Abstract
Background:
People with Huntington’s disease (HD) exhibit neurocognitive alterations throughout the disease, including deficits in social cognitive processes such as Theory of Mind (ToM).
Objective:
The aim is to identify methodologies and ToM instruments employed in HD, alongside relevant findings, within the scientific literature of the past two decades.
Methods:
We conducted a comprehensive search for relevant papers in the SCOPUS, PubMed, APA-PsyArticles, Web of Science, Redalyc, and SciELO databases. In the selection process, we specifically focused on studies that included individuals with a confirmed genetic status of HD and investigated ToM functioning in patients with and without motor symptoms. The systematic review followed the PRISMA protocol.
Results:
A total of 27 papers were selected for this systematic review, covering the period from 2003 to 2023. The findings consistently indicate that ToM is globally affected in patients with manifest motor symptoms. In individuals without motor symptoms, impairments are focused on the affective dimensions of ToM.
Conclusions:
Based on our analysis, affective ToM could be considered a potential biomarker for HD. Therefore, it is recommended that ToM assessment be included as part of neuropsychological evaluation protocols in clinical settings. Suchinclusion could aid in the identification of early stages of the disease and provide new opportunities for treatment, particularly with emerging drugs like antisense oligomers. The Prospero registration number for this review is CRD42020209769.
Keywords
INTRODUCTION
Huntington’s disease (HD) is an autosomal dominant genetic neurodegenerative disorder caused by the expansion of the CAG repeat length in the huntingtin (HTT) gene. This expansion occurs when there are 36 or more CAG repeat expansion, initiating a cascade of troubles manifested in progressive neurodegeneration. The neurodegenerative progression of HD initiates at a subcortical level within the basal ganglia during the early stages of the disease. Here, the mutated huntingtin protein begins to accumulate, triggering a process of cellular death that impacts neurons in the striatum (putamen and caudate). This leads to a reduction in the size of the basal ganglia and influences the production of neurotransmitters such as dopamine, glutamate, and GABA. These alterations contribute to the onset of both cognitive and motor symptoms. Beyond the striatum, other structures progressively affected in the course of the disease include the caudate nucleus, thalamus, corpus callosum, and neocortex [1, 2].
All of the aforementioned factors contribute to the gradual and progressive manifestation of motor problems, characterized by choreic movements, psychiatric symptoms, and neurocognitive deficits in patients. [3–5]. Regarding neurocognitive deficits, the most frequently reported symptoms in individuals with HD include attention and concentration problems, impaired processing speed, memory and language deficits, as well as difficulties in visuospatial perception, executive functioning, and social cognition, especially in terms of emotion recognition and Theory of Mind (ToM) [6–8].
To date, a significant number of studies have reported alterations in ToM, indicating that individuals with HD exhibit difficulties in understanding the behaviors of others. [9, 10]. ToM refers to the ability to attribute thoughts, desires, and beliefs to oneself and others [11, 12]. This ability is directly associated with social functioning [13–15] and serves as a mediator between executive functions and social interaction [16]. Thus, ToM facilitates the development of meaningful social relationships, which are often affected in individuals with HD [17].
The concept of ToM is a component of Social Cognition, but its definition remains quite heterogeneous, nonetheless it is generally accepted to have two components: affective and cognitive. The affective component, or Affective ToM, pertains to the ability to attribute emotional states and recognize how another person is feeling. Conversely, the cognitive component, or Cognitive ToM, involves the ability to attribute mental states and recognize what another person may be thinking [10, 18]. In recent years, efforts have been made to identify the associated neuroanatomical areas. For Affective ToM, the main associated areas include the right frontal cortex, reaching its peak in the inferior frontal gyrus, and expanding from the right insula and temporal pole to the pre and postcentral gyri, as well as the supramarginal gyrus. For Cognitive ToM, the main associated areas are the cortical midline and temporoparietal areas, with significant activation in the anterior cingulate cortex and medial prefrontal cortex. Additionally, there is a proposal for structures that mediate between Affective and Cognitive ToM, such as bilateral temporal lobes, ranging from the posterior superior temporal gyri to the anterior temporal lobes [19].
