Abstract
Background:
Spinal Muscular Atrophy (SMA) is the most common neurodegenerative disease in childhood. New therapeutic interventions have been developed to interrupt rapid motor deterioration. The current standard of clinical evaluation for severely weak infants is the Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP INTEND), originally developed for SMA type 1. This test however, remains subjective and requires extensive training to be performed reliably.
Objective:
Proof of principle of the motion tracking method for capturing complex movement patterns in ten children with SMA.
Methods:
We have developed a system for tracking full-body motion in infants (KineMAT) using a commercially available, low-cost RGB-depth sensor. Ten patients with SMA (2–46 months of age; CHOP INTEND score 10–50) were recorded for 2 minutes during unperturbed spontaneous whole-body activity. Five predefined motion parameters representing 56 degrees of freedom of upper, lower extremities and trunk joints were correlated with CHOP INTEND scores using Pearson product momentum correlation (r). Test-retest analysis in two patients used descriptive statistics.
Results:
4/5 preselected motion parameters highly correlated with CHOP INTEND: 1. Standard deviation of joint angles (r = 0.959, test-retest range 1.3–1.9%), 2. Standard deviation of joint position (r = 0.933, test-retest range 2.9%), 3. Absolute distance of hand/foot travelled (r = 0.937, test-retest range 6–10.5%), 4. Absolute distance of hand/foot travelled against gravity (r = 0.923; test-retest range 4.8–8.5%).
Conclusions:
Markerless whole-body motion capture using the KineMAT proved to objectively capture motor performance in infants and children with SMA across different severity and ages.
INTRODUCTION
Spinal Muscular Atrophy (SMA) is the most common neurodegenerative disease in childhood. Approximately 14 / 100.000 live-born infants are affected. In more than 95%of cases a homozygous deletion in the SMN1 gene is responsible for this autosomal recessive disorder [1, 2]. There are different types of SMA, which are clinically classified based on motor function capability and age of onset [3, 4]. SMA type 1 is the most severe form and presents in infants younger than 6 months of age. Progression of disease leads to severe muscle weakness, progressive respiratory insufficiency and if untreated most of the patients die before the age of 2 years. Patients with intermediate form (type 2 SMA) are less affected and learn to sit, but are too weak to achieve ambulation, whereas SMA type 3 manifests after children have accomplished independent ambulation.
Recently, splicing modifiers and gene-replacement therapy have been developed to interrupt the rapid motor deterioration. Nusinersen is an intrathecally injected antisense oligonucleotide which was approved by the European Medicines Agency (EMA) in 2017. In 2020, a single dose gene replacement approach (Onasemnogene Abeparvovec-Xioi) aiming to deliver the SMN 1 gene in patients with severe SMA became commercially available in the US and Europe. First promising results have demonstrated the potential effectiveness for infants with SMA [5–7].
The current standard of clinical evaluation for severely weak infants is based on the Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP INTEND) score, which was developed to evaluate motor function in infants with SMA type 1 [8, 9]. It has shown to track motor function over time in infants with SMA with better delineation on the weak side of SMA spectrum. Although initially developed for SMA type 1 it has been used in a wider spectrum of infants and children with neuromuscular disorders ranging from presymtomatic infants with SMA, children with myotubular myopathy to Pompes disease) [10–14]. CHOP INTEND evaluates a child’s ability to move his/her extremities, head and trunk by rating performance of 16 items using a 4-point scaling system. The maximum score of 64 represents normal performance on the test. The evaluator has to encourage the child to perform gross motor tasks (e.g. to roll onto their stomach) and documents the amount of support which is necessary to achieve the task. It has been shown, that the assessment remains subjective and requires extensive training to be performed reliably [9]. Additionally, some of these activities, may be not only challenging, but also profoundly disturbing for very weak children, especially for the ones with a fragile health condition. Therefore, alternative assessment tools are eagerly awaited to objectively quantify whole body movements in infants with SMA across time. Ideally, these should be applicable prior to disease onset and should be sensitive to disease progression as well as therapeutic effectiveness. Furthermore, they should be easy to use, not interfere with the patient's health, and be affordable.
