Abstract
Background:
Treatment decisions of neuromuscular diseases involve weighing clinical benefits and risks, as well as impact on patient social life, work status, other activities of daily living, and health-related quality of life.
Objective:
To conduct a systemic literature review of patient preferences for treatments of neuromuscular diseases.
Methods:
We searched Embase, Web of Science, and PubMed for full-text articles reporting results from studies of patient preferences for treatments of neuromuscular diseases. We excluded articles published before the year 2000, articles written in a language other than English, articles only reporting proxy-assessments of patient preferences, and studies reporting results for a sample comprising <5 patients.
Results:
The search resulted in the identification of 305 unique publications. Of these, 275 were excluded following title and abstract screening and 23 following full-text review. Seven articles were included for data synthesis. Preference data were identified for a hypothetical treatment with pulmonary benefits of Duchenne muscular dystrophy and Becker muscular dystrophy, pathways for different routes of opioid drug administration in motor neuron disease, wheelchair features in amyotrophic lateral sclerosis (ALS), ankle foot orthoses in patients with Charcot Marie Tooth disease, and mechanical ventilation in ALS and a mixed cohort of patients with neuromuscular diseases.
Conclusions:
Despite considerable research into the development of new health technologies targeting neuromuscular diseases, little is known of patients’ preferences for pharmacological interventions. More research is needed to help incorporate patient preferences in clinical decision-making to improve treatment satisfaction, medication compliance, and health outcomes.
Keywords
INTRODUCTION
In the medical management of most illnesses, therapeutic decisions involve trade-offs between expected benefits and risks [1]. For the patient, in addition to efficacy and safety, other less tangible aspects of available treatment options may be of importance, including impact on social life, work status, and other activities of daily living [2]. For this reason, shared decision-making – in which doctor and patient exchange information and preferences and reach a decision together – has been advocated as the gold standard model to help healthcare practitioners discriminate between marketed therapies and potentially enhance overall care satisfaction, increase medication compliance, improve health outcomes, and promote patient quality of life [3–5]. Knowledge of patient preferences at the population level may also inform patient-centered research and development of future health technologies, endpoints in clinical trials of novel therapies, clinical practice guidelines, and value-based resource allocation.
There are in broad terms two categories of approaches to preference-elicitation: (i) stated/revealed preference methods, grounded in consumer theory and the psychology of choice [6], and (ii) methods for the estimation of utilities for health states, consistent with the axioms of rational behavior and expected utility theory [7]. Stated preference methods typically involve collecting choice data (for e.g. different hypothetical treatments, usually as a discrete choice) or ranking of treatments or treatment attributes directly from respondents using surveys, in contrast to revealed preference methods that utilize empirical data of observed behavior. The second category of approaches comprises methods for the estimation of utilities representing preferences for health states, e.g. the standard gamble method [8], and are also based on data recorded directly from respondents. The main difference between stated/revealed preference methods and methods for the estimation of utilities for health states concern the analysis output, where the former accommodates estimation of e.g. the ranking and relative importance of treatment characteristics. The latter, on the other hand, produce preferences in terms of utilities representing quality of life (usually bounded between 0 = death and 1 = perfect health) that can be used to calculate quality-adjusted life years (QALYs) as applied in economic evaluations [6].
Neuromuscular disease is an umbrella term encompassing diseases, conditions, and disorders impairing the functioning of the muscles through the peripheral nervous system. Common manifestations include muscle weakness and antrophy, fasciculation, spasm and myokymia, and myalgia, and many neuromuscular diseases are debilitating and life-threating, accompanied by extensive morbidity and substantial healthcare needs. In addition, a large proportion of neuromuscular diseases are of genetic origin, with onset of symptoms in early childhood, e.g. Duchenne muscular dystrophy (DMD), spinal muscular atrophy (SMA), and congenital muscular dystrophy.
The aim of this study was to review the literature of patient preferences for treatments of neuromuscular diseases. Specifically, this systematic literature review sought to answer the following questions: For which neuromuscular diseases have patient preferences been elicited? For which neuromuscular treatments have patient preferences been elicited? What methods have been used for preference elicitation? What is known of patients’ preferences of treatments for neuromuscular diseases?
METHODS
This systematic literature review was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement [9].
