Abstract
Objectives:
Generally, neuropathies of peripheral nerves are a frequent condition (prevalence 2–3%) and most frequently due to alcoholism, diabetes, renal insufficiency, malignancy, toxins, or drugs. However, the vast majority of neuropathies has orphan status. This review focuses on the etiology, frequency, diagnosis, and treatment of orphan neuropathies.
Methods:
Literature review
Results:
Rareness of diseases is not uniformly defined but in the US an orphan disease is diagnosed if the prevalence is <1:200000, in Europe if <5:10000. Most acquired and hereditary neuropathies are orphan diseases. Often the causative variant has been reported only in a single patient or family, particularly the ones that are newly detected (e.g.
Conclusions:
Though orphan neuropathies are rare per definition they constitute the majority of neuropathies and should be considered as some of them are easy to identify and potentially treatable, as clarification of the underlying cause may contribute to the knowledge about etiology and pathophysiology of these conditions, and as the true prevalence may become obvious only if all ever diagnosed cases are reported.
ABBREVIATIONS
Autosomal dominant Allogenic hematopoetic stem cell transplantation Acute inflammatory demyelinating polyneuropathy Autosomal recessive Autologous stem cell transplantation Bruns Garland syndrome Chronic ataxic neuropathy with ophthalmoplegia, M-proteins, cold agglutinins and disialosyl antibodies Cerebellar ataxia, neuropathy, vestibular areflexia syndrome Contactin-associated protein-1 Chronic, inflammatory demyelinating polyneuropathy Chemotherapy-induced polyneuropathy Charcot-Marie-Tooth Central nervous system Contactin-1 Distal, acquired, demyelinating, symmetric neuropathy Eosinophilic granulomatosis with polyangitis Epithelioid hemangio-endothelioma Friedreich ataxia Giant axonal neuropathy Guillain-Barre syndrome Glial fibrillary acidic protein High density lipoprotein Hereditary motor neuropathy Hereditary motor and sensory neuropathy Hereditary neuralgic amyotrophy Hereditary neuropathy with liability to pressure palsies Hereditary sensory and autonomic neuropathy Hereditary spastic paraplegia Intermediate filaments Large-fiber neuropathy Lysosomal storage disorders Multifocal acquired demyelinating sensory and motor Myelin associated glycoprotein Mitochondrial disorder Miller-Fisher syndrome Myoclonus epilepsy, myopathy, and sensory ataxia Monoclonal gammopathy of unknown significance Multifocal motor neuropathy Mitochondrial neuropathy, gastrointestinal encephalopathy Maternal transmission Neuropathy, ataxia, retinitis pigmentosa Nerve conduction study Neurofascin-155 Next generation sequencing Non-systemic, vasculitic neuropathy Progressive myoclonus epilepsy Polyneuropathy Peripheral nervous system Polyneuropathy, organomegaly, endocrinopathy, monoclonal protein, skin changes Sensory ataxic neuropathy with dysarthria and ophthalmoparesis Spinocerebellar ataxia Spinocerebellar ataxia with epilepsy Small cell lung carcinoma Small fiber neuropathy Subacute sensory neuropathy Sympathetic skin response Transthyretin-related Vascular endothelial growth factor Whole exome sequencing X-linked
INTRODUCTION
Generally, neuropathies of peripheral nerves are a highly prevalent neurological problem, due to the high prevalence of diabetes, alcoholism, and renal insufficiency [1]. The remaining neuropathies are rare and there are a number of neuropathies, which have been only reported in a few patients or a single patient (orphan neuropathies). Concerning these rare neuropathies, prevalence figures are hardly available and often questionable due to limited coverage of a region or insufficient work-up or cooperation between centres. Generally, rare neuropathies occur among all subtypes, hereditary and acquired (metabolic, immune, neoplastic, infectious, toxic). This review focuses on recent advances concerning the etiology, clinical presentation, diagnosis, treatment (if available), and outcome of rare neuropathies of peripheral nerves.
DEFINITION OF RARENESS
The term “rare” is poorly defined in Medicine. It may implicate that only a “few” cases have been reported worldwide so far or it may implicate that the condition can be hardly found in a particular region but may occur with a much higher frequency in another region. Subjectively, a rare disease may be one that an individual physician has not encountered during his practice yet but this depends strongly on the duration of his experience and the setting he is working in. Quantitatively, rareness of a disease can be defined as a disorder with <10 hits on a broad PubMed search. With regard to prevalence rareness is defined as neuropathy occurring in less than 1:10000 subjects. The definition of an orphan disease is not uniform and varies from country to country [2]. Figures vary from 1:1000 to 1:200000 (e.g. USA) people nationwide [3]. In Europe, orphan diseases are those with a prevalence of <5:10000. Ultra-rare diseases have a prevalence of <1:50000. It is estimated that there are about 6000 (5000–7000) orphan diseases [3]. For the majority of rare neuropathies, figures about the true prevalence and incidence are unavailable. Thus, frequency figures largely rely only on estimations [4].
