Abstract
Background
Friedreich’s ataxia (FA) is an autosomal-recessive neurodegenerative disease characterised by neurologic, cardiac and endocrine abnormalities. Currently, Friedreich cardiomyopathy (FA-CM) staging is based on early ECG findings, high sensitivity troponin (hsTNT) ≥14 ng/ml and echocardiographic left ventricular (LV) morphologic and functional evaluation. However, further parameters, accessible only by cardiovascular magnetic resonance (CMR), such as myocardial oedema, perfusion defects, replacement and/or diffuse myocardial fibrosis, may have a place in the staging of FA-CA. Our aim was to elucidate the additive value of CMR in FA-CM.
Methods
Three FA cases were assessed using ECG, 24 h Holter recording, hsTNT, routine ECHO including wall dimension, valvular and ventricular function evaluation and CMR using 1.5T Ingenia system. Ventricular volumes-function, wall dimensions and fibrosis imaging using late gadolinium enhancement (LGE) was performed.
Results
All FA patients had non-specific ECG changes, almost normal 24 h Holter recording, mild hypertrophy with normal function assessed by echocardiography and increased hsTNT. However, the CMR evaluation revealed the presence of LGE >5% of LV mass, indicative of severe fibrosis. Therefore, the FA patients were re-categorized as having severe FA-CA, although their LVEF remained normal.
Conclusion
The combination of classical diagnostic indices and CMR may reveal early asymptomatic FA-CM and motivate the early initiation of cardiac treatment. Furthermore, these indices can be also used to validate specific treatment targets in FA, potentially useful in the prevention of FA-CM.
LIST OF ABBREVIATIONS
Friedrich Ataxia
Cardiovascular magnetic resonance
Late gadolinium enhancement
Angiotensin converting enzyme inhibitors
Angiotensin receptor blocker
Electrocardiogram
Left ventricular ejection fraction
Extracellular volume fraction
Steady-state free-precession sequence 3D–T1-TFE sequence,
Echo time
Repetition time
Inversion recovery time
BACKGROUND
Friedreich ataxia (FRDA), a multisystem autosomal recessive condition, affects approximately 1 in 29,000 individuals, with equal frequency in males and females, while a carrier frequency is 1:60 to 1:120 [1]. The hallmarks of FRDA include progressive sensory and cerebellar ataxia, dysarthria, impaired vibratory sense and proprioception, absent tendon reflexes in lower limbs, pyramidal weakness, scoliosis, foot deformity and cardiomyopathy [1–5]. Despite significant progress in disease modifying agents, the progressive nature of FRDA continues to have a serious impact on the life of these people. At present, there is no proven treatment to slow the progression of this disease. Thirty-nine expert clinicians critically appraised the published evidence related to FRDA care and provided a concise document with recommendations, based on expert consensus [1].
Progression of the neurological syndrome is well characterized using the ICARS score (International Consensus Ataxia Rating Scale) [6]. In contrast, Friedreich cardiomyopathy (FA-CM) is not well characterized, although recently, an algorithm for staging has been proposed [7]. This is based on echocardiographic (ECHO) assessment of left ventricular (LV). However, detailed characterization myocardial tissue involvement that can potentially contribute to the evolution of heart disease cannot be assessed by echocardiography [8]. Cardiovascular magnetic resonance imaging (CMR), a non-invasive, non-radiating, operator independent modality, providing both function and tissue characterisation, has been successfully used in other non-ischemic cardiomyopathies [9] and may have a place in the evaluation of FA-CM.
To define FA-CM the following parameters were used [10]: Left ventricular ejection fraction (LVEF) < 55% (for global LV function); Left ventricular posterior wall thickness (LVPWT) ≥11 mm (for hypertrophy); Myocardial late gadolinium enhancement (LGE) positivity, indicative of myocardial replacement fibrosis; High sensitivity troponin (hsTNT) ≥14 ng/ml (for myocyte damage) and, T-wave-inversion (for electrocardiographic (ECG) repolarization abnormalities).