While the majority of the neuroanatomical structures associated with ToM are cortical, there is also evidence suggesting the involvement of subcortical structures in this social cognitive function, such as the basal ganglia and the cerebellum [19, 20]. Notably, these structures are prominently implicated in movement disorders, such as HD. From this perspective, a more comprehensive understanding of the reported ToM alterations in HD can be achieved. However, these alterations vary among individuals according to disease severity, whether the patient already shows motor symptoms or carries the abnormal CAG repeat length without any motor symptoms.
Individuals with manifest motor symptoms exhibit alterations in all dimensions of ToM measured through various instruments used to assess it [21–23]; especially in the Faux Pas test and the Reading the Mind in the Eyes Test (RMET). The performance of patients with motor symptoms in these tests is usually reported as lower than that of healthy individuals, regardless of the disease stage or the number of CAG repeat length [21, 24–27]. Concerning patients without manifest motor symptoms the evidence is less clear. While some studies show that ToM performance between individuals without motor symptoms and healthy controls is similar [26, 28], other studies report that individuals without manifest motor symptoms perform worse than healthy controls, albeit to a lesser extent than individuals with manifest motor symptoms [21, 24]. Furthermore, individuals without motor symptoms exhibit increased apathy and language difficulties [29, 30], processes associated with ToM [31, 32]. On the other hand, patients carrying more copies of the CAG repeat length in the HTT gene have a high tendency to develop psychiatric disorders, several of them associated with ToM difficulties [33–35].
Regarding the above, a meta-analysis reports that HD patients with manifest motor symptoms exhibited significantly poorer performance compared to healthy individuals in the RMET and Faux Pas recognition. Additionally, in patients without manifest motor symptoms, a trend-level impairment in ToM was observed using the same assessment tool. Moreover, significant associations were found between ToM impairments, older age, and increased severity of motor symptoms [36]. This study is particularly intriguing as it suggests that ToM alterations could be considered an early biomarker for HD diagnosis.
However, the meta-analysis conducted in this study focused exclusively on the RMET and the Faux Pas test. While it represents a significant contribution, it may not be sufficient to address the functioning of ToM comprehensively. There are other complementary tests evaluating aspects of ToM that have also been employed in studies involving HD patients, such as Happé’s Strange Stories, assessing pragmatic language comprehension [37]; the Hinting task and The Awareness of Social Inference Test (TASIT), evaluating the ability to make social inferences [38, 39]; the Yoni Task, assessing the ability to judge mental states based on verbal cues [40]; and the Mental State Attribution Task (MSAT), serving to evaluate the skill in identifying situations involving deception or cooperation based on the principle of false beliefs [41]. All these tests could offer a more comprehensive insight into the functioning of ToM in individuals with HD, so they must also be considered.
It is imperative to understand the functioning of ToM in HD, as deficits in ToM could serve as an early precursor to symptomatic onset. This is an important advantage since these deficiencies can be identified more easily in the patient’s daily life than non-social cognitive alterations, taking into account that one of the first manifestations of HD is personality changes [6, 33] which lead to problems in interpersonal relationships that may be associated with how the mental and emotional states of others are understood.
Furthermore, comprehending the trajectory of ToM within the neurodegenerative process of HD is integral for the informed design of efficacious intervention programs at the clinical level. By incorporating the psychosocial dimension through the lens of social cognition, the treatment plan is complemented. Traditionally, from a neurocognitive viewpoint, the emphasis tends to be on non-social cognitive processes such as memory, attention, and perception. Consequently, the social dimension is relegated solely to psychotherapy, which, although effective, could amplify its impact by incorporating ToM and thus providing a more nuanced clinical description of the patient.