CLASSIFICATION OF NEUROPATHIES
Neuropathies of peripheral nerves may be classified according to various criteria (Fig. 1). The most important classification is the one according to the etiology, which differentiates between acquired or inherited forms. Among acquired and hereditary neuropathies further subdivision have been proposed (see below). Acquired and hereditary neuropathies are classified according to the course as acute or chronic. Acquired and hereditary neuropathies may be accompanied by involvement of other systems/organs/tissues or not. The latter are further classified according to the degree of co-affection of other organs or tissues as dominant or non-dominant. In non-dominant cases, neuropathy is overshadowed by manifestations of other organs. The course may be chronic progressive or relapsing remitting (Tangier’s disease, porphyrias). Progressive neuropathies may be slowly or rapidly progressive. According to the number of affected nerves, mono-neuropathies and polyneuropathies (PNPs) are distinguished. Among PNPs those with uniform affection of all nerves and those with predominant affection of several single nerves (multiplex neuropathy) are differentiated. PNPs are further classified according to the distribution as symmetric or asymmetric and as proximal or distal. Furthermore, neuropathies of the cranial nerves and the peripheral nerves are delineated. According to the direction of progression neuropathies can be ascending or descending. According to the types of nerve fibers affected they may be classified as motor, sensory, autonomic, or mixed neuropathies. According to the size of affected fibers, large- and small-fiber neuropathies (LFNs, SFNs) are delineated. Upon electrophysiological findings, axonal, demyelinating (with or without conduction block), or mixed neuropathies are differentiated. According to the prevalence, they may be classified as frequent or infrequent (rare). Neuropathies can be painful or painless. Neuropathies can be length-dependent (i.e. distal, the longer the more severe, dying-back), which is the case for most of the neuropathies, or length-independent. Lastly, neuropathies may have an infantile/childhood onset or may have an onset in adolescence/adulthood. Some neuropathies may be accessible to treatment, others may not.
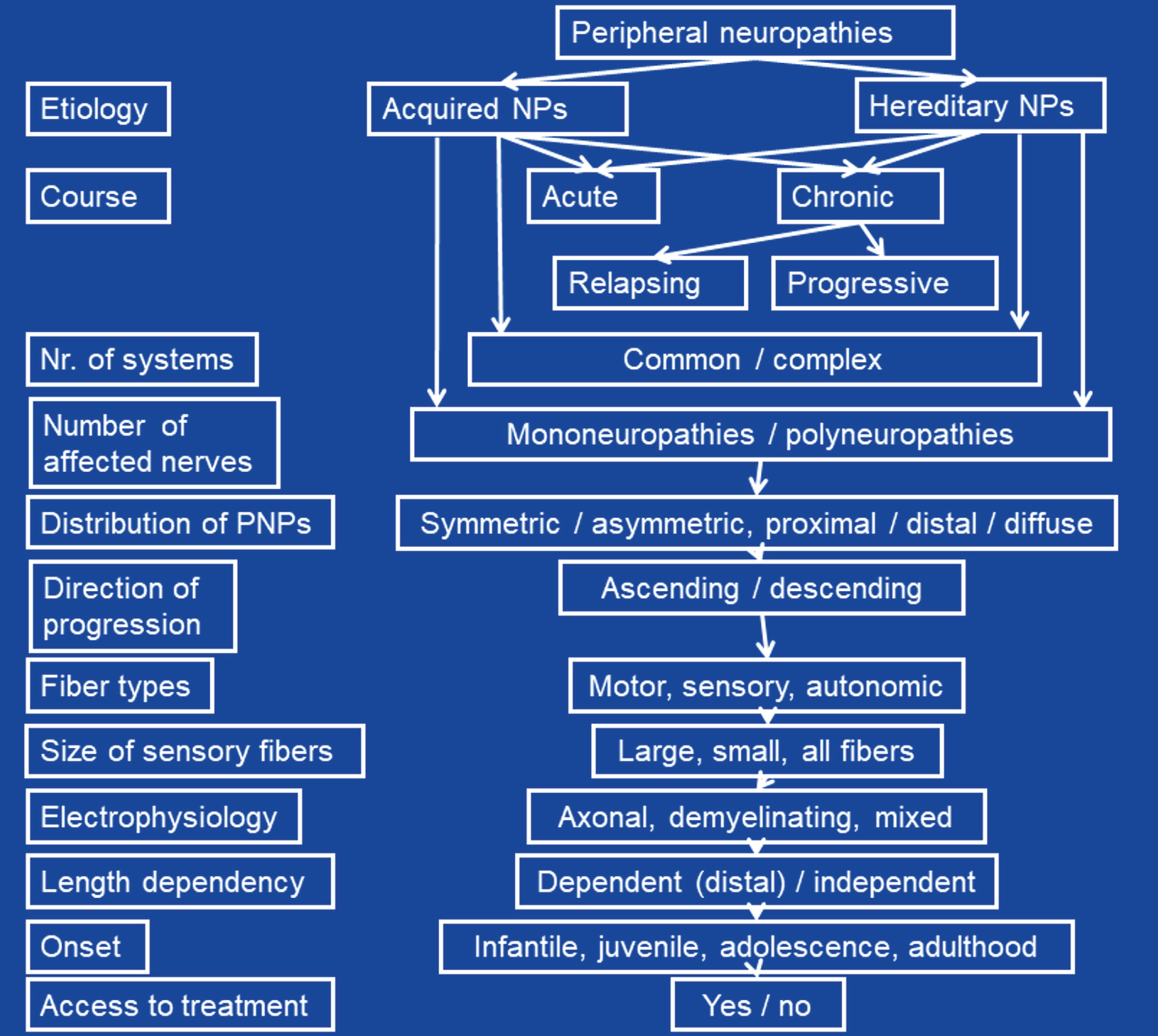
Classification of orphan peripheral neuropathies according to various criteria.