If all these parameters are normal, then by definition there is no cardiomyopathy. To define end-stage cardiomyopathy a global LV function with LVEF <55% was used based on previously reported studies [10, 11). Between these extreme variants, identification of LV hypertrophy (LVPWT ≥11 mm) was used to distinguish mild (=no hypertrophy) from intermediate cardiomyopathy (=with hypertrophy). In addition, significant LGE positivity is also used to define the presence of severe cardiomyopathy because it signifies an irreversible finding, while “pure” LV hypertrophy may regress after adequate treatment 10, 11]. Of importance, LGE positivity can be found even in the absence of severe LV hypertrophy [12].
In this study, we aimed to present our clinical experience in three FA patients, using ECG, 24 h Holter recording, ECHO, hsTNT and CMR, and discuss the potential role of CMR in the assessment of FA-CA.
METHODS
Three FA cases were assessed using ECG, 24 h Holter recording, hsTNT and routine ECHO including wall dimension, valvular and ventricular function evaluation. Then FA patients were referred for CMR that was performed using 1.5T Ingenia system. Ventricular volumes-function, wall dimensions and fibrosis imaging using LGE was performed.
Ventricular volumes and ejection fraction assessment was performed using SSFP sequence. LGE images were acquired using inversion recovery sequences. Quantification was performed according to previously described protocols [13] using the Philips processing system
RESULTS
Case 1
A twelve-year old boy with genetically documented FA was evaluated. His ECG showed T wave inversion in II, III, and AVF. Echocardiography revealed LVEF = 65%, intraventricular septum thickness /lateral wall thickness (IVS/LAT) = 12/12 mm. The CMR evaluation revealed left ventricular end-diastolic volume (LVEDV) = 85 ml, left ventricular end-systolic volume (LVESV) = 30 ml, left ventricular ejection fraction (LVEF) = 62%, left ventricular mass index (LVMI) = 120 g/m2, (lower = 64, mean = 87, upper value = 110 g/m2) [14] right ventricular end-diastolic volume (RVEDV) = 47.5 ml, right ventricular end-systolic volume (RVESV) = 12.5 ml, right ventricular ejection fraction (RVEF) = 73%, IVS/LAT = 14.5/8, late gadolinium enhanced area (LGE) = 6% of LV mass.
Case 2
A twelve-year old girl with genetically documented FA was evaluated. Her ECG showed T wave inversion in II, III, AVF and V4-V6. Her echocardiography showed LVEF = 61%, IVS = 13 mm, LAT = 11.4 mm, a normal RV, and no evidence of valvular disease. Her CMR evaluation revealed: left ventricular end-diastolic volume (LVEDV) = 87 ml, left ventricular end-systolic volume (LVESV) = 31 ml, left ventricular ejection fraction (LVEF) = 63%, LVMI = 103 gr/m2 (lower = 56, median = 79, upper normal = 101 g/m2) [14], right ventricular end-diastolic volume (RVEDV) = 110 ml, right ventricular end-systolic volume (RVESV) = 41 ml, right ventricular ejection fraction (RVEF) = 62%, IVS/LAT = 16/11, LGE area = 8% of LV mass.
Case 3
A twelve-year old girl with genetically documented FA was evaluated. Her ECG showed T wave inversion in II, III, AVF and V4-V6 (Fig. 1). Her echocardiography showed a LVEF = 67%, IVS/ LAT = 15/18, normal RV, no evidence of valvular disease). Her CMR evaluation revealed LVEDV = 76 ml, LVESV = 19 ml, LVEF = 75%, LVMI = 105 g/m2 ((lower = 56, median = 79, upper normal = 101 g/m2) [14], RVEDV = 76 ml, RVESV = 20 ml, RVEF = 56 %, IVS/LAT = 16/19, LGE area = 7% of LV mass (Fig. 2).
