Abstract
Background:
Myotonic dystrophy type 1 (DM1) is characterized by progressive and predominantly distal muscle atrophy and myotonia. Gait and balance impairments, resulting in falls, are frequently reported in this population. However, the extent to which individuals with DM1 rely more on a specific sensory system for balance than asymptomatic individuals (AI) is unknown.
Objective:
Evaluate postural control performance in individuals with DM1 and its dependence on vision compared to AI.
Methods:
20 participants with DM1, divided into two groups based on their diagnosis, i.e. adult and congenital phenotype, and 12 AI participants were recruited. Quiet standing postural control was assessed in two visual conditions: eyes-open and eyes-closed. The outcomes measures were: center of pressure (CoP), mean velocity, CoP range of displacement in anteroposterior and mediolateral axis, and the 95% confidence ellipse’s surface. Friedman and Kruskal-Wallis analysis of variance were used to compare outcomes between conditions and groups, respectively.
Results:
Significant group effect and condition effect were observed on postural control performance. No significant difference was observed between the two DM1 groups. The significant differences observed between the AI group and the two DM1 groups in the eyes-open condition were also observed in the eyes-closed condition.
Conclusions:
The result revealed poorer postural control performance in people with DM1 compared to AI. The DM1 group also showed similar decrease in performance than AI in eyes-closed condition, suggesting no excessive visual dependency.
INTRODUCTION
Myotonic dystrophy type 1 (DM1) is a dominant inherited autosomal disorder. It is the most common neuromuscular disorder in adults with the prevalence of 9-14/100000 individuals worldwide [1, 2] which increases up to 1/550 individuals in the northeastern part of Quebec (Canada) [3]. DM1 is characterized by progressive and predominantly distal muscle atrophy, resulting in muscle weakness [4]. Furthermore, to varying degrees, there is a presence of myotonia, somatosensory deficit, visual impairments and altered posture characterized by head protraction [5–7].
In daily clinical practice, a decrease in functional level including balance deficits is frequently reported in people with DM1 [6, 8–10]. More precisely, several studies highlighted balance impairments using clinical tests, such as the Berg Balance Scale, the modified Dynamic Gait Index, the Time-Up and Go, and the Limits of Stability [6, 12]. Differences in balance performance have been reported between fallers and non-faller individuals with DM1 [11]. Moreover, all studies reported lower balance scores in individuals with DM1 compared to asymptomatic individuals (AI). However, such clinical tests generally evaluate tasks without the possibility to isolate the cause of the decrease in functional level. Challenging balance task involves different underlying systems and the treatment of three sources of information, i.e. visual, somatosensorial, and vestibular, are required [13, 14]. A better understanding of the mechanisms related to balance performance is of utmost importance to improve therapeutic approaches in people with DM1.
Static posturography tests allow investigating mechanisms related to postural control and balance by the measurement of the center of pressure (CoP) characteristics during quiet standing [11]. Indeed, with different test conditions it is possible to isolate the impact of a sensory system on balance control [15]. In order to evaluate the effect of a pathology on balance mechanisms, data from AI are also essential to correctly interpret quiet standing tests. For example, comparison of the CoP velocity between AI and individuals with a balance deficit, under eyes-closed and eyes-open conditions, allows to specify if the proprioception is altered. The inhibition of a sensory system allows quantifying the efficiency of the other sensory systems [15].
To the author’s knowledge, only three studies evaluated postural control using a force platform to measure CoP characteristics in adults with DM1 [8, 17]. Missaoui et al. (2010) described a wider surface trajectory along with balance improvement after a rehabilitation program in a group of individuals with DM1 without comparison to a control group [8]. Bachasson et al. (2016) reported greater CoP sway velocity in individuals with DM1 compared to AI, but only in eyes-closed conditions [16]. Recently, Naro et al. (2019) demonstrated a lower CoP path length in individuals with DM1 compared to AI. They also compared the CoP path length performance in eyes-open and eyes-closed condition and reported a similar performance between the two groups [17]. Despite the valuable information provided by the latter study, only one CoP characteristic was reported. Hence, with this study, the interpretation of postural control deficits in the DM1 population is limited by the only variable reported, reflecting the overall postural control performance. Based on these studies, there is a need for an analysis comprising various CoP characteristics reflecting many aspects of the postural control performance including different test conditions and a performance comparison to an AI group.