FREQUENCY OF PNPS
It is estimated that the prevalence of peripheral neuropathies is 2–3% in the general population [5]. However, in patients >55 y of age the prevalence increases to 8% [5]. Most of the neuropathies of undetermined cause are rare neuropathies but many of the acquired and hereditary neuropathies are rare either. Exact prevalence/incidence data about rare neuropathies are hardly available. In the rare case that prevalence/incidence data about neuropathies are available, they are representative only for a certain region but usually not for a nation, a continent, or the globe. Low prevalence data may result from inadequate effort to detect the cause or insufficient tools to search for the etiology. For example, transthyretin-related (TTR)-amyloid neuropathy is infrequent (estimated world-wide prevalence: >10000) because it may remain undetected for some time as long as TTR is considered as a differential diagnosis of neuropathy of undetermined cause. The prevalence of TTR-amyloid neuropathy is 22.93/100000 in Portugal [6], 8.7–11/100000 in Japan [7], and 91–104/100000 in Sweden [8]. The prevalence of chronic, inflammatory demyelinating polyneuropathy (CIDP) is calculated as 2.8–3.0/100000.
DIAGNOSIS OF PERIPHERAL NEUROPATHIES
Multiple algorithms for diagnostic work-up of neuropathies are available [9]. They all rely on the history, clinical exam, blood tests, NCSs, CSF investigations, imaging, biopsy and genetic investigations. Selection of laboratory investigations is variable, depending on the clinical presentation, availability, costs, and expertise of the referring neurologist. Generally, answers to the questions “what” (which nerve fiber modalities are involved), “where” (which are the complaints), “when” (which is the temporal evolution), and “which setting” (unique clinical circumstances of a patient) should be obtained to classify the type of neuropathy [5]. For work-up of hereditary neuropathies, it is crucial that a thorough individual and family history is taken and that apparently asymptomatic family members are seen by the managing physician for subclinical weakness, wasting, or foot deformities, since the affected subject may not recognise subtle deficiencies himself. As soon as hereditary neuropathy is suspected, genetic work-up (e.g. multigene panel, next generation sequencing (NGS)) should be initiated. NGS has the disadvantage that it does not reliably detect duplications or deletions, splice-site variants, or intronic variants and is difficult to interpret in the context of non-specific point mutations (variants of unknown significance) with oligogenic inheritance [10]. Additionally, NGS has a cost-depth trade-off [10], that is, it can screen a small number of genes with good read depth or a large number with less depth [10]. A list of Charcot-Marie-Tooth (CMT) genes can be accessed via the inherited neuropathy variant browser (http://hihg.med.miami.edu/neuropathybrowser). Work-up for SFNs relies on skin biopsy as golden standard but can be supported by quantitative sensory testing, electrophysiology, and the skin-responsive Sudoscan®. After diagnosing SFN, work-up for the underlying cause must follow. In patients with prominent muscle atrophy but little muscle weakness or sensory deficits and preserved tendon reflexes, a vascular neuropathy should be considered. In asymmetric PNPs an infectious, immune-mediated, or neoplastic etiology should be considered. Nerve biopsy is indicated for vasculitis, amyloidosis, sarcoidosis, or leprosy, but may be helpful also for CIDP. A proposal for the diagnostic work-up of orphan neuropathies is provided in Fig. 2.
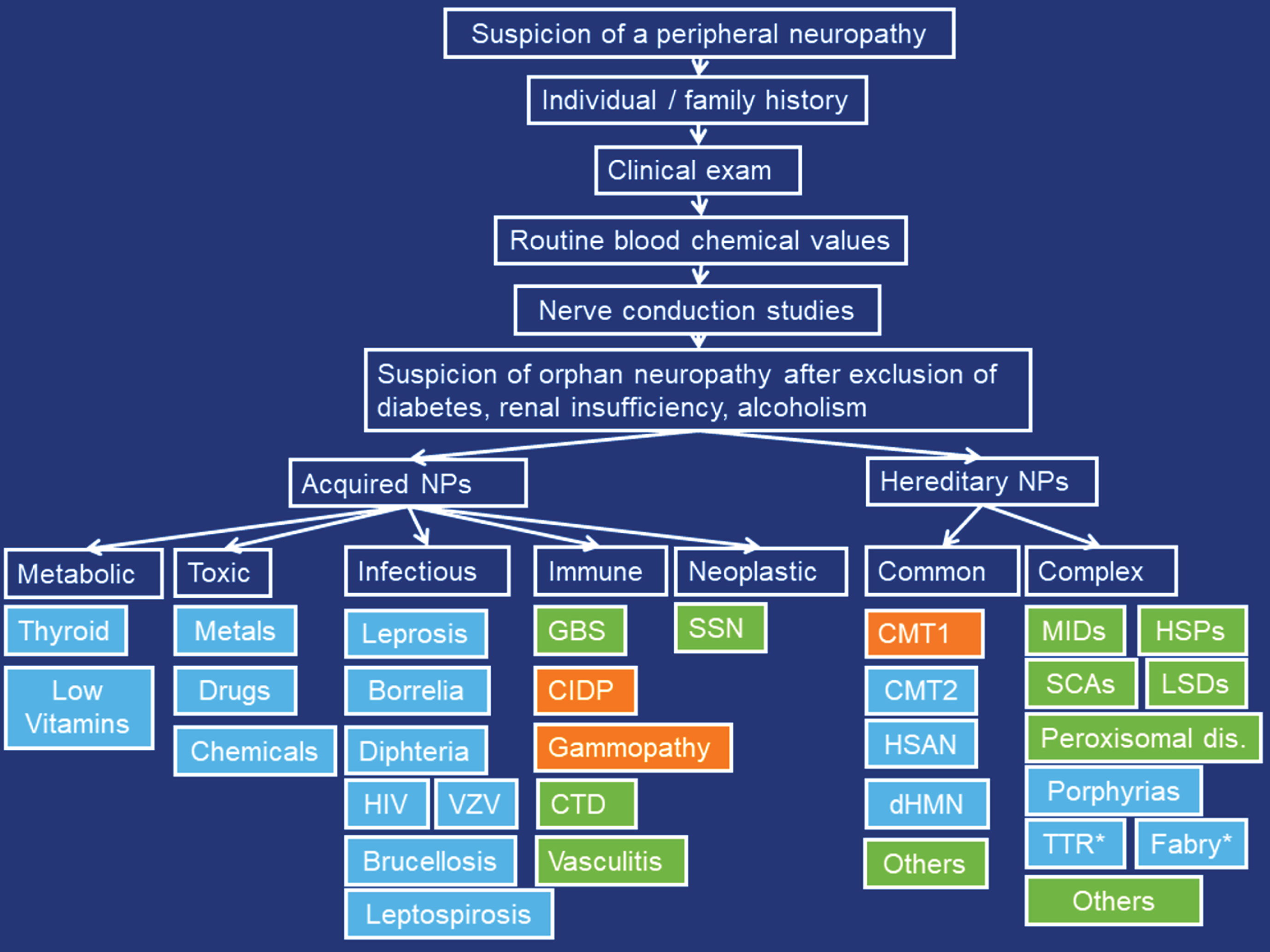
