Abstract
Background:
Expert guidelines recommend annual monitoring of 25-hydroxyvitamin D (25-OHD) and maintaining 25-OHD ≥30 ng/ml in patients with dystrophinopathies.
Objective:
We hypothesized that 25-OHD remains stable and requires less frequent monitoring in patients taking stable maintenance doses of vitamin D.
Methods:
We performed a retrospective cohort study, using the electronic health record to identify 26 patients with dystrophinopathies with a baseline 25-OHD ≥30 ng/mL and at least one additional 25-OHD measurement. These patients had received a stable dose of vitamin D for ≥3 months prior to their baseline 25-OHD measurement and throughout follow-up. The main outcome measured was the mean duration time the subjects spent with a 25-OHD ≥30 ng/mL.
Results:
Only 19% of patients dropped their 25-OHD to < 30 ng/ml, with a mean time to drop of 33 months and a median nadir 25-OHD of 28 ng/mL.
Conclusions:
These results suggest that measurement of 25-OHD every 2–2.5 years may be sufficient in patients with a baseline 25-OHD ≥30 ng/mL and who are on a stable maintenance dose of vitamin D. Other patients may require more frequent assessments.
ABBREVIATIONS
Body mass index
Vitamin D External Quality Assessment Scheme
Duchenne muscular dystrophy
Duchenne Muscular Dystrophy Care Considerations Working Group
Electronic medical records
25-hydroxyvitamin D
Kaplan-Meier
Liquid chromatography/tandem mass spectrometry
Parathyroid hormone
INTRODUCTION
Dystrophinopathy results from absence, deficiency or dysfunction of dystrophin, leading to muscular dystrophy with a continuum of clinical severity. Duchenne muscular dystrophy (DMD) is the most frequent childhood muscular dystrophy, affecting approximately 1.12 per 10,000 males aged 5–24 years in the United States [1]. At the milder end of the spectrum is Becker muscular dystrophy, which is less common and can present in childhood or adulthood. Patients with dystrophinopathy experience progressive muscle weakness, cardiomyopathy, restrictive lung disease, and for the more severe phenotypes, a shortened life expectancy [2–4].
Although there is no known cure for dystrophinopathy, glucocorticoids are the mainstay of treatment in DMD, leading to delayed loss of muscle strength, prolongation of walking, and preservation of pulmonary and cardiac function [5, 6]. However, long-term use of glucocorticoids along with multiple other factors including immobility, chronic illness, delayed puberty, lack of sun exposure with low vitamin D production, and possibly inflammatory changes from the disease itself, contribute to low bone mineral density and an increased risk for fractures in these patients [7–11].
Vitamin D plays a critical role in maintaining bone health by promoting calcium absorption from the gastrointestinal system, thus allowing normal mineralization of the bone. Supplementation with vitamin D might decrease the risk of fractures, though data testing this notion have been contradictory [12–15]. Current guidelines provided by the Duchenne Muscular Dystrophy Care Considerations Working Group (DMD-CCWG) recommend annual monitoring of 25-hydroxyvitamin D (25-OHD) and maintaining a 25-OHD concentration ≥30 ng/mL [4]. This recommendation is based on expert opinion due to lack of scientific evidence on the topic.
Obtaining annual blood tests can be challenging in patients with dystrophinopathy; pediatric patients are often anxious about needlesticks and later in life, patients often have contractures that can make phlebotomy difficult. While no studies have investigated the optimal frequency for monitoring 25-OHD concentrations, we observed that most patients who are vitamin D sufficient and on a stable dose of vitamin D have a stable concentration of 25-OHD over periods of time exceeding one year. If this clinical observation is accurate, we postulate that the time between 25-OHD measurements in this group of patients can be extended beyond one year. This may especially benefit patients who would otherwise not need phlebotomy. Here we present a retrospective cohort study that analyzes the degree of variability in 25-OHD concentrations in patients who are vitamin D sufficient and taking a stable dose of vitamin D supplementation.
MATERIALS AND METHODS
Data sources and patients
We used the electronic medical records (EMR) at the University of Iowa (Epic 2018 version UI 2, Epic Systems Corporation, Verona, WI, USA) to identify patients with dystrophinopathy who were evaluated at the Neuromuscular Clinic between January 1, 2009 and September 22, 2019.
