Abstract
Background:
Huntington’s disease (HD) is an autosomal dominant, neurodegenerative disease that involves dysfunction in the autonomic nervous system (ANS). Heart rate variability (HRV) is a valid and noninvasive measure for ANS dysfunction, yet no study has characterized HRV response to exercise in people with HD.
Objective:
Characterize HRV response to exercise in individuals with HD and explore its implications for exercise prescription and cardiac dysautonomia mechanisms.
Methods:
19 participants with HD were recruited as part of a cohort of individuals enrolled in the Physical Activity and Exercise Outcomes in Huntington’s Disease (PACE-HD) study at Teachers College, Columbia University (TC). 13 non-HD age- and gender-matched control participants were also recruited from TC. HRV was recorded with a Polar H10 heart rate (HR) monitor before, during, and after a ramp cycle-ergometer exercise test.
Results:
Participants with HD showed reduced HR peak (p < 0.01) and HR reserve (p < 0.001) compared with controls. Participants with HD demonstrated reduced root mean square of successive differences between normal-to-normal intervals (RMSSD) and successive differences of normal-to-normal intervals (SDSD) at rest (p < 0.001). Participants with HD also showed differences for low frequency (LF) power (p < 0.01), high frequency (HF) normalized units (nu) (p < 0.05), LF (nu) (p < 0.001), and HF/LF ratio (p < 0.05) compared with controls.
Conclusions:
We found reduced aerobic exercise capacity and sympathovagal dysautonomia both at rest and during post-exercise recovery in people with HD, suggesting modified exercise prescription may be required for people with HD. Further investigations focusing on cardiac dysautonomia and underlying mechanisms of sympathovagal dysautonomia in people with HD are warranted.
INTRODUCTION
Huntington’s disease (HD) is an autosomal dominant, neurodegenerative disease resulting in cognitive decline, emotional/behavioral disturbances, and motor dysfunction [1]. In addition, dysfunction of the autonomic nervous system (ANS) often accompanies HD, as people with HD frequently show signs and symptoms including orthostatic intolerance, excessive perspiration, swallowing difficulties, sleep disorders, and cardiac problems [2–4]. The underlying mechanism might be the damage caused by the progressive nerve cell apoptosis in the central autonomic network (CAN) that performs high-level autonomic control, including insular cortex and hypothalamus [5].
Heart rate variability (HRV), which refers to the variation in time of consecutive heartbeat intervals, is a noninvasive and widely used measure to determine the function and abnormality of ANS in a variety of populations [6, 7], including people with HD [2, 8–11]. Parasympathetic modulation in the ANS is associated with HRV time domain measures and frequency domain measures. Time domain measures include standard deviation of normal-to-normal intervals (SDNN), root mean square of successive differences of successive normal-to-normal intervals (RMSSD), and standard deviation of differences between successive differences of normal-to-normal intervals (SDSD). Each of these plays an important role in evaluating overall HRV and parasympathetic activity [12]. Frequency domain HRV measures such as high frequency (HF) power and HF normalized units (nu) represent parasympathetic activities; and sympathetic activity is associated with low frequency (LF) power, LF (nu), and LF/HF ratio. Together, frequency domain measures provide detailed insights into sympathetic and parasympathetic influences [12]. Employing both time and frequency domain HRV measures allows for a comprehensive assessment of ANS function, enhancing the understanding of its role in people with HD. Several studies in people with HD demonstrated altered HRV parameters, including reduced RMSSD, elevated LF/HF ratio, and LF power compared with controls, suggesting people with HD may have a reduction in vagal modulation and a shift in autonomic balance towards sympathetic predominance [2, 13–15]. Moreover, inverse correlations between the severity of clinical HD symptoms and the modulation of cardiovagal activity have been found [2, 9], suggesting ANS dysfunction severity increases as the disease progresses.
