Abstract
Background:
GNE myopathy (GNEM) is a rare, adult-onset, inclusion body myopathy that results from mutations in the GNE gene. GNE encodes UDP-GlcNAc epimerase/ManNAc-6 kinase, a protein with two enzymatic activities that comprise the committed step in biosynthesis of sialic acid (SA), an essential glycan that appears on the terminal positions of many extracellular oligosaccharide chains. These GNE mutations can cause a reduction of SA in many tissues, although pathology is restricted to skeletal muscles through a poorly understood mechanism.
Objective:
Despite recent advances in the field, it remains unclear which therapeutic avenue is most promising for the restoration of SA level in skeletal muscle affected by GNEM. Our objective was to assess dietary and gene therapy strategies for GNEM in Cmah-deficient GNED207V Tg Gne-/- mice, a model that allows for the visualization of orally delivered N-glycolylneuraminic acid (Neu5Gc), one of the two predominant SA forms in muscle.
Methods:
Methods included in situ physiology studies of the tibialis anterior muscle, studies of ambulation and limb grip strength, and muscle staining using MAA, SNA, and anti-Neu5Gc antibody, along with qPCR, qRT-PCR, western blot, and HPLC studies to assess virally introduced DNA, GNE gene expression, GNE protein expression, and SA expression.
Results:
We found that a diet enriched in Neu5Gc-containing glycoproteins had no impact on Neu5Gc immunostaining in muscles of GNEM model mice. Delivery of a single high dose oral Neu5Gc therapy, however, did increase Neu5Gc immunostaining, though to levels below those found in wild type mice. Delivery of a single dose of GNE gene therapy using a recombinant Adeno Associated Virus (rAAV) vector with a liver-specific or a muscle-specific promoter both caused increased muscle Neu5Gc immunostaining that exceeded that seen with single dose monosaccharide therapy.
Conclusions:
Our findings indicate that dietary loading of Neu5Gc-containing glycoproteins is not effective in increasing muscle Neu5Gc expression, while single dose oral Neu5Gc monosaccharide or GNE gene therapy are. Neu5Gc immunostaining, however, showed greater changes than did lectin staining or HPLC analysis. Taken together, these results suggest that Neu5Gc immunostaining may be more sensitive technique to follow SA expression than other more commonly used methods and that liver expression of GNE may contribute overall muscle SA content.
INTRODUCTION
GNE myopathy (GNEM) is a severe autosomal recessive myopathy characterized by progressive muscle weakness and atrophy. The age of onset is typically in the third or fourth decade of life, beginning with weakness in the tibialis anterior (TA) and hamstring muscles and often rendering patients non-ambulatory by the second decade after diagnosis [1]. Muscle pathology includes rimmed vacuoles and inclusion bodies that contain various proteins including the Aβ1 - 42 peptide [2]. GNEM is caused by mutations in GNE (UDP-GlcNAc-2 epimerase/ManNAc-6 kinase) [3], the gene that encodes a bifunctional enzyme required for synthesis of sialic acid (SA) [4]. The SA biosynthetic pathway culminates in the production of CMP-SA, which is utilized by sialyltransferases to transfer SA onto glycoproteins and glycolipids [5]. CMP-SA also acts as a negative feedback regulator of GNE through allosteric inhibition [6, 7]. SA addition is an essential post-translational modification, occurring on many glycoproteins and glycolipids in all cells throughout the body [5]. Common founder mutations in GNE exist in GNEM patients of Japanese (D207 V) and Middle Eastern (M743T) descent [1]. Mutations in the GNE gene that cause disease are typically missense mutations that lead to reduced GNE enzyme activity and consequently lower SA production [1]. Complete loss of Gne expression leads to embryonic lethality in the mouse [8]. While hyposialylation is often a finding in skeletal muscle of patients with GNEM, the pathophysiology remains unclear. Various studies have suggested Aβ1 - 42 cytotoxicity, induction of the unfolded protein response, and changes in reactive oxygen species levels as mechanisms that contribute to disease pathology [9–12].
Several efforts have been made to develop an animal model of GNEM. This includes GNED207V Tg Gne-/- mice, which are reported to show appropriate progressive muscle pathology, generalized muscle weakness, and reductions in lifespan. By 30 weeks, GNED207V Tg Gne-/- mice show reduced scores in rod climbing and constant speed treadmill walking, modest elevation in serum CK activity and muscle production of Aβ1 - 42 peptide [21, 41]. By 42 weeks, muscles are reported to exhibit rimmed vacuoles with congophilic inclusion bodies, as well as pathology in respiratory and cardiac muscles that are not observed in human GNEM patients [21, 41]. A knock-in model of the GneM743T mutation showed unexpectedly severe disease that was not related to GNEM, specifically perinatal lethality from kidney disease [13]. With extended breeding, however, this model generated mice with more subdued phenotypes, including some mice with no apparent disease and prolonged lifespan [14].
Given the hyposialylation present during GNEM, treatment strategies have focused on restoration of SA to the muscle membrane through oral glycan supplementation or gene therapy approaches. Most therapies in development for GNEM rely on oral supplementation of SA or its precursors. These have included N-Acetylmannosamine (ManNAc) [13, 15–18], which can also be converted to ManNAc-6 phosphate, the end product of GNE activity, through a GlcNAc kinase activity that is not mutated in GNEM, and aceneuraminic acid extended release capsules (Ace-ER) [19, 20]. Exogenous ManNAc supplementation has been shown to increase SA levels in serum and tissues and prevent formation of histopathology, atrophy, and muscle weakness with continuous administration over approximately one year in GNED207V Tg Gne-/- mice [16, 21]. ManNAc can also rescue perinatal lethality in the GneM743T mouse [15]. This approach is currently being tested in phase 2 clinical trial (NCT 04231266). While SA therapy was shown to have biological effects in the GNED207V Tg Gne-/- mouse model [17, 21], Ace-ER met none of the clinical milestones tested in a phase 3 clinical trial of GNEM patients despite a doubling of free SA in the serum of Ace-ER-treated trial participants [19]. These findings do not necessarily refute the efficacy of glycan therapy for GNEM, but they do suggest further knowledge is needed on SA dosing and the dynamics of SA expression and localization within skeletal muscles.
Gene therapy approaches are also in development for GNEM. An initial study tested GNE gene therapy delivered via lipoplex in a single patient [22]. This subject displayed evidence of GNE gene expression, stabilization of muscle function, and modest improvement in muscle sialylation. A thorough analysis of the efficacy using this approach is challenging, however, as only a single patient was treated. There have also been tests of systemic GNE gene replacement using recombinant Adeno Associated Viral (rAAV) vectors in wild-type mice [23].
While several studies have used HPLC to quantify increased SA in the muscle of mouse models after delivery of exogenous glycans [15, 24], these reports were not able to distinguish between endogenous SA and exogenously delivered SA. To visualize SA uptake in a manner that differentiates it from endogenous SA production, we tested oral SA therapy using N-glycolylneuraminic acid (Neu5Gc) in a GNEM model (GNED207V Tg Gne-/-) that is also deficient for the CMP-Neu5Ac hydroxylase gene (Cmah). The Cmah gene is required for CMP-Neu5Gc synthesis, and thereby for expression of all Neu5Gc on proteins and lipids [25–27]. Neu5Gc and N-acetylneuraminic acid (Neu5Ac) are the two most abundant forms of SA in most mammalian tissues [5], including mouse skeletal muscle [28], but modern humans lack the ability to make Neu5Gc due to an inactivating mutation in the human CMAH gene that is not present in other mammals, including non-human primates [27] and mice [26]. As a result, humans do not express Neu5Gc and instead replace this missing SA with additional Neu5Ac. Use of GNED207V Tg Gne-/-Cmah-/- mice, therefore, not only allows for visualization of delivered Neu5Gc, but creates a disease model with a more “humanized” SA repertoire. Neu5Gc differs from Neu5Ac only by having an additional single oxygen atom at the 5-N-acyl position. The elimination of this single oxygen from Neu5Gc removes perhaps a million oxygen atoms from the SA present in each cell, thereby creating unique SA epitopes that can be recognized by antibodies raised in birds (including chickens) who have evolved without a CMAH gene [29]. As GNEM is a disease involving SA biosynthesis, studies in mouse models lacking Neu5Gc could be important for understanding aspects of the human disease.
Modeling of human CMAH deficiency in Cmah-/- mice has shown absence of Neu5Gc to be an important disease modifier in multiple models for human diseases and disease processes, including inflammation [30], pathogen susceptibility [31–33], cancer [34, 35], diabetes [36], atherosclerosis [37], neuromuscular disease [38, 39], and muscle function [40]. For example, deletion of Cmah in the mdx mouse model of dystrophin-deficient Duchenne Muscular Dystrophy led to mice that displayed a more severe disease phenotype than did their mdx counterparts [38, 39]. Interestingly, a fraction of Cmah-/-mdx mice, but not mdx or Cmah-/- mice, developed serum antibody titers to Neu5Gc [39]. As regenerating skeletal muscle fibers in Cmah-/-mdx mice and in DMD muscles incorporate dietary Neu5Gc [28, 39], it is likely that this incorporated Neu5Gc acted as a xeno-autoantigen, triggering an immune response and exacerbating pathology. Here, we have used the novel GNED207V Tg Gne-/-Cmah-/- mouse model to address issues related to human Neu5Gc deficiency in GNEM as well as to visualize Neu5Gc delivered as a monosaccharide therapy or by dietary intake of glycoprotein.
MATERIALS AND METHODS
Mice
All animal experiments were conducted with approval from the Institutional Animal Use and Care Committee (IACUC) at The Research Institute at Nationwide Children’s Hospital. Mice lacking Cmah (Cmah-/-) were a generous gift from Dr. Ajit Varki (UC San Diego) [26]. Gne knockout mice transgenically expressing human GNE containing the D207 V mutation (GNED207V Tg Gne-/-) were provided by Dr. Satoru Noguchi and Dr. Ichizo Nishino [41]. GNED207V Tg Gne-/-Cmah-/- mice were obtained by interbreeding of Cmah-/- mice with GNED207V Tg Gne-/- mice. Mice were fed and watered ad libitum with standard mouse chow, (Cat # 2919, Teklad Global Rodent diet, Harlan, USA), which contains approximately 6μg of Neu5Gc per gram of chow [39], or with Neu5Gc-enriched mouse chow containing porcine submaxillary mucin (PSM), which contains 250μg of Neu5Gc per gram of chow (Dyets, Inc.; Bethlehem, PA). For high-dose oral sialic acid (SA) studies, mice received a single 2 g/kg dose of free N-acetylneuraminic acid (Sigma; St. Louis, MO) or 2 g/kg N-glycolylneuraminic acid (Sigma) diluted in drinking water by oral gavage.
