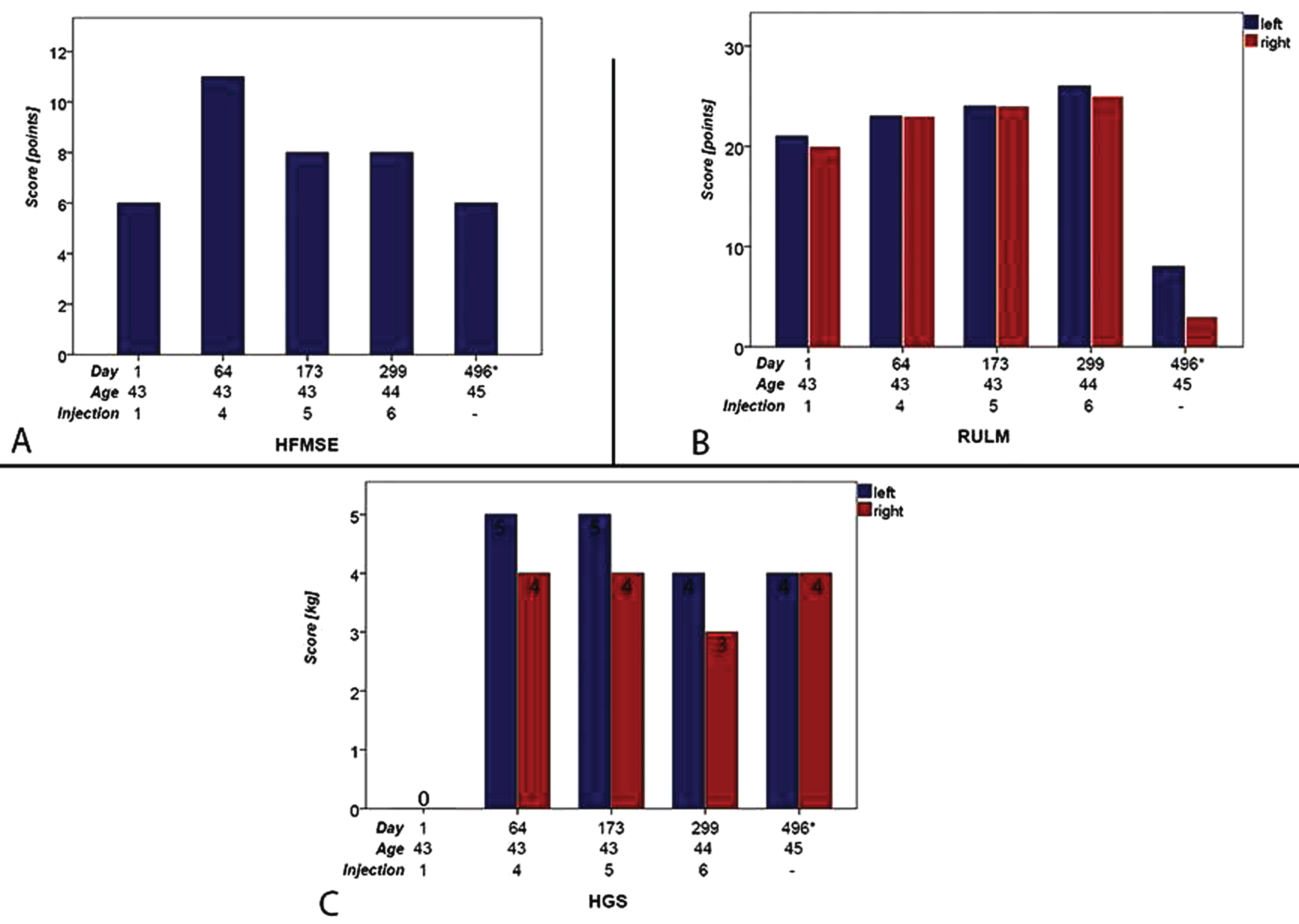
HFMSE (A), RULM (B) and HGS (C) during treatment and after discontinuation (*After discontinuation of Nusinersen).
In summary, from injection 1 to 6 the improvement in the HFMSE was mainly in the area of the upper extremities. Starting from the 5th injection, the hands (individual and bilaterally) could be brought to the head in sitting position again (score 1 to 2). In RULM, a button light could be pushed again with one hand (score 0 to 2 on both sides). In addition, a ziploc container could be opened again (score 0 to 1 on both sides). After discontinuation of therapy, the patient, now aged 45 years, presented again at our outpatient clinic in February 2020, 496 days after discontinuation of treatment. Subjectively, she reported a marked deterioration in upper extremity strength, clinical examination revealed MRC 2/5 in proximal arm muscles. Due to increasing weakness, the patient now was unable to bring a glass to her mouth without help. Since discontinuation of therapy, back pain and sleep disturbances persisted almost unchanged, subjectively even worsened. The lung function was nearly stable with FVC of 1,8l (54% of predicted). The RULM and HFMSE now showed a decrease in the achieved scores. In the RULM a decrease from 26 points on the right side at the sixth administration to now only 8 points could be shown. On the left side, the deterioration was even more pronounced with a decrease from 25 points to 3 points. Previously improved motor skills such as pushing a button light with one hand or opening a ziploc container were no longer possible.
The HFMSE deteriorated from 8 points to 6 points as well. The hand could no longer be brought to the head. HGS remained stable at 4 kg (see Table 1).