Proposed diagnostic work-up for orphan peripheral neuropathies. Orange: demyelinating, blue: axonal, green: mixed. CTD: connective tissue diseases, SSN: subacute sensory neuropathy, MIDs: mitochondrial disorders, HSPs: hereditary spastic paraplegias, SCAs: spinocerebellar ataxias, LSDs: lysosomal storage diseases, TTR: transthyretin-related neuropathy, *: Treatable.
ACQUIRED NEUROPATHIES
Acquired neuropathies include the metabolic, toxic, infectious, immune-mediated, and neoplastic/paraneoplastic neuropathies [5]. As with hereditary neuropathies, acquired neuropathies can be the exclusive manifestation of a disease or may occur together with affection of other organs. The majority of acquired neuropathies is frequently diagnosed but some have to be assessed as rare. Generally, acquired neuropathies are more frequent than hereditary neuropathies.
Metabolic neuropathies
Generally, metabolic neuropathies include diabetic, uremic, endocrine, and nutritive neuropathies. Particularly, among the nutritive neuropathies rare forms are known. Nutritive neuropathies are most frequently length-dependent, sensory neuropathies with the exception of vitamin-B12 deficiency neuropathy [11]. While diabetic and uremic neuropathies are highly prevalent, most of the other metabolic neuropathies are rare.
Endocrine
Vitamin deficiencies
Though vitamin deficiencies are frequently detected in the general population, neuropathy due to vitamin deficiency, with/without involvement of other systems, has been only rarely reported. Low vitamin levels may be due to dietary causes (malnutrition, malabsorption, diarrhea), autoimmune conditions (e.g. perniciosa), certain drugs, alcoholism, chronic colitis, bariatric surgery, or due to other gastrointestinal compromise. Low vitamins that can go along with neuropathy include vitamin-B12, folic acid, thiamine (vitamin-B1), vitamin-B6, and vitamin-E. Why only a small portion of patients with vitamin deficiency develops neuropathy, remains speculative but one hypothesis is that pre-existing nerve pathology may be necessary for the development of a vitamin-deficiency-related neuropathy.
Toxic neuropathies
Metals
Metal intoxications most frequently not only cause isolated neuropathy but also systemic manifestations. Thus, systemic features should be considered in the diagnostic work-up of suspected metal intoxications.
Drugs
Neuropathy can be a side effect of several drug intoxications. Most well-known for their neuropathic toxicity are chemotherapeutics and nucleoside analogs. However, neuropathy may be also a side effect of non-chemotherapeutic drugs.
Chemicals and plants
A number of chemicals (e.g. nitric oxide, vinyl-benzene, di-ethylene-glycol, hexa-carbon, acryl-amide, carbon-disulfide, and many others), is toxic to peripheral nerves [13]. Neuropathy due to intoxication with any of these molecules has been only rarely described. For example, acryl-amide causes, in addition to encephalopathy, dermatitis, and hyperhidrosis, a sensori-motor axonopathy [13]. Acryl-amide is neurotoxic as it directly attacks DNA or is transformed into glycidamid by liver enzymes [40]. Acrylamid and glycidamide bind to aminoacids and nucleotides and thus directly impair DNA functions. Hexa-carbon causes a subacute, length-dependent, sensori-motor axonopathy with secondary demyelination [13]. A length-dependent, sensori-motor axonopathy is also caused by carbon-disulfide. Diethylene-glycol causes an acute, rapidly progressive, sensori-motor axonopathy with demyelination [13]. Toxicity of vinyl-benzene manifests as painful, sensory, length-dependent SFN [13]. An example of a plant causing neuropathy is the buckthorn fruit, which occurs in south-west US and Mexico. Buckthorn causes a rapidly progressive, length-dependent, sensori-motor axonopathy with secondary demyelination [13]. The neuro-toxic agent in Buckthorn intoxication has been identified as T-544 [13].