Grip strength and treadmill ambulation
Forelimb and hindlimb grip strength were assessed in mice using a grip strength meter (Columbus Instruments, Columbus, OH). The highest three measurements were averaged from 10 forelimb repetitions and 10 hindlimb repetitions each day; mice were tested once per day for a week, and the daily measurements were averaged to produce a final forelimb and hindlimb measurements. To measure ambulation, the mice underwent a 75-min run on a treadmill with a 15° decline (Treadmill Simplex II; Columbus Instruments). The mice ran for 5 min at 5 m/min, which increased 1 m/min each minute until 15 m/min, and then remained at that speed for an additional 60 min. The time that the mice remained on the treadmill, up to a total time of 75 min, was recorded. After an initial week of training, mice were tested once per day for five days, and the daily measurements were averaged to produce a final measurement of treadmill running time before exhaustion. For all measures, GNED207V Tg Gne-/- and GNED207V Tg Gne-/-Cmah-/-mice were compared to littermate controls, which included Gne+/-, GNED207V Tg Gne+/-, Gne+/-Cmah-/-, and GNED207V Tg Gne+/-Cmah-/-mice.
Muscle physiology
Mice were anesthetized with ketamine/xylazine, and hindlimb skin was removed to expose the tibialis anterior (TA) muscle. The distal tendon was dissected out and sutured to a force transducer (Aurora Scientific, Aurora, ON, Canada). Muscle contractions were produced by stimulation of the sciatic nerve by bipolar platinum electrodes and the optimal length was determined by stretching the muscle until maximum twitch force was elicited. Maximum force was determined by successive stimulation at 50, 100, 150, and 200 Hz with a 1-minute rest period between each stimulation, and specific force was ascertained by dividing the maximum force by the muscle cross sectional area. Finally, the TA underwent 10 cycles of eccentric contractions, each in which the TA was stimulated for 350 ms total, the last 200 ms of this while being stretched by 10% before returning to optimum length. For these measurements, the maximum force generated in the first cycle prior to muscle lengthening is designated was 100%.
Histology
Mouse skeletal muscles were snap frozen in liquid nitrogen-cooled isopentane, mounted on corkboard, and cut in 10μm cross-sections on a cryostat. Hematoxylin and Eosin (H&E) staining was done as before [42]. Congo Red staining was performed by fixing sections in 95% ethanol and rinsing briefly, before subsequent incubations in Harris Hematoxylin with acetic acid, alkaline salt solution, and Congo Red solution (Poly Scientific R&D Corporation; Bay Shore, NY). Finally, slides were dehydrated through graded alcohols, cleared in xylene, and sandwiched with mounting media under a glass cover slip.
Fluorescence staining
Mouse skeletal muscles were snap-frozen in liquid nitrogen-cooled isopentane, mounted on corkboard, and cut in 10μm cross-sections on a cryostat. Slides being used for FLAG immunostaining were fixed in 4% paraformaldehyde, washed, incubated in 1% Triton, and washed again. All slides were blocked with phosphate-buffered saline (PBS) containing 10% goat serum (GS), for FLAG or lectin staining, or 10% Neu5Gc-free human serum (HS), for Neu5Gc staining, for 1 hour each. Sections were incubated with anti-FLAG (F7425, Sigma) or anti-Neu5Gc (Poly21469, Biolegend; San Diego, CA) antibody in blocking solution (10% GS or HS, as before) overnight at 4°C, and washed in PBS. Slides stained with anti-FLAG antibody were then incubated with goat anti-mouse IgG conjugated to Cy3 (111-165-144, Jackson ImmunoResearch; West Grove, PA), and slides with anti-Neu5Gc antibody were then incubated with donkey anti-chicken IgG conjugated to Cy3 (703-165-155, Jackson ImmunoResearch). Slides for lectin staining were incubated with fluorescein isothiocyanate (FITC)-labeled Maackia amurensis agglutinin (MAA) lectin (EY Laboratories; San Mateo, CA), Sambucus nigra agglutinin (SNA) lectin (EY Laboratories), or peanut agglutinin (PNA) lectin (EY Laboratories) at room temperature for 1 hour. All sections were washed and cover-slipped with mounting medium containing 4’,6-diamidino-2-phenylindole (DAPI). All imaging was done on a Zeiss Axiophot epifluorescence microscope using AxioVision LE 4.1 imaging software. Pixel density of staining in time-matched images of identical size (150,125μm2) was compared using NIH ImageJ software (imagej.nih.gov/ij/; version v1.47v, last access date 5/26/2021), as previously described [43].
Cloning and production of rAAV constructs
A cDNA encoding a C-terminal myc/FLAG-tagged wild-type GNE protein corresponding to the human GNE1 transcript [44], Genbank Accession Number: NM-005476; Origene; Rockville, MD), encoding a protein of 722 amino acids, was cloned into an rAAV.MCK vector and an rAAV.LSP vector using NotI sites. This yielded wild-type GNE under the control of a muscle-specific or liver-specific promoter, both with C-terminal myc and FLAG tags. rAAV vectors, rhesus 74 serotype, were produced by the Viral Vector Core at Nationwide Children’s Hospital. The standard triple transfection method was used to produce rAAV in HEK293 cells [45], and packaged vector was purified using sucrose density centrifugation and anion exchange chromatography, as previously described [46].
Injection of rAAV gene therapy vectors
For the liver- and muscle-specific expression of GNE, 2-month old GNED207V Tg Gne-/- or GNED207V Tg Gne-/-Cmah-/- mice were given unilateral intramuscular (IM) injections of 1×1011 vector genomes (vg) in the TA muscle and 5×1011 vg in the gastrocnemius muscle with rAAVrh74.MCK.GNE or rAAVrh74.LSP.GNE. Other mice were given intraperitoneal (IP) injection with 5×1011 vg of rAAVrh74.LSP.GNE or rAAVrh74.MCK.GNE . Uninjected age-matched mice served as controls in all instances. Mice were euthanized 10 months post-injection for analysis.
Quantitative polymerase chain reaction (qPCR)
TaqMan quantitative polymerase chain reaction (qPCR) was used to quantify rAAV vector genome (vg) copies in rAAV-treated muscles. Genomic DNA was extracted as previously described [47] from treated and untreated muscles or liver. DNA purity and quantity were measured using an ND-1000 spectrophotometer (NanoDrop; Thermo Fisher Scientific). Vector-specific primer/probe sets were used to amplify a portion of the vector DNA encompassing the 3′ end of the MCK promoter and the 5′ end of the GNE cDNA (forward: 5′-CGGAAGTGTTACTTCTGCTCTAA, probe: 5′-/56-FAM/CCGCCACCA/ZEN/TGGAGAAGAATG GAA/3IABkFQ/, and reverse: 5′-CGGTTACAAGTAGCAACACAAAC) for rAAVrh74.MCK.GNE or encompassing the 3′ end of the LSP promoter and the 5′ end of the GNE cDNA (forward: 5′-TTTCTCAGGATAACAAGAACGAAAC, probe: 5′-/56-FAM/AGCCACCGC/ZEN/TAGCAAGAAT TCGAT/3IABkFQ/, and reverse: 5′-CCGCAGC TTTCGGTTATTTC) for rAAVrh74.LSP.GNE. The plasmids used to make virus were linearized with MluI for rAAVrh74.MCK.GNE and rAAVrh74.LSP.GNE, re-purified, and utilized to generate a standard curve from 50 to 5 million copies in log increments. The correlation coefficient of the standard curve equaled or exceeded 0.99 for all runs. Samples were measured in duplicate, and data are reported as vgs per microgram of genomic DNA assayed. Calculations of vg/nucleus were based on an estimate of 1.7×105nuclei/μg genomic mouse DNA.
Semi-quantitative reverse transcription polymerase chain reaction (qRT-PCR)
Total RNA was isolated from frozen blocks of skeletal muscles or liver by use of TRIzol reagent (Invitrogen, Carlsbad, CA). RNA was purified on a silica gel-based membrane-containing column (RNeasy; Qiagen, Germantown, MD). Relative transcription levels were assessed by semiquantitative real-time PCR (qRT-PCR) using the 2-ΔΔCT method, with 18 S rRNA as an internal reference [48]. A high-capacity cDNA archive kit (Applied Biosystems, Foster City, CA) was utilized for the reverse transcription RNA as per the manufacturer’s guidelines. Samples underwent real-time PCR in duplicate using a TaqMan ABI 7500 sequence detection system (Applied Biosystems). Primer/probe sets for 18 S (assay ID: Hs99999901_s1), human GNE (Assay ID: Hs01103400_m1) and mouse Gne (assay ID: Mm00450174_m1) were purchased from Applied Biosystems (Thermo Fisher Scientific). A primer/probe set to recognize the myc/FLAG tag region common to both rAAVrh74.MCK.GNE and rAAVrh74.LSP.GNE was designed using PrimerQuest DNA software and synthesized by Integrated DNA Technologies (Coralville, IA)- 5’-CTACACAACACGCAGGATCT (forward), 5’-56-FAM/TCTCAGAAG/Zen/AGGATCTGGCAGCAA/ 3IABkFQ/ (probe), and 5’-ATCGTCGTC ATCCTTGTAATCC (reverse). This probe set was used to differentiate rAAV-driven GNE transgene expression from endogenous GNED207V transgene expression. In all cases, relative mRNA levels were averaged for each group.
Western blotting
Mouse skeletal muscle and liver samples were lysed in buffers containing 1% NP-40 (NP), 2% sodium dodecyl sulfate (SDS), or 2% SDS with 4% urea (Ur) where indicated. 40μg of protein from muscle lysate was diluted in SDS denaturing buffer and separated on 4–12% gradient SDS-PAGE gels and then transferred to nitrocellulose. After transfer, blots were blocked in TBST with 5% non-fat dry milk, then incubated with either anti-FLAG (F7425, Sigma) or anti-Gapdh (MAB374, MilliporeSigma; Burlington, Massachusetts), washed in TBST, incubated with appropriate horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (111-035-144, Jackson ImmunoResearch; Seattle, WA) or donkey anti-mouse IgG (715-035-150, Jackson ImmunoResearch) secondary antibody, washed again, and developed using an ECL developing kit (Amersham, Piscataway, NJ), much as previously described [49].
