Abstract
Monoclonal antibodies that target either PD-1 or PD-L1 have recently been approved for treatment of advanced non-small cell lung cancer. These antibodies are immune checkpoint inhibitors which have been shown to exacerbate Myasthenia Gravis (MG) and other autoimmune diseases. While effective in preventing tumor cells from evading immune attack, immune checkpoint inhibitors such as nivolumab, an antibody directed against the programmed cell death protein-1 (PD-1) receptor located on T-cells, may also cause immune dysregulation and could cause or potentiate pre-existing autoimmune conditions. We present a patient with latent ocular MG treated with nivolumab for her stage IV non-small cell lung cancer who developed generalized MG and severe myasthenic crisis. Providers must be aware of the risks inherent to these novel therapies since they can have life-threatening effects.
INTRODUCTION
Myasthenia gravis (MG) is an autoimmune disease in which pathogenic antibodies are most often directed against the acetylcholine receptors (AChR) at the neuromuscular junction, effectively causing a defect in neuromuscular transmission and fluctuating muscle weakness. It is widely known that a number of medications can unmask or exacerbate pre-existing MG, usually by adversely affecting neuromuscular transmission. The field of oncology has recently advanced the use of monoclonal antibodies targeted against immunologic inhibitory pathways in the treatment of various cancers. While effective in augmenting immune surveillance against cancer, this disruption of the immune autoregulatory mechanisms has the potential of unmasking or worsening autoimmune diseases, such as MG. We describe a case of a woman with ocular MG in virtual remission who developed severe generalized disease culminating in myasthenic crisis after receiving anti-programmed death-1 (PD-1) monoclonal antibody immune therapy for cancer.
CASE REPORT
A 68-year-old woman with anti-AChR antibody positive ocular MG presented initially with isolated fluctuating diplopia. She was treated with both pyridostigmine and prism eyeglasses and felt the latter was most helpful. She was eventually weaned off of pyridostigmine and remained essentially asymptomatic for several years. She subsequently developed benign essential blepharospasm and received incobotulinumtoxinA injections periodically for several years, remarkably with no clinical evidence of MG exacerbation, further attesting to disease stability.
In 2012 she presented with hemoptysis and a CT chest revealed two right upper lobe pulmonary masses and lymphadenopathy. A PET scan confirmed widely metastatic disease and a diagnostic biopsy led to the diagnosis of stage IV non-small cell lung cancer. Initial MRI brain was negative for metastasis but when she developed ataxia and dizziness in July 2014, imaging revealed metastases in the right cerebellar hemisphere and right parasagittal parieto-occipital region. She received several cycles of conventional chemotherapy and radiation. However, imaging demonstrated disease progression, so her regimen was modified and she was started on nivolumab in December 2015. She received 5 doses (291 mg) of nivolumab every other week for a total of 1455 mg.
She presented one month later with dysarthria, diplopia, and facial weakness. She had noted progressive difficulty walking that had begun approximately 2 weeks earlier. Neurologic exam revealed severe bilateral ophthalmoparesis with significant restriction of eye movement in all planes (somewhat worse horizontally), decreased upper and lower facial strength bilaterally, reduced palatal elevation, weak tongue movements, and mild bilateral proximal weakness of upper and lower extremities. MRI brain and spinal cord showed stable to minimally decreased size of known metastatic lesions. Slow repetitive nerve stimulation at a frequency of 3 Hz of the right ulnar and spinal accessory nerves showed significant decrements following 1-minute exercise consistent with a defect in the neuromuscular transmission. Maximal post-exercise decrement for the ulnar nerve was 17.5% noted 3 minutes post-exercise, while for the spinal accessory nerve was 11.1% noted 2 minutes after activation. Slow 3 Hz repetitive nerve stimulation following a 10 second burst of exercise failed to show a significant increment, arguing against a presynaptic pathology. Needle EMG of the right upper extremity was normal and showed no evidence for myopathy and no abnormal spontaneous discharges to suggest membrane irritability. The right frontalis muscle showed myopathic motor unit action potentials with early recruitment which was likely a sequela of patient’s known history of botulinum injections. AChR titers were checked at the time and showed binding antibodies were 0.28 nmol/L (normal range <0.25 nmol/L) and AChR modulating antibodies were 34% (normal <32%). Muscle-specific receptor tyrosine kinase (MuSK) titer was undetected.