Search strategy
We searched Embase, Web of Science, and PubMed for full-text articles reporting results from studies of patient preferences for treatments of neuromuscular diseases. The search string contained a combination of the Medical Subject Heading terms “Neuromuscular Diseases” and “Patient Preference”, the topic field tags “Neuromuscular Diseases”, “Neuromuscular Disease”, and preference*, as well as 94 topic field tags for specific diseases, conditions, and disorders (available as supplemental material online). In addition, for the purpose of this review, we also excluded (i) articles published before the year 2000 (to ensure that patient preferences reflect current treatment practices and health technologies), (ii) review and editorial articles, (iii) articles written in a language other than English, (iv) articles only reporting proxy-assessments of patient preferences, and (v) studies reporting results for a sample comprising <5 patients with a neuromuscular disease (to allow for meaningful inference). For studies including patients with different indications, we also required that preference data were reported separately for patients with neuromuscular disease. Due to the expected limited body of literature, we chose to include studies reporting patient preferences for medical devices and aids, such as mechanical ventilation, in addition to pharmacological interventions. However, we did not include studies that only assessed treatment satisfaction without analysing different treatments and/or treatment characteristics.
Screening, data extraction, and synthesis of results
The search was performed August 17 through August 18, 2017. Two independent investigators (EL and JE) initially screened article titles and abstracts for eligibility, and subsequently reviewed full-text versions of selected records. For all articles included in the review, the following data were extracted: Author, year of publication, title, indication, setting, sample, method for preference elicitation, and results. The reasons for article exclusion were recorded and potential disagreements were specified to be resolved by consensus or, if necessary, the involvement of a third investigator (HL). Result data from each article was synthesized and reported with respect to the four review questions (as stated in the Introduction).
RESULTS
The systematic literature review resulted in the identification of 457 publications (Fig. 1). Of these, 152 were duplicates, 275 records were excluded following title and abstract screening, and 30 articles were selected for full-text review. Finally, seven articles [10–16] were considered for data synthesis. Table 1 presents summary data of the included publications.
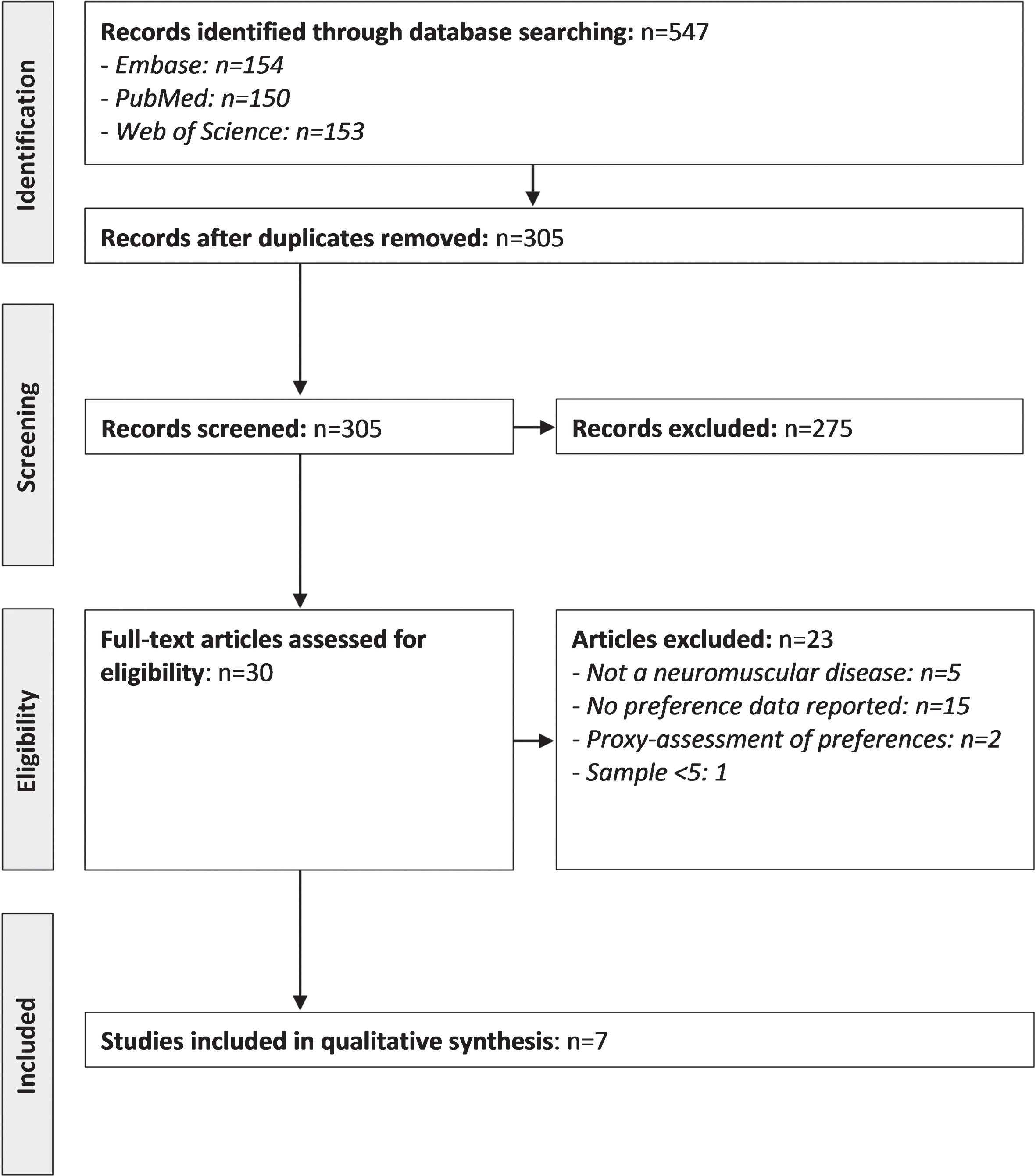
PRISMA diagram of the selection process of the included publications.
