Abstract
Background:
Weakness affects motor performance and causes skeletal deformities in spinal muscular atrophy (SMA). Scoliosis surgery decision-making is based on curve progression, pulmonary function, and skeletal maturity. Benefits include quality of life, sitting balance, and endurance. Post-operative functional decline has not been formally assessed.
Objective:
To assess the impact of scoliosis surgery on motor function in SMA types 2 and 3.
Methods:
Prospective data were acquired during a multicenter natural history study. Seventeen participants (12 type 2, 5 type 3 with 4 of the 5 having lost the ability to ambulate) had motor function assessed using the Hammersmith Functional Motor Scale Expanded (HFMSE) performed pre-operatively and at least 3 months post-operatively. Independent t-tests determined group differences based on post-operative HFMSE changes, age, and baseline HFMSE scores.
Results:
Three participants had minimal HFMSE changes (±2 points) representing stability (mean change = –0.7). Fourteen participants lost >3 points, representing a clinically meaningful progressive change (mean change = –12.1, SD = 8.9). No participant improved >2 points. There were no age differences between stable and progressive groups (p = 0.278), but there were significant differences between baseline HFMSE (p = 0.006) and change scores (p = 0.001). Post-operative changes were permanent over time.
Conclusions:
Scoliosis surgery has an immediate impact on function. Baseline HFMSE scores anticipate post-operative loss as higher motor function scores were associated with worse decline. Instrumentation that includes fixation to the pelvis reduces flexibility, limiting the ability for compensatory maneuvers. These observations provide information to alert clinicians regarding surgical risk and to counsel families.
Introduction
Spinal muscular atrophy (SMA) is a genetically determined neuromuscular disease of childhood characterized by progressive muscle weakness and caused by degeneration of lower motor neurons in the spinal cord and brainstem. The disease affects an estimated 10–16 per 100,000 infants [1]. SMA has 3 clinical phenotypes including: type 1, the most severely involved (never achieve the ability to sit unsupported); type 2, the moderately involved (achieve the ability to sit but never walk); and type 3, the less involved (achieve the ability to walk independently but have varying degrees of disability) [2].
The primary musculoskeletal issues for SMA patients include: 1) muscular imbalances and prolonged postures with the development of contractures in the lower and upper extremities; 2) weakness affecting motor performance, which leads to progressive spinal deformity and pelvic obliquity; and 3) fractures due to osteoporosis exacerbated by inactivity, lack of weight bearing, or falls [3]. Treatment for these musculoskeletal issues can range from conservative methods including palliative bracing with orthoses, using assistive devices, or rehabilitation including physiotherapy, to more invasive methods including surgery for spinal stabilization and lengthening procedures for contractures [4, 5].
Occurrence of scoliosis, age at onset, and severity of the curve are related to SMA disease severity [6]. Progressive scoliosis develops in nearly all non-ambulatory SMA patients. Immobility and weakness influences posture, chest-wall development, and respiratory function [6, 7]. Scoliosis and pelvic obliquity is observed on average at age 3 years in type 2 patients, with continual progression even after spinal growth is completed [5, 8]. Rates of progression for scoliosis have been reported from 8° to 20° annually when left untreated in SMA type 2 and 0.6° to 15° annually in ambulatory and non-ambulatory type 3 [3, 10]. Preferred radiologic measurement of scoliosis in type 2 patients has varied among clinicians from the sitting to supine position. A recent study reported SMA type was associated with the cumulative lifetime risk of scoliosis that requires surgery. Age at which surgery was necessary was associated with acquired milestones and their loss [10].
The surgical treatment of scoliosis is of great importance and decision-making is largely based on magnitude of the curve (i.e. major curve Cobb angle ≥50°) and rate of progression (≥10° per year) [11]. Severe underweight and failure to thrive may delay time to surgery in some cases. Related musculoskeletal issues includes parasol rib and thoracic deformity, pelvic obliquity, and hip dislocation. Impaired pulmonary function, nutritional status, and adverse effects on motor function including loss of stability in sitting, trunk imbalances, pain, and skeletal maturity are other factors which may be considered [3, 11].