We have developed a system for tracking full-body motion in infants using a commercially available, low-cost RGB-depth sensor (Kinematic Motion Analysis Tool; KineMAT). To track the motions of infants we use the Skinned Multi-Infant Linear body model (SMIL model), a 3-D-full body model relying on the body surface of infants (see Fig. 1; for details on method development and clinical prior applications see [15–18] The SMIL model provides time series of angles in three degrees of freedom for all body joints, as well as positional information for all joints and arbitrary points on the body.
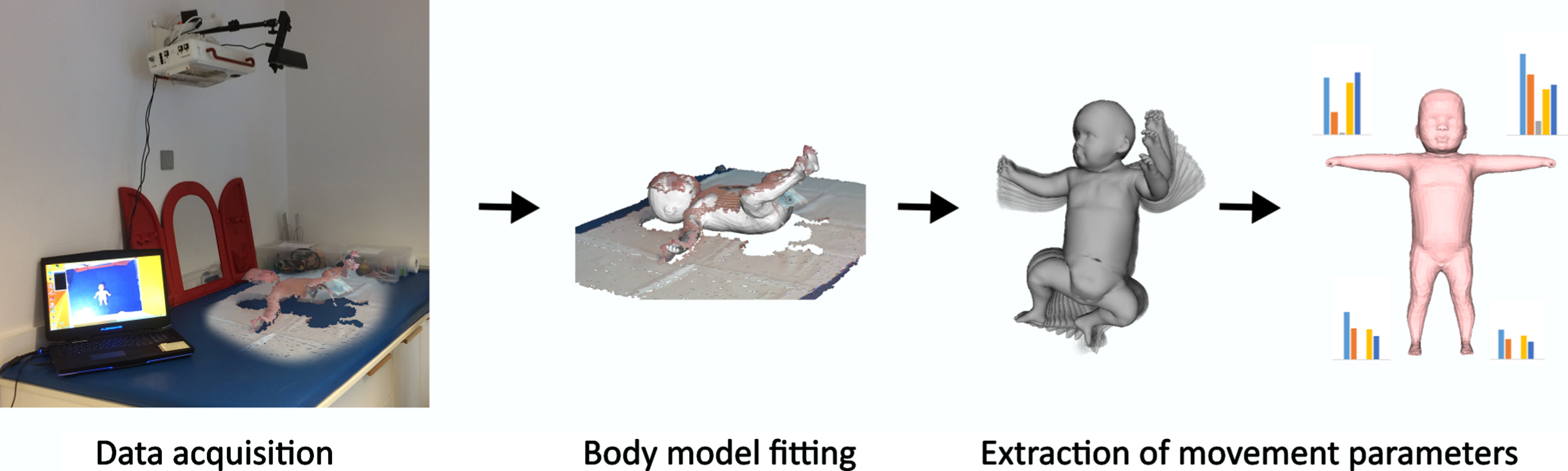
Examination set up. Legend: Data processing pipeline. From left to right: Data acquisition: the patient is recorded in supine position with an RGB-D sensor. Body model fitting: The SMIL model is aligned to the 3D data so that it best matches the pose and shape of the real child. Extraction of movement parameters: From the angles and joint positions of the complete sequence, different movement parameters are computed for the extremities.
We showed that the amount of motion detail captured with our method allows accurate assessment of general movements when the virtual infant reproduces the captured motions [18], and can be used to capture different movement patterns in different disease entities (e.g. Cerebral Palsy, Trisomy 21, neuromuscular disorders and genetic disorders (e.g. Smith-Lemli-Opitz Syndrom and SMA) [17]. The hallmark of muscle weakness in children with SMA is characterized as proximal weakness with predominant involvement of lower limbs. Thus, motor tasks of extremities should be a good measure to reflect the typical clinical phenotype in SMA.
The primary aim of this study was to demonstrate the proof of principle of the motion tracking method using the SMIL model for capturing complex movement patterns in ten children with SMA representing a broad spectrum of age and disease severity and to correlate quantitative and qualitative motion parameters derived from the SMIL model using the CHOP INTEND score as the present clinical gold standard.