Summary data of included publications
Note: Duchenne muscular dystrophy (DMD). Becker muscular dystrophy (BMD). Amyotrophic lateral sclerosis (ALS). Non-invasive positive pressure ventilation (NPPV). Invasive ventilation by tracheostomy (TPPV). Ankle foot orthoses (AFOs). Visual analogue scale (VAS).
For which neuromuscular diseases have patient preferences been elicited?
In total, two articles reported preference data for patients with ALS [14, 16], two for motor neuron disease [13, 15], two for DMD and Becker muscular dystrophy (BMD) [10], one for Charcot Marie Tooth disease [12], and one for a cohort of patients with neuromuscular diseases (e.g. DMD, BMD, myotonic dystrophy, and spinal muscular atrophy, although results were not stratified by indication) [11]. In other words, out of the 94 specific diseases, disorders, and conditions included in this systematic literature review, patient preference data was identified for only 5% (5/94).
For which neuromuscular treatments have patient preferences been elicited?
Of the seven included articles, two reported patient preference data for a pharmacological intervention (one study of opioid treatment of breathlessness in patients with motor neuron disease [15] and one of a hypothetical treatment with pulmonary benefits in patients with DMD and BMD [10]. Of the remaining publications, three reported preferences for mechanical ventilation [11, 14], one for wheelchairs [16], and one for ankle foot orthoses (AFOs) [12].
What methods have been used for preference elicitation?
Three studies elicited patient preferences through patient interviews (either as part of routine clinical follow-up/assessment or scheduled separately as part of the study procedure) [12, 15] and four through surveys composing discrete- or multiple-choice questions [11, 16], visual analogue scales (VAS) [11, 16], and best-worst scaling [10].
What is known of patients’ preferences of treatments for neuromuscular diseases?
Overall, data on patient preferences for treatments of neuromuscular diseases is scarce. Evident from Table 1, the current body of evidence is limited to ranking of benefits and risks of a hypothetical treatment of DMD and BMD (e.g. cough strength, risk of lung infection, and risk of diarrhoea) [10], preferences for alternative pathways for opioid drug administration in motor neuron disease (e.g. oral, inhaled, and sublingual) [15], preferences for different types and features of ventilation support (e.g. non-invasive positive pressure ventilation [NPPV] and invasive ventilation by tracheostomy [TPPV], and ventilator interface, humidification and heating, and mouthpiece condition) [11, 14], preferences for wheelchair features in ALS (e.g. comfort, ease of manoeuvring, and portability) [16], and preferences for benefits and disadvantages with AFOs in patients with Charcot Marie Tooth disease [12].
DISCUSSION
The aim of this study was to conduct a systematic literature review of patient preferences for treatments of neuromuscular diseases. In summary, our review and synthesis show that patient preference data is lacking for treatments of neuromuscular diseases in general and for pharmacological interventions in particular. In total, we identified five full-text articles reporting patient preference data for DMD and BMD, ALS, motor neuron disease, and Charcot Marie Tooth disease, respectively, and one for a mixed cohort of patients with neuromuscular disease. Thus, for most neuromuscular diseases, disorders, and conditions, preference data are not known at the population level. Moreover, with the exception of the study by Simon et al. [15] (which reports preference data for different routes of opioid applications), the identified study publications estimated preferences for either a hypothetic treatment [10] or medical devices [11–14, 16, 11–14, 16]. Thus, preference data is particularly lacking with respect to drugs included in the standard of care treatment algorithm of neuromuscular diseases (e.g. glucocorticoids in the medical management of DMD, antisense oligonucleotide treatment in SMA, enzyme replacement therapy in Pompe disease, and anti-myotonic mediation in myotonic dystrophy type 1). These results emphasize that more research is needed to help align therapeutic decisions and clinical practice guidelines with patients’ preferences to help improve treatment satisfaction, compliance, and health outcomes, and inform development of new health technologies of neuromuscular diseases.
