Abstract
Andersen-Tawil syndrome (ATS) is a rare autosomal dominant neuromuscular disorder due to mutations in the KCNJ2 gene. The classical phenotype of ATS consists of a triad of periodic paralysis, cardiac conduction abnormalities and dysmorphic features. Episodes of either muscle weakness or cardiac arrhythmia may predominate however, and dysmorphic features may be subtle, masking the true breadth of the clinical presentation, and posing a diagnostic challenge. The severity of cardiac involvement varies but includes reports of life-threatening events or sudden cardiac death, usually attributed to ventricular tachyarrhythmias. We report the first case of advanced atrioventricular (AV) block in ATS and highlight clinical factors that may delay diagnosis.
ABBREVIATIONS
Andersen-Tawil syndrome
INTRODUCTION
Andersen-Tawil syndrome (ATS) is a rare autosomal dominant neuromuscular disorder due to mutations in the KCNJ2 gene that codes for the inwardly rectifying potassium channel Kir2.1 [1]. This channel is expressed in both cardiac and skeletal muscle as well as some bony structures. The characteristic phenotype of ATS is a triad of periodic muscle paralysis, dysmorphic features and cardiac rhythm abnormalities related to QT prolongation [1]. Cardiac or neuromuscular symptoms may dominate the clinical picture in any individual however dysmorphic features may be subtle, making a unifying diagnosis challenging. Cardiac involvement varies, ranging from asymptomatic individuals with an incidentally noted abnormal electrocardiogram to reports of sudden cardiac death, usually attributed to ventricular tachyarrhythmias [2]. Advanced atrioventricular (AV) block has not been previously reported in ATS.
CASE REPORT
A 57 year old gentleman was referred to the UK National referral centre for neuromuscular channelopathies with symptoms of episodic weakness. Early history and development were unremarkable. In his teens he began to complain of episodes of “mild” weakness predominantly affecting the legs e.g. walking home from school was difficult but he was still able to mobilise. These episodes persisted throughout life but were not investigated. In his early 50’s he complained of an increase in these symptoms but also new episodic breathlessness, fatigue and pre-syncope. Witnesses described him as looking “ashen” during these.
During a severe event he was admitted to hospital. Complete AV block was diagnosed (Fig. 1A), and a pacemaker was implanted. Coronary angiography demonstrated no significant coronary artery disease. His symptoms of dizziness ceased but one year later he represented complaining of increasing intermittent limb weakness with fatigue, occurring every few weeks. Episodes were often provoked by exercise or cold weather and could last from hours to several days.
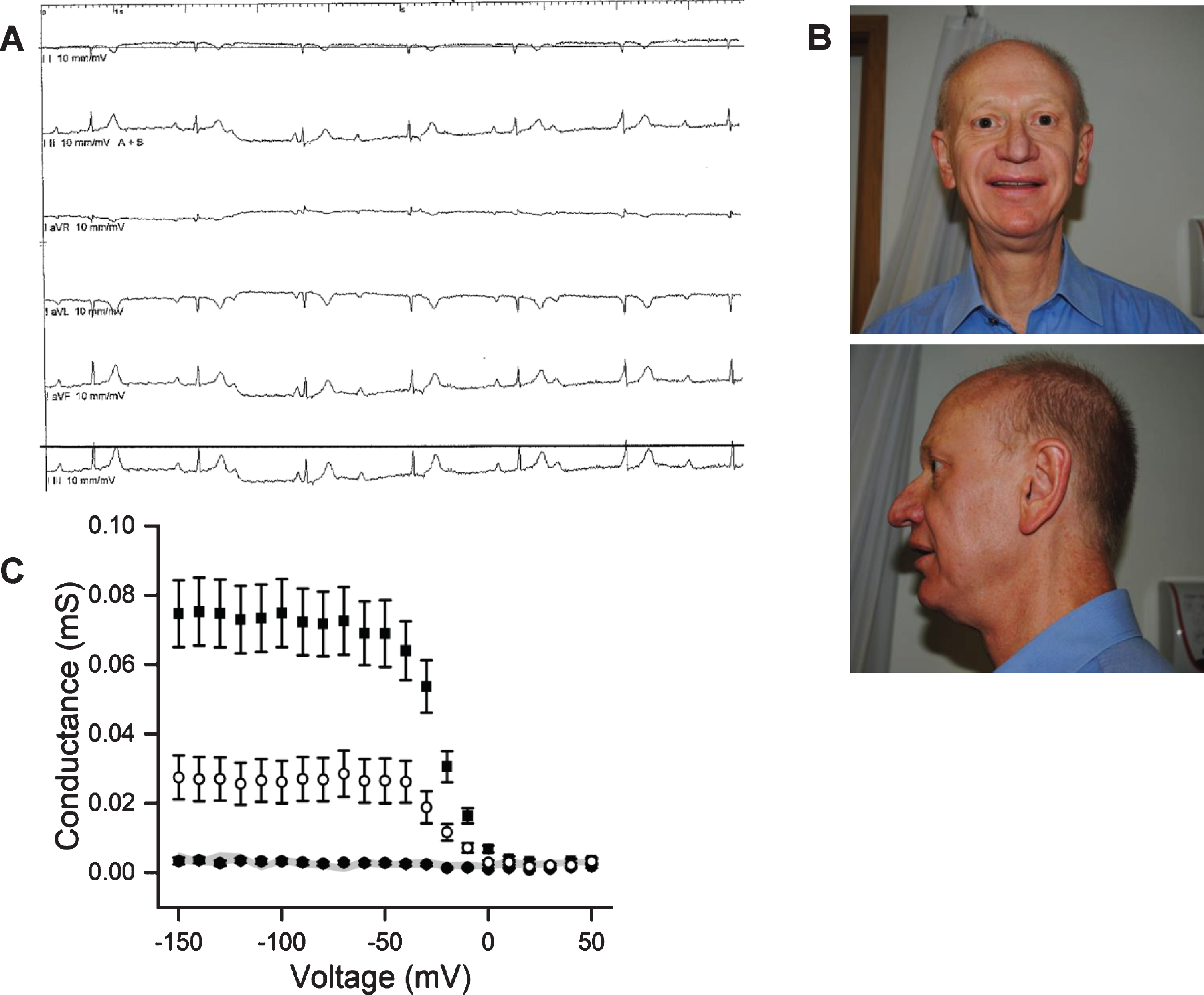
A: ECG showing complete heart block pre-pacemaker insertion. B: Facial characteristics of our patient demonstrating micrognathia. C: Oocytes injected with WT Kir2.1 RNA (squares, n = 24), R260H Kir2.1 RNA (solid circles, n = 20) or 1:1 mixture of these (open circles,
His pacemaker was interrogated and showed no arrhythmias. An echocardiogram confirmed a structurally normal heart. He completed a treadmill test after nine minutes, complaining of fatigue and dizziness with normal chronotropic response being documented. A tilt-table test showed a normal blood pressure response.
Following the exclusion of a cardiac cause of his symptoms he was investigated extensively at his local neurology centre with normal brain imaging, MR intracranial angiogram and EEG.