From a neuropsychological assessment perspective, understanding the ToM profile in HD patients enhances the accuracy of assessments aligned with the Diagnostic and Statistical Manual of Mental Disorders (DSM-5). The DSM-5 recognizes Social Cognition (and consequently ToM) as a pivotal clinical factor for diagnosing neurocognitive disorders in neurodegenerative diseases [42], even being considered a potential biomarker [7, 43].
In this study, we conducted a systematic review to examine the functioning of ToM in individuals with HD, both with and without manifest motor symptoms. The aim is to identify, within the scientific literature of the past 20 years, the methodologies and ToM instruments employed in this neurodegenerative condition, alongside the most pertinent findings. This involves an evaluation of their relevance and prospective application in clinical settings.
METHODS
Studies selection
This work was conducted following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guide [44] and the PRISMA protocol [45]. We searched research papers in English and Spanish in the SCOPUS, PubMed, APA-PsyArticles, Web of Science, Redalyc, and SciELO databases. Reference lists from previous research related to the topic were also reviewed. Because the concept of ToM is still heterogeneous in the literature and different terms can be found that are often used as synonyms, ToM keywords were taken from thesauri and previous systematic reviews and meta-analyses on the topic [46, 47]. Keywords and booleans in English were “Theory of Mind” OR “mentalizing” OR “mentalising” OR “Mindreading” OR “Mindread” OR “false belief” OR “mental attribution” OR “Social Cognition” OR “Reading the mind in the eyes” OR “The eye test” AND “Huntington” OR “Huntington's Disease” OR “Pre-manifest Huntington's Disease” OR “Pre-clinical Huntington's Disease” OR “Prodromal Huntington's Disease”. Similarly, in Spanish the following keywords and booleans were used: “Teoría de la Mente” OR “Mentalización” OR “Falsa creencia” OR “Falsas creencias” OR “atribuciones mentales” OR “Cognición social” OR “Lectura de la mente en los ojos” OR “Test de la Mirada” AND “Huntington” OR “Enfermedad de Huntington” OR “Enfermedad de Huntington pre-manifiesta” OR “Enfermedad de Huntington pre-clínica” OR “Enfermedad de Huntington prodrómica”.
For this systematic review, articles published up to May 2023 that explored ToM functioning in patients with HD were included. Research studies on other neurodegenerative disorders were also included if they had at least one group of patients with manifest or prodromal HD. We excluded 1) studies that did not use any ToM clinical instrument/measure; 2) studies that did not include unaffected healthy control group; 3) case-only studies; 4) studies in which the group of patients with HD did not have a confirmed diagnosis by genetic analysis; 5) studies with other motor disorders not associated with HD; and 6) clinical trials that did not report ToM clinical measures before treatment. Research papers meeting the inclusion criteria were organized and filtered using the PRISMA flowchart (Fig. 1) using the following procedure. First, the titles of papers detected in the identification phase were exported to a spreadsheet to delete duplicates. Secondly, in the tracing phase, abstracts of not duplicated titles were carefully read to verify the inclusion and exclusion criteria. Finally, in the eligibility phase, the full text of selected papers in the tracing phase was carefully read. The review protocol is registered in the international prospective register of systematic reviews (PROSPERO) with registration number CRD42020209769.