Infectious neuropathies
The prevalence of infectious neuropathies is quite variable and strongly depends on the region of investigation [41]. What may be rare in one region, country, or continent, may be highly prevalent in another country. The prevalence of infectious neuropathies may also increase in association with epidemic outbreaks. Infectious agents most frequently causing neuropathy include M. Leprae (South America, Asia, Africa), Borrelia burgdorferi (Europe, America), C. diphteriae (Asia), HIV (worldwide), VZV (worldwide), hepatitis-C (worldwide), HTLV1 (worldwide), Zika (GBS) (South America), influenza-A (GBS) (Europe), CMV (Europe, America), dengue (GBS) (Asia, Africa), Toscana-virus (Europe, Africa), and other arboviruses.
Immune-mediated neuropathies
Guillain Barre syndrome
The term GBS includes a group of autoimmune disorders that share a common presentation of acute/subacute, progressive poly-radiculo-neuropathy [42]. The incidence of GBS varies between regions and countries from 1.1–1.8/100000/y to 2.66/100000/y [42]. There are several subtypes of GBS classified according to the underlying pathology, clinical presentation, and NCS features [42]. The most common subtype in Europe is acute, inflammatory, demyelinating poly-radiculo-neuropathy (AIDP) with primarily demyelinating features and a favourable prognosis [42]. Less common is acute, axonal, motor neuropathy (AMAN) with primary axonal injury, pure motor involvement, and a worse prognosis [42]. AMAN is the most prevalent subtype in East Asia. A rare cause of dysimmune and acquired neuropathy is acute painful autoimmune neuropathy, an acute small fiber neuropathy resembling GBS [43]. Acute, motor, sensory, axonal neuropathy (AMSAN) shares a similar pathogenesis with AMAN with additional sensory involvement. Miller-Fisher syndrome (MFS), is characterised by opthalmoparesis, areflexia, and ataxia. Bickkerstaff encephalitis presents similarly to MFS but additionally with impaired consciousness due to brainstem involvement. Less common variants are the pharyngeal-cervico-brachial variant, associated with GQ1b and GD1a antibodies and pandysautonomia, associated with GT1a antibodies [42, 44]. GBS is often preceded by an infection with
CIDP
CIDP is a chronic, demyelinating, poly-radiculo-neuropathy, which manifests clinically with symmetric weakness and sensory disturbances. CIDP is diagnosed according to the EFNS/AAN/INCAT criteria if there is progressive or recurrent symmetric weakness of proximal and distal upper and lower extremities for at least 2 months. Frequently, there is dissociation cytoalbuminique and there is demyelination on NCSs (reduced or normal motor conduction velocity, conduction block, temporal dispersion, increased F-wave latency, prolongation of distal latency and of the CMAP duration) [52]. According to these findings definite, probable, and possible CIDP are delineated. Several rare variants have been identified since the original description, including multifocal acquired demyelinating sensory and motor (Lewis-Sumner syndrome, MADSAM) neuropathy, a pure sensory form, distal, acquired, demyelinating, symmetric CIDP (DADS) neuropathy, and syndromes associated with monoclonal gammopathy [44]. Rarely, nerves innervating respiratory muscles or involvement of the autonomic nerves (bowel or bladder dysfunction, dry mouth/eyes, orthostatic intolerance, sexual dysfunction, flushing, hyperhidrosis) have been described [52]. In 2–6% of the cases CIDP is associated with elevated titers of auto-antibodies against the paranodal protein contactin-1 (CNTN-1), neurofascin-155 (NF-155), or contactin-associated protein-1 (Caspr1) [52]. These patients more frequently present with an acute onset, tremor and ataxia than CIDP patients without these antibodies and often poorly respond to standard immunotherapy [52]. Anti-CNTN-1 antibodies have been shown to interfere with the structural integrity of the paranodal region and are known to induce conduction block [53]. Occasionally, antibodies against gangliosides or MAG, are elevated in CIDP [44]. Most frequently these antibodies are found in patients with multifocal motor neuropathy (MMN) (GM1), DADS (anti-MAG), or chronic ataxic neuropathy with ophthalmoplegia, M-proteins, cold agglutinins and disialosyl antibodies (CANOMAD). The other parameters lack sufficient sensitivity and specificity to be used as reliable biomarkers. First-line treatment of CIDP includes steroids, intravenous immunoglobulins, and plasmapheresis.
Neuropathies related to monoclonal gammopathies (paraproteinemias)
Monoclonal gammopathies are a heterogeneous group of lymphoproliferative disorders characterised by overproduction and deposition of paraproteins (light chains, immunoglobulins). Monoclonal gammopathies include monoclonal gammopathy of unknown significance (MGUS), multiple myeloma, amyloidosis, Waldenström macroglobulinemia, and CANOMAD, polyneuropathy, organomegaly, endocrinopathy, monoclonal protein, and skin changes (POEMS) syndrome [13]. Neuropathy is a common manifestation in monoclonal gammopathies and attributed to a cross-reaction of the paraprotein with neural antigens, deposition of paraproteins in nerve or myelin, or due to a neurotoxic effect of the paraprotein [13]. Other pathomechanisms include vasculitis due to cryoglobulinemia or infiltration of the neve roots by a lymphoma. Neuropathy is particularly common in IgM-MGUS, multiple myeloma, and amyloidosis. More rarely, neuropathy develops in the remaining four types.