As a predetermined inclusion criterion, only patients who had a 25-OHD concentration ≥30 ng/mL after consistently taking a stable dose of vitamin D for at least three months were considered for this study. This was considered their “baseline” 25-OHD concentration and this measurement was defined as the time of entry into the study. In addition, patients had to have at least one subsequent 25-OHD measurement on the same vitamin D dose to be included in the analysis. We defined vitamin D sufficiency by a 25-OHD ≥30 ng/ml based on the inverse relationship between parathyroid hormone (PTH) and 25-OHD, and the fact that PTH plateaus when the 25-OHD reaches ∼30 ng/ml [16, 17]. This was also consistent with the DMD-CCWG’s recommendation to maintain the 25-OHD concentrations ≥30 ng/mL in patients with DMD [4]. Consequently, 25-OHD concentrations ≥30 ng/mL were considered “normal” and concentrations < 30 ng/mL were considered “low”.
Patients were excluded if their dose of vitamin D or compliance with vitamin D were not clearly documented in the EMR. Since vitamin D is an over-the-counter medication that does not require a prescription, information on dose and compliance through the EMR medication list is not always accurate. For that reason, we only included records where vitamin D dose and compliance were directly addressed by the medical provider and documented in their clinical notes. Other exclusion criteria included a daily dose of vitamin D exceeding 10,000 units (since daily doses of up to 10,000 units are unlikely to result in vitamin D toxicity, while the risk of toxicity may increase with daily doses above 10,000 units) [18, 19], use of anti-epileptic medications, and concomitant malabsorptive disorders. If a patient took vitamin D at a frequency other than daily (such as weekly), an average daily dose was calculated.
The University of Iowa EMR contains complete demographic, clinical, laboratory, and medication data. For subjects who were included in the study, the following variables were recorded: age at diagnosis of dystrophinopathy, age at which patient had a baseline 25-OHD concentration of ≥30 ng/mL, subsequent 25-OHD concentrations and measurement dates, ambulatory status, and glucocorticoid use.
All data were extracted from the patients’ EMR by one investigator (N.V.W.) and cross-checked by another investigator (A.S.R.).
Laboratory assays
Prior to 2012, the University of Iowa Hospitals and Clinics (UIHC) core clinical laboratories referred serum/plasma 25-OHD measurement to a commercial reference laboratory (ARUP Laboratories, Salt Lake City, UT), which used the DiaSorin Liaison chemiluminescence immunoassay technology [lower detection limit, 0.8 ng/mL; intra-assay coefficient of variation (CV) 6.7 %; inter-assay CV 6.3 %]. Starting in 2012, 25-OHD measurements were performed at the UIHC core clinical laboratories using the Roche Diagnostics electrochemiluminescence immunoassay technology [lower detection limit 3.0 ng/mL; intra-assay CV 2.9%; inter-assay CV 6.0%]. Both assays cross-react similarly with 25-OHD2 and 25-OHD3. The laboratory participated in regular DEQAS (the Vitamin D External Quality Assessment Scheme) performance assessments. The core clinical laboratories are accredited by the College of American Pathologists.
Eight patients had some of their 25-OHD measurements performed at pathology laboratories outside the University of Iowa. These accounted for 10 out of 107 (9%) total measurements. The techniques used for these tests included liquid chromatography/tandem mass spectrometry (LC/MS/MS) in 1 measurement, and immunoassays for the other 9 measurements (DiaSorin chemiluminescence in 5, Roche electrochemiluminescence in 2, Siemens Diagnostics competitive immunoassay in 1 and Beckman competitive binding immunoassay in 1).
Despite the variability in the assays used, we included all results in this analysis since the measurement disagreement between different immunoassays are mostly significant for extreme values and less so for mid-range values [20, 21]. Additionally, disagreement between assays was projected to play a smaller role with “clustering” of the results into two main outcome groups (< 30 and ≥30 ng/mL).
Statistical analyses
Statistical analyses were conducted using SAS for Windows, version 9.3 (SAS Institute Inc., Cary, NC, USA).
Data are presented as a percentage or median (IQR) given the non-normal distribution and the small study sample. We began by examining patient characteristics and calculating the median length of follow-up for the cohort, the median age at diagnosis of dystrophinopathy, the proportion of individuals treated with glucocorticoids, the proportion of individual who are wheelchair dependent, the median vitamin D dose received, and the median baseline 25-OHD concentration.
For the survival analysis, the outcome of interest was “low 25-OHD” (i.e. 25-OHD < 30 ng/mL). In subjects where the 25-OHD did not drop below 30 ng/mL, data were censored at the time of the last recorded 25-OHD found in the EMR. A univariate descriptive statistical analysis was conducted using a non-parametric procedure, Kaplan-Meier (KM) method to estimate overall survival (OS).