Treatment with pyridostigmine and prednisone at 60 mg was initiated and the patient was discharged home. Nivolumab was discontinued. She returned to clinic several weeks later with minimal improvement in symptoms and was re-admitted. Formal speech and swallow evaluation documented persistent oropharyngeal weakness resulting in impaired oral motor function. Respiratory status had worsened from initial negative inspiratory force (NIF) and forced vital capacity (FVC) of –40 and 1.7L respectively to a NIF/FVC of –20/1.5L. Plasmapheresis was initiated and she completed 5 exchanges but symptoms remained refractory, culminating in neuromuscular respiratory failure. She did not wish to be intubated, so noninvasive positive pressure ventilation was utilized. This resulted in rupture of a pulmonary bleb, causing pneumomediastinum, pneumoperitoneum, and pneumoretroperitoneum and ultimately leading to sepsis. On hospital day 18, in light of her critical condition and poor prognosis due to her underlying cancer, her family decided to pursue comfort care measures and the patient was transferred to the hospice unit.
Discussion
Myasthenia gravis (MG) is an autoimmune disease characterized by fluctuating, fatigable muscle weakness caused by pathogenic autoantibodies that target the post-synaptic muscle membrane, in most cases the skeletal muscle AChR, impairing neuromuscular transmission. Weakness in MG has a predilection for, and often initially involves muscles controlling extraocular movements and eyelid elevation. Ocular MG is defined as disease limited to extraocular muscles and eyelid elevators without more generalized involvement for a period of greater than two years [1]. These patients have a very low incidence of developing generalized MG. Our patient fulfilled the criteria for ocular MG and had very stable disease without requiring either symptomatic therapy with acetylcholinesterase inhibitors or immunomodulation. She also received serial incobotulinumtoxinA injections for concurrent blepharospasm without evidence of disease exacerbation.
Nivolumab is a fully human monoclonal IgG4 antibody directed against the programmed death-1 (PD-1) receptor and is used for the treatment of cancers such as melanoma, lymphoma and more recently non-small-cell lung cancer [2, 3]. PD-1 is a cell-surface receptor that is expressed on thymocytes, mature T and B cells as well as myeloid cells [4]. It binds to two ligands, programmed death ligand-1 (PD-L1) and programmed death ligand-2 (PD-L2), which are part of the B7 family of cell surface protein ligands [3, 5]. As demonstrated in Fig. 1A, this binding prevents the activation of T-cells and their production of cytokines, effectively inhibiting the function of the immune system [6, 7]. By this mechanism, PD-1 serves as an immune system checkpoint and protects against autoimmunity [8]. Many cancer cells also express PD-L1, which allows the tumor cells to evade an immune attack [2]. Nivolumab binds to the PD-1 receptor and blocks interaction with PD-L1 and PD-L2 (Fig. 1B). By blocking the interaction of the PD-1 receptor with the PD-L1 ligand on tumor cells, nivolumab enhances the ability of the immune system to detect and eliminate tumor cells.