The reported benefits of scoliosis surgery have included improved quality of life, preserved trunk sitting balance and endurance, enhanced respiratory function by re-aligning the thorax, as well as cosmesis [11, 12]. Post-operative functional decline has been described retrospectively for activities of daily living and an increased use of upper extremity aides [13, 14], but the impact of scoliosis surgery on motor function has not been formally assessed. The aim of this study is to assess the impact of scoliosis surgery on motor function in SMA types 2 and 3 and to quantify the precipitous motor decline as one potential morbidity that often accompanies scoliosis surgery.
Materials and Methods
Data was acquired prospectively as part of an IRB-approved multicenter natural history study. All patients with SMA participating in the Pediatric Neuromuscular Clinical Research (PNCR) Network sites were eligible to participate. The study was approved by the institutional review board at each clinical site (Columbia University, The Children’s Hospital of Philadelphia and University of Pennsylvania, and Boston Children’s Hospital and Harvard Medical School) and informed consent was obtained prior to enrollment [15, 16]. This study is in accord with the Helsinki Declaration of 1975.
Subjects
Seventeen participants with SMN1-associated SMA type 2 or 3, diagnosed before age 19 years, were included between December 2005 and September 2015 (Fig. 1). Exclusion criteria for PNCR natural history study participation were unstable medical conditions that would preclude participation, severe respiratory compromise at baseline, or other conditions precluding safe participation and travel exceeding a reasonable distance [15]. No participants received any investigational or approved disease-modifying therapies for SMA.
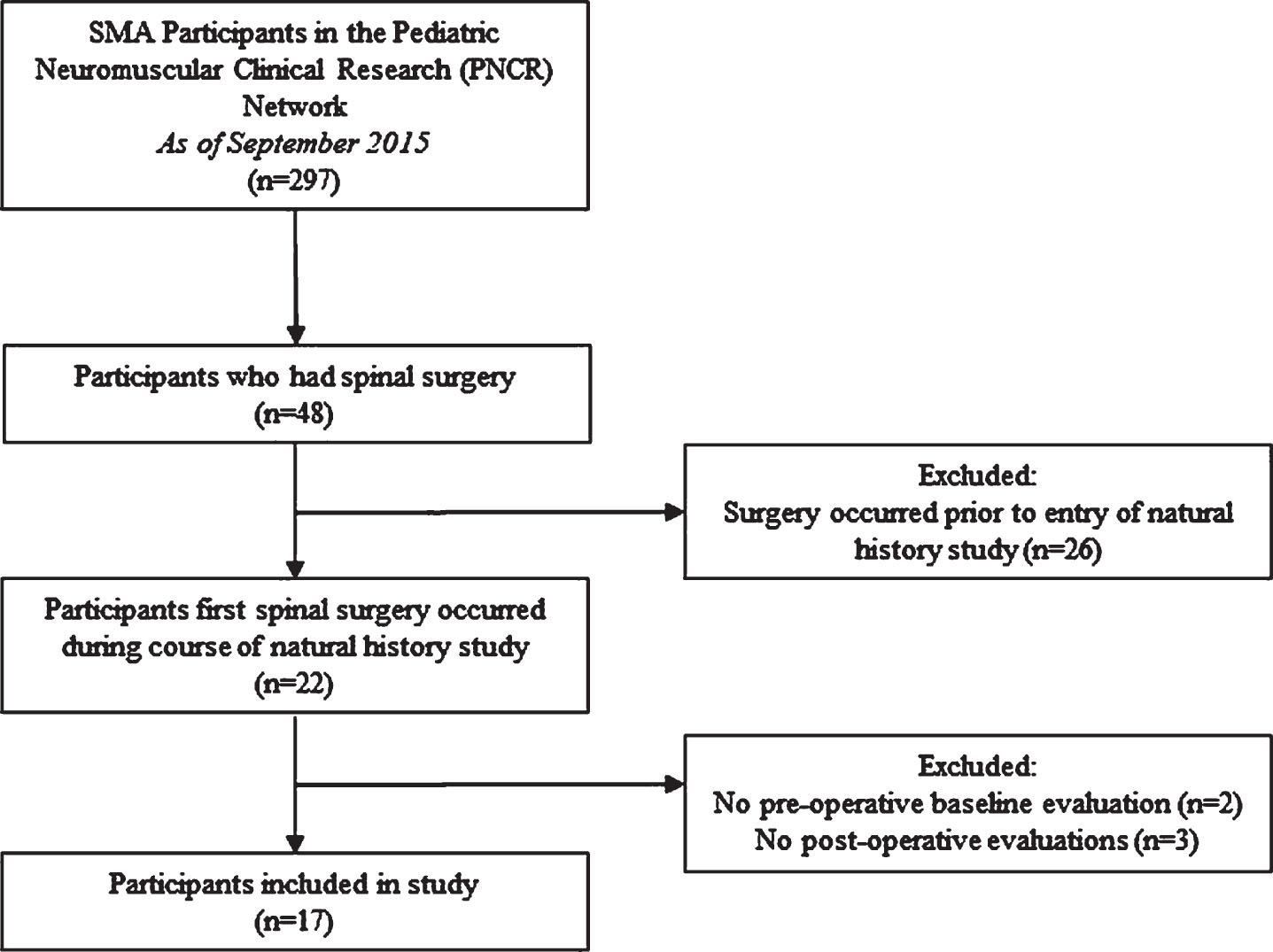
Participant recruitment.