The studies included in our review were all based on methods of stated preference (as opposed to revealed preferences derived from observed behaviour). Specifically, Hollin et al. [10] collected preference data using best-worst scaling and subsequently predicted utility scores using mixed logistic regression. In the other studies, the analysis of interview and survey data was more descriptive in nature, involving rankings of attributes, summary of outcomes of discrete- or multiple-choice questions, or assessment of patient preferences using VAS. For example, Simon et al. [15] conducted face-to-face interviews with patients and asked them to rank different administration routes of opioids illustrated using photographs on laminated cards, Rabkin et al. [14] asked patient with ALS a discrete-choice question with three response options regarding their preference for mechanical ventilation, and Lofaso et al. [11] elicited preferences for specific features of mechanical ventilation using VAS. Although it is challenging to assess the relative validity of these approaches to preference elicitation, given that none of the methods encompass decision-making under uncertainty, they are not consistent with the axioms of rational behaviour and expected utility theory [7]. As a consequence, although representing important contributions to the literature, reported preference data should therefore be interpreted as values rather than utilities, and not be confused with e.g. utilities derived for health states using the standard gamble method [8].
Moreover, with respect to differences in study design and methodology, our synthesis of data also illustrates challenges relating to comparison of results. Although conducted in the same therapeutic area, the included articles investigated different treatments and/or treatment attributes using different approaches. Therefore, as the estimated preferences only are valid relative to the attributes or treatments studied, direct comparisons of results across studies are not straightforward. It should be noted, however, that this issue also is present when comparing studies utilizing identical methodology, for example conjoint analysis (as the different utility values only can be interpreted relative to the other studied attributes and levels), and is thus not an issue specific to preference research of neuromuscular diseases. In addition, challenges with comparison of preference data may also be present when relating studies of the same disease and treatment using identical methodology due to differences in patient characteristics, e.g. disease status and comorbidity profiles [17]. This would be expected to be particularly relevant for progressive diseases such as DMD, as patients’ preferences would be expected to change over time as their muscle strength and functional ability deteriorates.
Good research practice guidelines for the design, implementation, and execution of stated preference studies developed by the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) Preference-based Methods Special Interest Group’s Conjoint Analysis Working Group [6] suggest that the selection of treatment attributes should be guided by evidence of the potential range of preferences and values that the target population may hold and the research question. In other words, it is central that patients’ views are taken into account when designing preference studies. In our systematic review, we found that only two studies [10, 12] directly involved patients in the identification and selection of treatments attributes. Specifically, Hollin et al. utilized a community-engaged approach to elicit feedback from key stakeholders, including patients, caregivers, clinicians, advocacy leadership, and industry representatives, and Philips et al. used the nominal group technique, a qualitative methodology for obtaining priority ranking on a list of items generated by study participants. Of the other studies, Lofaso et al. [11] designed the study questionnaire with input from a scientific committee comprising e.g. clinicians, representatives from patient-support organizations, and representatives from the study sponsor, Rabkin et al. [14] formulated questions “based on previous findings and clinical observations”, Simon et al. [15] adapted questions from a previous study about acceptability of different routes of administration in pain, and Raaphorst et al. [13] and Trail et al. [16] did not disclose the process of treatment attribute selection/survey development. In our opinion, to help assess validity, both internal and external, it is important that future studies seek to fully disclose the processes involved in choosing attributes and levels.
Many neuromuscular diseases present early in life and a non-trivial proportion are also accompanied by some degree of cognitive impairment and/or elevated risks of e.g. autism spectrum disorder and obsessive-compulsive disorder. As a result, as patients may not be fully capable of providing reliable self-assessments of their preferences, it may be appropriate to allow caregivers or other close relatives to make proxy-assessments of their preferences. However, to facilitate interpretation and assessment of generalizability, it would be helpful if researchers disclose if such proxy-assessments reflect caregiver preferences or patients’, and that results are reported separately for these two strata, if possible. Moreover, in agreement with what has been advocated for assessment of patient health-related quality of life in paediatric populations [18, 19], we argue that proxy-reports should be encouraged to obtain valuable, complementary preference data.
In conclusion, despite considerable research into the development of new health technologies targeting neuromuscular diseases, little is known of patients’ preferences for pharmacological interventions. More research is needed to help incorporate patient preferences in clinical decision-making to improve treatment satisfaction, medication compliance, and health outcomes.
FUNDING
None.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