Unlike most studies in HD that were conducted only under vagus-dominated conditions such as at rest and during deep respiration [2, 15], Bär et al. assessed the HRV parameters both under rest and postural change conditions. They reported no difference at rest, but significant differences in RMSSD and LF/HF ratio during postural change (i.e., head-up tilt) between people with early-to-mid stage HD and controls [13]. This finding indicates that, compared with vagus-dominated conditions, putting participants in sympathetic dominant conditions (e.g., postural change, cardiovascular stress tests) might be a better way to uncover vagal modulation reduction in people with early-to-mid stage HD.
Using exercise as the stressor to put the body in a sympathetic dominant condition while monitoring HRV responses might be a valuable approach to investigate autonomic stress reactivity [16–18]. Although several studies have demonstrated altered metabolic and physiological responses to exercise in people with HD during exercise testing and training [19–21], none applied HRV as an outcome measure. In this study, we evaluated HRV in people with early-to-mid stage HD and controls during a maximal incremental exercise test, which enabled us to assess the cardiovagal modulation ability in people with HD by characterizing their HRV parameters during post-exercise recovery.
METHODS
Participants
19 participants with HD and 13 controls were recruited. The HD participants were part of a non-intervention cohort of individuals enrolled in the Physical Activity and Exercise Outcomes in Huntington’s Disease (PACE-HD) study at Teachers College, Columbia University [22]. Inclusion criteria were: (1) prior diagnosis of HD, confirmed by genetic testing, (2) above the age of 18, (3) currently or newly enrolled in Enroll-HD, an integrated clinical research platform [23], (4) up to and including stage 2 disease status, as determined by a total functional capacity (TFC) scale score of 7–13. Exclusion criteria were (1) a diagnosis of juvenile HD, (2) a history of comorbid neurological conditions (e.g., multiple sclerosis or stroke), and (3) an acute orthopedic condition within the previous month (e.g., ankle sprain or fracture) that could limit exercise. Controls were matched for age and sex. It is worth noting that we aimed to enroll 19 control participants, but recruitment was curtailed by the COVID-19 pandemic. Despite considering the resumption of recruitment post-pandemic, we faced logistical challenges, constraints in funding, and a notable decrease in participant availability. Thus, only 13 controls were recruited. Due to the exploratory nature of the study, a formal power calculation was not conducted. Written informed consent was obtained from all the participants. The study was approved by Teachers College, Columbia University Institutional Review Board (18–194 and 19–267).
Procedure
Participants were first asked to complete a series of questionnaires, detailed below, and then proceeded to HRV recording and exercise testing.
Baseline and clinical measures
For both groups of participants, depression, anxiety, and self-reported irritability that might affect HRV measures [24] were rated using the combined Hospital Anxiety and Depression/Snaith Irritability Scale (HADS-SIS) [25]. Self-reported physical activity levels were rated using the International Physical Activity Questionnaire (IPAQ) short form to determine if both groups had the same physical activity level [26]. In addition to demographic data, height, weight, resting heart rate (HR), and blood pressure were also recorded in a sitting position.
For participants with HD, motor function and functional capacity were evaluated using the United Huntington’s Disease Rating Scale (UHDRS) Total Motor Score and Total Functional Capacity measures, respectively [27]. All participants were screened for risk factors, and signs and symptoms of disease by completing the Physical Activity Readiness Questionnaire (PAR-Q+) [28] to ensure safety before exercise testing. Participants were also asked to abstain from smoking, alcohol, and caffeine for at least 8 hours before the exercise test to avoid potential confounding effects on HRV [6].
HRV recording and exercise testing
All participants wore a Polar H10 HR monitor with a V800 watch (Polar Electro Oy, Kempele, Finland) for continuous RR interval data recording throughout the test. The Polar H10 HR monitor has been shown to have a good RR interval (the time elapsed between two successive R-waves of the QRS signal on the electrocardiogram) signal recording quality at rest and during exercise [29]. RR interval data were first recorded for 4 min under resting condition, with participants lying in supine position. Next, participants were positioned on an electromechanically braked cycle ergometer (Lode B.V., Groningen, the Netherlands) with seat height and handle position adjusted to comfort. Then, participants received the description of the exercise test and were instructed in the use of Borg Modified rating of perceived exertion (RPE) on a scale of 1 to 10 [30].