Procedures
Consecutive participants in the natural history study who underwent scoliosis surgery had age, SMA type, type of scoliosis surgery (vertical expandable prosthetic titanium rib [VEPTR] growing rods, traditional growing rods, or spinal fusion), and standardized motor function assessed pre-operatively and at least 3 months post-operatively. Longitudinal post-operative motor function data collection continued if available for the duration of the natural history study. Motor function was assessed using the Hammersmith Functional Motor Scale Expanded (HFMSE) which is a disease-specific gross motor measure including 33 items performed in a hierarchical fashion ranging from sitting, to rolling, to crawling, standing, and jumping, for a maximum score of 66, with a higher score representing higher function [17, 18].
Data analysis
Demographic and clinical characteristics were summarized using means and standard deviations for continuous variables or using frequencies and percentages for categorical variables. For analytic purposes, group comparisons occurred in 3 ways: 1) Participants were grouped by post-operative HFMSE changes, those participants who lost within±2 points, representing no change (stable group), and those who lost >3 points representing a functionally meaningful change (progressive group); 2) Participants were grouped by their age at the pre-operative/baseline visit (<10 years old and >10 years old); and 3) Participants were grouped by baseline HFMSE score of <10 points (weaker group) and those who had a score >10 points (stronger group). A score of less than 10 points was an exclusion criteria for phase 3 SMA clinical trials (Clinicaltrials.gov identifier: NCT02292537 and NCT02644668) and is descriptive of a patient who can sit, bring hands to head in sitting, and roll side-to-side, but is unable to roll over fully, crawl or stand. Independent t-tests were performed using HFMSE change scores from pre- to post-operative visit to determine group differences.
Results
Seventeen participants (mean age 9.9 years, range 5–18) underwent scoliosis surgery (at mean age 10.4 years, range 6–19) during the course of the study. Clinical characteristics are described in Table 1. Majority of type 2 and 3 patients were able to sit independently and were non-ambulatory (n = 15). Only one type 2 patient unable to sit (non-sitter) pre-operatively gained the ability to sit independently at the first assessment post-operatively. Only one type 3 patient able to walk was only able to take several steps independently pre-operatively and lost this ability at the first assessment post-operatively which remained permanent. The type of scoliosis surgery between VEPTR growing rods and spinal fusion was nearly equal. No patients had traditional growing rods placed. The mean duration between the baseline assessment and scoliosis surgery was 5.8 months (SD 2.8, range 1–11 months). Post-operative changes were assessed at a mean of 13.4 months after scoliosis surgery (SD 4.0, range 12–24 months). Mean pre-operative and post-operative HFMSE score, and mean change scores for the whole group, by SMA type, and by type of scoliosis surgery are shown in Table 1.
Clinical Characteristics
HFMSE, Hammersmith Functional Motor Scale Expanded; SD, Standard Deviation; VEPTR, Vertical Expandable Prosthetic Titanium Rib.
Group comparisons are highlighted in Table 2. Three of 17 participants had minimal HFMSE changes within±2 points, representing no change or stability (age = 9.1 years, SD = 3.8). The remaining 14 participants lost >3 points on the HFMSE, representing a functionally meaningful change (age = 10.1 years, SD = 3.7). No participant had improvement greater than 2 points post-operatively (Table 2).
Group Comparisons
* significant difference (p = 0.001); ** significant difference (p < 0.01). HFMSE, Hammersmith Functional Motor Scale Expanded; SD, Standard Deviation.