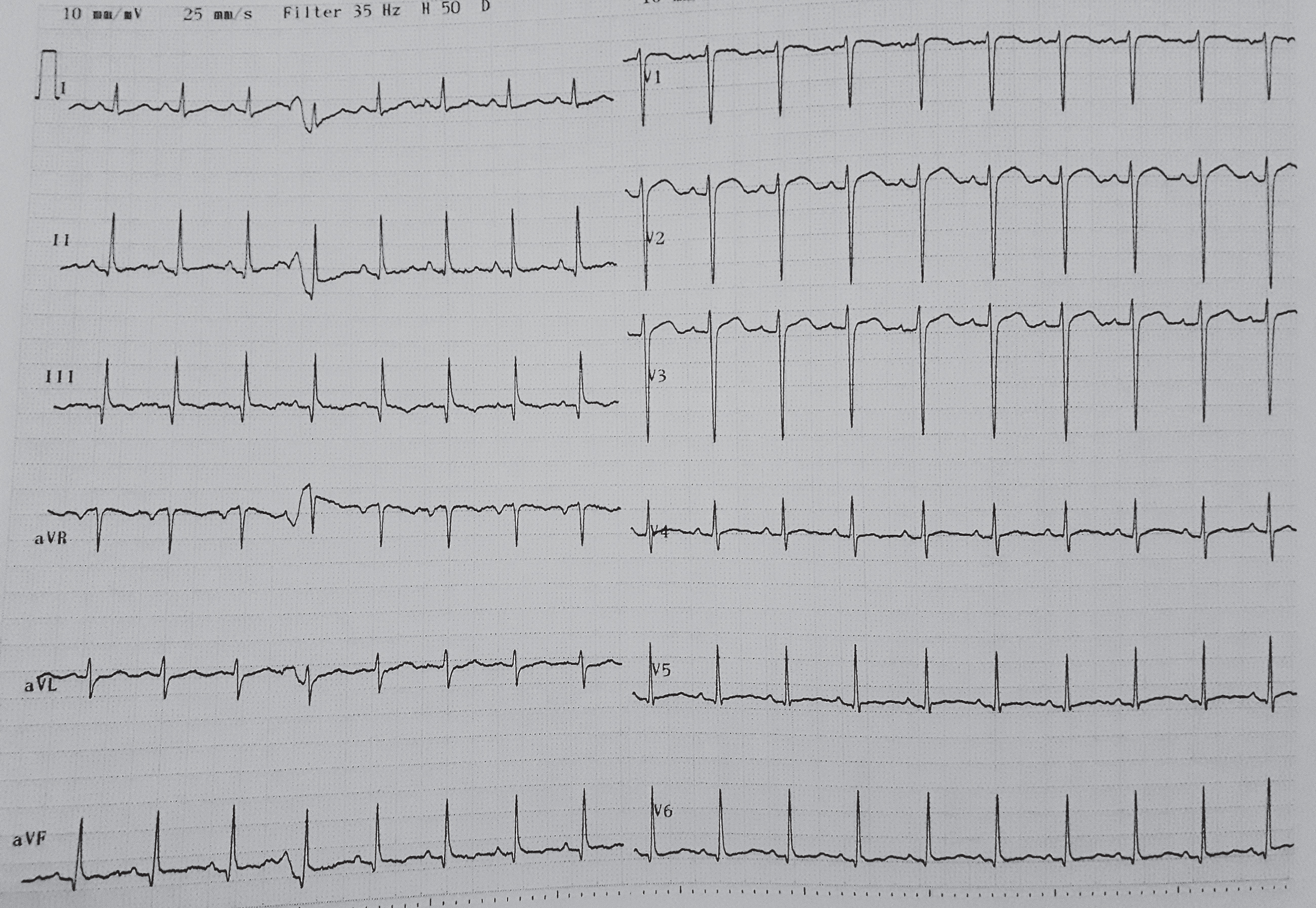
T wave abnormalities in a patient with FA-CM.