Connective tissue disorders
Connective tissue disorders are multisystem, autoimmune disorders that show common features of organ inflammation (most commonly joints and skin) [13]. In many of the connective tissue disorders the CNS or PNS are involved. Neuropathy has been most frequently reported in lupus erythematosus and rheumatoid arthritis (75% of patients). Rarely, neuropathy occurs in sarcoidosis, Sjögren syndrome, or systemic sclerosis (scleroderma). Neuropathy in sarcoidosis frequently starts as SFN with prominent pain, areas of tenderness, numbness, and dysesthesias, responding only to immunoglobulins or TNF-alpha blockers [13]. LFN is much less common and manifests as length-independent PNP or polyradiculopathy, or rarely as mononeuritis multiplex, GBS (AIDP), symmetric sensori-motor neuropathy, or as pure sensory neuropathy [13]. Neuropathy in Sjögren’s syndrome is usually a sensory, length-dependent PNP. Occasionally, sensory symptoms are patchy and asymmetric, suggesting ganglionopathy [13]. More rarely, neuropathy is sensori-motor taking a subacute or chronic course. Rarely, neuropathy presents as polyradiculopathy, mononeuritis multiplex, or autonomic neuropathy [13]. Neuropathy in systemic sclerosis (scleroderma) manifests as length-independent numbness and paresthesias of hands and feet [13].
Vasculitis
Vasculitic neuropathies result from non-infectious inflammation of the vasa nervorum [63]. Vasculitic neuropathy may occur in the context of systemic inflammation or from vasculitis confined to the peripheral nerves (non-systemic vasculitic neuropathy (NSVN)) [63]. Vasculitis may be primary (arteritis originates from the arteries) or secondary (due to conditions such as connective tissue diseases, infections, paraneoplastic, immune-mediated conditions, or drug-induced) [63]. Vasculitic neuropathy typically presents with an acute or subacute onset of multiple, painful, sensory, or sensori-motor mononeuropathies [63]. All sensory modalities are affected as pure SFNs are rarely vasculitic [63].
Primary vasculitis
Neoplastic
Neuropathies due to neoplasms are rare but have been occasionally reported as acute, demyelinating neuropathy in neurolymphomatosis due to direct infiltration of the PNS [68], as upper limb neuropathies in epithelioid hemangio-endothelioma (EHE) [69], as infiltration of the plexus by lung cancer (Pancoast) [70], as plexopathy in breast cancer, or as peripheral nerve metastasis [71].
Paraneoplastic
Paraneoplastic neuropathies are regarded as a secondary complication of a primary neoplasm due to reaction of the immune system against the neoplasm. Paraneoplastic neuropathies develop prior or after detection of a malignoma and are attributed to a remote effect of the malignoma, irrespective of neoplastic infiltration, chemotherapy, infection, or metabolic complications [72–74]. Paraneoplastic neuropathies occur in <1% of patients with malignoma [72]. Definite and possible paraneoplastic neuropathies are delineated [72]. Definite paraneoplastic neuropathies include those with a direct link between malignoma and neuropathy in the presence of an antibody, those representing a well-established paraneoplastic syndrome but without an identified antibody, and those which improve upon successful treatment of malignancy [72]. With regard to the antigen localisation, two types of antigens are delineated, intracellular and membrane-bound antigens [72]. The most frequent intracellular antigens are HuD and CRMP5. The most frequent membrane bound antigen is Caspr2 [72].
Paraneoplastic neuropathies are classified as neuronopathies (subacute sensory neuropathy (SSN (Denny Brown syndrome), lower motor neuron disease, sensory-motor neuropathy, autonomic neuropathy), as sensorimotor neuropathies without gammopathy (axonal, mixed axonal/demyelinating, demyelinating, vasculitic), as sensorimotor neuropathies with gammopathy (axonal, sensory and painful (AL-amyloidosis, myeloma), demyelinating (Waldenström, non-Hodgkin lymphoma), POEMS, vasculitic), and as neuromyotonia. An estimation of the number of these subtypes so far reported is provided by Antoine et al. 2017 [72].
The most common of the paraneoplastic neuropathies is SSN [72]. It is probably T-cell mediated and targets neurons in dorsal root ganglia [72]. Sensory disturbances are multifocal, asymmetric, involve the upper limbs in a length-independent manner, and progress rapidly over weeks [72]. NCSs show reduced or absent sensory nerve action potentials. SSN is associated with small cell lung cancer (SCLC) in 70–80% of cases [75]. Most patients have anti-Hu antibodies, some CRMP5 antibodies, and in 10% no antibodies are found. Among the sensory-motor neuropathies without gammopathy the mixed axonal/demyelinating subtype is the most frequent and associated with CRMP5 antibodies. Among those with CRMP5 antibodies about 50% develop neuropathy. Only anecdotally anti-Yo, or anti-Ma2 antibodies have been reported.
Acquired neuro-myotonia (peripheral nerve hyper-excitability), clinically characterised by cramps, stiffness, twitching, spasms, weakness, paresthesias, hyperhidrosis, and abnormal relaxation, occurs most frequently in association with thymoma, SCLC, or lymphoma. Frequently, Caspr2 antibodies and rarely netrin-1 antibodies can be found.
Pure sensory neuropathies
Pure sensory neuropathies (PSNs) are usually acquired but can be also hereditary. A subtype of acquired PSN is sensory neuropathy with anti-FGFR3-autoantibodies. PSN can be also associated with vitamin-B12 deficiency. Chronic immune sensory polyradiculopathy (CISP) is a pure sensory form of CIDP. There are pure sensory forms of LFNs associated with connective tissue disorders. PSN may be a manifestation of NSVN. Among the mononeuritis multiplex neuropathies, a pure sensory form with IgG1 deficiency has been reported. As a counterpart to MMN, multifocal sensory demyelinating neuropathy has been described. Statins are known to cause a pure sensory and autonomic ganglionopathy. Pure sensory GBS is a rare entity.