There was no age difference between stable and progressive groups (p = 0.278), but there were significant differences in baseline/pre-operative HFMSE scores (p = 0.006) and change scores (p = 0.001) (Table 2).
Nearly half of all 17 participants lost function on 9 items on the HFMSE (items 2, 3, 4, 5, 8, 9, 11, 21, & 22) (Fig. 2). These 9 items fall into 5 functions which include long sitting, hands to head in sitting, rolling supine to prone, prone props on elbows, and hip flexion in supine. In participants who had a baseline HFMSE score <10 points (weaker group), the most vulnerable functions lost post-operatively were long sitting, sitting one hand to head, and rolling supine to side-lying. In participants who had a baseline HFMSE score >10 points (stronger group), the most vulnerable functions lost post-operatively were long sitting, sitting hands to head, rolling, sitting to lying, prone prop, and hip flexion. Similarly, in participants who were stable (±2 points change), the most vulnerable functions lost post-operatively were long sitting, sitting one hand to head, and rolling supine to side-lying. In participants who were progressive (≥3 point change), functions lost post-operatively occurred in all 9 items (Table 3).
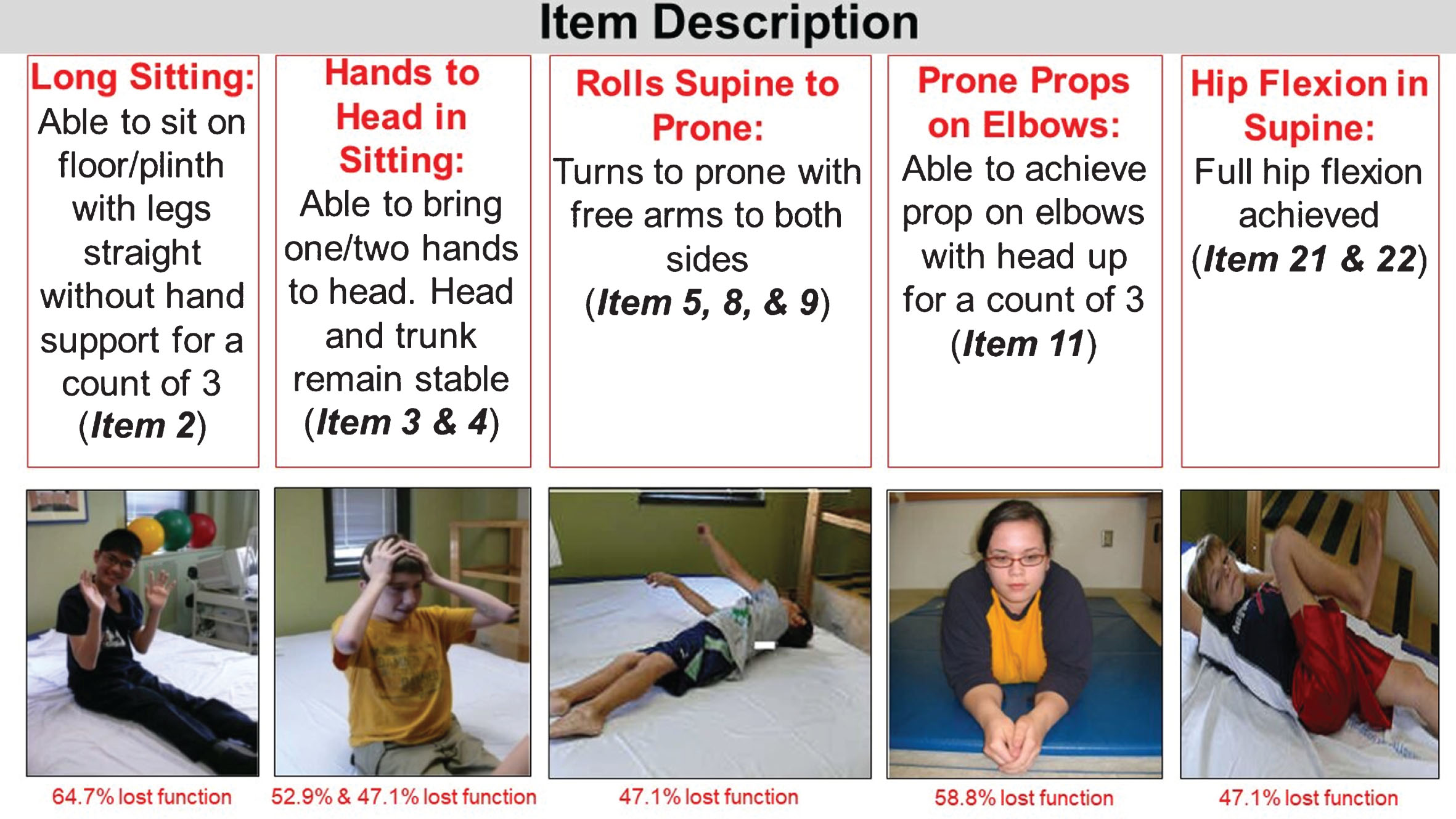
Most vulnerable motor functions (HFMSE items) lost post-operatively. Permissions for use of figures published granted.