Measurement of free and total sialic acid using High Performance Liquid Chromatography
Muscle tissue was homogenized in 200 mM ammonium acetate, pH 4.4 (approximately 1 mL/100 mg of tissue) containing cOmplete mini protease inhibitor (Sigma Aldrich, St. Louis, MO). Free sialic acid (SA) was measured using a portion of the homogenate by precipitation of proteins in 60% ethanol at –20°C for one hour. Precipitated proteins were centrifuged at 16000 g for 10 minutes and removed, after which the supernatant was dried by Speedvac. Free sialic acids were then derivatized using 1,2-diamino-4,5-methylenediaoxy-benzene dihydrochloride (DMB). Samples were incubated with DMB reagent (a mixture of 1.5 mL distilled water, 172μL glacial acetic acid, 112μL of 2M mercaptoethanol, 4.9 mg of sodium hydrosulfite, and 3.5 mg of DMB (dissolved first in water)) for 2.5 hours at 50°C, after which reaction was stopped by freezing at –40°C. Total sialic acid was measured from another portion of the lysate. For this portion, sample proteins were lysed in 2M acetic acid at 80°C for 2 hours. Lysates were then centrifuged at 4000 g for 10 minutes and supernatants dried by Speedvac and labelled using DMB derivatization as above.
Concentrations of free and total SA were measured in the muscle samples by separation using high performance liquid chromatography (HPLC). Samples were separated on an C18 column (Agilent ZORBAX C18, 100×4.6 mm, 3.5μm). Analytes were eluted isocratically at a flow rate of 1 mL/min with water/acetonitrile/methanol 85/8/7 (v/v/v). Labelled SA was detected at 373 nm excitation/488 nm emission and compared to a calibration curve of SA standards of 2.5, 5, 12, 5, 25, and 50nM. The quantity of free or total (free+bound) SA was calculated by linear interpolation of the relevant area to the calibration curve. Each concentration of sialic acid standard was treated with the same procedure as the sample to minimize the impact of ethanol precipitation or acid hydrolysis.
Statistical analysis
Comparison of two groups was assessed by Student’s two-tailed, unpaired t test, and comparison of more than two groups was assessed using one-way analysis of variance (ANOVA) with post-hoc Tukey’s pairwise comparison. Survival curves were assessed using the log-rank test, and R square values were determined by linear regression. Measures with p < 0.05 were considered significant in all cases. Statistics were analyzed using GraphPad Prism Version 6.03 (GraphPad Software Inc., La Jolla, CA).
RESULTS
Creation of a GNEM mouse model with a humanized sialoglycome
We began by breeding the GNED207VTgGne-/- GNEM mouse model [41] to mice lacking the CMP-Neu5Ac hydroxylase (Cmah) gene [26]. In the resulting double knockout transgenic mice (GNED207VTgGne-/-Cmah-/-), the absence of Cmah gene expression precludes biosynthesis of N-glycolyl-neuraminic acid (Neu5Gc) [28]. Absence of Neu5Gc expression in skeletal muscles was confirmed by staining using a purified antibody with high specificity and sensitivity for Neu5Gc [29] (Fig. 1). Quantification of pixel staining density also showed an overall reduction of 36±6% in GNED207VTgGne-/- tibialis anterior muscle compared to wild type (p < 0.001, n = 6-7 per group).
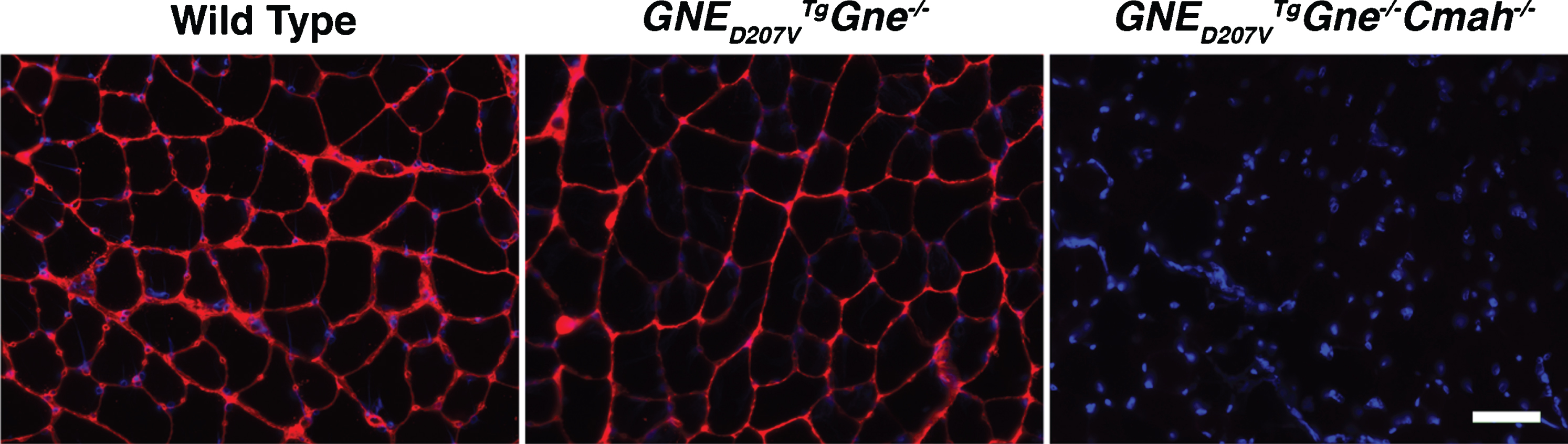
Endogenous Neu5Gc expression is absent in GNED207V Tg Gne-/-Cmah-/- mouse muscle. Tibialis anterior (TA) muscle from wild type (WT), GNED207V Tg Gne-/-, or GNED207V Tg Gne-/-Cmah-/- mice was stained with an antibody specific for the Neu5Gc form of sialic acid (red). All images are time-matched exposures and images shown are merged with DAPI, a nuclear marker (blue). Scale bar = 50μm.
We next assayed various disease phenotypes in GNED207VTgGne-/- mice and GNED207VTgGne-/-Cmah-/- mice compared to normal controls. We found no change in lifespan (Fig. S1A) or in male mouse weight (Fig. S1B) up to 15 months of age. Female GNED207VTgGne-/- and GNED207VTgGne-/-Cmah-/- mice, however, showed lower mouse weights relative to controls at some time points beginning at 9 months (Fig. S1C). With equally mixed genders, there was no difference between groups at 15 months in muscle: body weight ratios for the tibialis anterior (TA), gastrocnemius, quadriceps femoris or triceps brachii (Fig. S1D), but there was a significant increase in heart weight and spleen weight for both GNED207VTgGne-/- and GNED207VTgGne-/-Cmah-/- mice (Fig. S1E). Hindlimb and forelimb grip strength showed no significant difference between the three groups, with two minor exceptions, between 6 and 15 months, and ambulation also showed no significant changes (Fig. S2). At 15 months, we found no evidence of inclusion bodies by Congo Red staining in skeletal muscle sections, suggesting a delayed phenotype onset relative to previous studies [21, 41] (Fig. S3). Here, we used sections from the hippocampus of an APPswePSEN1del9 double transgenic mouse, a model for Alzheimer’s disease [50, 51], as a positive staining control. Hematoxylin and Eosin staining of GNED207VTgGne-/- and GNED207VTgGne-/-Cmah-/- muscle sections also showed no evidence of disease pathology and appeared similar to Gne+/- and Gne+/-Cmah-/- controls (Fig. S3). At 15 months of age, we found no significant decrease in absolute and specific force in the TA muscle of GNED207VTgGne-/- mice relative to littermate control mice. Likewise, there was no significant increase in force drop during 10 eccentric contractions (Fig. S4). Last, we observed serum anti-Neu5Gc antibodies in only 2 of 5 GNED207VTgGne-/-Cmah-/- mice, and these levels were quite low (at or below 2μg/mL, data not shown). Thus, deletion of Cmah in GNED207VTgGne-/- mice did not alter muscle phenotypes at the ages studied and did not induce a significant muscle inflammatory response, unlike mouse models of Cmah-deficient muscular dystrophy [38, 39].
Visualization of Neu5Gc in GNED207VTgGne-/-Cmah-/- mice after prolonged feeding of Neu5Gc-enriched glycoprotein or after single oral gavage with high-dose Neu5Gc monosaccharide
We next used GNED207VTgGne-/-Cmah-/- mice to assess Neu5Gc uptake from diet. Normal mouse chow contains modest amounts of Neu5Gc, but likely does not reflect the full capacity of human diet to contain Neu5Gc protein sources. To address this, we fed GNED207VTgGne-/-Cmah-/-mice a specialized mouse chow containing porcine submaxillary mucin (PSM) to increase dietary Neu5Gc. Based on a 4.5 g diet per day for an adult 25 g mouse, normal mouse chow provides 1.1 mg/kg/day of dietary Neu5Gc, while PSM chow provides 45 mg/kg/day, or over 40 times as much Neu5Gc, a diet analogous to a human diet high in red meat [52]. GNED207VTgGne-/-Cmah-/- mice were fed regular mouse chow or PSM-enriched mouse chow until 10 months of age (Figs. 2 and 3). Neu5Gc expression was then analyzed by immunostaining.