In people with DM1, several factors including muscle weakness, sensory-motor peripheral neuropathy, and accelerated aging of the eyes, can have a detrimental effect on balance control, as those factors can affect the proprioceptive and the visual system [15]. Deficit of these sensory systems can lead to instability, but systems can also partly compensate each other and limit the impact on postural control [15]. Therefore, a better understanding of the contribution of each factor is of utmost importance to improve physical therapy approach.
As the first step for exploring the contribution of sensory systems on postural control in individuals with DM1, the aim of the present study was to evaluate postural control performance and its dependence on vision compared to AI. We hypothesized that the decline in postural control performance between two visual conditions will be greater in individuals with DM1 compared to AI.
MATERIAL AND METHODS
Study population
A sample of 20 participants with DM1 was recruited through a tertiary rehabilitation center. The DM1 group included 10 participants with the adult phenotype and 10 participants with the congenital phenotype. A clinician screened the potential participants based on their cognitive and behavioral functions to ensure that they were able to understand the research protocol and provide informed consent. The inclusion criteria for the DM1 groups were aged between 18 and 60 years, a genetically confirmed diagnosis of DM1, and the ability to stand upright without support. Along with the DM1 groups, 12 AI individuals were recruited. The inclusion criteria for the AI group were aged between 25 and 60 years, and no history of neuromuscular disorder or any musculoskeletal conditions, which may influence balance. The exclusion criteria for both groups were aged older than 60 years to avoid the effect of aging on balance and postural control [18, 19]. Descriptive data for the DM1 groups and the AI group are presented in Tables 1 2 respectively.
Characteristics of participants with Myotonic Dystrophy type 1
BMI (Body Mass Index); M (Male); F (Female); TUG (Time Up and Go); nd (no data); nt (Not-tested); IQR (interquartile, 1st and 3rd); * Significant difference (p < 0.05).
Characteristics of asymptomatic participants
BMI (Body Mass Index); M (Male); F (Female); IQR (Interquartile, 1st and 3rd).
The local ethics committee approved the study, and all participants provided written informed consent.
Characteristics
Baseline characteristics were collected including age, gender, body mass, height, Body Mass Index (BMI), and disease duration (years since the first significant symptom). The Timed Up and Go (TUG) and isometric muscle strength of the lower limbs were also measured and included in the population description to characterize the clinical impact of the disease (see Table 1).
The dynamic balance was assessed by the TUG [20]. Participants were asked to get up from a seated position, walk at a comfortable speed to a mark 3 m away, turn around, walk back to the chair, turn around and sit back.
Maximal isometric muscle strength was measured, by an experienced assessor, using a handheld dynamometer (Lafayette Instruments, Lafayette, IN). Hip and knee flexors and extensors, as well as hip abductors, were measured. Knee flexion and extension were measured in a sitting position, hip abductors and flexors were measured in a supine position and hip extensors were measured in a prone position [9, 21]. Two trials were completed for each muscle group, and the mean of the two trials was kept. Force was multiplied by the lever arm length to obtain joint torques, and then normalized to body mass. The results are expressed in Newton*meter per kg.
Test procedures
The center of pressure displacement was recorded using a force platform embedded in the floor (AMTI, Watertown, MA, USA) with a sampling frequency of 1000 Hz. Postural control tests were conducted without shoes. Feet were placed at a natural width, and they were marked to ensure the same feet placement between trials and conditions. Participants were asked to maintain a quiet upright standing posture, with arms comfortably at the sides, and to remain as stable as possible for the duration of each trial.