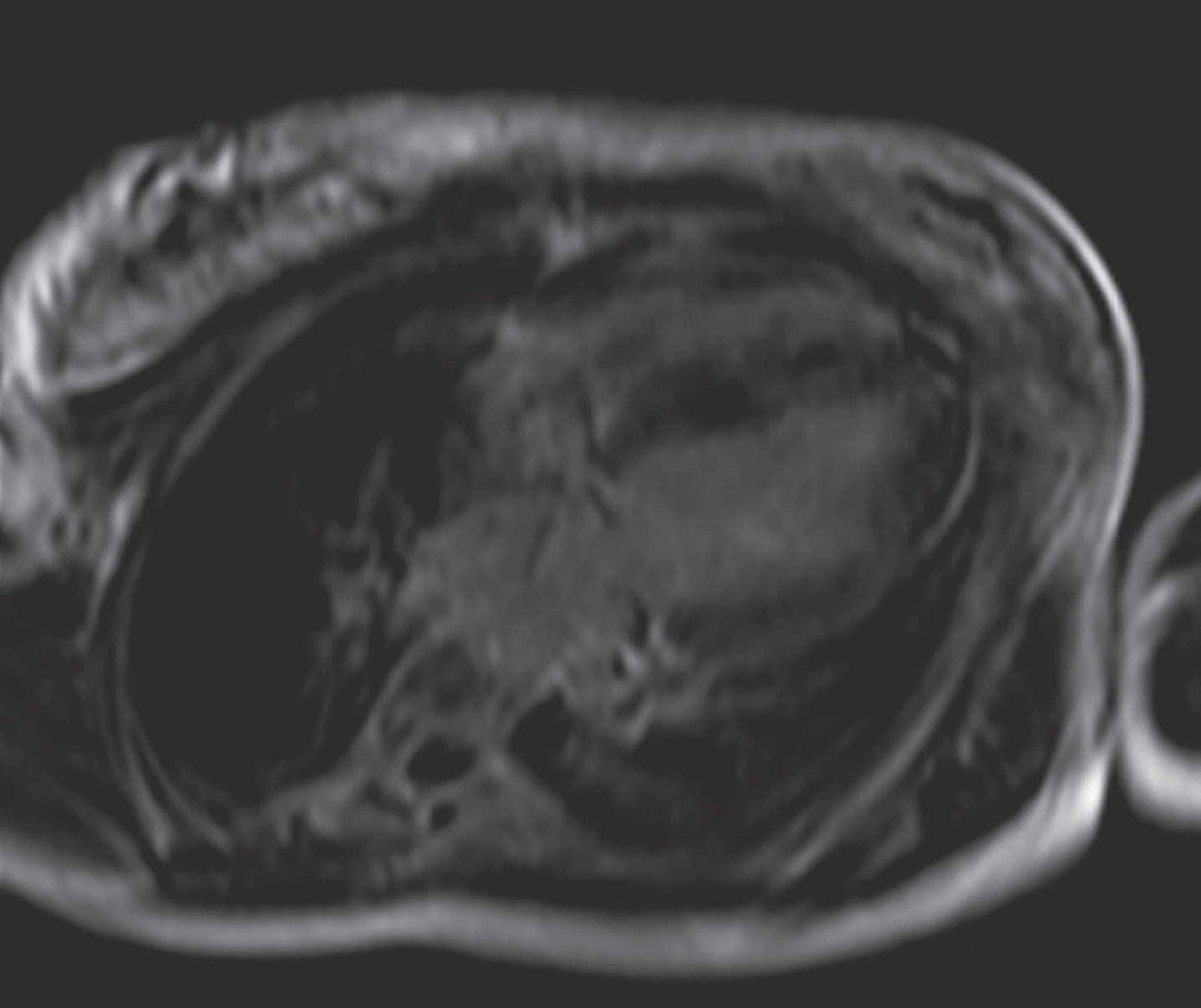
Late gadolinium enhancement in a patient with FA-CM and mild hypertrophy.