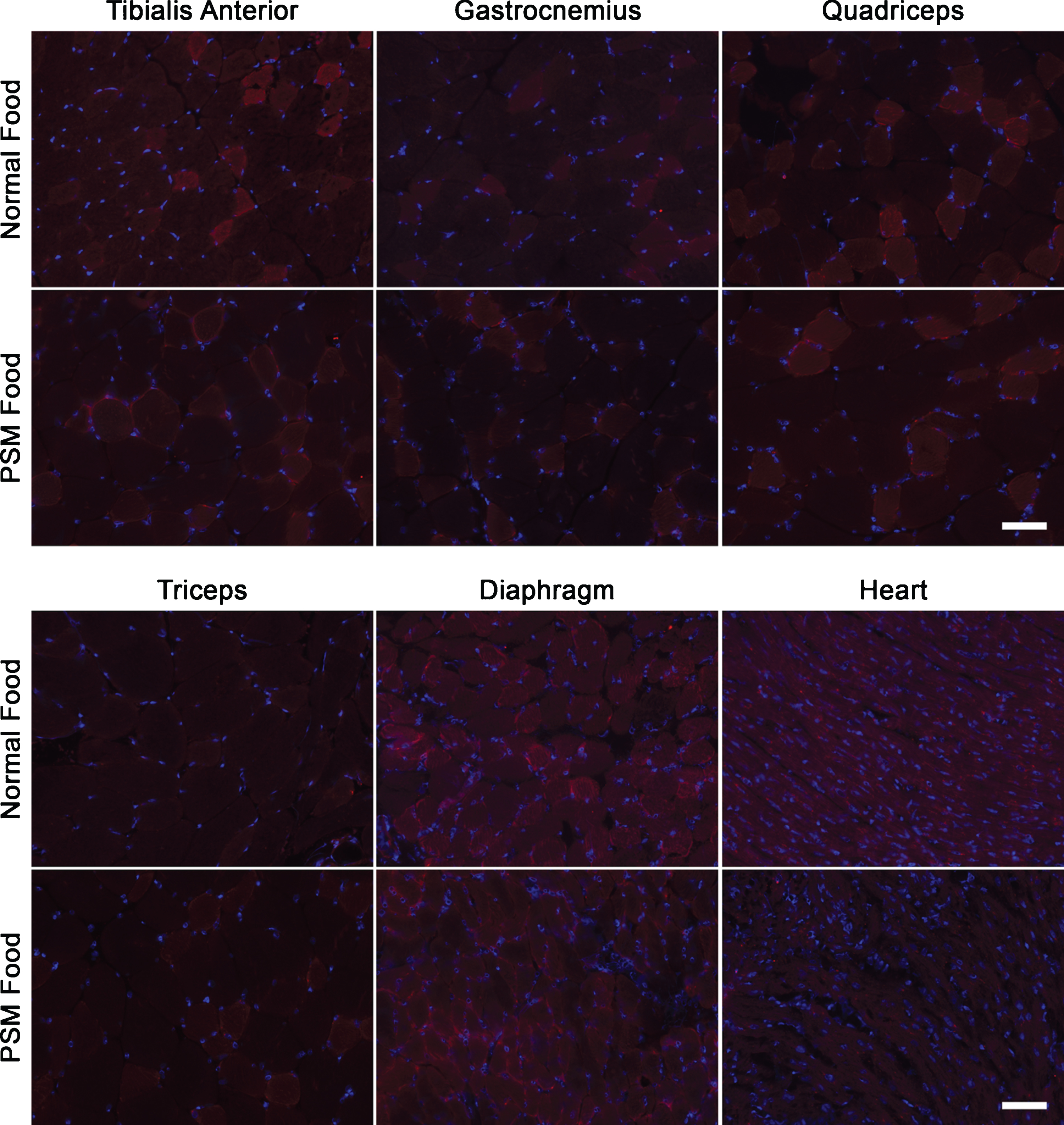
Neu5Gc is not appreciably expressed in GNED207V Tg Gne-/-Cmah-/- muscle after feeding with normal or Neu5Gc-enriched mouse chow. GNED207V Tg Gne-/-Cmah-/- mice were fed normal mouse chow or Neu5Gc-enriched mouse food formulated with porcine submaxillary mucin (PSM) from conception onward until 10 months of age. Muscles sections from tibialis anterior, gastrocnemius, quadriceps femoris, triceps brachii, diaphragm, and heart were stained with anti-Neu5Gc antibody (red). Exposures are time-matched within muscle types and relative to Figures 3, 4 and 5. Images shown are merged with DAPI, a nuclear marker (blue). Scale bar is 50μm for each panel.
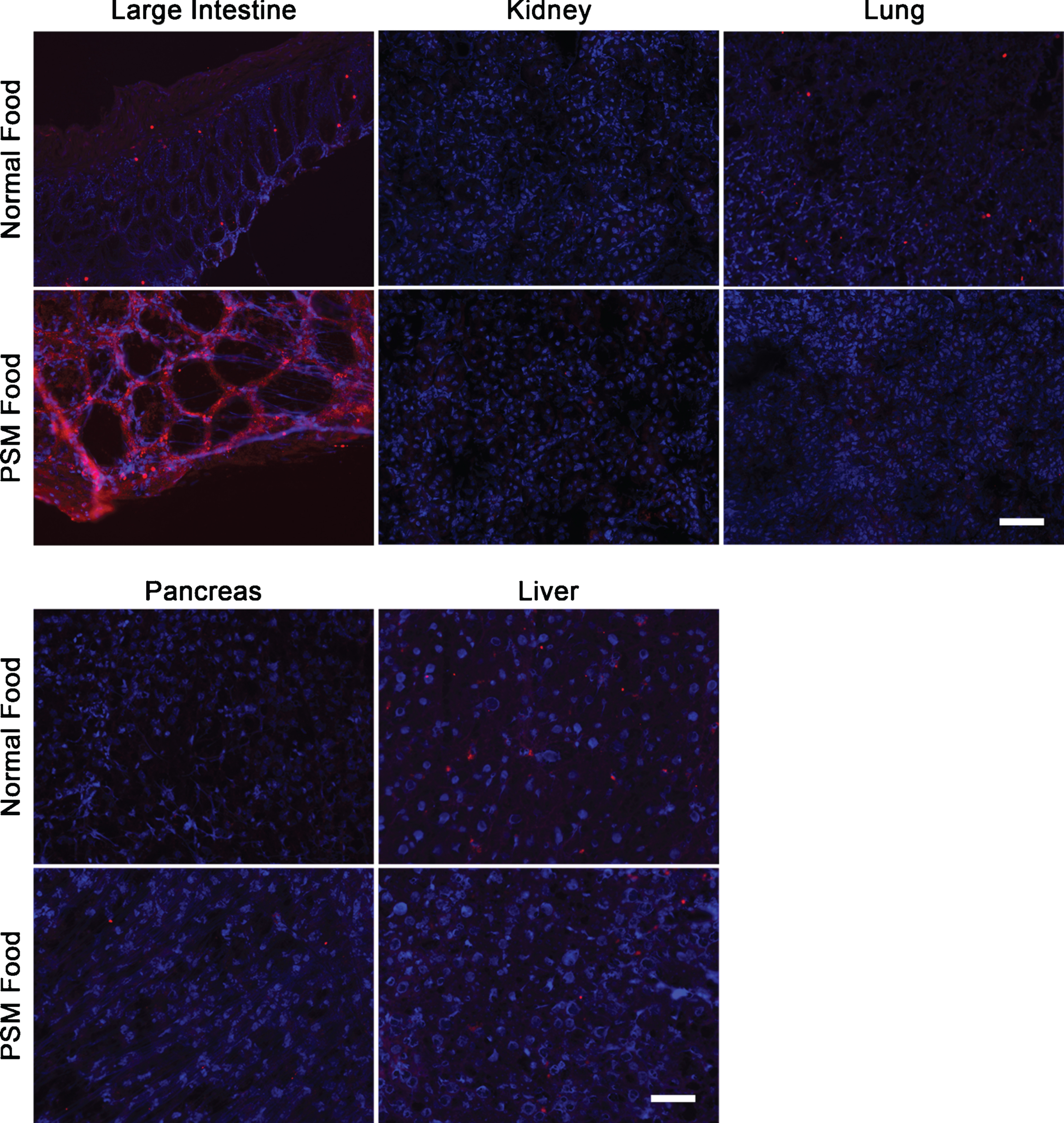
Neu5Gc is not appreciably expressed in GNED207V Tg Gne-/-Cmah-/- non-muscle organs, save large intestine, after feeding with Neu5Gc-enriched mouse chow. GNED207V Tg Gne-/-Cmah-/- mice were fed normal mouse food or Neu5Gc-enriched mouse food formulated with porcine submaxillary mucin (PSM) from conception onward until 10 months of age. Tissues sections from large intestine, kidney, lung, pancreas, and liver were stained with anti-Neu5Gc antibody (red). Exposures are time-matched within muscle types and relative to Figs. 2, 4, and 5. Images shown are merged with DAPI, a nuclear marker (blue). Scale bar is 50μm for each panel.
None of the skeletal muscles studied (TA, gastrocnemius, quadriceps femoris, triceps brachii, or diaphragm) or heart showed any Neu5Gc staining even after 10 months of feeding with normal or PSM (Neu5Gc-rich) chow (Fig. 2). Wild type mouse muscle sections, by contrast, were positive for Neu5Gc staining under the same conditions (e.g., Fig. 1). There was also no Neu5Gc staining observed in kidney, lung, pancreas or liver, but there was significant staining in the large intestine when mice were fed PSM chow (Fig. 3). This staining was consistent with previous studies showing intestine can take up dietary Neu5Gc [53] and is consistent with the higher Neu5Gc content in PSM chow. These data suggest that dietary Neu5Gc is unlikely to provide significant amounts of SA to skeletal muscles when presented as a glycoprotein source, even when Neu5Gc is enriched relative to amounts found in a normal mouse diet.
To ensure that we had a positive control for dietary SA uptake, we next delivered a single 2 g/kg dose of Neu5Gc to adult GNED207VTgGne-/-Cmah-/- mice by oral gavage. This very high dose of monosaccharide is similar to the highest doses used in previous pre-clinical studies [21]. Control mice were given a 2 g/kg dose of Neu5Ac. After 7 days, mice were euthanized and tissues were studied by anti-Neu5Gc immunostaining, with images time-matched to those taken for the dietary glycoprotein experiment (Figs. 2 and 3). Here, we now observed increased Neu5Gc expression in skeletal muscles including the TA, gastrocnemius, quadriceps femoris, triceps brachii and diaphragm (Fig. 4). Some sarcolemmal membrane staining was evident in all skeletal muscles studied, with diaphragm showing the highest level of staining. All limb muscles, however, showed lower and more intermittent staining than was found in diaphragm, and some intracellular accumulations of Neu5Gc staining were evident, particularly in the quadriceps (Fig. 4). Heart showed a predominantly intracellular staining pattern within cardiomyocytes, with little or no sarcolemmal membrane staining (Fig. 4). Neu5Gc staining of non-muscle organs showed differences as well, with stomach and kidney having high staining in a subset of cells, lung having intermediate (and more uniform) staining, and pancreas and liver having low to absent staining (Fig. 5). In kidney, heterogeneity of cell staining in the tubules was very striking.