Most vulnerable motor functions (HFMSE items) lost post-operatively by stable and progressive groups
HFMSE, Hammersmith Functional Motor Scale Expanded; R, right; L, left; SD, Standard Deviation.
Longitudinal post-operative data collection occurred up to 102 months (mean 44 months, SD 28.4). Longitudinal post-operative changes transpire immediately after surgery and plateau thereafter (Fig. 3). A drop in score from baseline to the first post-operative assessment occurs for each participant which remains permanent (minimum of 12-months post-operative follow-up). Three sitters eventually lost the ability to sit independently post-operatively during their longitudinal assessments.
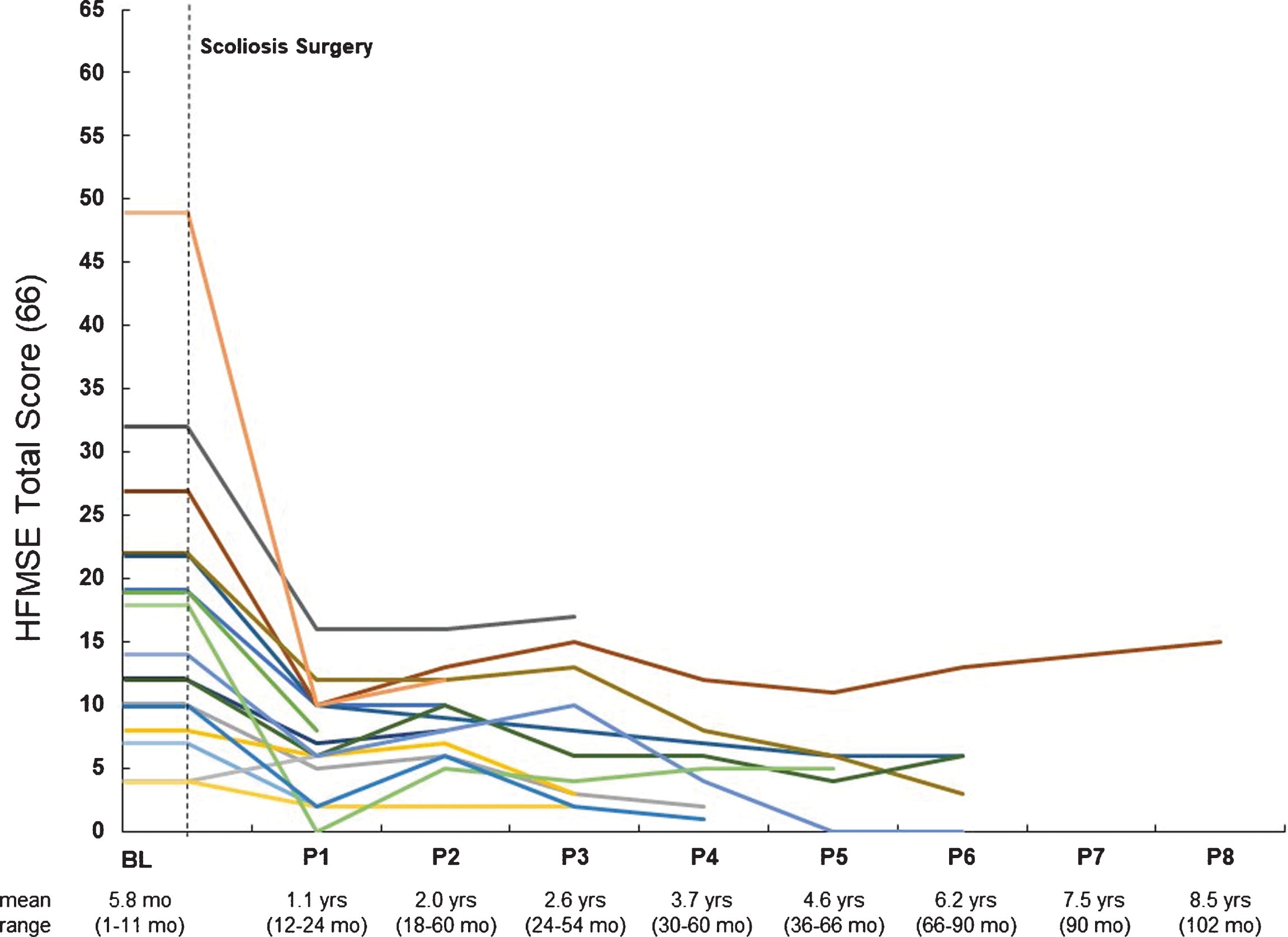
Post-operative HFMSE scores for individual participants over time. Abbreviations: HFMSE, Hammersmith Functional Motor Scale Expanded.