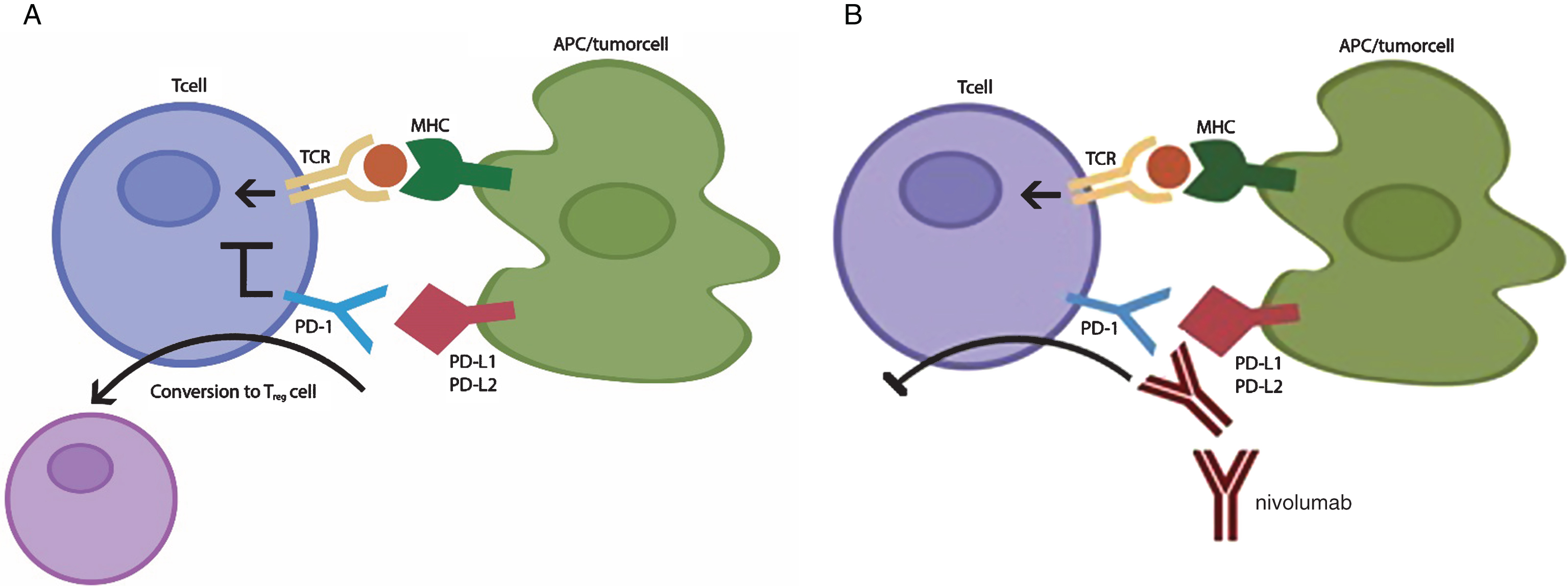
A) Normal function of PD-1 pathway. Binding of PD-1 with PD-L1 or PD-L2 on tumor cell or antigen presenting cell (APC) inhibits activation of T-cell. Binding of PD-1 with PD-L1 on APC induces conversion of T cell to Treg cell. B) Binding of nivolumab with PD-1 receptor on T-cell blocks binding with ligands and activates T-cell. PD-L1 blockade inhibits the induction of Treg cells in the periphery thereby tipping the Th:Treg balance favoring autoimmunity. TCR – T cell receptor; MHC – major histocompatiliby complex; APC – antigen presenting cell; PD-1 – programmed death-1; PD-L1- programmed death ligand-1; PD-L2 – programmed death ligand 2; Treg – regulatory T cell; Th – helper T cell.
Inhibition of the PD-1-PD-L1/PD-L2 pathway may not only enhance the body’s response against tumor cells, but also result in unchecked immune activation targeting host tissue [9]. PD-1 deficient mice develop immune mediated arthritis and glomerulonephritis [10] and cardiomyopathy [11]. Salama et al. have demonstrated in murine models of experimental autoimmune encephalomyelitis that blocking the PD-1 pathway led to more fulminant disease with increase in lymphocytic infiltration in the central nervous system (CNS) [7]. Neurologic complications affecting all levels of the neuroaxis and peripheral nervous system have been reported with the use of these agents: autoimmune meningitis, encephalitis, hypophysitis, polyradiculopathy, neuropathy, neuromuscular junction (NMJ) disease and myositis have been described [8].
There have been several recent reports of MG or myasthenic-like syndromes developing after treatment with immune checkpoint inhibitors. Maeda et al. discussed a patient with pre-existing ocular MG treated with nivolumab who developed generalized MG as well as elevated CK level but his symptoms improved without intervention and despite continuation of nivolumab therapy [9]. A patient with no prior history of MG was treated for non-small cell lung cancer (NSCLC) and developed a myasthenic-like syndrome that similarly improved without intervention [12]. Sciacca et al. presented further evidence of nivolumab triggering MG in a patient with no prior history of a neuromuscular junction dysfunction who, after 3 infusions of nivolumab, developed AChR-antibody-positive MG [13]. While these 3 cases had relatively favorable outcomes, Shirai et al. recently discussed a case similar to our patient in which treatment for melanoma was initiated with nivolumab; subsequently the patient developed MG and died of respiratory complications [14]. This patient was found to have subclinical myasthenia gravis with presence of elevated AChR antibodies in retrospective serum analysis prior to treatment. Similarly Kimura et al. reported a patient with MG crisis after receiving a single dose of nivolumab [15]. Overall, there has been a general trend of worse outcome in patients who had preexisting AChR antibodies, possibly suggesting a role for pre-treatment screening.