The exercise test was a ramp incremental exercise test performed following the PACE-HD fitness assessment protocol [22]. During the test, participants were instructed to maintain a cycling cadence of 50 revolutions per minute (rpm), starting at a work rate of 10 W with a ramp protocol of 12.5 W/min increase until termination, and to report RPE at the end of every minute. The exercise test was terminated when participants reached volitional exhaustion, an RPE of 10, or the cadence dropped by 10 rpm twice (participants would receive a warning when the cadence dropped by 10 rpm for the first time). After termination, participants cooled down at 40% of maximum resistance for 2 minutes.
After the exercise test participants laid down in a supine position again for a 4 min recovery period. Thereafter, RR interval recording was terminated. In addition to RR interval recording, peak work rate, and HR peak were also recorded for data analysis. Predicted VO2max was calculated using ACSM Leg Ergometry Equation [31].
HRV parameter calculation and signal processing
HRV parameter calculation and signal processing were conducted using VivoSense software version 3.4 (VIVONOETICS, San Diego, CA). Artifact management was performed through VivoSense automatic artifact marking by setting up the max and min HR to 220 and 30 beats per minute (bpm) and setting noise filtering to medium level [32]. A visual inspection of RR interval data was performed to identify ectopic beats and noise. Regions with the presence of ectopic beats or noise for more than 3 seconds were removed from the analysis [33]. Any other identified ectopic beats or noise with shorter duration were replaced by interpolations from adjacent RR intervals [34].
Both time and frequency domain HRV measures were calculated. For time domain measures, the following parameters were calculated: SDNN, RMSSD, and SDSD. For frequency domain measures, the following parameters were calculated: HF power (0.15–0.4 Hz), LF power (0.04–0.15 Hz), LF/HF ratio, HF (nu), and LF (nu). Fast Fourier transformation was conducted as the method of power spectral density analysis. All HRV parameter calculations were performed following the Task Force of the European Society of Cardiology guideline [35].
Statistical analyses
Data were imported into SPSS version 27.0 (IBM, Chicago, USA) software for statistical analysis. Shapiro–Wilk test was used to evaluate the normality of data. Levene’s test was used to assess the homogeneity of variance. Independent t-tests (for parametric data), Chi-square (for gender), or Mann Whitney U test (for non-parametric data) were conducted for descriptive and clinical data to compare the differences between groups.
We calculated the HR and HRV measures of three phases for data analysis: 1) rest phase (4 min rest period before exercise test); 2) 30 s post-exercise phase (30 s window after reaching HR peak); and 3) recovery phase (4 min recovery period after exercise test).
A 2 (group) * 3 (phase) repeated measures ANOVA was conducted to test the differences of HR and HRV time domain measures. A 2 (group) * 2 (phase) repeated measures ANOVA was conducted to test the differences of HR and HRV frequency domain measures. Frequency domain measures such as HF may not be suitable during exercise where higher respiratory frequencies are present, which might affect the accuracy of the measurement [16, 36]. Moreover, the 30 s post-exercise phase is an ultra-short period (<1 min), which was not sufficient to achieve the power spectrum analysis for frequency domain measures [37]. Thus, only two phases (rest and recovery) were analyzed for HRV frequency domain measures.
HRV data were excluded if one of the phases (i.e., rest, 30 s-Post-Exercise, or recovery) had < 65% acceptable data (i.e., percentage of data that passed both automatic artifact marking and visual inspection) [38]. HRV measures that were not normally distributed were log-transformed to achieve normality before performing repeated measures ANOVA. Alpha level of p < 0.05 was set for all statistical analyses.