Discussion
Scoliosis surgery is the standard of care for progressive spinal deformity in SMA. Cobb angle, rate of progression, pulmonary function, and age are all important factors to determine the optimal timing [11]. Surgery stabilizes the spine, prevents further curvature and mitigates long-term clinical worsening. Pre-operative motor function can anticipate the degree of functional decline seen immediately post-operatively. Patients with higher motor function scores experience a greater decline and may be at greater risk of a functionally meaningful post-operative loss. The resulting fixation, including the pelvis, following spinal surgery decreases spinal and lower extremity flexibility as documented in the post-operative motor function scores, thereby limiting the ability for compensatory maneuvers. In light of our findings, the long-term benefits and permanent risks should be weighed by clinicians when advising the family regarding the timing and consequences of scoliosis surgery in SMA.
Few studies have reported the possible adverse effects of scoliosis surgery. The reported detrimental effects include reduced independence and functional ability [14]. Despite improved curve correction, some patients lose the ability to sit and others may require a thoracic lumbar sacral orthosis to improve sitting and increase time in a seated position [3]. Early reports placed an emphasis on the loss of muscle strength, loss of a particular motor skill (including loss of walking ability, functional status, or head control), or experience a decrease in pulmonary function post-operatively [19–21]. Furumasu et al. found the loss of function to be attributed to the post-operative lack of spinal flexibility, and the longer lever arm of the trunk rather than disuse atrophy [13]. This complication is similar to our findings as those who had greater functional abilities pre-operatively had the most to lose. Eighty-two percent of participants experienced a loss of function which represented a meaningful change (≥3 points). The most vulnerable functions lost post-operatively for the weaker group were long sitting, sitting one hand to head, and rolling supine to side-lying. These items can interfere with daily functions such as sitting up in bed, scratching an itch on their forehead, and turning their body at night while sleeping. The most vulnerable functions lost post-operatively for the stronger group were long sitting, sitting hands to head, rolling, sitting to lying transition, prone prop positioning, and active hip flexion. Similar to the weaker group, these items can translate into daily functions such as lying themselves down in bed independently and putting on pants. Conversely, past literature reported that many patients felt improvements in comfort and stability of the spine post-surgery outweighed these functional declines [19, 21–25].
One study, remarkably, reported improved functional benefits after spinal surgery. Thacker et al. noted improved functional status in 63% of neuromuscular patients, including SMA, post-operatively. The most notable improvement occurred in the dependent sitters as all gained the ability to sit independently (when placed) and some could now use their upper limbs for activities of daily living such as feeding, transfers, and helping in their own care. Additionally, caregivers reported a dramatic decrease in the time needed for care [26]. This was not our experience, however, as most patients lost functional ability and only one (6.1 year old female, SMA type 2) regained the ability to sit independently. This apparent difference might reflect differences in study design. Our study was prospective and clinical assessment used a standardized motor function scale for SMA. Thacker et al. used a retrospective chart review examining hospital records and included other neuromuscular diseases with variable courses of progression [26].