HEREDITARY NEUROPATHIES
Hereditary neuropathies can be common or complex (Tables 1 and 2). Common hereditary neuropathies are characterised by isolated or predominant affection of the peripheral nerves, whereas complex hereditary neuropathies are characterised by being part of a hereditary multisystem disorder, which overshadows neuropathy [10]. Generally, hereditary neuropathies are classified according to the 1) clinical manifestations (hereditary sensory-motor neuropathies (HMSN), hereditary sensory-autonomic neuropathies (HSAN), distal hereditary motor neuropathies (dHMN)), 2) nerve conduction studies (NCSs, axonal, demyelinating, mixed), 3) mode of inheritance (autosomal dominant (AD), autosomal recessive (AR), X-linked (XL), maternally transmitted), and 4) mutated gene. HMSN types are also known as Charcot-Marie-Tooth (CMT) disease. The most widely used classification system is the one proposed by Dyck et al. [1]. This classification system has been repeatedly revised and the current version also includes the mutated genes.
Overview about hereditary neuropathies (updated until 31st August 2020)
CHN: congenital hypomyelinating neuropathy, DSD: Dejerine-Sottas disease, HMN: hereditary motor neuropathy, im: intermediate, SFN: small fiber neuropathy, SMA: spinal muscular atrophy.
Overview of orphan, complex hereditary neuropathies
ERT: enzyme replacement therapy, PNP: polyneuropathy, PSD: peroxisomal disorders.
Common hereditary neuropathies
Among common hereditary neuropathies the frequency of rare forms is particularly high. A large number of common hereditary neuropathies has been reported only in a single family or a few families. [76]. Often the pathogenic variant has been detected only in one or a few family members. Rare hereditary neuropathies have been most frequently identified by panel investigations or whole exome sequencing (WES). Common hereditary neuropathies are classified as CMT/HMSN (∼75% of common hereditary neuropathies), with predominant motor and sensory involvement, as HSAN (∼3% of common hereditary neuropathies) with predominant sensory or autonomic involvement, or as distal hereditary motor neuropathy (HMN) (∼3% of common hereditary neuropathies) [77]. There is a significant overlap between HMN, HMSN, and HSAN [10]. Other common hereditary neuropathies include hereditary neuropathy with liability to pressure palsies (HNPP) (∼3% of hereditary neuropathies), hereditary plexopathy, giant axonal neuropathy (GAN), and the hereditary SFNs [77]. Several subtypes derive from inclusion of the underlying mutated gene in the classification. Currently, mutations in >170 genes have been identified as causes of hereditary neuropathies [76].
CMT (HMSN)
More than 120 genes have been identified, which are associated with the CMT/HMSN phenotype (Table 1) [78]. Genes most frequently mutated in CMT1 phenotypes (AD, demyelinating) are
An example of a rare CMT is CMT4C which is due to mutations in
HSAN
HSAN represent ∼3% of hereditary neuropathies [77]. Currently, mutations in >25 genes are made responsible for the respective HSAN subtypes (Table 1) [76]. All HSANs have to be regarded as rare or ultra-rare diseases (Table 1).
Distal HMN
Distal HMNs are rare. Like the HSANs, HMNs constitute ∼3% of the hereditary neuropathies [77]. Currently, mutations in >35 genes are made responsible for distal HMNs (Table 1) [76]. All of them have orphan status.
Other common hereditary neuropathies
These include HNPP, GAN, hereditary plexopathy, and the hereditary SFNs. With the exception of HNPP, which is due to duplications/deletions in
Complex hereditary neuropathies
Complex hereditary neuropathies can be more easily diagnosed than common hereditary neuropathies as they go along with concomitant pathology in systems other than the peripheral nerves. Accordingly, not only the neurologist may be involved or the first being confronted with such a phenotype but many other specialities depending on the time point at which other organs/tissues/structure become affected. Since some of the manifestations may favourably respond to symptomatic treatment, it is crucial that the managing physician refers these patients to other responsible specialities. Complex hereditary neuropathies are most prevalent among MIDs, hereditary spastic paraplegias (HSPs), spinocerebellar ataxias (SCAs), lysosomal storage disorders (LSDs), porphyrias, and other rare genetic conditions (Table 2).
Mitochondrial neuropathies
Many of the syndromic and non-syndromic MIDs manifest with neuropathy. Neuropathy may dominate the phenotype or may be only a collateral feature. Particularly, among the non-syndromic MIDs, neuropathy can be the only manifestation, usually at onset of the MID. Generally, the prevalence of neuropathies in MIDs is high but compared with the general population mitochondrial neuropathies are rare. Mitochondrial neuropathies more frequently occur with nDNA than mDNA variants, while neuropathies are rare with single mtDNA deletions.
