Abstract
Background:
Mutations in the Huntingtin (
Objective:
To investigate the developmental component of HD, with a specific emphasis on understanding the functions of wild-type and mutant
Methods:
We used CRISPR/Cas9 gene-editing technology to disrupt the ATG region of the
Results:
We showed that the loss of wild-type, mutant, or both HTT isoforms does not affect the pluripotency of iPSCs or their transition into neural cells. However, we observed that HTT loss causes division impairments in forebrain neuro-epithelial cells and alters maturation of striatal projection neurons (SPNs) particularly in the acquisition of DARPP32 expression, a key functional marker of SPNs. Finally, young post-mitotic neurons derived from HTT-/- human iPSCs display cellular dysfunctions observed in adult HD neurons.
Conclusions:
We described a novel collection of isogenic clones with mono- and biallelic
Keywords
INTRODUCTION
The human huntingtin (
While typically diagnosed in adulthood, a growing body of literature describes a significant developmental component to HD [10–18]. Wild-type HTT is involved in multiple developmental processes including gastrulation [19–21], spermatogenesis [7], epithelial morphogenesis and epithelial-to-mesenchymal transition [13, 23]. In the developing brain, HTT is implicated in neural tube formation and is essential for the differentiation and migration of neuroblasts in mice [10, 17]. HTT is also linked to forebrain developmental defects in human fetuses [11, 24] confirming previous data in the cortex and striatum of HD animals and in human induced pluripotent stem cell (iPSC) models [13, 26]. Moreover, severe reduction of HTT protein causes an extremely rare, yet dramatic, neurodevelopmental disorder [27, 28]. The impacts of complete loss of HTT, HTT haplo-insufficiency and HTT mutations on early steps of human neural development and later on forebrain cells maturation and functions, the brain region most affected in HD, remain poorly understood.
In this study, we have tackled this question employing patient–specific iPSCs and present the generation of a collection of isogenic clones. These clones encompass mono- or biallelic inactivation of
MATERIALS AND METHODS
Human iPSC and hESC cultures
The HD-hiPSC “109Q” line ND42222 (XX, 109 CAG, passage 42) was obtained from Coriell repository. This line is heterozygous for
Generation of an isogenic series from 109Q-iPSCs
We used CRISPR/Cas9 technology to generate one series of isogenic hiPSC lines with different HTT proteins dosages (HTTwt/-; HTT-/mut and HTT-/-). The ND42222_109Q line (Coriell) was used as a parental line. To create this series, CRISPR/Cas9 was used to alter the ATG in the first exon of
Neural and neuronal differentiations
For neural differentiation, hiPSC colonies were treated (DIV0) as previously described [30] in N2B27 medium consisting of 50% DMEMF-12 Glutamax, 50% Neurobasal medium, 2% B27 supplement 50×minus vitamin A, 1% N2 supplement and 50μM β-mercaptoethanol (Thermo Fisher Scientific) supplemented with SB431542 (20μM; Tocris), LDN-193189 (100 nM; Sigma-Aldrich), XAV-939 (1μM; Tocris), and 10μM ROCK inhibitor (Y27632, Calbiochem). For medium spiny neurons (MSN) and cortical neurons differentiation, hiPSC or hESC colonies were treated (DIV0) as previously described in [30, 34] and [29], respectively.
Protein extraction and western blotting
Protein extracts (5–10μg) were loaded on a 3–8% (NuPage Tris-Acetate gels, Invitrogen®) or 10% (NuPage Bis–Tris gels, Invitrogen®) gels and transferred onto Gel Transfer Stacks Nitrocellulose membranes (Invitrogen®) using the iBlot2 Dry Blotting System (Invitrogen®). Antibody binding was quantified using a LiCor Odyssee CLx imager and Image Studio Lite 5.2 software. All antibodies used are listed in the Supplementary Material.