All our patients had increased hsTNT 2-5 ng/ml (normal values <0.02), but their 24 h Holter recording showed only rare monomorphic, ventricular beats. The detection of LGE motivated the early start of ACE-inhibitors and b-blockers according to ESC/AHA guidelines, even if the LVEF is normal [15].
DISCUSSION
In this case series, we present the non-invasive cardiac evaluation of three pediatric patients with FA. In our cases, although ECG, 24 h Holter recording and echocardiographic evaluation categorized the patients as having mild cardiac disease, the values of hsTNT suggested a more severe cardiac involvement. CMR further reinforced the diagnosis of severe cardiac disease by assessing more severe LV hypertrophy with concurrent evidence of LGE >5% of LV mass and therefore, the patients were categorized as having severe FA-CA, although the LVEF remained normal [16].
The current evaluation of FA patients includes resting ECG to detect repolarization abnormalities, 24-hour Holter recording to detect arrhythmias, transthoracic echocardiography for morphologic and functional assessment, speckle-tracking imaging for early regional deformation, CMR for oedema, perfusion, fibrosis detection, 24-hour blood pressure recording and blood test for hsTNT [12].
Subtle ECG changes could be among the earliest findings of FA-CM and include non-specific T-wave abnormalities in the lateral leads; however, these findings are not diagnostic of FA-CM and does not offer any predictive value [11]. Furthermore, although 24 h Holter can reliably assess supraventricular and ventricular tachycardia, no individual ECG parameter has been proven adequate to stratify patients with regard to risk for sudden cardiac death, due to ventricular tachycardia /ventricular fibrillation (VT/VF) [10, 11]. Additionally, the diagnostic efficacy of 24 h Holter recording is high only in patients exhibiting daily symptoms, but significantly lower compared with the newer continuous recording devices [17, 18]. Furthermore, it has become clear that Lown’s grading of premature ventricular contractions (PVCs) into six categories, despite initially having been proposed as a prognostic classification, is of value only in patients with acute myocardial infarction and of little prognostic relevance in other cardiac disorders [19, 20].
Although ECHO is the cornerstone for assessment of both systolic and diastolic function in cardiomyopathies, it has the disadvantage of being operator- and acoustic window-dependent and unable to perform tissue characterization [21].
Increased hsTNT can provide important information in acute coronary syndromes, stable coronary artery disease, myocarditis and heart failure; however, increased sensitivity comes at a cost of decreased specificity [22]. However, advanced stages of FA-CA can be characterized by elevated hsTNT in conjunction with replacement fibrosis leading to apparent improvement of hypertrophy, but concomitant reduction of global myocardial function, and electrical instability [16].
CMR has been recently applied in the evaluation of FA-CM, because it provides an accurate assessment of cardiac hypertrophy, myocardial perfusion [23] and fibrosis [24–26], which is an early manifestation of FA-CM, even in the absence of significant hypertrophy [27, 28]. CMR, using cine sequences, can measure ventricular volumes and ejection fraction without contrast agent, while it provides 3-dimensional images of the heart, also feasible with 3D echocardiography [24]. While ECHO remains the every day, bedside, inexpensive, widely available technique, CMR is ideal for the serial evaluation of volumes, mass and ejection fraction, due to its high reproducibility and operator independency, especially for the RV parameters that cannot be always reliably assessed by echocardiography [24]. Furthermore, it is the most reliable imaging technique to detect and quantify fibrotic tissue (viability study). Fibrotic tissue retains the paramagnetic contrast agent gadolinium and, therefore, appears bright [25]. Both the presence and the extent of LGE play an important role in patient prognosis, because even a small area of LGE (<2% of LV mass) was associated with >7-fold increase in risk for a major adverse cardiac event in patients with coronary artery disease [26]. A recent CMR assessment of patients with non-ischemic cardiomyopathy documented that LGE was an independent predictor of malignant arrhythmias, while its amount and distribution did not provide additional prognostic value; it may therefore help identifying those patients for ICD therapy that do not fulfil the current criteria based onLVEF [27].