The institutional review board at the University of Iowa, Carver College of Medicine, Iowa City, IA, USA approved this study (IRB ID #: 201909793).
RESULTS
A total of 211 patients with dystrophinopathy followed at the University of Iowa Neuromuscular Clinic between January 1, 2009 and September 22, 2019 were identified. Of those, only 30 met the inclusion criteria of at least one 25-OHD measurement ≥30 ng/mL and at least one subsequent 25-OHD measurement while on the same dose of vitamin D. These measurements were obtained at different times of the year. We subsequently excluded 4 additional patients for the following reasons: poor medication compliance as documented in clinical notes (1), vitamin D dosage of > 10,000 units per day (1), ulcerative colitis which could affect the absorption of vitamin D (1), and the use of anti-epileptic medications (1). One patient was taking a stable weekly dose of vitamin D, so an equivalent average daily dose was calculated. Among the 185 patients who did not qualify for the study, 90% were white, their median age of diagnosis of dystrophinopathy was 4 years and at the time of data review 16 years, and 52% were wheelchair dependent at the time of data review (the median age when these patients became wheelchair dependent was 10 years).
A total of 26 unique subjects thus met all inclusion and exclusion criteria. Four of these unique subjects were classified as having Becker or intermediate phenotypes whereas the remainder of the subjects had the Duchenne phenotype. Five of the 26 subjects were studied twice on different doses of vitamin D. These patients met enrollment criteria as they were on a stable dose of vitamin D with a follow up vitamin D level, and at a later time point, were on a stable higher dosage of vitamin D with follow up level. Thus, they were analyzed twice on different doses. Therefore, our study sample totaled 31 entries. The 5 patients who were analyzed twice all had the Duchenne phenotype. The reason for the change in the vitamin D dose was a drop in 25-OHD while on the lower dose in 4 of them and was unclear in the fifth (since he had a normal 25-OHD on both doses). Table 1 summarizes the demographic and treatment information for the full study population, and separately for subjects with the Duchenne phenotype and those with the Becker or intermediate phenotypes. Demographic and treatment information for patients included twice is provided in Table 2.
General characteristics and treatment distribution
BMD: Becker muscular dystrophy; DMD: Duchenne muscular dystrophy, IQR: interquartile range, 25-OHD: 25 hydroxyvitamin D. * Total number of entries analyzed 31(including subjects analyzed twice). Other statistics provided for 26 individual subjects.
General characteristics and treatment distribution of patients analyzed twice (labeled as entry 1 and 2)
25-OHD: 25-hydroxyvitamin D.
The median daily vitamin D dose for all subjects was 2000 (IQR 1000) units. The median initial 25-OHD concentration was 40 ng/mL (IQR 10). The median age of entry into the study (i.e. when the first 25-OHD concentration of D ≥30 ng/mL was achieved while on a stable dose of vitamin D) was 16 years (IQR 6). The median duration of follow-up for all study patients was 1120 days (∼37 months, IQR 1089), and the median time between 25-OHD measurements was 413 days (∼ 13.8 months, IQR 520). All patients received a stable dose of glucocorticoids throughout the study follow-up. During follow up, 3 patients sustained fractures upon falls, 2 in the group that maintained a normal 25-OHD (femoral neck and humerus fractures respectively) and 1 in the group whose vitamin D dropped to < 30 ng/mL (elbow and L4 vertebral fractures).
A survival analysis was performed with “survival” defined as a 25-OHD measurement that remained ≥30 ng/mL throughout the duration of follow-up. The 25-OHD concentration dropped to < 30 ng/mL in 6 of the 31 entries that constituted our study population (19%). In these cases, the individual nadir concentrations (defined as the lowest 25-OHD concentration below 30 ng/mL that subjects reached) ranged between 26 and 29 ng/mL, with a median of 28 ng/mL (IQR 3). Kaplan-Meier estimated overall mean time before 25-OHD decreased below 30 ng/mL (i.e. mean time-to-event) was 987 days (∼33 months, SE 46) (Fig. 1). Similar results were obtained when the survival analysis was conducted without the five duplicate entries.