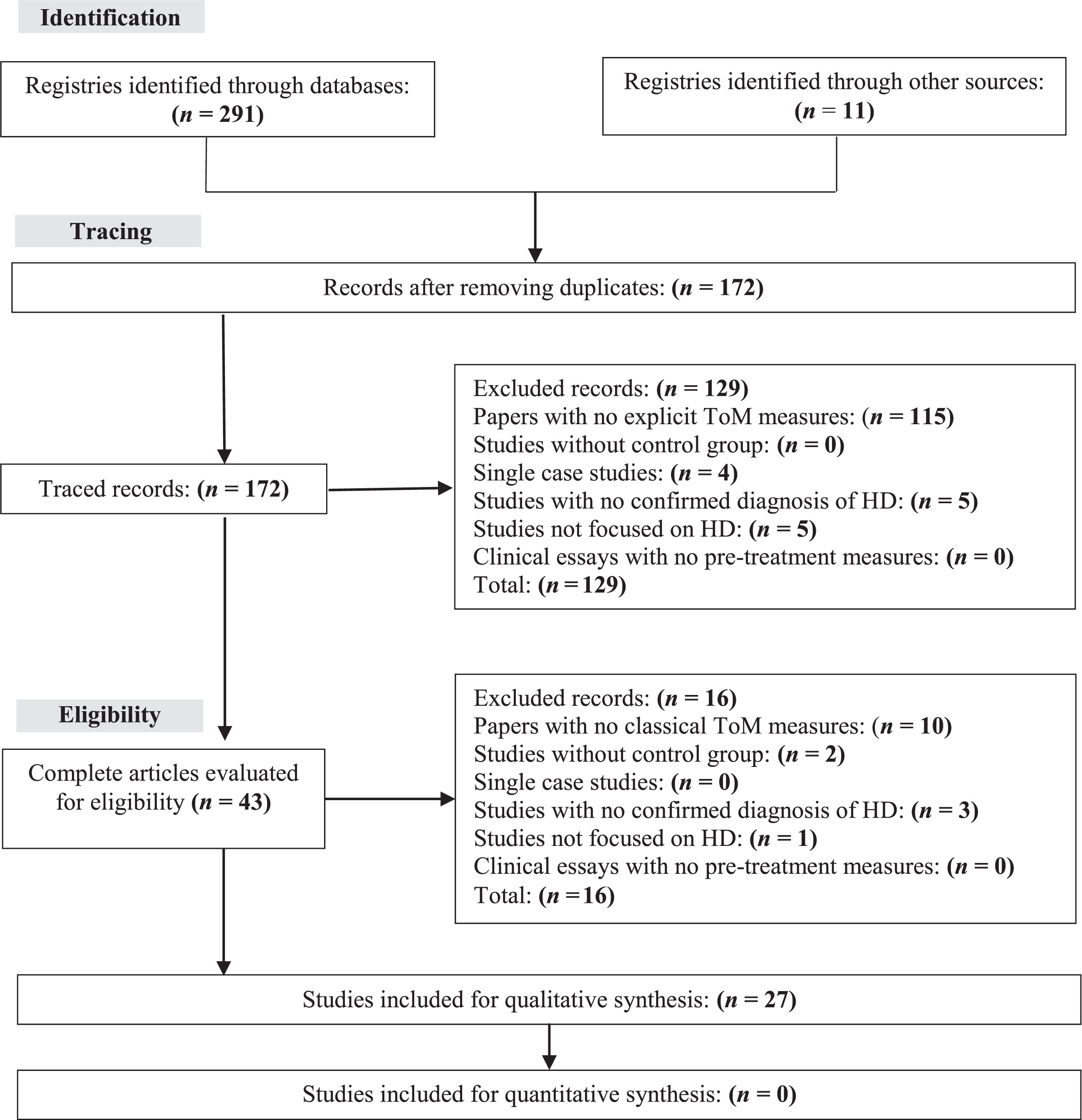
PRISMA diagram for the selection process of research papers.
A meta-analysis could not be conducted for three primary reasons. Firstly, there was a significant variation in the instruments used to assess ToM. Secondly, despite initially considering the inclusion of papers that utilized the Reading the Mind in the Eyes Test, we discovered a high degree of heterogeneity (I2=94%) during the meta-analysis. Lastly, we identified a substantial risk of publication bias. These reasons, particularly the second and third factors, rendered the results unreliable, leading us to conduct this systematic review instead.
RESULTS
A total of 302 papers were found in the Identification phase, 291 directly from databases, and 11 from a previously published meta-analysis [36] (Fig. 1). Of these, 172 (57.3%) papers remained after removing duplicates. In the Tracing phase, 129 papers were excluded after applying the exclusion criteria to the abstracts (Fig. 1). In the Eligibility phase, 16 papers were excluded. The remaining 27 papers were included in the qualitative analysis of this systematic review.