DISCUSSION
Our 45-year-old SMA3 patient discontinued Nusi-nersen treatment after the sixth injection (299 days after treatment initiation) due to worsening of an anxiety disorder, although objective improvement of HFMSE and RULM scores was noted, along with progress in patient reported outcomes, e.g. lifting a glass and moving around in the wheelchair independently. Unfortunately, stop of treatment resulted in further disease progression, with a loss of additional 2 score points in HFMSE and up to 22 score points in RULM. However, HGS remained largely stable after discontinuation of Nusinersen, in line with the slower progress of weakness in distal muscles.
It should be noted that there already was a deterioration of HFMSE by 3 points during Nusinersen treatment. To what extent these fluctuations are caused by the natural course of the disease or the patient’s daily form (e.g. chronic back pain worsening mobility) cannot be clarified conclusively.
Compared to the natural history of the disease, characterised by a continuous deterioration of motor function, the course in our patient during Nusinersen was characterised by a continuous improvement prior to discontinuation of therapy. A natural history study in patients with SMA 2 and 3 by Kaufmann et al. [14] showed a mean decrease of HFMSE of –1.71 points after 36 months. Additionally the study shows a largely stable course over 12 months in HFMSE, only then increasing deterioration could be documented. In contrast, our patient showed a 2-point improvement in HFMSE after 11 months of treatment, in line with our findings in a cohort of adult SMA3 patients with longstanding disease, where a mean HFMSE improvement of + 4.33 points after 10 months was seen [5]. In 2020, Hagenacker et al. [7] also demonstrated a mean HFMSE increase of + 2.58 points after 10 months in a more severely affected cohort of 139 adult patients with SMA2 and 3, and Maggi et al. [6] were able to demonstrate a mean increase of + 2 points in a similar cohort of 116 patients. It should be noted that generally an improvement of at least +3 points is considered to be of clinical significance [15].
Our patient achieved a bilateral improvement of + 5 points in RULM 10 months after initiation of treatment; notably, a RULM improvement of at least + 2 points is seen as clinically meaningful [16]. Hagenacker et al. [7] demonstrated a mean improvement of + 0.59 points after 10 months of treatment, while our cohort [5] improved + 0.74 points, in line with the data of Maggi et al. [3] with an improvement of + 0.61 points (see Table 2).
Comparison of HFMSE and RULM in different studies and our patient
So far, there is no published data on the clinical course of a patient after discontinuation of Nusinersen treatment available. After discontinuation the RULM in our patient showed a marked deterioration of the score on both sides, whereas only a decrease of 2 points in the HFMSE was noted. The only slight loss of points in the HFMSE is possibly explained by the already low baseline level before therapy, especially since the patient had not subjectively noticed any improvement in the motor function of the lower extremities even during therapy. In the RULM, a clear increase in the score could be seen after only a few doses, and subjectively, everyday performance in the area of the upper extremities was also clearly improved. However, it must be noted that the patient already had a better baseline level of motor function of the upper extremities in the RULM than in the HFMSE before start of therapy.
On the other side, significant improvement in HGS during Nusinersen treatment had been recently reported [17], and was equally seen in our patient, most likely due to the better preserved distal muscle function in the natural history of the disease. Our group [5] and Hagenacker et al. [7] had recently shown that the preserved level of motor function prior to onset of treatment is a major factor for treatment efficacy and outcome. In our patient, HGS improvement remained constant for 16 months after Nusinersen discontinuation, suggesting a fairly long-term benefit.
Response to treatment in our patient was not only mirrored in HFMSE and RULM despite a long-standing disease and an advanced disease stage prior to onset of therapy in our patient, but also in patient reported outcomes. In line with the recently published real-world data [5–7], even patients in an advanced stage of the disease can benefit from Nusinersen. In addition to an improvement in hand grip strength [17], improvement in HFMSE and RULM was cumulative with ongoing treatment duration [5, 7].
In conclusion, treatment with Nusinersen was effective in our patient with long-standing SMA3, and the effect could be partly sustained for some time after treatment discontinuation, indicating a benefit on the patient’s quality of life even after a short treatment duration.
It is possible that the deterioration of motor functions, apart from the discontinuation of treatment with Nusinersen, is also due to the natural course of the disease. Ultimately, it is not possible to distinguish precisely between effects due to the discontinuation of treatment and the natural course of the disease. Unfortunately, the patient could only be re-examined 496 days after discontinuation of Nusinersen, and we were therefore unable to collect continuous data on the course of the disease directly after discontinuation.
Our study adds to the increasing evidence of real-word data in adult SMA patients showing efficacy of Nusinersen through different types and stages of the disease, and highlights again that the preserved motor function prior to onset of treatment is key to predict treatment efficacy and durability. These findings should be considered with regard to the ongoing cost-benefit discussion of Nusinersen treatment [18, 19].
Footnotes
ACKNOWLEDGMENTS
We wish to thank the patient for participation in our study.
FINANCIAL DISCLOSURE
Miriam Hiebeler received Speaker Honoraria from Biogen. Angela Abicht has served on advisory boards for Sarepta and PTC Therapeutics. Maggie C. Walter has served on advisory boards for Avexis, Biogen, Novartis, Pfizer, Roche, Santhera, Sarepta, Pharnext, PTC Therapeutics, Ultragenyx, Wave Sciences, received funding for Travel or Speaker Honoraria from Avexis, Biogen, PTC Therapeutics, Ultragenyx, Santhera, Sarepta, and worked as an ad-hoc consultant for AskBio, Audentes Therapeutics, Avexis, Biogen Pharma GmbH, Fulcrum Therapeutics, GLG Consult, Guidepoint Global, Gruenenthal Pharma, Novartis, Pharnext, PTC Therapeutics, Roche.