Immunocytochemistry
Cells were fixed with 4% paraformaldehyde (PFA) with 4% sucrose and further permeabilized with 0.1% Triton X-100 (Sigma) and 2% bovine serum albumin (BSA) in phosphate-buffered saline (PBS, Sigma). Primary antibodies were then added and the samples incubated at 4°C overnight in PBS with 2% BSA and 0.1% Triton. Species-specific secondary antibodies coupled to Alexa 350, 488, 555, and 647 (1/1000, Invitrogen) and DAPI counterstain were applied for 1 h at room temperature. Primary Antibodies: CALB (Origene; TA318675; 1/500); CTIP2 (abcam; ab18465; 1/500); DARPP32 (Abcam; ab40801; 1/500); FOXP1 (Abcam; ab16645; 1/800); MAP2 (Biolegend; 822501; 1/1500); NANOG (Abcam; ab62734; 1/500); OCT4 (Cell signaling; 28405; 1/500); Pericentrin (Abcam; ab28144; 1/800); SSEA3 (Biolegend; 330312; 1/500); TBR1 (Abcam; ab31940; 1/500); γ-H2AX (Millipore; 05-636; 1/500).
DNA damage analyses
DNA damage was analyzed in undifferentiated iPSCs, striatal neurons, and cortical neurons stained with γ-H2AX and DAPI to visualize double-strand DNA breaks and nuclei. Twenty images were taken per experiment and processed using the HCS CellInsight CX7 Platform (Thermo Fisher Scientific). The software analyzed each image to determine the number of cells (DAPI positive nuclei) and the number of γ-H2AX foci in each nucleus. The number of foci per cell was calculated and normalized to the number of foci in HTT wt/mut cells.
Spindle orientation quantification, lumen size determination, and mitosis count
Spindle angle in metaphase cells were stained for pericentrin and DAPI to visualize the spindle poles, the lumen outer limit and chromatin, was calculated using ImageJ software (http://rsb.info.nih.gov/ij/, NIH, USA). The images were captured with a Leica DMI6000 confocal optical microscope (TCS SPE) equipped with a 63x oil-immersion objective controlled by LAS X software. Z-stack steps were of 0.64μm. For hiPSCs, the angle between the pole-pole axis and the substratum plane was calculated. Using imageJ software, a line crossing both spindle poles was drawn on the Z projection pictures and repositioned along the Z-axis using the stack of Z-sections. For R-NSCs, one line crossing both spindle poles and the tangent of the lumen outer limit were drawn on the Z projection pictures to determine the angle. Lumen area was calculated using Z-projection images. The outer boundary of the lumen was manually traced using pericentrin staining, and the perimeter was measured using ImageJ software. For mitosis counting, Z-projection images were employed to count the number of round DAPI-positive nuclei and cells in M-phase, characterized by condensed DAPI + chromosomes within rosette structures.
BDNF transport
Cortical progenitors were infected with BDNF-mCherry lentivirus upon seeding. At DIV17, we used an inverted microscope (Axio Observer, Zeiss) coupled to a spinning-disk confocal system (CSU-W1-T3, Yokogawa) connected to a wide-field electron-multiplying CCD camera (ProEM + 1024, Princeton Instrument) and maintained at 37°C and 5% CO2. We took images every 200 ms for 30 s BDNF-mCherry trafficking (×63 oil-immersion objective, 1.46 NA). Images were analyzed with the KymoToolBox plugin for ImageJ [35–37].