Two trials of 40 seconds were performed for each condition, i.e. (1) eyes-open and (2) eyes-closed [22]. For the eyes-open condition, participants were asked to look at a target placed 3 meters in front of them at eye level. For the eyes-closed condition, participants were asked to keep their head in the direction of the target. These two conditions were selected to evaluate the importance of visual inputs on postural control [15]. Two trials of 40 seconds are sufficient for reliable postural control measurement [23]. A 1-minute rest period in a seated position was given to participants between trials and conditions. The first and the last 5-second of data acquisitions were trimmed in data analysis. This adjustment allows removing the stabilization phase that could occur immediately after the beginning of the test and at the end, because of the transient nature of these phases [24, 25]. Therefore, a 30-second time frame was left for analysis, which is sufficient to produce reliable measurement [23]. Raw data were low-pass filtered at 10 Hz using a dual-pass second-order Butterworth digital filter. The two trials were averaged, and the mean value was used for statistical analysis.
Three postural control parameters were selected to quantify the individual’s stability performance: (1) the CoP mean velocity defined as the ratio between the path length and the total duration of the test (30 seconds), this parameter is recognized as one of the most sensitive [26] and reliable [27, 28] measure to assess postural parameters; (2) the CoP range in the anteroposterior and mediolateral axis; (3) the 95% confidence ellipse’s surface defined as the surface covered by the ellipse that contains the CoP center of points with a 95% probability [29]. The ellipse’s surface provides a general measure of performance, i.e., smaller surface reflects a more efficient postural control.
Statistical analysis
Considering the small sample size in each group, non-parametric analyses were conducted. The data were reported as median and interquartile (IQR, 1st and 3rd). Kruskal-Wallis one-way ANOVA was used to compare anthropometric data and postural control parameters between the groups. Friedman’s two-way ANOVA was used to compare postural control outcomes between the conditions, i.e., eyes-open and eyes-closed. If the tests were significant, post hoc Wilcoxon or Mann-Whitney U tests with the Bonferroni correction were realized for paired and unpaired comparison respectively. A p-value of < 0.05 was considered significant. Statistical analyses were performed using R (version 3.6.2) and RStudio (version 1.2.5033).
RESULTS
All reported characteristics were not significantly different between the two DM1 groups, i.e. the adult and the congenital phenotypes except for muscle strength. The congenital phenotype group had significantly lower muscle strength at the knee flexors and extensors, and at the hip flexors and abductors than the adult phenotype group (Table 1). No significant difference was observed through the groups for age (p = 0.223), body mass (p = 0.455), and BMI (p = 0.050). The AI group was significantly taller than the congenital phenotype group (p = 0.005) but not the adult phenotype group (p = 0.729).
One DM1 participant (DM1_11), in the congenital phenotype group, was unable to complete the quiet standing test. The participant was unable to maintain an upright posture for 40 seconds without taking a step in the two visual conditions. Therefore, this participant was excluded from statistical analysis.
Median and IQR for all postural control variables are presented in Table 3. A significant condition effect (i.e., eyes-open and eyes-closed) was observed on the CoP range in the mediolateral (p = 0.011) and anteroposterior axis (p = 0.011) and on the mean CoP velocity (p < 0.001). No significant condition-effect was observed on the 95% Ellipse’s surface (p = 0.068). The CoP range in the mediolateral axis was significantly higher in the eyes-closed condition compared to the eyes-open condition for the AI group (p = 0.012) and the adult phenotype group (p = 0.014). No significant difference was observed for the congenital phenotype group between the eyes-open and eyes-closed conditions (p = 0.652). The CoP range in the anteroposterior axis was significantly higher in the eyes-closed condition compared to the eyes-open condition in the AI group (p = 0.002). No significant difference was observed on the CoP range in the anteroposterior axis for the two DM1 groups. All group significantly increased their CoP mean velocity in the eyes-closed condition compared to the eyes-open condition (AI: p < 0.001; Adult: p = 0.002; Congenital: p = 0.020).
Postural control performance eyes-open and eyes-closed in the DM1 groups and the asymptomatic group
DM1 (Myotonic Dystrophy type 1); IQR (Interquartile. 1st and 3rd); CoP (Center of Pressure); AP (Anteroposterior); ML (Mediolateral).