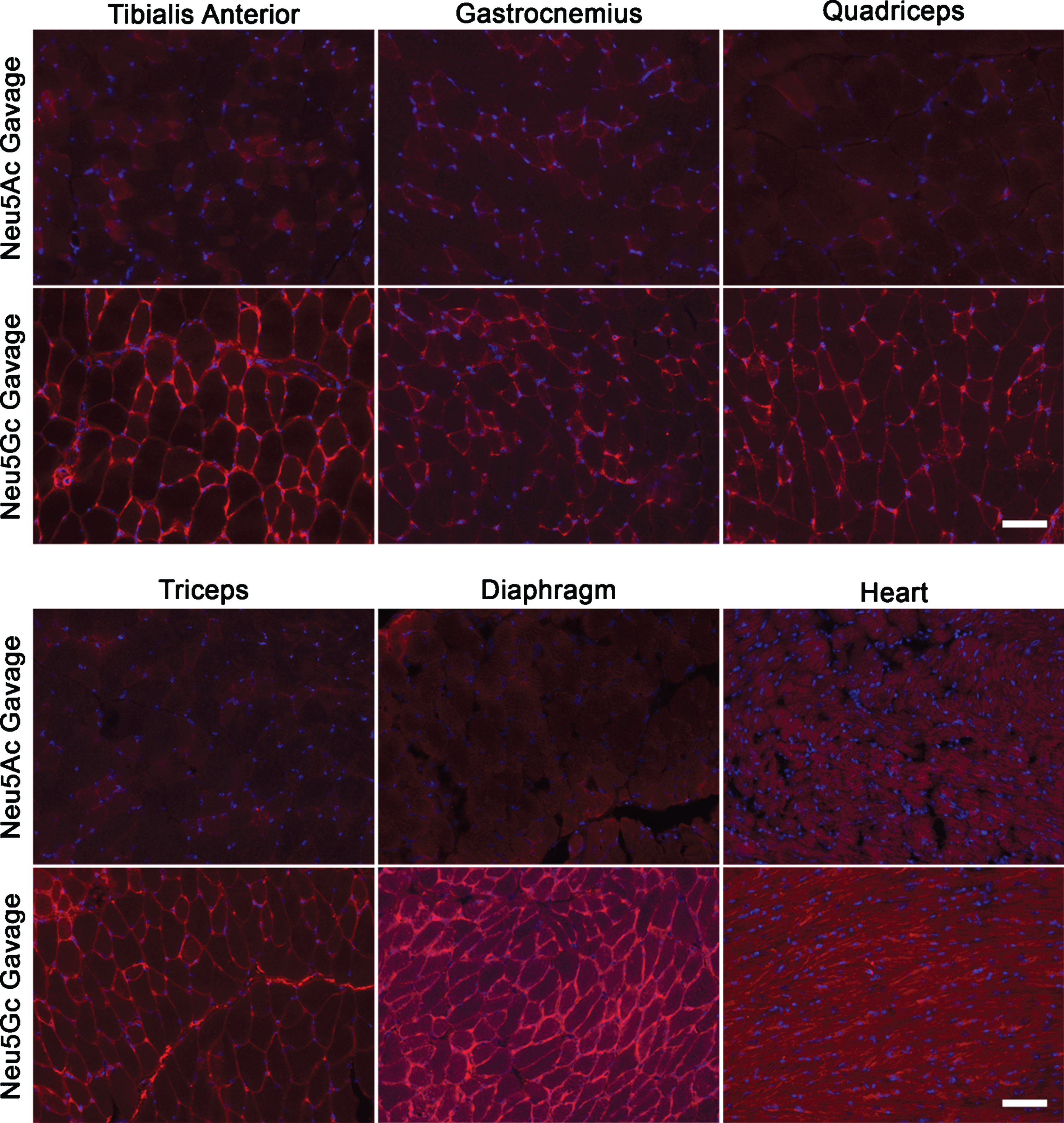
Neu5Gc is expressed in GNED207V Tg Gne-/-Cmah-/- muscle after oral gavage with Neu5Gc monosaccharide. 2-month-old GNED207V Tg Gne-/-Cmah-/- mice were given 2 g/kg Neu5Ac or 2 g/kg Neu5Gc by oral gavage. Muscle sections from the tibialis anterior, gastrocnemius, quadriceps femoris, triceps brachii, diaphragm, and heart were stained with an anti-Neu5Gc antibody (red) 7 days after feeding. Exposures are time-matched within muscle types, and images shown are merged with DAPI, a nuclear marker (blue). Scale bar is 50μm.
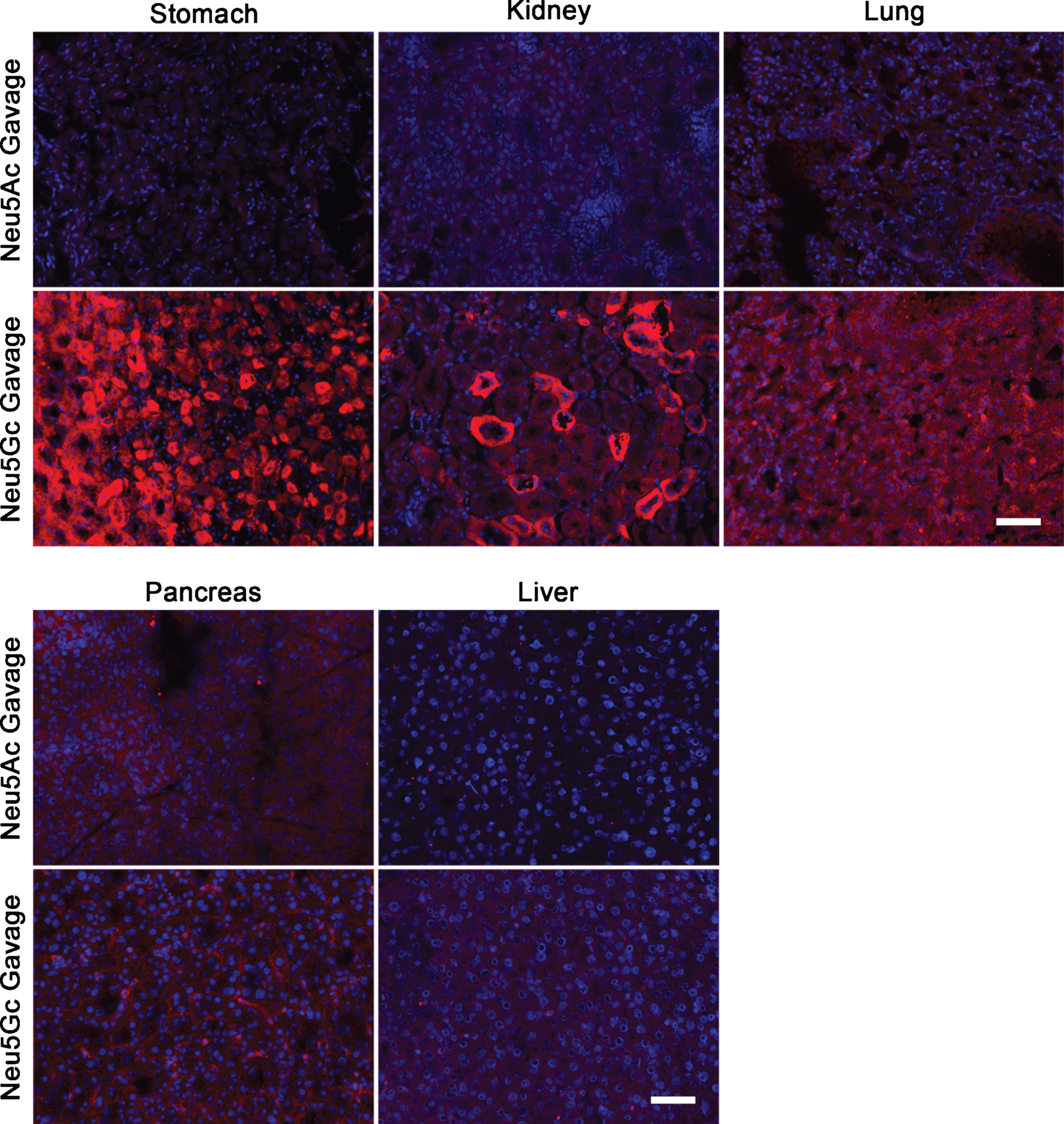
Neu5Gc is expressed in GNED207V Tg Gne-/-Cmah-/- non-muscle tissues after oral gavage with Neu5Gc monosaccharide. 2-month-old GNED207V Tg Gne-/-Cmah-/- mice were given 2 g/kg Neu5Ac or 2 g/kg Neu5Gc by oral gavage. Tissue sections from stomach, kidney, lung, pancreas, and liver were stained with anti-Neu5Gc antibody (red) 7 days after feeding. Exposures are time-matched and images shown are merged with DAPI, a nuclear marker (blue). Scale bar is 50μm.
A quantification of anti-Neu5Gc immunostaining in Neu5Gc-fed GNED207VTgGne-/-Cmah-/- skeletal muscles compared to Neu5Ac-fed GNED207VTgGne-/-Cmah-/- skeletal muscles showed that the cumulative increase in staining was only 3–14% above background in limb muscles with Neu5Gc feeding, but that staining doubled in the diaphragm (Fig. 6). Staining also doubled in kidney and lung and was increased more than 4-fold in stomach, but there was almost no increase in liver or pancreas (Fig. 6). The small level of increased Neu5Gc staining in Neu5Gc-fed GNED207VTgGne-/-Cmah-/- limb muscles never exceeded 14% of the Neu5Gc staining level observed in wild type muscle, suggesting that Neu5Gc muscle incorporation, even when given at a very high oral dose, was not robust. No gender-specific differences were seen in any staining measure.