Statistical analysis
GraphPad Prism (GraphPad Software, Inc.) software was used for statistical analysis. All experiments were conducted blindly and consisted of at least three independent replicates. Data are expressed as the median. The criterion for statistical significance was set to
RESULTS
In order to model the loss of wt-HTT, mut-HTT, or both isoforms during human neural and striatal development, we used CRISPR-Cas9 technology to inactivate the
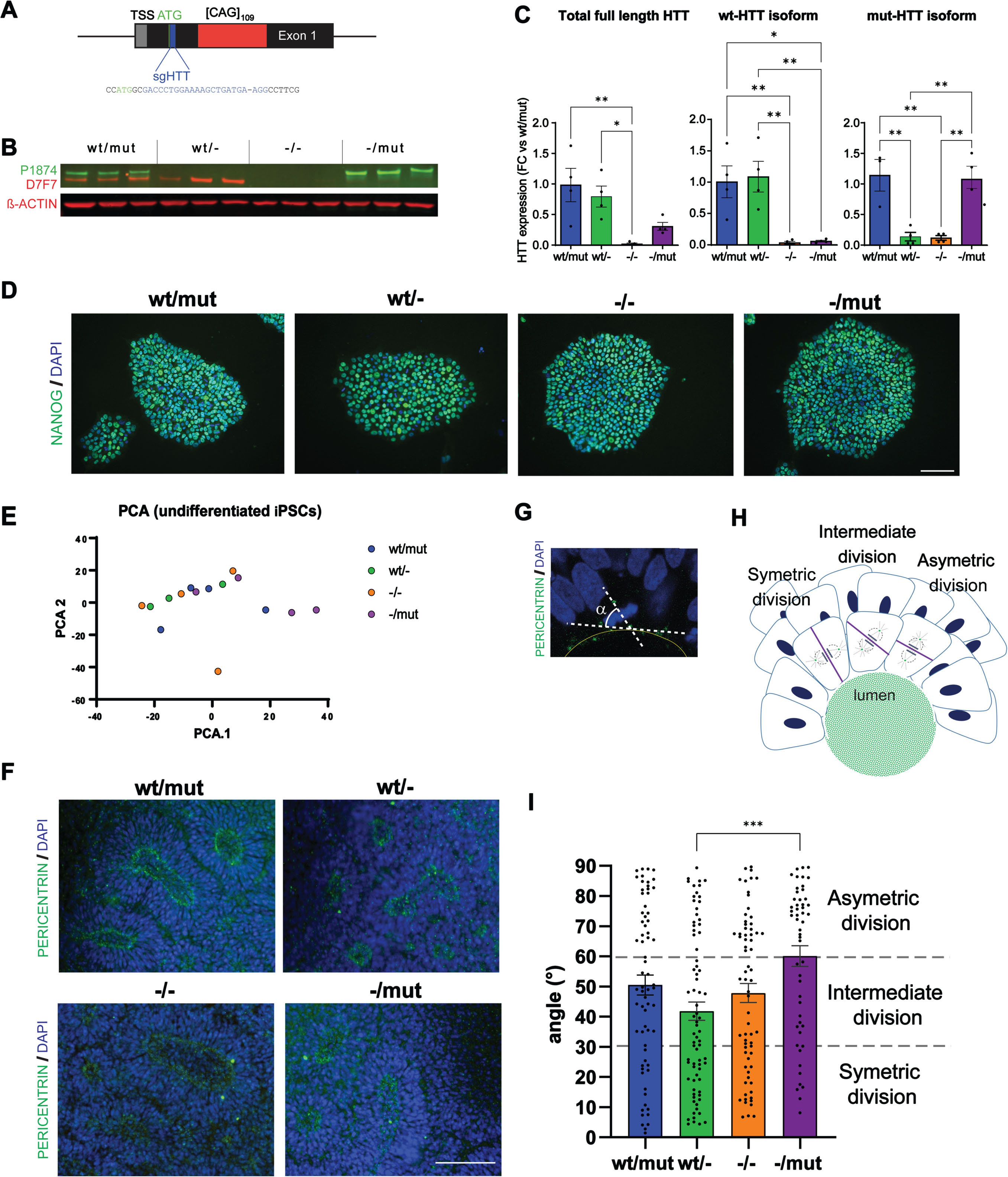
Generation of isogenic clones of 109Q-iPSC with monoallelic or biallelic HTT inactivation and alteration of neural rosette derived from these clones. A) Schematic diagram of the
We then used our clones to record the effect of wt and/or mut-HTT loss on human neural induction and neuro-epithelial cells formation and organization. We did not record any change in the transition from pluripotency to neuroectodermal fate when monitoring OCT4 and PAX6 levels between DIV0 to 8 (Supplementary Figure 4A). Investigating the next stage when neuro-epithelial cells progressively emerge into rosette-like structures (R-NSC), the lumen size of DIV7 rosettes remained unchanged across genotypes (Fig. 1I; Supplementary Figure 4B). In contrast, we detected statistically higher cell division in HTT-/- rosettes compared with HTT-/mut (Supplementary Figure 4C) as well as changes in the orientation of cell division of cells adjacent to R-NSC lumen. Spindle orientation drives the self-organization of R-NSCs as symmetric division gives rise to two daughter neuroepithelial cells and permits the expansion of rosette size while asymmetric division (alpha spindle angle 0–30°) generates a daughter more likely to mature into post-mitotic neurons [26]. Ruzo and collaborators reported that wt human embryonic stem cells (hESC) derived R-NSCs (DIV28) show a bias toward symmetric division that is significantly reduced in rosettes derived from HD-hESCs [26]. In our R-NSC cultures, we observed that HTT-/mut R-NSCs are biased towards asymmetric divisions while HTTwt/- cells are biased towards symmetric division (Fig. 1F–H; Supplementary Figure 4D). These conclusions are consistent with our previous observations of HD-hESC derived neural stem cells (DIV > 50) treated with RNAi silencing both