When referred to our centre micrognathia was noted (Fig. 1B) but the remainder of the examination was normal. Given the description of episodic muscle weakness, the micrognathia and history of cardiac disease he was investigated for the possibility of ATS. An EMG with long-exercise test (LET) was within normal limits. Genetic testing identified a novel heterozygous KCNJ2 variant c.779G>A p. Arg260His.
Two electrode voltage clamp recordings made from xenopus oocytes transfected with human KCNJ2 Arg260His mRNA demonstrated typical loss of function changes seen in ATS variants (Fig. 1C). No current was recorded from cells expressing Arg260His alone but when co-expressed with wild-type mRNA reduced currents were detected indicating a dominant negative effect of the Arg260His variant. Another substitution of the same amino acid, Arg260Pro, has been previously reported in association with ATS and functional studies confirmed its pathogenicity [3].
DISCUSSION
In a study in 96 patients with ATS, conduction disturbances were found in 23% of cases, including 1st degree atrioventricular block, right bundle branch block, left bundle branch block and non-specific intraventricular conduction delay [4]. Cardiomyocytes expressing a dominant-negative subunit of Kir2.1 exhibited a 95% reduction in the inward rectifying potassium current, resulting in significant action potential prolongation. Mouse models of Andersen-Tawil syndrome exhibited a slower heart rate and significant slowing of conduction [5, 6]. Therefore it is likely that the complete AV block in our case is attributable to the
Our report illustrates factors that can delay the diagnosis of ATS but are important for clinicians to be aware of, namely that dysmorphic features can be minimal; either cardiac or muscle symptoms may dominate the clinical picture and a negative long exercise test does not exclude the diagnosis of ATS. A negative LET has been reported in 18% of genetically confirmed patients in one series [7] and recurrent muscle weakness in only 35% of another series presenting predominantly with cardiac complaints [2]. Complete heart block has not previously been described in ATS meaning it may not be considered in the differential diagnosis of patients presenting with unexplained AV block. Additionally, patients with ATS often receive b-blockers as treatment for QT prolongation and the possible propensity for advanced conduction disease needs to be taken into account in decision making. Sudden cardiac death or aborted cardiac death has been described in ATS patients, mainly driven by ventricular arrhythmias [2]. It is possible however that some cases of unexplained sudden cardiac death may have been due to high degree atrioventricular block rather than tachyarrhythmia. This has important implications for both the diagnosis and cardiac management of ATS patients.
Study funding and disclosures
Part of this work was undertaken at University College London Hospitals/University College London, which received a proportion of funding from the Department of Health’s National Institute for Health Research Biomedical Research Centres funding scheme. Dr Suetterlin is supported by an MRC Clinical Research Training Fellowship (MR/M01827X/1). Dr Matthews is supported by a Wellcome Clinical Research Career Development Fellowship (209583/Z/17/Z). Prof Hanna is supported by the UCLH Biomedical Research Centre, and the National Highly Specialised Service (HSS) Department of Health UK.
Patient consent
The patient signed a written informed consent stating that data can be used for research and education, approved by The Joint National Hospital for Neurology and Institure of Neurology Research ethics Committee.