PARTICIPANTS AND METHODS
Participants
Ten representative patients from our outpatient clinic at the iSPZ Hauner, University Children’s Hospital, Munich, Germany were chosen for the analysis. CHOP INTEND values and the patient’s age were selected to best represent a broad clinical spectrum of children with SMA. Patient specific data was tabulated (e.g. type of SMA, age at recording, SMN2 copy number, medical therapy, additional medical conditions; see Table 1) and correlated with the patients’ CHOP INTEND score at the time of the video recording.
Clinical characteristics of each patient
Methods
Two minutes of uninfluenced spontaneous activity of patients was recorded in a peaceful, quiet environment during routine outpatient visits. The video recording was performed prior to the general neurological examination and the structured CHOP INTEND assessment. Both, a 2D RGB video and a 3D depth video were recorded simultaneously using a Kinect 1.0 (Microsoft, USA). The camera was placed 100 cm directly above the child. No markers were positioned on the patient’s body.
The SMIL model is registered to the RGB-D sequences, where registration denotes the process of automatic adjustment of shape and pose of the model so that the resulting virtual body best matches the surface of the real child in the 3D point cloud data, as demonstrated in Fig. 1. Further details on the development of the method of SMIL model registration to RGB-D sequences are described in detail in previous publications [15, 16]. The registered SMIL Model allows the extraction of angles of all body joints, 3D locations of arbitrary points on the body, as well as velocities and accelerations of different body parts. Although the model is specifically adapted to infantile body proportions between the age of two and four months, we observe accurate tracking results for older children included in this study. Measures based on 3D locations are highly dependent on body size and comparisons between children of different age and size would be highly unreliable without further processing. The result of our tracking method is not only the motion, but also the body shape of the recorded child. Before the extraction of 3D positional data, we can set the captured body shape to be the same for all children, while leaving the captured motion untouched. This allows comparing the movements of children of different sizes without the need for normalization using additional measures like body size or limb lengths. Our tracking method captures fast movements and handles self-occlusions, which are a common problem when capturing motions of freely moving humans with only one camera [15].
The clinical phenotype of patients with SMA is characterized by progressive and proximally pronounced muscle weakness. Therefore, the clinical motion pattern shows movements in the head and neck muscles, and the distal upper and distal lower limb, in which the upper limb shows more activity compared to the lower limb. We therefore extracted five motion parameters which should track SMA specific proximal muscle weakness in 2D and 3D space over time.
We extract parameters for each of the 3 degrees of freedom of the joints. For upper limb, we use shoulder, elbow, and wrist joints. Lower limb includes hip, knee, and ankle joints, and torso/head is comprised of head, neck, spine and collarbone joints (as defined by the SMIL model [16]. The parameters are averaged to create one score per extremity. We evaluated each extremity individually as well as combined in a whole-body sum score: Standard deviation of joint angles for each extremity over complete sequence (measured in degrees°) Standard deviation of joint position over complete sequence (cm) Volume covered by hand/foot in complete sequence (cm3) Absolute distance of hand/foot travelled per minute (cm/min) Absolute distance of hand/foot travelled against gravity per minute (cm/min)
STATISTICAL ANALYSIS
Descriptive statistics were used to describe the children’s clinical information on single subject level. Pearson product momentum correlation (denoted r) was calculated as a measure of the linear correlation between CHOP INTEND values and the five motion parameters. For the given set of 10 patients, the power to detect a statistically significant correlation would be a Pearson correlation above 0.828 (given a statistical power to be set to 0.9 and a significance level of α= 0.05) [19].
The local ethics committee approved this study prior to the inclusion of the first patient (Ethikkommission LMU, Project Nr. 454-16). Parents gave written informed consent prior to participating in this study. No family withdrew consent during the study period.
RESULTS
Eight of ten children were classified as having severe form of SMA (SMA type 1) with lacking ability to sit, respiratory involvement ranging from frequent (severe) infections to permanent invasive ventilation. The remaining two patients were classified as type 2 SMA, since they gained the ability to sit independently. Mean CHOP INTEND value in the assessed patients was 34 (range 10–50). The mean age of all patients was 17 months (range 2–46 months of age). Apart from one patient, in which decision for palliative care was reached, all patients were receiving specific treatment for SMA (i.e. regular intrathecal injection of Nusinersen; details see Table 1).