Considering that the alteration in rosette self-organization might influence their differentiation into post-mitotic neurons, we investigated the maturation of R-NSCs from all genotypes (HTTwt/mut, wt/-, -/mut, -/-) into striatal neurons, the subtype of neurons most affected in HD. We assessed the cultures at DIV55 by immunocytochemistry (Fig. 2A, B; Supplementary Figure 5A, B), western blot (Fig. 2C, D), and RNAseq (Supplementary Figure 6) for markers of post-mitotic neurons (MAP2), and striatal projecting GABAergic neurons (SPN: DARPP32/PPP1R1B, CTIP2/BCL11B). Overall, all cultures produced neuronal populations enriched in striatal cells. Proportion of positive cells and/or the protein level of DARPP32 and CTIP2 (BCL11B) cells, a transcription factor essential for the formation of DARPP32 + SPN, was significantly higher in HTTwt/- than in HTT-/mut. (Fig. 2A, B). To determine if the loss of wt- or wt/mut-HTT would impair DARPP32 expression in post-mitotic striatal neurons expressing this SPN marker, we used lentiviruses expressing short hairpin RNA (shRNA) targeting
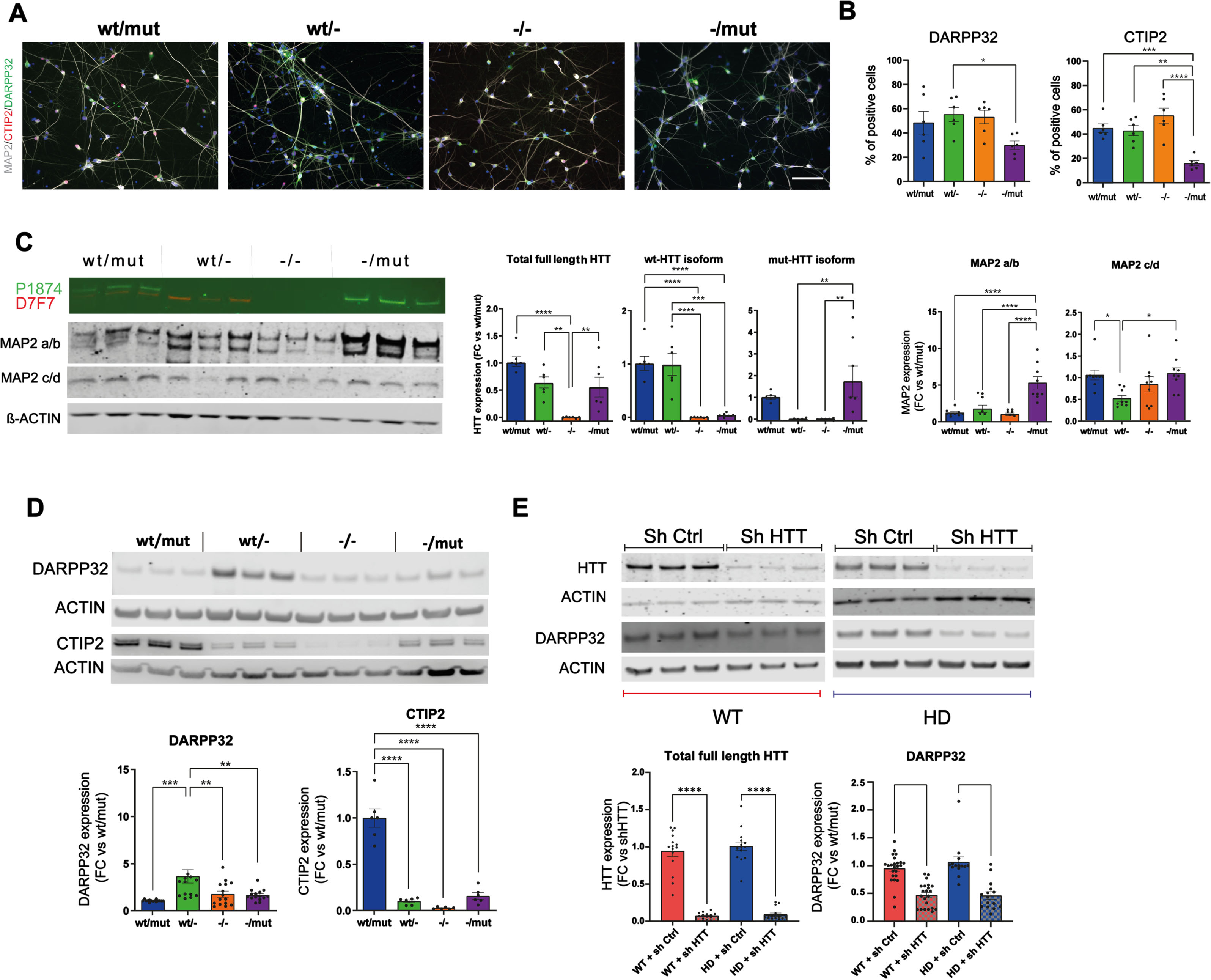
Loss of HTT impairs striatal differentiation of human iPSCs. A) Representative immunostaining of neuronal marker (MAP2) and SPN marker (DARPP32, and CTIP2) at DIV55 (scale bar: 100μm). B) Percentage of cells expressing DARPP32 and CTIP2 (1 clone per genotype,
While impaired functions of striatal and cortical neurons are well described in adult HD mice this is far less the case in patient-derived iPSC neuronal derivatives. We explored two functions: DNA damage and BDNF transport in neurons in the derivatives of our iPSC clones. Increased levels of DNA damage are found in HD neural progenitor cells derived from a panel of isogenic, allelic human ESCs [38]. In order to assess whether the loss of wt- or wt/mut-HTT