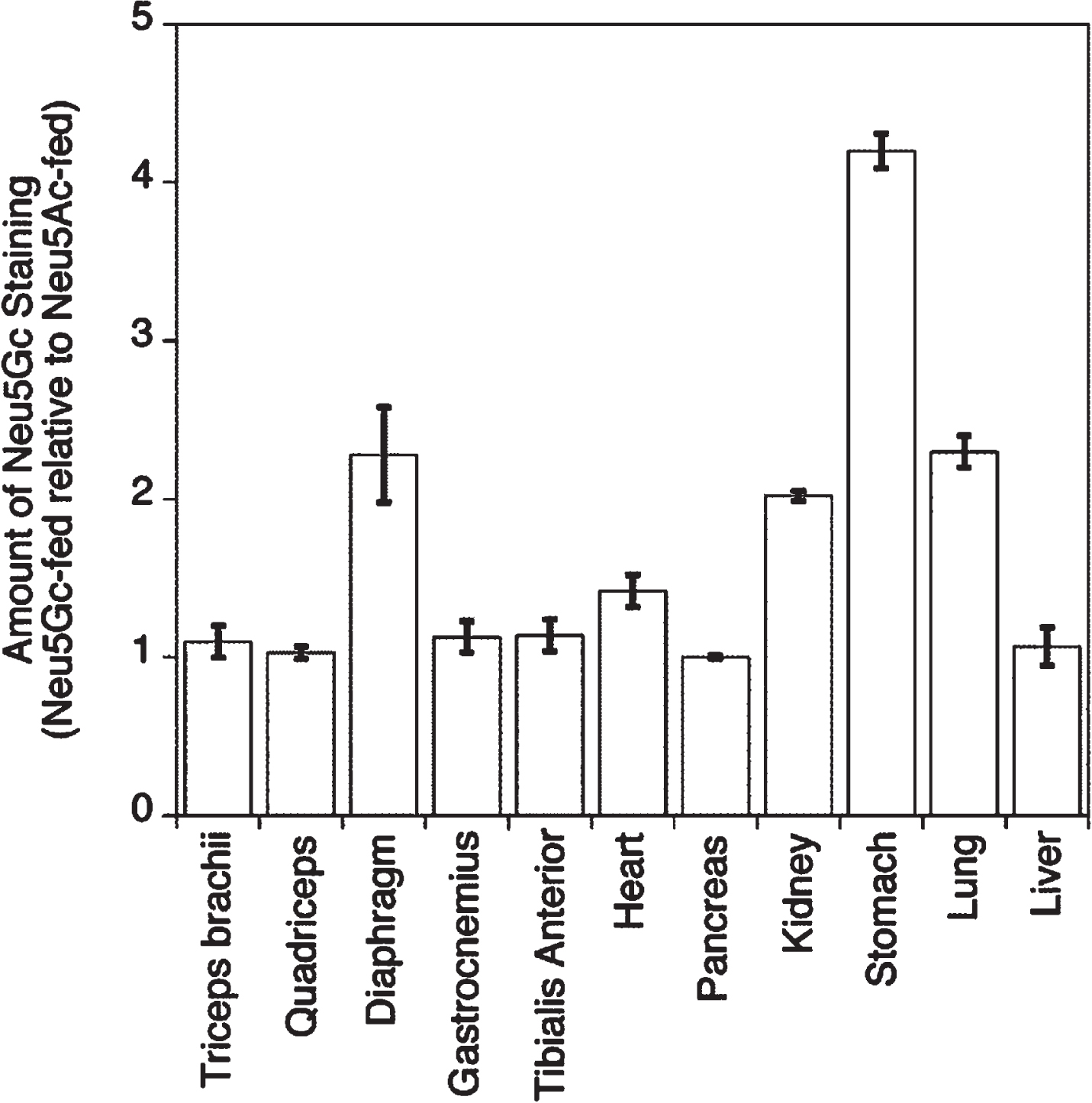
Quantification of Neu5Gc staining after oral gavage with Neu5Gc monosaccharide. Relative anti-Neu5Gc tissue staining intensity was compared between Neu5Gc-fed and Neu5Ac-fed GNED207V Tg Gne-/-Cmah-/- mice for experiments shown in Figs. 4 and 5. Staining intensity for Neu5Ac-fed muscles is set at 1 for each tissue. Errors are SD for n = 2–4/grp.
GNE expression in liver increases muscle SA expression
Because Neu5Gc staining resulting from oral SA therapy was very low in the liver compared to staining in the stomach and kidney (Figs. 5 and 6), and because SA in serum is reduced in some GNEM patients compared to controls [54] (suggesting a contribution of liver to SA serum deficiency), we examined the contribution of liver to muscle sialylation further using a gene therapy approach. We developed recombinant (r) Adeno Associated Virus (AAV) vectors expressing the wild type human GNE gene with FLAG and myc epitope tags at the C-terminus. These rAAVs were made using the rhesus 74 serotype (rh74) rAAV capsid protein, and the GNE transgene was driven by either a liver-specific promoter (LSP) [55] or a muscle-specific promoter (MCK) [56]. We then injected these vectors directly into the TA or the gastrocnemius muscle using intramuscular injection (IM) or using intraperitoneal injection (IP, to allow for high delivery to the liver). Because GNE is required for biosynthesis of both Neu5Ac and Neu5Gc, we chose to include GNED207VTgGne-/- mice for these experiments, as these mice express a functional Cmah gene that would allow us to perform Neu5Gc immunostaining to assess SA levels. No gender-specific differences were observed for any of the resulting measures shown below.
We measured rAAV transduction in muscles and liver by qPCR (Fig. S5) and measured GNE gene overexpression by qRT-PCR (Fig. 7). Transduction of liver with vector genomes (vgs) of rAAV approached 1vg per nucleus after IP injection of either rAAVrh74.MCK.GNE or rAAVrh74.LSP.GNE, both of which were injected at 5×1011vg, while transduction of muscle was > 1vg per nucleus after IM injection of either vector (Fig. S5). Only IP injection of rAAVrh74.LSP.GNE led to increased GNE gene expression in liver, showing an over 8-fold increase relative to control, while only IM injections of rAAVrh74.MCK.GNE led to increased GNE gene expression in muscle, with a 17-fold increase in the TA and a 35-fold increase in the gastrocnemius. We also tested the LSP promoter by IM injection in skeletal muscle and the MCK promoter by IP injection in liver, neither of which resulted in increased GNE gene expression. Interestingly, none of the injection schemes with either vector reduced the enlargement in heart and spleen seen in untreated GNED207V Tg Gne-/- mice (data not shown).
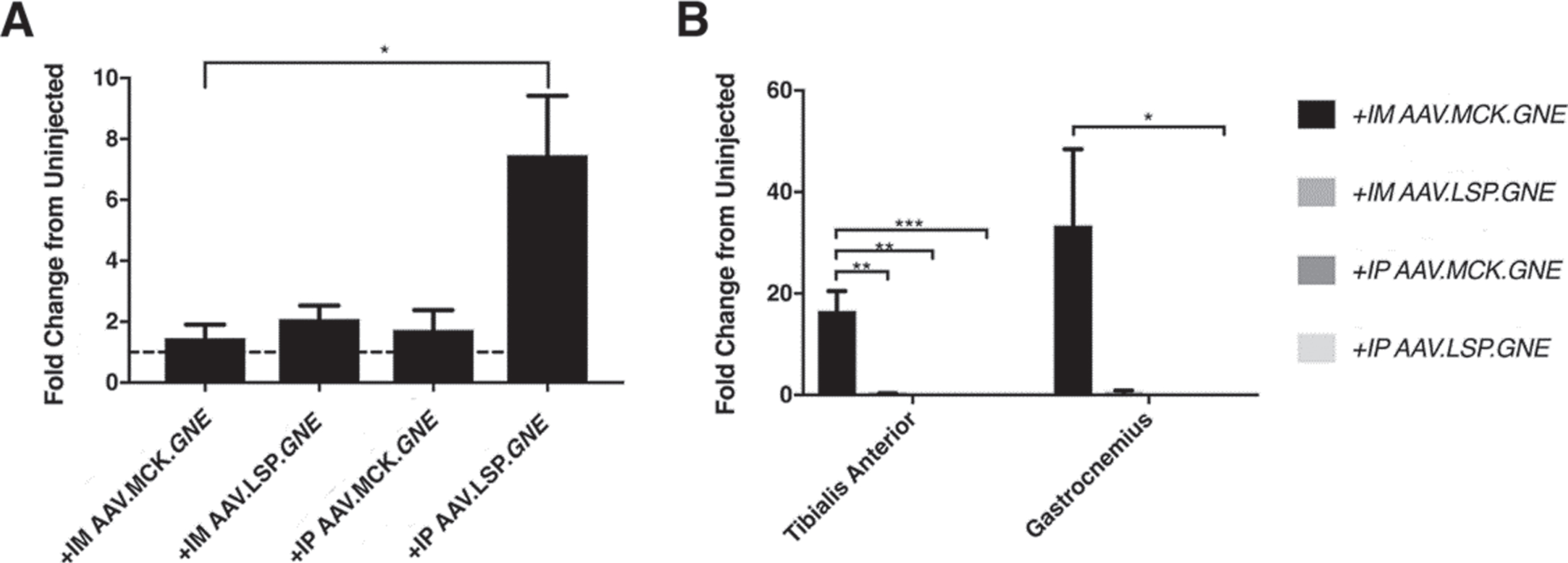
GNE gene expression after AAV.GNE gene therapy using tissue-specific promoters. qRT-PCR analysis of GNE transgene expression in liver (A) or skeletal muscle (B) resulting from rAAV.GNE gene therapy in GNED207V Tg Gne-/-Cmah-/- mice. Fold-change in GNE transgene expression is reported relative to endogenous GNED207V expression in uninjected GNED207V Tg Gne-/- liver (A) or relative to endogenous Gne expression in uninjected wild type skeletal muscle (B). Mice were either injected intramuscularly (IM) with 1×1011 vg/TA and 5×1011 vg/gastrocnemius of rAAVrh74.MCK.GNE or rAAVrh74.LSP.GNE or were injected intraperitoneally (IP) with 5×1011 vg rAAVrh74.MCK.GNE or rAAVrh74.LSP.GNE at 2 months of age. Gene expression was analyzed in liver and skeletal muscle (gastrocnemius) at 10 months post-injection. Dashed line in (A) represents baseline (no change) endogenous GNED207 V expression in GNED207V Tg Gne-/- mouse liver. Data are mean±SEM for 3–5 mice per group. *p < 0.05, **p < 0.01, ***p < 0.001.
Use of the LSP and MCK promoters led to liver- and muscle-specific GNE protein expression, respectively. Comparison of staining for the FLAG epitope tag on the GNE protein after different injection paradigms confirmed that FLAG staining was increased in liver only after IP injection of rAAVrh74.LSP.GNE and in muscle only after IM injection of rAAVrh74.MCK.GNE (Fig. 8A). Western blotting of whole muscle lysates made by extraction in non-denaturing (1% NP40) or in denaturing buffers (2% SDS or 2% SDS with 4M urea) also confirmed appropriate GNE protein expression (Fig. 8B). No GNE protein was detected in liver after IM injection of rAAVrh74.MCK.GNE and no protein was detected in muscle after IP injection of rAAVrh74.LSP.GNE (not shown). Staining of liver sections with the SA-binding lectin Sambucus nigra agglutinin (SNA) also indicated an increase in SA after IP injection of rAAVrh74.LSP.GNE (Fig. S6). Similarly, Peanut agglutinin (PNA) staining, which would stain Galβ1,3GalNAc glycans not capped by sialic acid, was decreased in rAAVrh74.LSP.GNE-treated liver tissue.