All five predefined motion parameters correlated with CHOP INTEND results (Table 2; raw data see Suppl Table S1a-S1e). The values for each individual extremity showed more variation compared to the whole-body average scores, and the upper extremity joints usually show higher absolute values than the ones for the lower extremities. All but one parameter (volume covered r = 0.773) reached significance for the Pearson product momentum (r > 0.828; Table 2). Figure 2 depicts graphically an example of the tabularized results for the parameter standard deviation of joint angles. Correlation to the CHOP INTEND values (r = 0.959) (Fig. 2A) and the raw data for the motion parameter for each individual patient (Fig. 2B) are shown.
Correlation CHOP INTEND versus KineMAT motion parameters
Legend: Pearson product momentum correlation (r) of CHOP with each individual motion parameter. Significant values are printed in
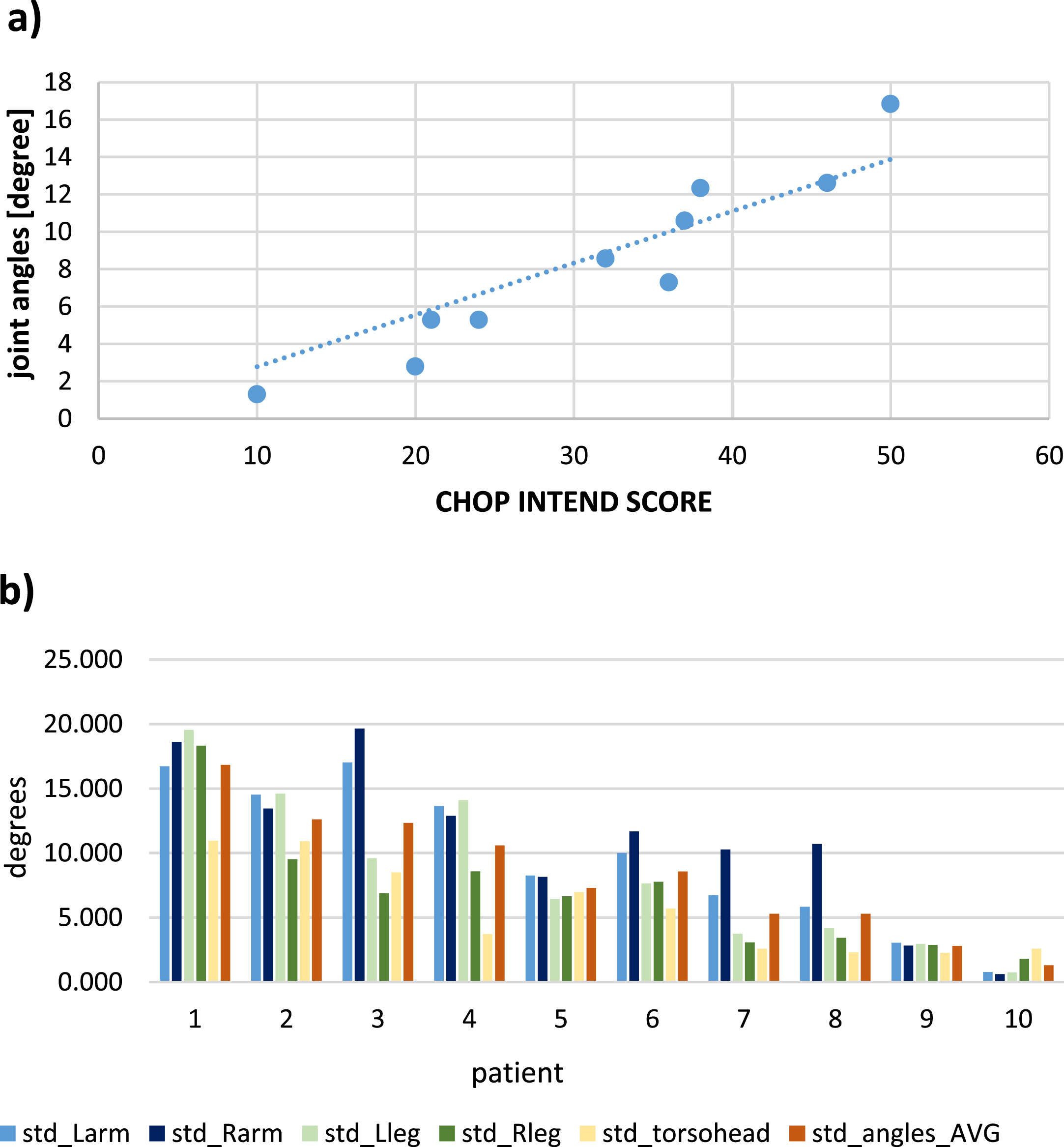
“Joint angles” versus CHOP-INTEND Score of all 10 patients. 2a: correlation of all patients. 2b: distribution across extremities. Legend: std: standard deviation, of value over complete sequence, Larm: left arm, Rarm: right arm, Lleg: left leg, Rleg right leg, AVG: average over all measurements.