Progressive scoliosis can cause deterioration in functional capacity and independence by affecting limb movements, gait, standing, sitting, rolling, trunk stability, balance, activities of daily living, bilateral hand use, pain, and positioning [19, 27]. Conservative treatment of scoliosis is complex and ineffective. Early physiotherapy may play an important role including maintenance of walking and standing ability (including passively with external support such as standing frames) and well-regulated strength training [7, 28], although little supportive evidence is available. Seating supports and wheelchair modifications may also help support and position a progressive spine into a more optimal alignment while providing a more functional orientation and increased comfort [27, 28].
Bracing may not influence the progression of scoliosis for non-ambulatory SMA patients. However, this intervention may have value as a temporary remedy, assuming the ability to maintain a proper fit to improve stability when seated and function of the upper extremities (primarily for those ages 4 through 10 years during the growth period) up to the point when surgery (growing rods or fusion) is indicated [3, 28]. Bracing is rarely indicated in ambulatory patients because of the possible loss of walking ability due to limited trunk mobility within the device which interferes with the biomechanics of movement [7].
Lifetime probability of scoliosis surgery differs between SMA types as well as acquired motor milestones and their loss with age at scoliosis surgery [10]. Surgical stabilization is the most effective and permanent method of choice to treat scoliosis. Growing rod instrumentation is considered for younger skeletally immature patients to allow continued spinal growth while stabilizing and improving spinal deformity. Posterior spinal fusions are implemented in nearly skeletally mature patients [11], although recent results suggest routine formal fusion is unnecessary for growing rod graduates with SMA [29]. The aim of spinal stabilization in children with SMA types 2 and 3 is not solely for preservation of pulmonary function, but can also include a variety of additional benefits. These may include improved sitting balance, use of arms for activities of daily living, improved pelvic obliquity, improved ease of transfers, decreased caregiver burden, increased comfort, reduced pain, and improved cosmesis and self-esteem [19, 30–32].
Spinal surgery at an earlier age tends to have better results because the curve is less and there is greater flexibility of the spine. These attributes allow for better correction to be achieved at the time of surgery [7]. We now anticipate newer, milder SMA phenotypes with the advent of disease modifying therapies [33]. The clinical course of SMA is now altered with treatments. Any therapeutic response will depend on timing of drug administration and the magnitude of preexisting motor neuron degeneration [33]. Newborn screening for SMA encourages early treatment [34] and will potentially change our present knowledge of primary musculoskeletal issues including scoliosis progression and chest wall development. Early spinal surgery may adversely affect such a patient’s ability to gain new motor milestones and functional skills by limiting spinal flexibility and truncal compensatory mechanisms. Spine flexibility, upper extremity use, and positioning due to enhanced contractures are all carefully connected and interdependent. Flexible spines and weakness allow trunk collapse to increase the forward reach of the arms [14]. Maintaining flexibility of the spine and extremities allows a patient’s center of gravity to be placed at the greatest mechanical advantage for weak muscles to function (e.g. Gowers sign) [13]. Spinal fixation creates a longer leaver arm making weak muscles more difficult to control and requiring additional support [14]. In addition, ambulatory SMA type 3 patients need spinal mobility in order to walk, and scoliosis surgery would likely lead to the loss of ambulation. Our one ambulatory patient (10.7 year old female, SMA type 3) was able to take only several steps pre-operatively and was otherwise wheelchair dependent. Clinically, surgery was indicated as her spinal curvature progressed to 44 degrees wearing a brace and 76 degrees out of the brace as well as requiring increased support with stepping. Post-operatively she lost the ability to take steps, however, given she was a marginal walker pre-operatively, natural history progression would lead us to believe she would have lost this ability not long after, regardless of the spinal surgery. These risks should be discussed in the timing of interventions as well as anticipated development of function with the available treatment methods as historically [14] and in this study, surgical patients never returned to their pre-operative skill levels.
Significant improvements in quality of life domains of sitting related items, bodily pain, and social functioning was reported post-operatively in patients with progressive neuromuscular scoliosis [35, 36]. One important limitation of this study includes not asking patients or caregivers if the improvements seen post-surgery outweigh the adverse effects. Future studies should explore the impact of these functional changes on quality of life and their clinically meaningfulness prospectively in a larger cohort. Future studies should also assess the impact of disease modifying therapies seen on function post-operatively. Patient registries should include scoliosis development and progression as an important data collection opportunity.