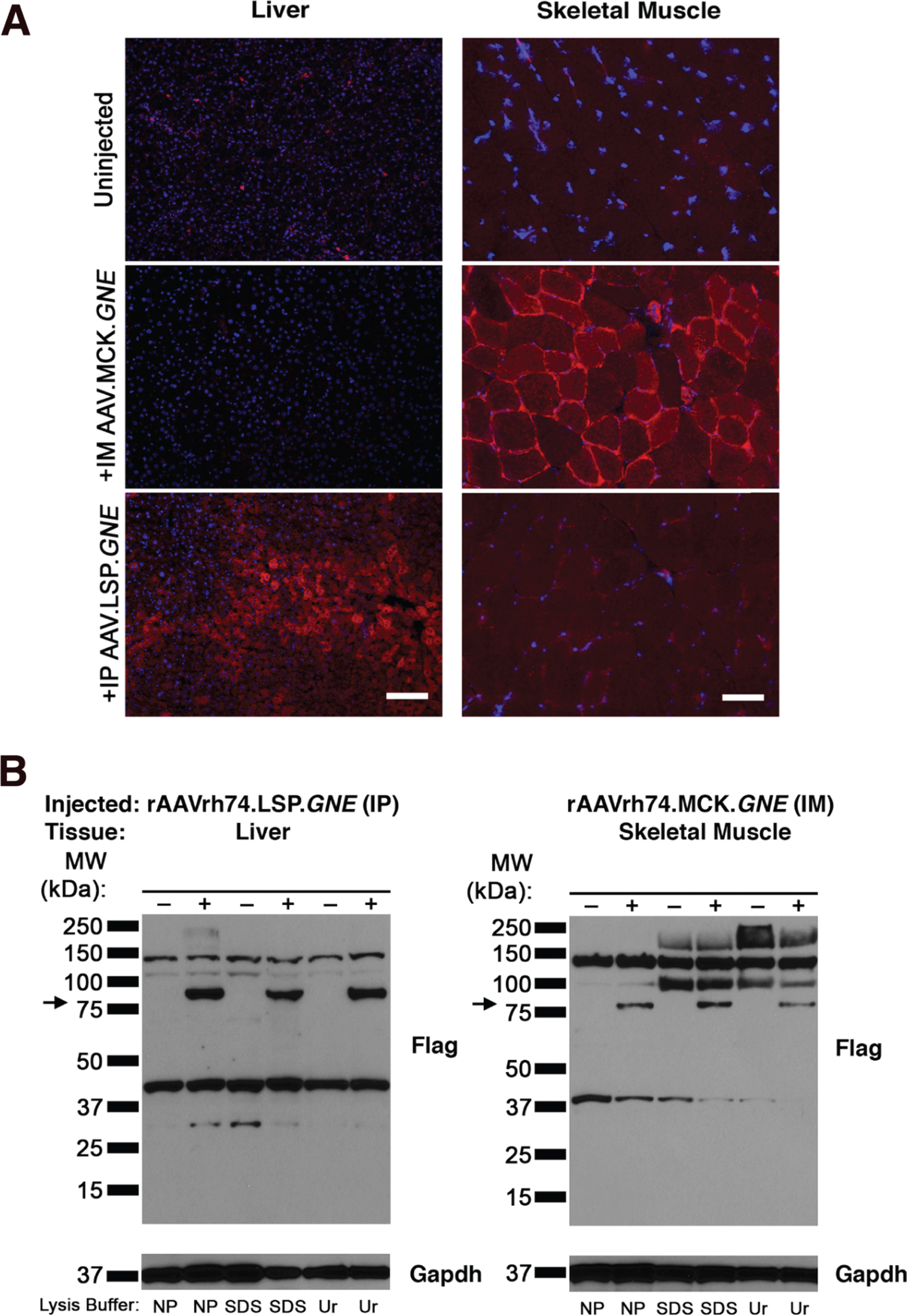
Immunostaining and immunoblotting of FLAG-tagged GNE protein in liver and skeletal muscle of GNED207V Tg Gne-/-Cmah-/- mice after GNE gene overexpression using tissue-specific promoters. (A) Liver and muscle (gastrocnemius) sections from GNED207V Tg Gne-/-Cmah-/- mice were stained with anti-FLAG antibody (red), to identify the epitope tag on GNE protein produced by AAV-mediated GNE gene therapy, and with DAPI (blue), to identify nuclei. Mice were injected intramuscularly with 5×1011 rAAVrh74.MCK.GNE or intraperitoneally with 5×1011 vg of rAAVrh74.LSP.GNE at 2 months of age and analyzed at 10 months post-injection. Scale bar is 100μm for liver and 50μm for skeletal muscle.(B) Liver or muscle tissue from the same experiment was extracted with 1% NP 40 (NP), 2% SDS (SDS), or 2% SDS with 4M urea (Ur), and probed with an antibody to the FLAG epitope tag on AAV-driven transgenic GNE protein. Arrow indicates the molecular weight of monomeric GNE (79kDa). Blots were stripped and probed with antibody to GAPDH (below) to control for protein loading and transfer.
We next assessed SA expression in skeletal muscle using Neu5Gc immunostaining. Surprisingly, both IM injection of rAAVrh74.MCK.GNE and IP injection of rAAVrh74.LSP.GNE showed increased Neu5Gc staining in muscle sarcolemmal membranes, while only IP injection of rAAVrh74.LSP.GNE showed the expected increase in Neu5Gc staining in liver (Fig. 9). Overall muscle Neu5Gc immunostaining for rAAVrh74.LSP.GNE-treated GNED207V Tg Gne-/- mice was 207±43% of untreated GNED207V Tg Gne-/- control, while muscle staining for rAAVrh74.MCK.GNE-treated mice was 165±28% of control (Fig. 10A). To further explore this issue, we stained tissues with Maackia amurensis agglutinin (MAA), a SA-binding lectin (Fig. S7). Here, we did not see the dramatic increases seen with anti-Neu5Gc immunostaining (Fig. 9); rAAVrh74.LSP.GNE-treated muscle staining with MAA was increased only to 118±11% of untreated control (Fig. 10B). To further understand SA changes, we next measured free SA and total (free + bound) SA levels by HPLC (Fig. 10C). Here, rAAVrh74.MCK.GNE and rAAVrh74.LSP.GNE injection showed a 23% and 17% increase, respectively, in total muscle SA compared to untreated GNED207V Tg Gne-/-mice. Only rAAVrh74.MCK.GNE injection, however, showed a sizable increase in free SA (+48%). Thus, while rAAVrh74.LSP.GNE increased overall SA, it did not increase free SA as much as rAAVrh74.MCK.GNE did. In addition, free SA in untreated GNED207V Tg Gne-/- muscle was reduced by only 14% relative to wild type. Regardless, overexpression of GNE using the LSP promoter demonstrated that liver-specific GNE expression could increase SA staining in skeletal muscle over the course of 10 months.
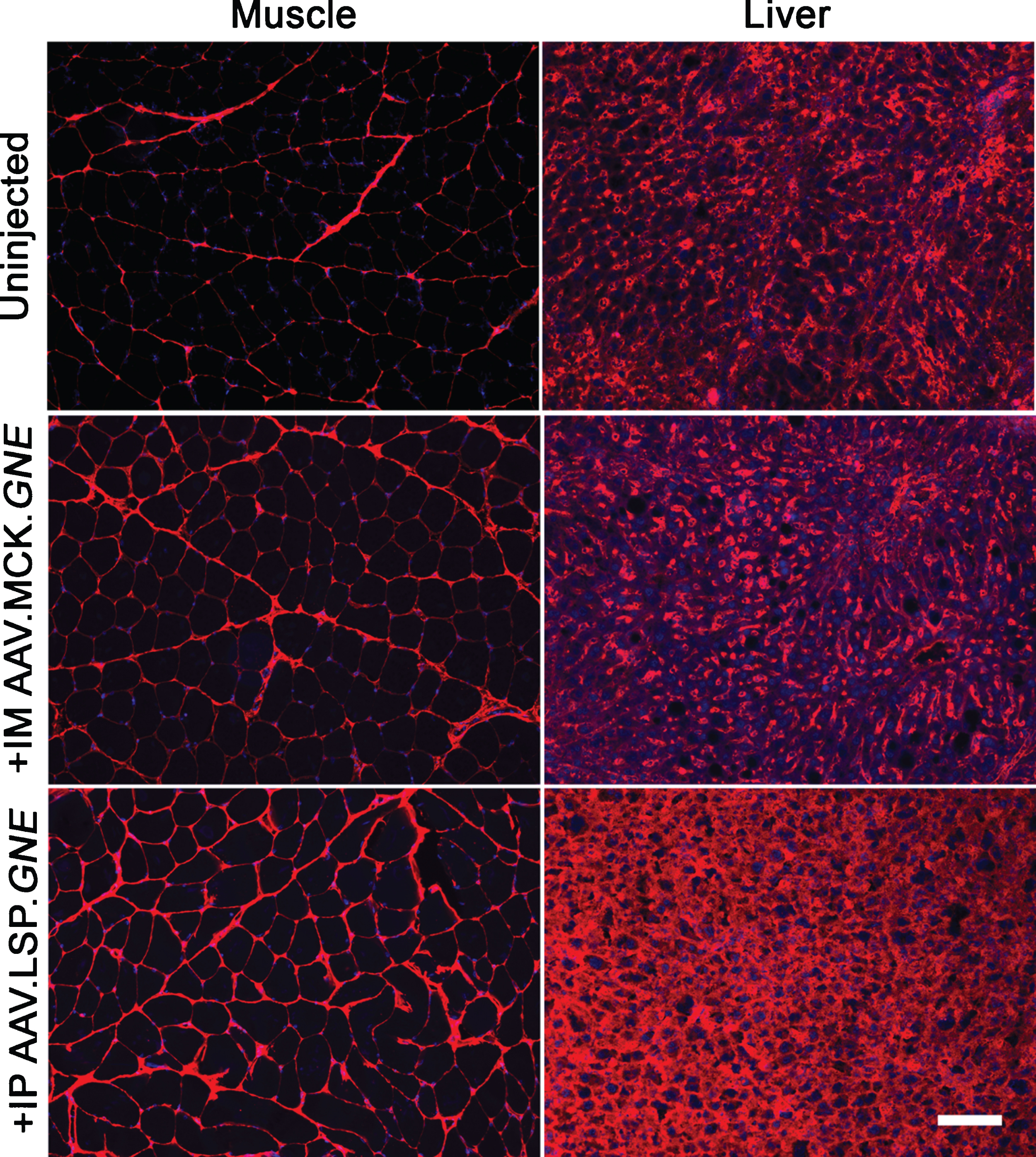
Overexpression of GNE in liver increases Neu5Gc staining in GNED207V Tg Gne-/- skeletal muscle. Gastrocnemius muscle (left) or liver (right) from 2-month-old GNED207V Tg Gne-/- mice were stained with an anti-Neu5Gc antibody at 10 months after intramuscular injection with 5×1011 rAAVrh74.MCK.GNE or intraperitoneal injection with 5×1011 vg of rAAVrh74.LSP.GNE. Exposures are time-matched, and images shown are merged with DAPI, a nuclear marker (blue). Scale bar is 100μm.