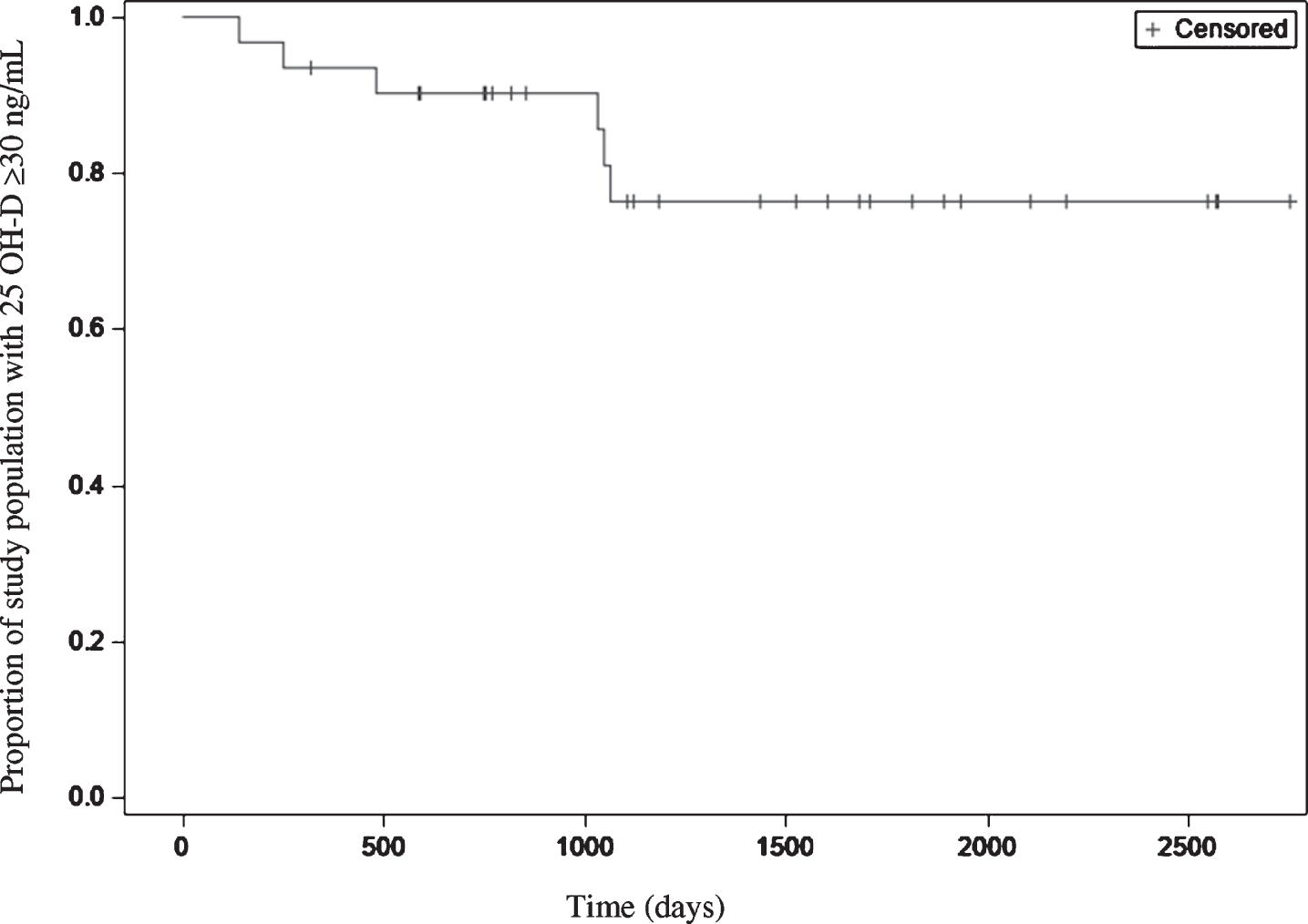
Kaplan-Meier survival curve with all subjects at a baseline 25-OHD measurement ≥30 ng/mL.
We then eliminated the four patients with Becker or intermediate phenotypes from the study group and repeated the survival analyses on those with the Duchenne phenotype only (22 individual patients, 5 analyzed twice, total DMD population 27). All six of the entries where the 25-OHD concentration dropped to < 30 ng/mL belonged to this group (22%). Kaplan-Meier estimated overall mean time before 25-OHD decreased below 30 ng/mL (i.e. mean time-to-event) was 975 days (∼33 months, SE 52). The median age at start of follow-up and weight gain during follow up were 16 years and 4.6 Kg respectively in the patients who maintained a stable 25-OHD, and 13 years and 10.7 Kg respectively in those whose 25-OHD concentration dropped to < 30 ng/mL.