A group effect was observed on all postural control variables (p < 0.001). The 95% ellipse’s surface was significantly lower in the AI group compared to the adult phenotype group (eyes-open: p < 0.001; eyes-closed: p < 0.001) and the congenital phenotype group (eyes-open: p = 0.001; eyes-closed: p = 0.013). No significant difference was observed between the two DM1 groups (eyes-open: p = 1.000; eyes-closed: p = 0.834). A similar effect was observed in the mediolateral CoP range, as no difference was observed between the two DM1 groups (eyes-open: p = 1.000; eyes-closed: p = 0.468) and a significant lower range was observed between the AI group and the adult phenotype group (eyes-open: p < 0.001; eyes-closed: p = 0.001). A significant difference was observed in the CoP range in the mediolateral axis in the eyes-open condition between the AI group and the congenital phenotype group (p = 0.007) but no significant difference in the eyes-closed condition (p = 0.083). The anteroposterior CoP range was not significantly different between the two DM1 group (eyes-open: p = 0.729; eyes-closed: p = 1.000) and between the AI group and the congenital phenotype group (Eyes-open: p = 0.208; Eyes-closed: p = 0.244). The anteroposterior CoP range was significantly lower in the AI group compared to the adult phenotype group (eyes-open: p < 0.001; eyes-closed: p = 0.006). Finally, the AI group had a significant lower CoP mean velocity compared to the adult phenotype group (eyes-open: p < 0.001; eyes-closed: p = 0.006) and the congenital phenotype group, only in the eyes-open condition (p < 0.001). No difference was observed between the two DM1 groups (eyes-open: p = 0.160; eyes-closed: p = 0.196). The boxplots of all CoP variables are presented in Fig. 1.
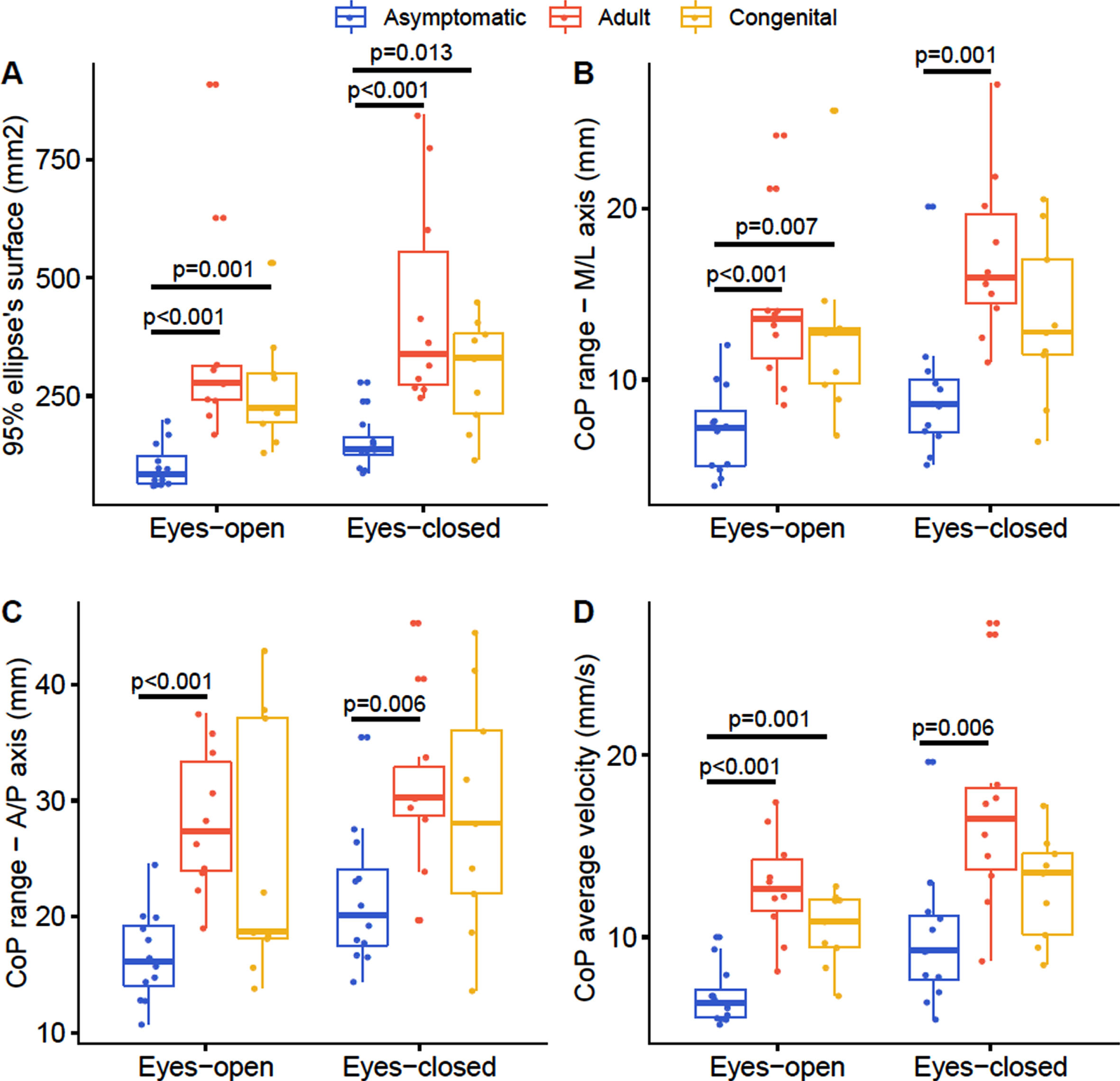
Boxplot of postural control parameters for the two visual conditions considering the three groups, i.e. asymptomatic Individuals, adult phenotype and congenital phenotype. The results of the Kruskal-Wallis one-way ANOVA are presented in the figure representing the group effect for each conditions.
DISCUSSION
The goal of the present study was to evaluate postural control performance in individuals with DM1 and its dependence on vision in comparison to AI. The results showed that individuals with DM1, in both the congenital and adult phenotype groups, had poorer postural control performance than the AI group in both eyes-open condition and eyes-closed condition. No difference between the two DM1 groups was observed. The adult phenotype group had significantly lower performance in all postural control parameters than the AI