Table 1 summarizes the main characteristics of the selected papers. The oldest research study dates from 2003 [37], suggesting that, up to 2023, only 20 years have passed in ToM research in HD. Interestingly, between 2003 and 2011, research papers were published every four years on average; this number increased to at least one paper per year since 2012.
All samples in the reviewed papers were composed of mostly male adults, with ages ranging from 34.9 years and older for participants without manifest motor symptoms, and 42.7 years and older for participants with manifest motor symptoms. Although not reported in all studies, individuals had eight or more years of education in both groups. The design of all the articles was strictly cross-sectional, except for the works of Unti et al. [48] who conducted a longitudinal study with 2-year time point measurements, and Hendel et al. [49] who worked in a 6-year follow-up study.
Table 1 shows the clinical features used in all research papers to diagnose HD. Except for one work where it was used, who utilized the Unified Parkinson’s Disease Rating Scale (UPDRS) [50], all studies reported motor symptoms using the motor subscale of the Unified Huntington’s Disease Rating Scale (UHDRS). In addition, all reviewed papers reported the quantification of the CAG repeat length for diagnosing HD. However, 23 (85.1%) studies reported the average size of the CAG expansion, 10 (37%) reported the burden, and 14 (51.89%) reported the average number of years since the HD diagnosis.
We found that, although most of the studies employed task groups to examine ToM, the instruments to assess ToM were quite heterogeneous. In this sense, the RMET task was the most frequently used (n = 19; 70.3%), followed by the Faux Pas (n = 7; 25.9%) and Heppe’s Strange Stories (n = 3; 14%). Other ToM tasks used were TASIT (n = 3; 11.1%), the Cartoon Stories (n = 2; 7.4%), and Judgment of Preferences, the Hinting Task, the MSAT, and the Yoni Task, with one study each (Table 1). Interestingly, among the studies reviewed, only one reported the use of psychometrically-validated instruments to assess ToM [51]. Thus, standardized scores were not reported.
Characteristics of the papers reviewed
AA, average age; AEY, average education years; CAG-E, CAG-Expansion; DY, duration in years; G-M/F: Gender-male/female; HD, Huntington’s disease; MSAT, Mental State Attribution Task; MSG, Motor Symptoms Group; N/R, not reported; NMSG, No Motor Symptoms Group; RMET, Reading the Mind in the Eyes Test; S-UHDRSM, Severity-Unified Huntington’s Disease Rating Scale Motor subscale; SPTT, Spatial Perspective-Taking Task; TASIT, The Awareness of Social Inference Test; ToM, Theory of Mind; UPDRS, Unified Parkinson’s Disease Rating Scale.
Finally, all studies that included individuals with manifest motor symptoms showed poorer ToM performance than healthy controls. However, in one study was found that, although ToM was affected in these patients compared to healthy controls, such change was moderate as no difference in the preference judgments task was found [37]. Regarding studies involving participants without motor symptoms, a total of 13 works were identified with more diverse results, as 8 studies (61.5%) documented ToM impairments, while 5 (38.5%) did not provide information on such alterations.
In order to better comprehend the diversity in the results of studies focusing on individuals with HD without motor symptoms, an analysis of these works was conducted in Table 2. It reveals a tendency wherein studies lacking ToM difficulties predominantly featured participants under 37.7 years of age. Conversely, studies involving participants under 37.7 years old, exhibiting ToM difficulties, reported scores exceeding 5 on the UHDRS scale. Hendel’s work stood as the sole exception to this pattern [49]. Conversely, ToM impairments commenced in studies involving participants aged 41.6 years and above, except for Mason’s study [27], where participants had an average age of 43.5 years. However, their UHDRS score was significantly lower compared to other studies reporting impairments. This trend persisted irrespective of the ToM measurement tests employed, as most studies utilized the same assessments (RMET, Faux Pas test, and the Yoni Task), as well as the repeat expansion, which were distributed proportionally across all studies.