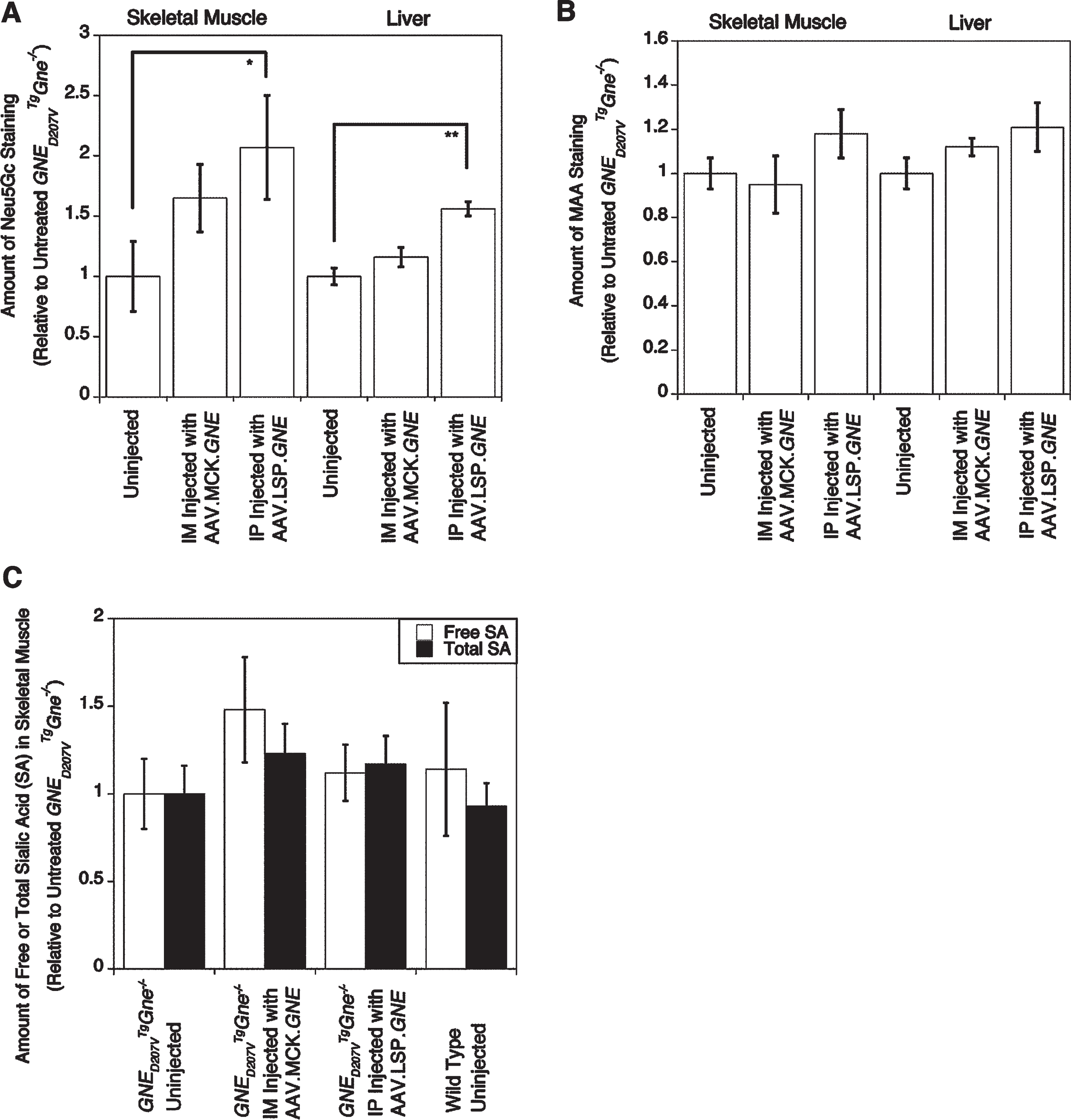
Quantification of muscle Neu5Gc and MAA staining and of sialic acid levels by HPLC. Gastrocnemius muscle or liver from 2-month-old GNED207V Tg Gne-/- mice were stained with an anti-Neu5Gc antibody (A) or MAA lectin (B) at 10 months post-injection, after which pixel density of staining was quantified. (C) The same muscles were also quantified for free and total (free+ bound) sialic acid (SA) by High Performance Liquid Chromatography (HPLC). Errors are SD for n = 2–5/grp. *p < 0.05, **p < 0.01.
DISCUSSION
While there is evidence to suggest that sialic acid (SA) deficits occur in GNEM [54], studies of oral glycan therapies have been hindered by the inability to discern expression of delivered glycan from the endogenous glycan. We addressed this challenge by creating a GNEM disease mouse model with a humanized, N-glycolylneuraminic acid (Neu5Gc)-free, sialoglycome. This has allowed us to visualize orally delivered Neu5Gc using a highly specific and sensitive anti-Neu5Gc antibody [29] in the absence of endogenous Neu5Gc expression.
We present two novel findings in this study. The first is that prolonged feeding with an enriched dietary source of Neu5Gc-containing glycoprotein does not lead to appreciable expression of SA in skeletal muscle. We enriched for Neu5Gc over 40-fold relative to the amount of this glycan normally found in mouse chow, essentially mimicking a human diet high in red meat [52]. Unfortunately, our study suggests that such a diet would not allow for loading of significant SA into muscle over time, as 10 months of this diet led to no detectable muscle Neu5Gc expression. Oral delivery of high-dose Neu5Gc monosaccharide, however, did cause Neu5Gc expression in the sarcolemmal membranes of muscles throughout the body at 7 days after treatment. A single high 2 g/kg dose of Neu5Gc led to sarcolemmal staining in several skeletal muscles, including forelimb muscles and hindlimb muscles, as well as in diaphragm. Thus, oral monosaccharide can deliver SA to muscles, unlike a glycoprotein diet.
We learned several additional things by studying Neu5Gc monosaccharide uptake by anti-Neu5Gc immunostaining. First, Neu5Gc expression after oral Neu5Gc gavage did not appear to be equal among all cells studied, in muscle or in non-muscle organs. Such differences could be due to differential glycan loading into cells within these tissues or could be due to differential vascular access. Unlike skeletal muscles, Neu5Gc was not appreciably present in the sarcolemmal membrane of cardiomyocytes in the heart at 7 days post-dosing. Faster membrane turnover or slower membrane uptake of SA in heart versus skeletal muscle could explain the altered expression pattern. Importantly, overall levels of Neu5Gc staining in limb muscles studied were well below levels found in wild type muscles (no more than 14% of normal). This suggests that oral Neu5Gc, even when given at a very high dose, cannot fully compensate for loss of muscle SA, at least when given as a single dose. While SA levels may be improved by increasing the frequency of oral dosing or by the addition of SA glycan modifications [16, 24], these current results suggest that loading of SA monosaccharide into muscle membranes is not an efficient process.
Our second finding is that liver sialylation can contribute to overall muscle SA expression. In GNED207V Tg Gne-/- mice, use of a liver-specific promoter to drive GNE gene expression actually led to higher levels of Neu5Gc staining in skeletal muscle than did use of a muscle-specific promoter when assayed 10 months after injection. While this result seems counterintuitive, the liver is responsible for the expression of many serum glycoproteins. Such proteins may bind into the muscle extracellular matrix or membrane over time, thereby altering muscle SA. In the original studies on GNED207V Tg Gne-/- mice, there were greater reductions in liver, spleen and serum sialylation than there were in skeletal muscle [21], which may, in turn, have contributed to some of the loss of muscle SA as well. As such, use of a promoter that allows for liver GNE transgene expression could potentially benefit muscles in GNEM patients. Importantly, while liver expression of GNE increased muscle Neu5Gc staining, it did not increase free SA levels in muscle as much as muscle-specific GNE expression did. Thus, liver GNE expression likely would not correct for all deficits in muscle SA metabolism in GNEM.
Our studies present two caveats that suggest potential problems with current methods of analysis for GNEM studies. The first is that SA changes were most easily seen with anti-Neu5Gc immunostaining, while changes were less obvious with MAA staining or HPLC analysis. The anti-Neu5Gc antibody, raised in chickens and purified to increase specificity, is a highly sensitive and specific antibody reagent that recognizes single oxygen differences in SA [29]. This antibody reagent likely derives its specificity, at least in part, from its ability to recognize highly packed oxygens from Neu5Gc within the glycocalyx. As such, anti-Neu5Gc immunostaining may be more sensitive to SA concentration differences than plant lectins like MAA. An alternative explanation would be that Cmah expression in muscle is altered by GNE gene therapy, which could also alter Neu5Gc expression. The trend toward increased SA with MAA staining and HPLC analysis, neither of which specifically measure Neu5Gc, however, suggests that this is not the case.
The second caveat is that the GNED207V Tg Gne-/- mouse model used to study this disease has lost some disease characteristics, at least at the developmental time points used here, relative to those described in previous studies [21, 41]. Further, such disease phenotypes were unaltered in GNED207V Tg Gne-/-Cmah-/- mice compared to GNED207V Tg Gne-/- mice. Having a transgenic disease model may lead to genetic drift over many generations due to changes in transgene copy number or to alterations in genetic background. Previous studies suggest that deletion of Cmah worsens disease phenotypes in mouse models of muscular dystrophy such as the mdx mouse by increasing muscle inflammation and muscle auto-immunity to Neu5Gc [39]. As GNEM is a myopathy without a significant muscle inflammatory component, it may be that auto-immunity to Neu5Gc has no modulatory role in this disease. Indeed, we found little to no induction of anti-Neu5Gc antibodies in GNED207V Tg Gne-/-Cmah-/- mice. Such a lack of contribution to muscle phenotypes by Cmah deletion, however, has allowed us to investigate the role of Neu5Gc localization and expression in the absence of changed muscle disease.
Footnotes
ACKNOWLEDGMENTS
This work was supported by NIH grants R21 AR067366, R01 AR049722 and R01 AR060949 to PTM. KEC was supported by the Jeffry J. Seilhamer Foundation Graduate Student Fellowship from Ohio State University. The authors would like to thank Anna Ashbrook for technical support and Parastoo Azadi and En Tzu Lu (both from the Complex Carbohydrate Research Center at the University of Georgia) for HPLC analysis of sialic acids.
CONFLICT OF INTEREST
PTM receives licensing fees from Sarepta Therapeutics for rAAVrh74.MCK.GALGT2. PTM is also President and CSO of Genosera Inc.