Hereditary spastic paraplegias (HSPs)
HSPs are rare genetic disorders, clinically manifesting with spasticity predominantly of the lower limbs and urinary dysfunction (pure HSPs) [102]. Some of the HSPs, however, may manifest with additional phenotypic features, including neuropathy (HSP-plus) [102]. Currently, 80 different types of HSPs are differentiated, which are classified according to their mode of inheritance as AD, AR, XL, or maternal transmission (MT). HSPs, which not only manifest with paraspasticity and bowel or bladder dysfunction, but also with neuropathy alone or with other manifestations (HSP-plus), include SPG1–7, SPG9–11, SPG14-15, SPG25, SPG27, SPG30-31, SPG36, SPG38-39, SPG43. SPG47. SPG49. SPG55–57, and SPG76 [102]. Except for SPG4, SPG3, SPG31 (most frequent of the AD forms) and SGG11 (most frequent of the AR forms), all other SPGs are regarded as rare.
Spinocerebellar ataxias (SCAs)
Currently (8/2020), 37 types of SCAs (SCA1–SCA37) are known. Some of the SCAs may occasionally go along with neuropathy. These include SCA1 (sensory neuropathy) [103], SCA2 (axonal neuropathy) [104], SCA3 (Machado-Joseph disease) [105], SCA4 [106], SCA6 [78], SCA7 [107], SCA10 [108], SCA11 [109], and SCA18 [110]. Neuropathy may be also a feature in non-classified SCAs, such as those due to mutations in
Porphyrias
Porphyrias are a group of ultrarare, mostly hereditary metabolic disorders due to a defective hem biosynthesis pathway [112]. Primary (hereditary) and secondary (acquired) porphyrias are differentiated. Primary porphyrias are subdivided into hepatic and erythropoetic forms. Among the hepatic porphyrias, acute porphyrias (acute intermittent porphyria, variegate porphyria, coproporphyria, DOSS porphyria (ALA-dehydratase (porphobilinogen synthase) deficiency), the most common of the porphyrias termed after Dr. Doss) and chronic porphyrias (porphyria cutanea tarda, hepato-erythropoetic porphyria) are delineated. The phenotype depends on the defective enzyme within the pathway and is due to accumulation of intermediate metabolites. Neuro-visceral porphyrias are characterized by recurrent, acute attacks, triggered by excessive hem synthesis, clinically manifesting as severe abdominal pain, vomiting, tachycardia, hypertension, hyponatremia, mild cognitive impairment, and peripheral neuropathy [112]. Severe attacks may manifest with seizures, psychosis, quadruparesis, respiratory failure, coma, or death [112]. Neuropathy occurs particularly in aminolevulinic acid dehydratase (
Lysosomal disorders
Lysosomal storage disorders (LSDs) predominantly affect the brain but in some of the about 80 subtypes, neuropathy has been occasionally described. Since LSDs are rare conditions and neuropathy does not occur in all of them, neuropathy in LSDs are generally rare. LSDs in which neuropathy has been described include Schindler’s disease [114], Ohtahara syndrome [115], metachromatic leucodystrophy [116], Fabry disease [117], hereditary β-mannosidosis [118], Krabbe disease [119], Sandhoff disease [120], Gaucher disease [121], Niemann-Pick disease type C [122], galactosialidosis [123], mucolipidosis [124], ceroid lipofuscinosis [125], and rarely Pompe disease [126]. In Tay-Sachs disease axonal neuropathy can be found in a quarter of patients. The most frequent of the LSDs with neuropathy is Fabry’s disease. Though Fabry disease is rare, all other LSDs are even rarer and thus neuropathy in these other LSDs has orphan status.
Peroxisomal disorders
Peroxisomal disorders form a group of rare, multisystem metabolic disorders, which are generally classified as either due to impaired peroxisomal biogenesis (PBDs) or due to deficiency of a single perioxismal enzyme (Zellweger spectrum disorders). The prototype of PBDs is Zellweger syndrome, clinically characterised by hypotonia, developmental delay, neuropathy, facial dysmorphism, epilepsy, rhizomelia, abnormal calcification, hepatomegaly, and abnormal liver function. Neuropathy in Zellweger syndrome is not well characterised but in the majority of the cases it is of the axonal type. The most well-known of the Zellweger spectrum disorders are neonatal, X-linked adrenoleukodystrophy and Refsum’s disease. Adenoleukodystrophy is due to mutations in the
Other complex hereditary neuropathies
Complex, hereditary neuropathies occur in a number of other genetic conditions. These include Tangier’s disease, TTR-amyloid neuropathy, progressive myoclonus epilepsy due to
CONCLUSIONS
Rare (orphan) neuropathies of peripheral nerves need to be studied not only to assess the true prevalence of these conditions but also to delineate similarities and differences concerning clinical presentation and genetic heterogeneity from other more frequent types of neuropathies and to discover etiology and pathophysiology. Diagnosis of orphan neuropathies can be improved by refering neuropathies of unknown etiology to a reference center. Diagnosis of hereditary neuropathies can be improved by application of advanced genetic testing methods as early as possible. Treatment of acquired neuropathies can be improved by accurate, early diagnosis. Among the hereditary neuropathies TTR-PNP, Fabry disease, and neuropathies due to mutations in the riboflavin transporter need to be detected as early as possibly to provide available treatment as early as possible. Though rare neuropathies are challenging with regard to diagnosis, epidemiology, and treatment, we should nonetheless meet this challenge as it may help elucidating etiology, pathophysiology, range of clinical presentation, and potential treatment options for these conditions.
CONFLICTS OF INTEREST
There are no conflicts of interest.
FUNDING
No funding was received.
AUTHOR CONTRIBUTION
JF: design, literature search, discussion, first draft, critical comments.
All authors have read the journal’s position on issues involved in ethical publication.
The authors have read the Journal’s position on issues involved in ethical publication.