RESULTS
Descriptive and clinical data
After data processing and applying the exclusion criterion of having less than 65% acceptable data (i.e., percentage of data that passed both automatic artifact marking and visual inspection) available in any phase [38], there were 14 participants with HD and 12 controls included in analyses. For participants with HD, 2 people were excluded due to less than 65% data acceptable in 30 s post-exercise phase and 3 people were excluded due to less than 65% acceptable data available in both 30 s post-exercise phase and recovery phases; for controls, 1 person was excluded due to less than 65% acceptable data in 30 s post-exercise phase. Descriptive and clinical data of participants with HD and controls are shown in Table 1. Compared with controls, participants with HD showed reduced HR peak (p < 0.01) and HR reserve (p < 0.001). There were no other significant differences in descriptive data between groups (p > 0.05). 11 out of the 14 participants with HD were taking medication, either to directly treat their HD or for other conditions. A full list of medication type is included in the Supplementary Material.
Descriptive and clinical data of HD and controls group
Table values are mean±standard deviations or n (%) HD, participants with Huntington’s disease; bpm, beats per minute; BP, blood pressure; HR, heart rate; W, Watts; IPAQ, International Physical Activity Questionnaire; MET-min/week, metabolic equivalent of task minutes per week; UHDRS, United Huntington’s Disease Rating Scale **p < 0.01, ***p < 0.001.
HRV time domain measures
The means and standard deviations of HRV time domain measures during each phase for both groups are presented in Table 2 and Fig. 1. Significant main effects of phase were found in SDNN, RMSSD, and SDSD (p < 0.001). The group * phase interaction effects were significant for both RMSSD and SDSD (p < 0.01). Simple effect analysis showed that compared with controls, participants with HD demonstrated significantly reduced RMSSD and SDSD at rest phase (p < 0.001) but not at other phases (p > 0.05).
HRV Time domain measures phase of HD and controls group at rest, 30 s-post-exercise, and recovery