could modulate this phenotype, we quantified nuclear foci in striatal (Fig. 3A, B; Supplementary Figure 7D) and cortical neurons (Fig. 3C; Supplementary Figure 7A–C) at DIV55 with γ-H2AX, a double-stranded DNA breaks marker. We identified a significant increase in basal DNA damage in both cortical and striatal neurons derived from HTT-/- clones while no change in DNA damage marks were observed in undifferentiated iPSCs of different genotypes (Supplementary Figure 7E, F). These results suggest that DNA damage phenotype in HD might be resulting from a loss of wt-HTT function in HD neurons. Impaired vesicular transport of BDNF along axons of cortical neurons projecting onto striatal SPN has been described in HD mice and in immature neurons derived from HD-hESCs [39]. As impaired transport and release of BDNF have profound consequences on the survival of the cortico-striatal pathway in HD, we examined the dynamics of BDNF-mCherry containing vesicles recording their trafficking (Fig. 1D). We analyzed the segmental velocity (the speed of a given vesicle without pauses and according to their direction), number of vesicles migrating in a given direction (anterograde or retrograde), global velocity (speed, including pausing and static vesicles), and linear flow. Linear flow accounts for the velocity and number of motile vesicles, providing an estimated numerical value of the overall transport within the dendrites (Fig. 3E–H). The majority of BDNF transport parameters were improved in cortical neurons derived from HTTwt/- clones relative to neurons from parental line. Together, these results support the dominant-negative activity of mutant HTT on BDNF transport in HD neurons. Considering the relative immaturity of human iPSC-derived neurons and because increased γH2AX staining may result from increased DNA damage and/or reduced DNA damage repair processes, our results suggest that alterations of BDNF transport and DNA damage and/or DNA damage repair already occur in young forebrain neurons in the developing fetus carrying the HD mutation.