Myocardial inflammation (myocarditis) can be present as a precursor of FA-CM and may remain subclinical, i.e. undetectable by blood inflammatory indices. This subclinical inflammation may then progress to clinically overt dilated cardiomyopathy, as documented from autopsy data of patients with FA-CM [28]. Progressive cardiomyopathy in FA is the end-point of iron-catalyzed mitochondrial damage, followed by muscle fiber necrosis and chronic reactive myocarditis [29]. CMR may help detect myocarditis using Lake Louise criteria [30]. Myocarditis, missed by other non-invasive techniques, has been identified in our FA patients using CMR (unpublished data), which may have important implications for early patient risk stratification [31, 32]. However, prospecitve detailed data will be needed to address this point further.
Abnormal myocardial perfusion reserve /fibrosis also represent early manifestations of FA-CM that are detectable by CMR [33] and have been identified in FA patients with normal epicardial coronaries in FA patients supporting FA-CM [33].
Finally, CMR has the capability to characterize myocardial tissue composition using T1 and T2 mapping techniques and extracellular volume (ECV). These indices are considered as a new, reliable tool to diagnose diffuse myocardial involvement in various cardiomyopathies [34]. These findings support the considerable potential of CMR with its excellent reproducibility to offer information beyond morphology and function that are unavailable by other modalities. It has thus also the potential to be used as a biomarker for treatment evaluation in clinical trials [35]. This is of particular importance given that interventional trials including gene replacement are imminent. At the moment only LGE is acknowledged in the evaluation of FA-CM so that further studies are needed to validate the complete repertoire of CMR indices in FA-CM. This additive information may also be relevant for the classification of FA-CM in the context of the MOGE(S)-classification [36].
In summary, our three cases were characterised by non-specific ECG findings, normal 24 h Holter recording and mild to moderate LV hypertrophy with normal LVEF, assessed by ECHO. The increased hsTNT suggested the presence of FA-CM. The utilization of CMR identified a more advanced staging of FA-CA, compared with that suggested by ECHO. While ours is a still preliminary observation without long term follow up, it suggests that the careful use of CMR indices warrants further evaluation as potentially useful tool for early recognition and hence proactive treatment of FA-CM, stratification of the disease, as well as a biomarker of cardiac involvement for clinical trials in FA.
DECLARATION
Ethics approval and consent to participate.
Patient data have been performed in accordance with the Declaration of Helsinki and have been approved by the Onassis Cardiac Surgery Center ethics committee.
Consent for publication.
Written Informed consent to participate in the study was obtained from parents or legal guardian of all examined patients.
Availability of data and material.
Data are available upon request.
COMPETING INTERESTS
MN and SM are members of the Editorial Board of BMC Cardiovascular Disorders. MN has received grants by the Deutsche Forschungsgemeinschaft (DFG) through the Sonderforschungsbereich Transregio 19 “Inflammatory Cardiomyopathy” (SFB TR19) (TP B2), and by the University Hospital Giessen and Marburg Foundation Grant “T cell functionality” (UKGM 10/2009). MN has been consultant to the IKDT (Institute for Cardiac Diagnosis and Therapy GmbH, Berlin) 2004–2008, and has received honoraria for presentations and/or participated in advisory boards from AstraZeneca, Bayer, Fresenius, Miltenyi Biotech, Novartis, Pfizer and Zoll.
FUNDING
Not applicable.
DISCLOSURE
There is no disclosure for any of the authors.
AUTHORS’ CONTRIBUTIONS
SM: Patients scanning evaluation and manuscript writing AG: Cardiac data assessment and writing AP: Neurologic data assessment and writing RP: Neurologic data assessment and writing MK: Neurologic data assessment and writing EK: Cardiac data assessment and writing MN: Manuscript writing GK: Coordination of CMR assessment, Manuscript writing GP: Coordination of paediatric cardiac assessment, Manuscript writing GC: Manuscript writing coordination.
CONFLICT OF INTEREST
There is no conflict of interest for any of the authors.
Footnotes
ACKNOWLEDGMENTS
Not applicable.