For two patients (No. 6 and 7) a test-retest analysis was possible with two independent sequences being obtained at the same day. Table 3 shows the average scores of each motion parameter (raw data available in supplementary Table S2). Test-retest session values differed between 0.9%for the parameter volume covered in space and 10.5%for average distance travelled. Due to low numbers statistical analysis was kept at the descriptive level.
Test retest analysis
Legend: Test-retest comparison in two patients recorded twice at the same visit. Variation: difference between baseline and follow-up measurement/range of values in all patients of each motion parameter (%).
DISCUSSION
With this case-study we present first data on the clinical application of markerless full-body motion tracking in children with SMA. Ten patients with a broad spectrum of disease severity based on CHOP INTEND values and a wide range of age [2–46 months] were selected for this pilot study.
By assessing just two minutes of RGB-D video recordings of unperturbed patients, we demonstrate, that four out of five preselected simple motion parameters show an excellent positive linear correlation (i.e. r = 0.923 to 0.959) with the established gold standard of clinical assessment tool (CHOP INTEND score). Only one motion feature (volume covered in space, r = 0.773) did not reach the predefined statistically significant correlation with CHOP INTEND scores (r > 0.828), however by standard definitions it is considered a strong correlation.
Motion tracking in infants / KineMAT system
Multiple systems have tried to quantify body movements in infants by using markers or sensors that are attached to the infants’ body [20–22]. Since 2014 we have developed an automated system that captures 3D joint positions and head rotation of infants without attached markers or sensors. [15, 24]. Selected motion parameters represent measures of body joint angles and positions, volume and distances covered in 2-D and 3-D space. By this, an objective substitute measure describes a wide range of motor abilities which are characteristic for the muscle weakness in children with SMA. Even the greater extend of weakness in the lower limbs can be clearly detected by the system (e.g. Fig. 2b). After analysis of this pilot data, however, we suggest that in SMA, analysis of average values (i.e. the sum of measures for all body regions) for our predefined motion features seems to be superior compared to the assessment of single joints or limbs (for detailed comparison see raw data in supplementary Tables S1 & S2). This seems reasonable, as asymmetry of motor performance is not a typical feature in children with SMA.
In summary KineMAT allows to analyze data from a commercially available RGB-D sensor (Microsoft Kinect system) which is taken while the child is placed on a mattress on the floor with diapers on. With this setting a comfortable environment can be realized which considers the special needs of infants (e.g. warmth, comfort). Short scanning time permits reduction of artefacts from fatigue or mood changes. Thus, the examination itself can easily be integrated into the routine outpatient clinics.
Quantitative motion measurement with KineMAT in comparison to CHOP INTEND
The CHOP INTEND is the gold standard to quantify motor capacities over time in children with SMA in clinical routine as well as in scientific settings. Initially developed for severely weak infants with SMA type 1 it is now widely used in infants and small children ranging from trials with presymptomatic infants with genetically proven SMA [10, 14] to patients with myotubular myopathy or Pompes disease [11, 13]. During the last years a change by four points was accepted as clinically relevant [25]. The main disadvantage of the CHOP INTEND is the need for extensive and repeating training of examiners in addition to long duration of the assessment itself. To maintain high quality standards CHOP INTEND requires a tight, repetitive schedule of staff training [9], not only in the light of multi-centered, international clinical trials. Despite all efforts to achieve high quality ratings with the CHOP INTEND, it remains a subjective assessment tool prone to human bias. In addition, the CHOP INTEND shows a ceiling effect towards better motor function with limited variability in scores between subjects with well-preserved motor function [8]. For these patients additional assessments e.g. Hammersmith infant neurological exam (HINE) are chosen to accurately assess motor abilities.