group. The congenital phenotype group also had lower postural control performance than the AI group, however, no significant differences were observed between the groups in the anteroposterior CoP range and no significant differences were observed in the eyes-closed condition for both the mediolateral CoP range and the CoP mean velocity in the eyes-closed conditions. A wider performance variability was observed in the eyes-closed conditions for both DM1 groups and for the anteroposterior CoP range in the eyes-open condition, which could affect the analysis. However, postural control performance decreased by approximately the same magnitude in individuals with DM1 and in AI when vision was retrieved as significant differences observed in the eyes-open condition were also observed in the eyes-closed conditions when comparing DM1 groups to AI. Hence, this result suggests no excessive dependency on the visual system for maintaining an upright posture in individuals with DM1.
In previous studies, little has been reported concerning force platform measurement of postural control performance in individuals with DM1 [8, 16, 17], especially when considering DM1 phenotypes. Measurement of CoP trajectory and orientation allows a more precise interpretation of balance performance. Our results showed a significant effect of the pathology on postural control and among others on the mediolateral sway range. As lateral sway range is the best predictor of falling risk [30] and that it requires greater neural responses than anteroposterior sway control [31], our results suggest, that the DM1 population with congenital and adult phenotype is at higher risk for falling.
In other studies, lower scores on various clinical tests have shown lower mobility abilities and balance in individuals with DM1 [6, 9]. Missaoui et al. (2010) measured static balance on two different kinds of surfaces (hard surface and foam) using a force platform in individuals with DM1 [8]. In addition to a wider CoP’s surface in both visual conditions, i.e., eyes-open and eyes-closed, they observed that participants with DM1 had a very limited balance on a foam surface. However, they did not aim to study postural control impairments but the effect of an intervention on postural control. Therefore, they only reported descriptive comparison to normative values on a restricted number of postural control parameters. Our results confirm a decline in performance between visual conditions and suggest that, in absence of vision and despite a somatosensory deficit, individuals with DM1 do not have a greater decline in balance performance compared to AI.