Characteristics of studies with HD participant with no motor Symptoms*
*Studies are classified by participants’ age. HD, Huntington’s disease; NMSG, No Motor Symptoms Group; RMET, Reading the Mind in the Eyes Test; TASIT, The Awareness of Social Inference Test; ToM, Theory of Mind; UHDRSM, Severity-Unified Huntington’s Disease Rating Scale Motor subscale.
DISCUSSION
This study systematically reviewed the last 20 years of research on ToM in HD. Our findings reveal that the investigation of ToM in HD is relatively recent compared to other neurocognitive and social-cognitive functions. The earliest study identified dates back to 2003 [37], Subsequently, from this pioneering work, we observe a conservative and sustained growth in ToM research, with sporadic publications until 2012. From 2012 onwards, there has been a noticeable increase, averaging one to two articles per year.
Considering that HD is a rare condition affecting 1–10 individuals per 100,000 people [52, 53] and the assessment of ToM in neurodegenerative diseases and adults is a relatively recent endeavor [7, 43], we anticipated a limited number of studies to be included in this systematic review. Such studies reported modest sample sizes and were recently published. However, the escalating number of publications indicates a growing interest in investigating ToM in the context of HD.
This surge in interest may be attributed to several factors. Firstly, there is a rising emphasis on identifying neurocognitive biomarkers, particularly in the ongoing landscape of pharmacological clinical trials aimed at mitigating or delaying the disease’s symptoms [54–56], with ToM emerging as a noteworthy candidate [7, 36]. Secondly, since the DSM-5 includes ToM within the criteria for diagnosing neurocognitive disorders [42], this necessitates the development of assessment techniques for ToM in HD, involving the identification of more sensitive and specific tools to detect ToM impairments at any stage of the disease. And thirdly, detecting the disease in the prodromal phase has become increasingly feasible due to the refinement of both clinical and molecular methods [35, 58]. Therefore, in addition to pharmacological strategies, it becomes necessary to develop other interventions to assist the patient in improving their quality of life. These strategies should encompass the psychosocial aspect, enabling the patient to maintain existing support networks and even establish new ones. In this context, identifying early impairments in ToM and subsequently intervening will aid the patient in strengthening their social skills, resulting in a more favorable prognosis in the psychosocial domain.
About the instruments used to assess ToM in patients with HD, we encountered an extensive array of tools addressing both the cognitive and affective dimensions of ToM. Among them, the most frequently employed are the Faux Pas test and the RMET test, considered to evaluate mainly the affective aspect of ToM [18, 19]. This finding regarding the popularity of these tests aligns with other studies investigating ToM in various neurodegenerative and neuropsychiatric disorders [59–62], indicating a trend toward to detect affective impairments in neurodegenerative disorders, and supporting the findings of Bora et al. [36] who report that it is the affective domain that is mainly affected in HD.
This makes these instruments likely to have greater sensitivity in detecting ToM alterations in the early stages. Our results support this notion, as we found that during the prodromal phase of the disease, the RMET and Faux Pas test exhibited greater sensitivity in detecting ToM alterations. In contrast, tests such as TASIT, Yoni task, or ToM cartoon stories showed sensitivity to ToM alterations when HD had already entered the phase where motor symptoms began to manifest (see Table 1).
One explanation for this might be that test like TASIT, ToM cartoon stories, or Yoni task are of such complexity that they require the support of other cognitive processes, such as executive functions, language, or memory, to be resolved. Considering that ToM does not always operate in isolation but interacts with other supporting cognitive functions that share cortical structures, such as the lateral and medial prefrontal cortex and temporal and parietal lobes [63, 64], it is possible that in early stages of HD, these cognitive functions supporting ToM compensate for its alterations. However, in the more advanced stages of the disease, when these supporting functions are affected by the natural course of the disease, they cease to compensate for ToM, and its alterations become apparent.