In contrast to the CHOP INTEND assessment, we did not observe a ceiling effect in the quantification of motor abilities. This is in line with previously published data in which we could demonstrate the capability of the KineMAT to reliably detect very subtle movements (such as fidgety movements in infants three months of age) [18]. Thus the KineMAT system could be a good addition to standard motor function tests in SMA, especially in the light of asymptomatically treated infants with its short examination time of 2–3 minutes with no loss of accuracy compared to CHOP INTEND.
Computerized CHOP INTEND calculation
Recently, different methods were published to automatically predict CHOP INTEND scores for SMA from motion tracking data [26, 27]. Both used motion tracking in combination with computerized analyses such as elastic net regression [26] or convolutional neural network (CNN) models [27]. Both applications were able to predict the CHOP INTEND score with an average error of 7 (out of a maximum of 72) points [27], respectively up to 8.55 points [26] compared to the manually taken test. Chen et al. showed a progression plot of tests taken from three individual patients over time which show a fairly good concordance of automated vs manually derived CHOP INTEND scores, however on single subject levels the automated score differed considerably from the clinical rating [26]. Taken into consideration that a change of four points in pivotal SMA trials was considered clinically relevant [25], the observed differences of up to 20 points predicted vs. manually scored CHOP INTEND seems undoubtedly too large. As soon as data from more patients and multiple time points are available, we plan to test an automated CHOP INTEND prediction using our motion features.
LIMITATIONS AND FUTURE WORK TO DO
As of now, there are several significant limitations, which have to be kept in mind. The main limitations are: (1) this is only a pilot study in ten preselected patients. We aimed to prove the principal possibility to measure movements in SMA infant with an markerless motion tracking system (KineMAT). Our selected motion parameters, therefore, have to be validated in a larger unselected SMA population prospectively and in an intraindividual long-term follow up including test-retest reliability. (2) No normative values for our motion parameters have been established. This would allow to define a cut off for normality in different age groups and would be of utmost importance for high functioning babies who received the diagnosis of SMA via a newborn screening entering modern therapies while still being clinically asymptomatic. By using the assessment of General Movements in the age group up to 4 months after birth we will address this question in more detail. (3) The KineMAT is not yet publicly available to clinical and scientific community, however comparative studies will be possible in the future as raw data on full body motion activity is stored and may be used for further analyses.
CONCLUSION
Markerless full whole-body motion capture using the KineMAT proved to objectively capture motor performance in infants and children with SMA across different severity and ages. The virtual full body infant models derived from raw data acquired with low-cost commercially available RGB-D camera are very accurate, resistant to measurement errors and require low technical skills. Quantitative assessment of full body movements showed an excellent correlation with established gold standards (CHOP INTEND). The portable system will allow to record consecutive data of children with SMA in a home-based fashion which minimizes the stress and effort for the child itself and its parents. Consequently, the KineMAT system permits long term investigation of motor development in SMA potentially comparing efficiency of drug intervention.
Footnotes
ACKNOWLEDGMENTS
We would like to thank all our families for their active participation in this study. WMF, KV und AB are members of the European reference network (ERN) Neuromucular diseases.
CONFLICT OF INTEREST
A. Blaschek reports personal fees (speaker honoraria) from Sanofi-Aventis, Merck, Biogen and Novartis outside the submitted work.
N. Hesse provides consulting services for Meshcapade GmbH, outside the submitted work; In addition, Dr. Hesse has a patent US20200058137A1 pending.
W. Müller-Felber is serving on a scientific advisory board or data safety monitoring board for Biogen, Novartis, PTC, Sanofi Aventis and Cytokinetics and received travel and speaker honoraria from Biogen, Novartis, PTC and Sanofi-Aventis, all outside the submitted work.
S.A. Schroeder reports grants from Lions Club Bavaria, during the conduct of the study.
K. Vill; F. Heinen; B. Warken; T. Well and C. Hodek have nothing to disclose.