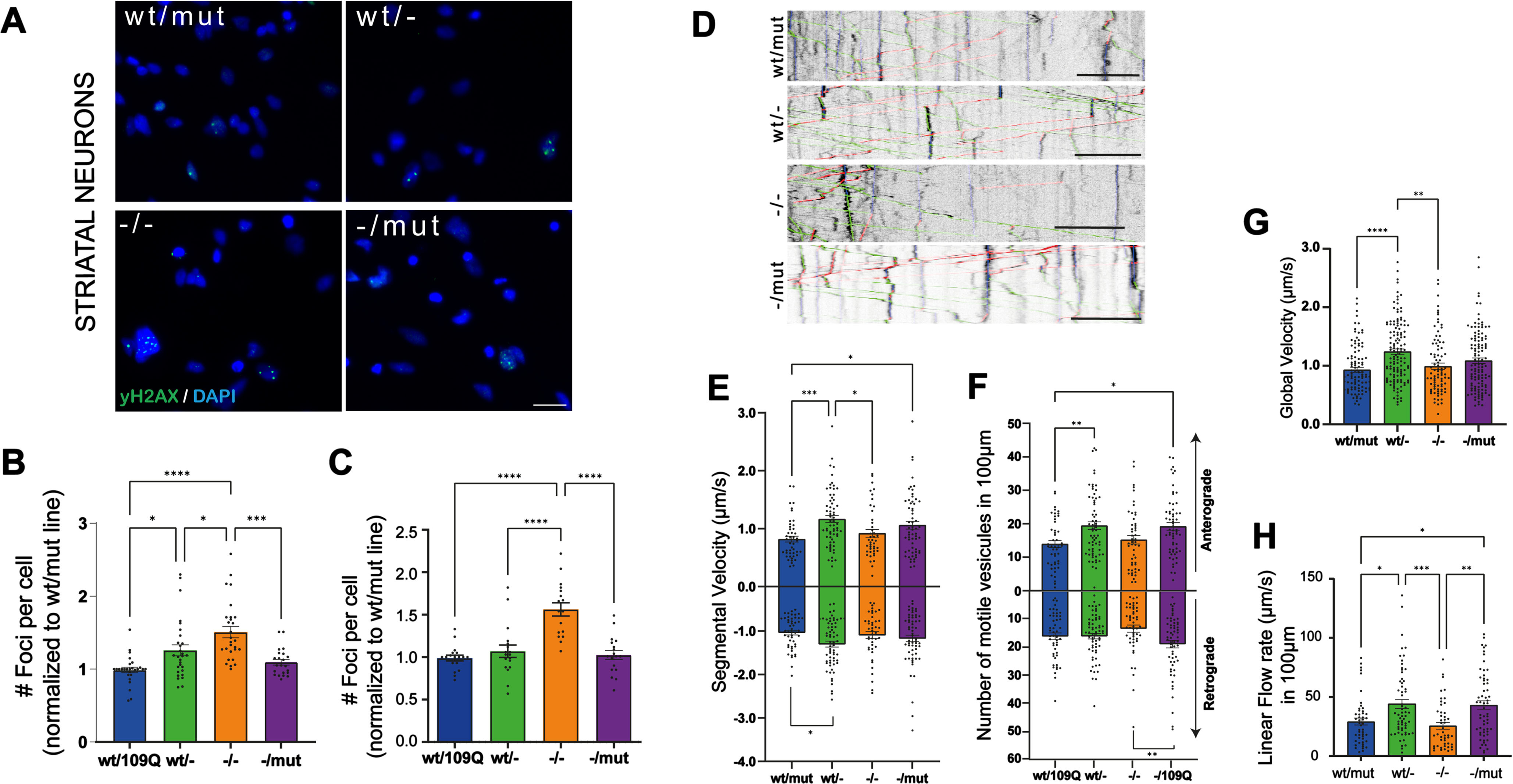
Mutation or loss of HTT isoforms impair DNA damage response and BDNF transport in human neurons. A) Immunostaining of yH2AX marker (green) to identify double-stranded DNA breaks in striatal neurons at DIV 55 (scale bar 20μm). Number of foci per cell normalized to HTTwt/mut line in striatal neurons (B) and cortical neurons (C). (Individual data points (mean of 20 pictures/well), mean and SEM are shown; n indicates the total number of neurons per condition in at least four independent experiments;
DISCUSSION
To date, although extensive research has been conducted on HTT inactivation [40] or lowering [41] as a potential strategy to improve HD pathogenesis, the translation into the clinic of this obvious approach for HD has not yet been substantiated. The largest clinical trial thus far (NCT03761849) by Roche examined non-allele selective antisense oligonucleotide (ASO) targeting
We created a novel collection of isogenic mono- or biallelic
Characterizing neural and neuronal derivatives of human iPSCs of this collection, we show evidence that HTT loss or mutation has impacts on neuro-epithelial and striatal neurons maturation in line with the growing body of literature in support of a role for HTT protein in specific steps of human neurodevelopment and for a developmental component to HD. A case of hypomorphic wt-HTT caused by a t(4;12) balanced chromosome translocation, reducing by half the expression of HTT, was not associated with any detectable abnormal phenotype in the translocation carriers at least up to 46 years old [45]. Likewise, heterozygote carriers of two types of single
Previously, we reported dominant-negative effects of HD mutations leading to alteration of the division of human ESC-derived neural cells [47]. These human neural stem cells present limited apico-basal polarization features oriented perpendicular to the substratum of culture. In the present study, we analyzed both undifferentiated iPSCs, which are not polarized cells, and DIV7 early neuro-epithelial cells that self-organized into rosette structures composed of highly polarized forebrain neural cells which apico-basal polarity oriented radially to the rosette structure [48]. While polarized neural cells present significant difference in the orientation of their cell division based on their HTT genotype, undifferentiated iPSCs from the four genotypes tested did not and divided uniformly perpendicular to their substratum. Although the rate of division of HTT-/- rosette cells was the highest, similar lumen areas were measured across genotypes unlike previous description of HD-hESC/iPSC derived rosettes [26, 49] and HTT-/- hESC-derived rosettes at later stage (DIV28) [26]. Overall, our data are consistent and expand results of studies that show mitotic angle and cell polarity defects in different epithelial neural, or mammary cells [15, 49] and suggest that HTT loss or mutation has significant impact on cell division, polarity and self-organization of developing structures by human epithelial cells but may have less or no impact on cell division of non-polarized cells during development.