In any case, considering these results, it is advisable to further refine the RMET and the Faux Pas test. Although they were the most sensitive in detecting ToM alterations in the prodromal phases of HD, there were still studies in which these same instruments did not detect alterations (see Table 2). This indicates the necessity of conducting studies to analyze the psychometric properties of these tests to make them more robust and, consequently, more reliable in detecting ToM alterations from the early stages of HD.
ToM in individuals with manifest motor symptoms
In the studies published in the last 20 years investigating ToM functioning in patients with HD who have manifested motor symptoms, we have observed that this aspect of social cognition is reported as impaired. Similar findings were reported by Bora et al. in their meta-analysis published in 2016 [36]. They noted that patients with manifest motor symptoms performed lower than their controls in the RMET and Faux Pas test. This finding is noteworthy as both the RMET and Faux Pas test are considered assessments for evaluating the affective aspect of ToM but operate at different levels of processing. The RMET operates at an automatic processing level, while the Faux Pas test operates at a controlled processing level [18]. However, this affective domain is not the only one affected in HD. According to our review, we found that the cognitive domain is also impaired, evident in the reviewed studies reporting lower performance in tasks such as Happé’s Strange Stories, TASIT, Judgment of Preference, Attribution of Intentions Task, ToM Cartoon Stories, Hinting Task, and the MSAT (Table 1). These tasks, except for TASIT, are classified as cognitive ToM tasks, and although TASIT is not strictly a cognitive domain task, like the Faux Pas test, it relies on controlled processing for execution [18, 65].
From a neuroanatomical perspective, all the findings align with the neuropathology of HD. To date, it is known that the initial affected structures are subcortical, such as the basal ganglia and limbic system [2]. The latter is linked to emotional processing and the affective domain of ToM [66, 67]. Subsequently, cortical structures are affected, including the inferior frontal cortex, premotor and sensorimotor cortex, medial cingulate cortex, and frontoparietal and temporoparietal cortices. All these structures are related to the cognitive domain of ToM and involve processes such as attributing false beliefs, understanding pragmatic language, and comprehending social situations. Furthermore, these processes rely on other neurocognitive functions such as executive functions, language, and memory, which are also associated with the mentioned cortical circuits [68–71]. All of this suggests that, in patients with HD in the phase of manifest motor symptoms, ToM impairment is global.
ToM in individuals without manifest motor symptoms
In HD patients without motor symptoms, ToM is also affected; this is evident in 8 out of the 13 articles that reported it (Table 2). However, this alteration occurs differently than in patients who do present motor symptoms. According to our results, while in patients with manifest motor symptoms, ToM alteration is global, in those without them, it is primarily affected in the affective dimension of ToM. This is supported by the fact that studies reporting ToM alterations mainly used RMET and Faux Pas test, and those employing cognitive domain tests like Yoni task or ToM Cartoon Stories did not find difficulties. This could be because in the prodromal stage of the disease, neocortical structures related to cognitive ToM, such as the temporo-parietal junction and the prefrontal lobe, do not show clear signs of deterioration, unlike structures related to affective ToM, such as the limbic system [27, 72].
However, it’s interesting to note that not all studies using affective ToM tests reported alterations. In fact, of the five studies that did not report ToM impairments in prodromal phase, three used RMET. Yet, upon reviewing the characteristics of the population in these studies, it was observed that these were patients either younger than 37.7 years or scored higher than 5 on the UHDRS scale (Table 2). This association between ToM alterations, age, and motor symptoms was proposed by Bora et al., who, through meta-regressions, concluded that the severity of ToM alterations, specifically assessed with RMET and Faux Pas test, was positively associated with the severity of motor symptoms and age [36]. This phenomenon can be explained by the neurodegenerative process in the disease, where cell death begins subcortically and gradually spreads to cortical areas as the patient’s symptomatology evolves due to abnormalities in the striatum and white matter, mainly in the prefrontal tract of the corpus callosum, the anterior thalamic radiations, the inferior frontal-occipital fasciculus, and the uncinate fasciculus [1, 74].