Like nivolumab, pembrolizumab is an anti-PD-1 monoclonal antibody approved for the treatment of melanoma. A recent case of pre-existing MG exacerbated by pembrolizumab has been reported, though interestingly the patient had positive antibodies at time of initial diagnosis of MG in her 30 s, and despite worsening of symptoms in her 50 s after pembrolizumab treatment, antibody titers were no longer elevated [16]. At time of submission of this case report, another case of pembrolizumab-induced MG was reported, showing that the patient did not develop AChR antibodies but was found to have elevated serum CK level, which the authors attributed to possible myositis [17]. Like nivolumab and pembrolizumab, ipilimumab is an immune checkpoint inhibitor used in the treatment of solid tumors, though its target is against a different surface molecule called cytotoxic T-lymphocyte associated protein-4 (CTLA-4). There has been one case of MG in a patient treated for small-cell lung cancer with both ipilimumab and nivolumab [18]. There have been at least 3 other cases of patients developing MG after treatment with ipilimumab [19, 20], one of whom developed concurrent myositis, as well as two other patients who developed transverse myelitis and chronic inflammatory demyelinating polyneuropathy from ipilimumab [20]. The increasing number of cases of nivolumab-associated MG calls into question whether the patient presented by Loochtan et al. developed MG as an additive effect of the two chemotherapeutic agents, which target different immune checkpoints [18].
Regulatory T cells (Tregs) protect against autoimmunity by suppressing self-reactive T cells that have eluded thymic selection. As seen in Fig. 1A, the normal binding of PD-1 with PD-L1 induces conversion of T cells to Tregs. Accordingly blockade of PD-L1 expressed by antigen-presenting cells (APCs) impairs production and function of peripherally derived Tregs [21, 22] potentially contributing to disease propagation (Fig. 1B). Dysfunction of Tregs has been described in acquired MG [23]. Aberrant production of follicular helper (Tfh) T cells and subsequent B cell activation are thought to be contributory to autoimmunity in MG. Recently Zhang et al. identified a population of circulating Tfh cells in ocular and generalized myasthenia gravis which expressed CD4+CXCR5-PD-1+, suggesting a role of PD-1 receptor in mediating T cell responses in MG [24].T cell expansion in muscle was also noted by Kimura et al. from a patient treated with nivolumab who developed MG and myositis which was attributed to inhibition of PD-1 binding with PD-L1 [15]. They also studied gene expression levels before and after treatment and found that nivolumab treatment changed the ratio of gene expression, resulting in the differentiation of T cells towards autoreactive cytotoxic T cells rather than regulatory T cells [15].
Nivolumab likely caused immune dysregulation in our patient. While she had a pre-existing diagnosis of seropositive ocular MG, she had been asymptomatic without treatment for many years. As noted, patient data from retrospective studies has suggested that risk of generalization of ocular MG is highest in the first two years [25]. Prevention of the normal immune-checkpoint function may have led to activation of autoreactive T-cells and resulted in disease generalization and severe clinical MG exacerbation. Of interest, anti-AChR antibodies in our case as well as in other reported cases were either weakly elevated or negative [12, 13] suggesting that the T-cell-driven autoreactivity may lead to autoantibodies with distinct specificities, perhaps directed at other constituents of the neuromuscular junction [16]. While these novel chemotherapy agents have been found to be useful in the treatment of previously refractory cancers, they can have potentially life-threatening complications. Physicians should be aware of the possibility that exposing susceptible patients to these medications may trigger autoimmune diseases or worsen pre-existing or latent autoimmunity. As the use of immune checkpoint inhibitors increases, it will be necessary to monitor closely for the development of other unforeseen complications related to dysregulation of the immune system.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