Therefore, the general lower postural control performance observed in the DM1 group suggests a deficit in proprioceptive or vestibular information processing. Indeed, many impairments associated with DM1 are likely to affect proprioceptive sensory information, such as muscle weakness, somatosensory deficit, and the altered posture of the head and trunk. However, muscle spindle proprioceptive function has been suggested as not impaired in muscular dystrophies, meaning that lower postural control may be due to muscle weakness rather than proprioceptive impairments [32].
Muscle weakness and myotonia could partly explain the lower postural control performance in individuals with DM1. An association between muscle strength and balance measurement has been previously demonstrated in individuals with DM1 [9, 16]. As observed in other populations, such as the elderly and individuals with neuromuscular disease, muscle weakness can influence balance [33, 34]. During an eyes-closed condition, Bachasson et al. (2016) found a greater CoP velocity in individuals with DM1, which was associated with neck and ankle muscle weakness [16]. Moreover, the severity of muscle weakness was associated with lower upright standing balance performance and gait impairments. However, the direct effect of myotonia on postural control has not been investigated. Myotonia can be defined as involuntary repetitive muscle depolarization causing a delay in muscle relaxation after contraction [35, 36]. Considering that postural control implies phasic and tonic postural muscle activity [37], a delay in muscle relaxation due to myotonia could affect stabilization mechanisms and alter adequate balance performance.
Pathologies affecting sensory output, force and movement control and spatial orientation logically affect postural control [38]. Previously, it has been suggested that individuals with DM1 demonstrated impairments in the parietal cortex as well as in several motor areas including the premotor area and the supplementary motor area [39]. Therefore, as the parietal cortex is involved in the spatial organization, impairment of this area can contribute to the lower postural control performance observed in this study.
Interestingly, no difference was observed in postural control performance between the DM1 phenotype both in the eyes-open and eyes-closed condition, but differences were observed in muscle strength. Furthermore, no difference was observed between the two DM1 groups on the TUG performance, which could indicate a similar overall function in the sample studied as the TUG provides information on balance and functional mobility. The differences or similarities in postural control and balance performance are scare between the DM1 phenotype and should be further investigated.
Study limitations
Three study limitations need to be considered in the interpretation of the results. First, the control group was not matched for age and height despite that both factors can affect postural control performance. However, no significant differences between groups were observed on age. Second, the measurement of proprioceptive and vestibular information processing was not assessed in the study design. Investigating these conditions would strengthen the knowledge on the contribution of sensory systems on postural control in individuals with DM1. Finally, the limited sample size and the few women included in the study (n = 4) did not allow to consider gender in the analysis. As previously demonstrated, gender influences the clinical profile of individuals with DM1 and should be considered in study design [40]. Therefore, the results of the present study should be interpreted cautiously concerning the gender. Further studies should address this limitation.
CONCLUSION
Postural sway is higher in individuals with DM1 compared to asymptomatic ones. A decrease in postural control performance is observed in individuals with DM1 when the vision is retrieved. However, the decrease is about the same magnitude for the DM1 and the AI group suggesting no excessive visual dependency. No differences in postural control performance were observed between the DM1 phenotype. Further studies investigating postural control in various surface conditions and evaluating the efficiency of the vestibular system are required to completely understand the underlying postural control impairment in this population. Increased knowledge of the underlying impairment of postural control in DM1 individuals will help to target interventions.
Footnotes
ACKNOWLEDGMENTS
The authors thank the participants who participated in this study. They also want to thank Benjamin Sinclair BSc, Maxime Gosselin BSc and Mathieu Bissonnette BSc for their implication in the project.
FUNDING
This research did not receive any specific grant from commercial or not-for-profit sectors. AP, LB, and APL received funding from the Fonds de recherche Québec –Santé.
DECLARATIONS OF INTEREST
None.