Limitations
Like any other systematic review, this study has some limitations that invite us to read its results with caution. First, the search for articles for the systematic review was conducted only in English and Spanish, leaving aside literature in other languages that could have provided important information. Secondly, it was not possible to conduct a meta-analysis, which implies that the results in this paper are more qualitative than quantitative. While this is not a problem, a meta-analysis offers more precise information about the performance of ToM in HD. Lastly, it is important to note that the limitations reported in each reviewed paper could influence our results.
Conclusions, final considerations, and future research
After reviewing scientific publications from the past 20 years on the functioning of ToM in HD, several conclusions can be drawn. First, ToM is affected in both the prodromal and manifest motor symptom phases of the disease. However, data trends indicate that, in the prodromal phase, only the affective dimension of ToM is affected, while in the motor symptom phase, the alteration is global, affecting both the affective and cognitive dimensions of ToM. This is significant as it allows for tracking the deterioration of ToM as the disease progresses, facilitating therapeutic approaches tailored to the patient’s condition. Timely intervention in the appropriate dimension is clinically relevant, potentially preserving and strengthening the patient’s interpersonal skills, enabling them to stay within a supportive social network.
Second, the RMET along with the Faux Pas test appear to be the most sensitive ToM instruments for detecting alterations in this social cognition ability at very early stages. Therefore, they are promising candidates for inclusion in neuropsychological protocols for evaluating HD patients in clinical settings. However, further refinement from a psychometric perspective is necessary to enhance their robustness through validation and standardization, akin to other neuropsychological instruments used for examining HD patients [75, 76].
Third, the study of ToM in the prodromal phase is clearly in an exploratory stage. We consider it so because the first study of this kind was published in 2013 [77], and only 10 years have passed, during which, although the number of publications has increased, much remains to be studied. For example, the course of ToM deterioration in relation to the neuropathology of the disease through longitudinal studies or the degree of interaction with other neurocognitive functions, such as executive functions, which also have extensive literature describing their impact on HD [6, 8]. Moreover, there is a close relationship between these functions and ToM [11, 64].
Finally, eight years after the publication of Bora et al.’s meta-analysis [36], the results of this study continue to support the proposition of ToM as a biomarker for HD. However, we propose that it should be the affective dimension of ToM studied as a prodromal biomarker rather than the cognitive dimension, which appears affected only in later stages, even when motor symptoms are already present.
For future research, we recommend working with longitudinal designs that allow tracking the course of ToM alteration over time as neurodegenerative changes unfold. Implementing neuroimaging studies with functional magnetic resonance imaging to observe brain region activation during ToM tasks would provide valuable insights. Considering the influence of sociodemographic variables such as sex, education, and socioeconomic status is crucial, as there is ample evidence that these variables influence ToM. Lastly, we recommend designing studies that analyze the interaction between Executive Functions and ToM in HD, as it has been reported that ToM and executive functions are closely intertwined, necessitating the identification of their distinct boundaries.
Footnotes
ACKNOWLEDGMENTS
JE-C and WP-A thank María Camila Pineda-Escudero for her constant motivation and inspiration. On the other hand, with this work, the authors pay tribute to Dr. Manuel Sánchez, Neurosurgeon and Director of the Research Project from which this article emanates. His insistence, perseverance, impetus, and dedication for more than 20 years made this project become a reality. We are and will ever be, in debt with you, Dr. Sánchez. Thank you!.
FUNDING
This study was financed by COLCIENCIAS, project “Identificación de Marcadores Tempranos de Tipo Neurológico, Neurofisiológico, Neurocognitivo y Neuropsiquiátrico en Población Presintomática con Riesgo de Enfermedad de Huntington en el Departamento del Atlántico”, grant # 839–2017, contract RC 1253-7775–7992, granted to Grupo de Neurociencias del Caribe, Universidad Simón Bolívar, Barranquilla, Colombia. The sponsor of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the paper.
CONFLICT OF INTEREST
The authors declare no conflict of interest. The results of this paper have not been shared partially or totally. We complied with APA ethical standards in treating our data.