The striatal projection neurons (SPN), which are the largest neuronal population in the striatum and most prominently affected in HD [51], arise from the Gsx2-positive progenitors in the lateral ganglionic eminence [52] and express DARPP32, a central mediator of dopamine signaling and other first messengers in these cells [53]. Pluripotent Hdh-/- cells injected in the mouse blastocyst mostly fail to colonize the striatum of chimeric mice [17]. Conversely, loss of HTT in the Gsx2 lineage leads to late-life neuronal loss in the striatum and is accompanied by reduced DARPP32 immunoreactivity [54]. Conditional deletion of HTT in the brain or specifically in SPNs produces similar results on genesis, differentiation, and long-term survival of SPNs [7, 55]. Likewise, striatal development in HD mice revealed defective SPN neurogenesis in the striatum [14]. Using our collection of
Altered survival of neurons in the telencephalon of HD patients has been linked to several neuronal dysfunctions [56]. Disruptions of the homeostasis of BDNF and of the DNA damage repair machinery play key roles in the pathological cascades that lead to neuronal loss.
In summary, the newly generated collection of isogenic iPSC clones provides a valuable resource for studying HTT functions during human development and in adult cells. It complements existing collections of isogenic hESC and h-iPSC clones with different CAG length variations in exon 1 of
Footnotes
ACKNOWLEDGMENTS
We would like to thank Anne de Cian and Jean-Paul Concordet from TACGENE: MNHN-CNRS UMR 7196/INSERM U1154 for the SpCas9 purified protein.
FUNDING
This work benefited from support from European Commission H2020 Project Joint Programme –Neurodegenerative Disease Research (JPND) ModelPolyQ grant 643417), NeurATRlS ANR-11-INBS-0011, of the French lnvestissements d’Avenir Program run by the Agence Nationale pour la Recherche.”, the Laboratoire d’Excellence Revive (ANR-10-LABX-73), Parkington project ANR-17-CE18-0026, Neurolead project BPI France and Institut national de la santé et de la recherche médicale (Inserm) (A.L.P). I-Stem and CECS are supported by the Association Française contre les Myopathies (AFM).
CONFLICT OF INTEREST
Frédéric Saudou is an Editorial Board member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer review.
DATA AVAILABILITY
The whole-genome sequence data have been uploaded to the Sequence Read Archive at NCBI under Accession number GSE228254. Other data supporting the findings of this study are available on request from the corresponding author.