This cohort was a small, mixed sample size of SMA types 2 and 3 with no control group. Due to clinical care, the timing of outcome measures performed were variable as well as mixed surgical techniques (VEPTR growing rods and spinal fusion) were performed in these participants. Upper limb function and pulmonary function (forced vital capacity, forced expiratory volume, and episodes of respiratory infections) were not captured in this data set. Future studies should assess the impact of spinal surgery on arm and pulmonary function post-operatively.
In conclusion, SMA patients experience a change in function post-operatively. Rehabilitation evaluations of gross motor activities and muscle weakness can be important in predicting functional loss after spinal surgery. Flexibility of the spine is functionally advantageous in SMA to compensate for proximal muscle weakness. Surgical stabilization compromises function and must be weighed against the long-term functional loss from severe scoliosis. Patients and families should be educated on the anticipated changes in function that follow after spinal surgery. These concerns must be weighed thoughtfully when considering the timing of scoliosis surgery in the modern era now that SMA is a “treatable” disease.
Conflicts of interest
S. D. Y. has been a member of advisory boards for Biogen, Roche/Genentech, and Scholar Rock; received personal compensation for activities with Biogen, Cure SMA, and Scholar Rock as a consultant; and received research support from the SMA Foundation.
J. M. has been a member of advisory boards for Astellas, Biogen, Cytokinetics, Roche, and Scholar Rock; consultant to Biogen and Ionis Pharmaceuticals, Inc.; and received research support from the Muscular Dystrophy Association and the Eunice Kennedy Shriver National Institute of Child Health and Human Development/National Institutes of Health.
R. S. has been a member of advisory boards for AveXis, Biogen, PTC Therapeutics and Sarepta Therapeutics; received personal compensation for activities with Biogen as a consultant; and received research support from the SMA Foundation.
A. M. G. received travel/lodging and compensation from Biogen to serve on an advisory board; licensing fees from Children’s Hospital of Philadelphia for development of the CHOP INTEND motor scale; institutional support from AveXis, Biogen, and Roche for trial training; and personal compensation from ATOM International and Mallinckrodt for trial training.
A. P. has received research support from the SMA Foundation; serves as a consultant for Biogen; and has served on advisory boards for AveXis, Biogen, Roche, and Scholar Rock.
E. C. has no disclosures to report.
W. M. has no disclosures to report.
R. S. F. reports grants and advisor fees from Biogen and Ionis Pharmaceuticals, Inc. during ENDEAR and CHERISH and from Biogen during NURTURE and SHINE; grants from Cytokinetics; outside the submitted work: advisor and grants from AveXis and Roche, and advisor to Novartis; advisory capacity to nonprofit organizations: Cure SMA, SMA Europe, the SMA Foundation, and SMA Reach (UK); data safety monitoring board for the AveXis AVXS-101 phase I gene transfer study and the Roche Moonfish phase IIb study; personal compensation for serving as coeditor of Swaiman’s Pediatric Neurology text (2018); licensing fees from Children’s Hospital of Philadelphia for development of the CHOP INTEND motor scale; and grants from AveXis, Cytokinetics, Ionis Pharmaceuticals, Inc./Biogen, and Roche.
B. T. D. has been a member of advisory boards for AveXis, Biogen, Cytokinetics, Dynacure, PTC, Roche, and Sarepta; received research support from Cure SMA, the National Institutes of Health/National Institute of Neurological Disorders and Stroke, the Slaney Family Fund for SMA, the SMA Foundation, and the Working on Walking Fund; and received grants from Biogen and Ionis Pharmaceuticals, Inc. during the ENDEAR, CHERISH, CS2, CS12, and CS11 studies, and from Cytokinetics, Fibrogen, PTC, Roche, Santhera, Sarepta, and Summit, and reports no personal financial interests in these companies.
D. C. D. has been an advisor/consultant for AveXis, Biogen, Cytokinetics, Ionis Pharmaceuticals, Inc., Metafora, Roche, Sanofi, Sarepta, and the SMA Foundation, with no financial interests in these companies; received grants from the Department of Defense, Hope for Children Research Foundation, the National Institutes of Health, and the SMA Foundation; and received clinical trial funding from Biogen, Mallinckrodt, PTC, Sarepta, Scholar Rock and Ultragenyx.
Footnotes
Acknowledgments
This work was supported by the SMA Foundation and had no involvement in the preparation of the article, study design, collection, analysis and interpretation of data, or in the writing of the report. We gratefully acknowledge the families and children who participated in this study.