DISCUSSION
To our knowledge, this is the first study that assesses the stability of 25-OHD concentrations over time in patients with dystrophinopathy who are taking a stable dose of vitamin D. Our findings demonstrate that most (81%) individuals with a 25-OHD concentration ≥30 ng/mL on a stable dose of vitamin D for at least three months maintained a 25-OHD ≥30 ng/mL for around 3 years, while continuing the same dose of vitamin D. Those whose 25-OHD concentration dropped to < 30 ng/mL had their lowest value at approximately 2–2.5 years after the start of follow-up, with the lowest 25-OHD concentration being 26 ng/mL (and the median at 28 ng/mL). This rather mild drop is of uncertain clinical significance because while 25-OHD concentrations between 21 and 29 ng/mL are classified as vitamin D insufficiency, it is unclear whether a 25-OHD concentration of 25–29 ng/mL increases the risk for fractures [22, 23].
Based on these results, we believe that in patients with dystrophinopathy who are taking a stable dose of vitamin D and whose 25-OHD concentration is ≥30 ng/mL, measurement of the 25-OHD concentration every 2 years may be sufficient. This can benefit patients who would otherwise not need phlebotomy performed for other laboratory tests. Patients with a 25-OHD < 30 ng/mL will continue to require more frequent monitoring until they achieve a 25-OHD concentration of at least 30 ng/mL while taking a stable dose of vitamin D for at least three months.
Some limitations to this study are worth noting. First, the sample size is small and from a single geographic area. It is possible that there would be less vitamin D level stability in a larger or different population. Our patient population is almost exclusively Caucasian, so these findings may not be generalizable to a population of darker skinned individuals where vitamin D sufficiency may not be maintained for as long due to reduced absorption of ultraviolet rays in sunlight by the skin. In addition, compliance tends to be high in our clinic population and only subjects with high compliance were included in this study, so our conclusions might not be generalizable if low compliance is suspected. Another limitation is the lack adjustment for body mass index (BMI). We recognize that a change in BMI over time in these patients (due to chronic treatment with glucocorticoids, limited physical activity or other factors) may affect 25-OHD concentrations. However, BMI is not accurate in non-ambulatory patients using estimated heights, so we could not use it for statistical adjustments. In Table 1 we present the patients’ weights at the start of follow up. There was some difference in weight between patients who maintained a stable 25-OHD and those whose 25-OHD dropped to < 30 ng/mL (median weight 56 Kg and 44 Kg, respectively), but these data are of limited clinical significance in the absence of height measurements, given the wide age range of our patients. Only one of the patients reported here was ambulatory throughout the study period, and his BMI was 21.7 at study entry and 23.6 after a follow-up period of 3 years with a stable 25-OHD concentration. Furthermore, this was a retrospective study spanning 10 years, so there was variability in the frequency of vitamin D monitoring as well as in clinical decision-making regarding the adjustment of vitamin D dosage and the specific vitamin D formulation used.
It is also important to note that the 25-OHD measurements in this study were performed by multiple methodologies, although the majority were either by Roche Diagnostics electrochemiluminescence immunoassay or DiaSorin chemiluminescence. There are a variety of factors that can influence 25-OHD measurements including analytical variations between assays, proportion of 25-OHD2 relative to 25-OHD3 in the patient sample, and presence of the C-3 epimer. While there has been ongoing work on standardizing 25-OHD assays, results can vary between assays, especially at extremely low or high 25-OHD values [21]. Analytical assay variation is likely not a major factor in the present study, since the 25-OHD concentrations analyzed in the study population are generally within the range where 25-OHD immunoassays correlate well with one another [20]. Some immunoassays cross-react less with 25-OHD2 compared to 25-OHD3 and this may be a factor when vitamin D2 supplements are used. The C-3 epimeric forms of vitamin D3 are most common in infants and young children [24], and is thus less likely to affect the results of the current study since our patients entered the study as adolescents or young adults. This epimer is detected in full by mass spectrometry-based assays but cross-reacts variably by marketed immunoassays. This can lead to variability in measurements in infants and young children.
In conclusion, our results suggest that vitamin D monitoring frequency can be reduced to every 2 years in patients with dystrophinopathy who meet the following criteria: older than 5 years; on a stable dose of vitamin D that does not exceed 10,000 units daily for at least three months; with high medication compliance; have no concomitant conditions that can impair vitamin D absorption, and have a 25-OHD concentration ≥30 ng/mL. Decreasing the frequency of 25-OHD concentration monitoring in this group can reduce healthcare costs and eliminate the discomfort of phlebotomy -which is difficult in some patients with contractures- if other laboratory tests are not required. Pending additional studies on the topic, patients who do not meet the criteria specified above should continue to receive annual or more frequent monitoring as dictated by their clinical status.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