Table values are mean±standard deviations. HRV, heart rate variability; HD, participants with Huntington’s disease; SDNN, standard deviation of normal-to-normal intervals; RMSSD, root mean square of successive differences of successive intervals; SDSD, standard deviation of successive differences of normal-to-normal intervals. **p < 0.01, ***p < 0.001 †p-values for comparison between groups (simple effect of group) ‡p-values for main effect of phase §p-values for interaction effect (group * phase).
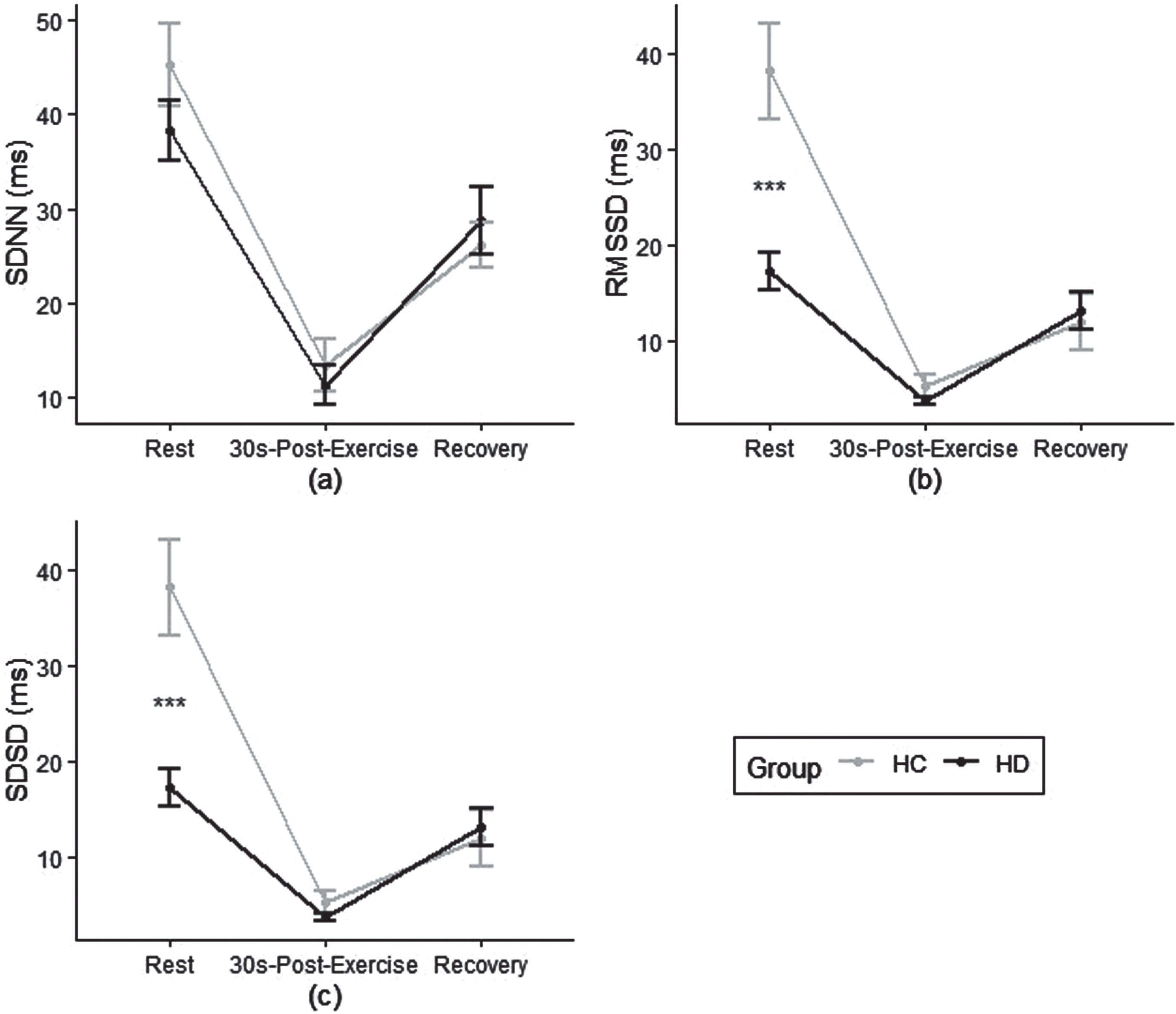
Heart Rate Variability (HRV) Time Domain Measures a) standard deviation of normal-to-normal intervals (SDNN); b) root mean square of successive differences of intervals (RMSSD); and c) standard deviation of successive intervalsc (SDSD) for HD and control groups at rest, 30s-post exercise and recovery phase. HC, healthy controls; SDNN, standard deviation of normal-to-normal intervals; RMSSD, root mean square of successive differences of successive intervals; SDSD, standard deviation of successive differences of normal-to-normal intervals; HR: heart rate. ***p < 0.001 between groups.
HRV frequency domain measures
The means and standard deviations of HRV frequency domain measures during rest and recovery phase for both groups are presented in Table 3 and Fig. 2. Significant main effects of phase were found in HF power (p < 0.001), LF power (p < 0.001), HF (nu) (p < 005), and HF/LF ratio (p < 0.05). The group * phase interaction effect was significant for LF power (p < 0.05). Simple effect analysis showed significant differences between groups for LF power (p < 0.01), HF (nu) (p < 0.05), LF (nu) (p < 0.001), and HF/LF ratio (p < 0.05), but only at recovery phase, not rest phase (p > 0.05).
HRV frequency domain measures of HD and controls group at rest and recovery phase
Table values are mean±standard deviations. HRV, heart rate variability; HD, participants with Huntington’s disease; HF, high frequency; LF, low frequency; nu, normalized units. *p < 0.05, **p < 0.01, ***p < 0.001 †p-values for comparison between groups (simple effect of group) #p-values for main effect of phase §p-values for interaction effect (group * phase).
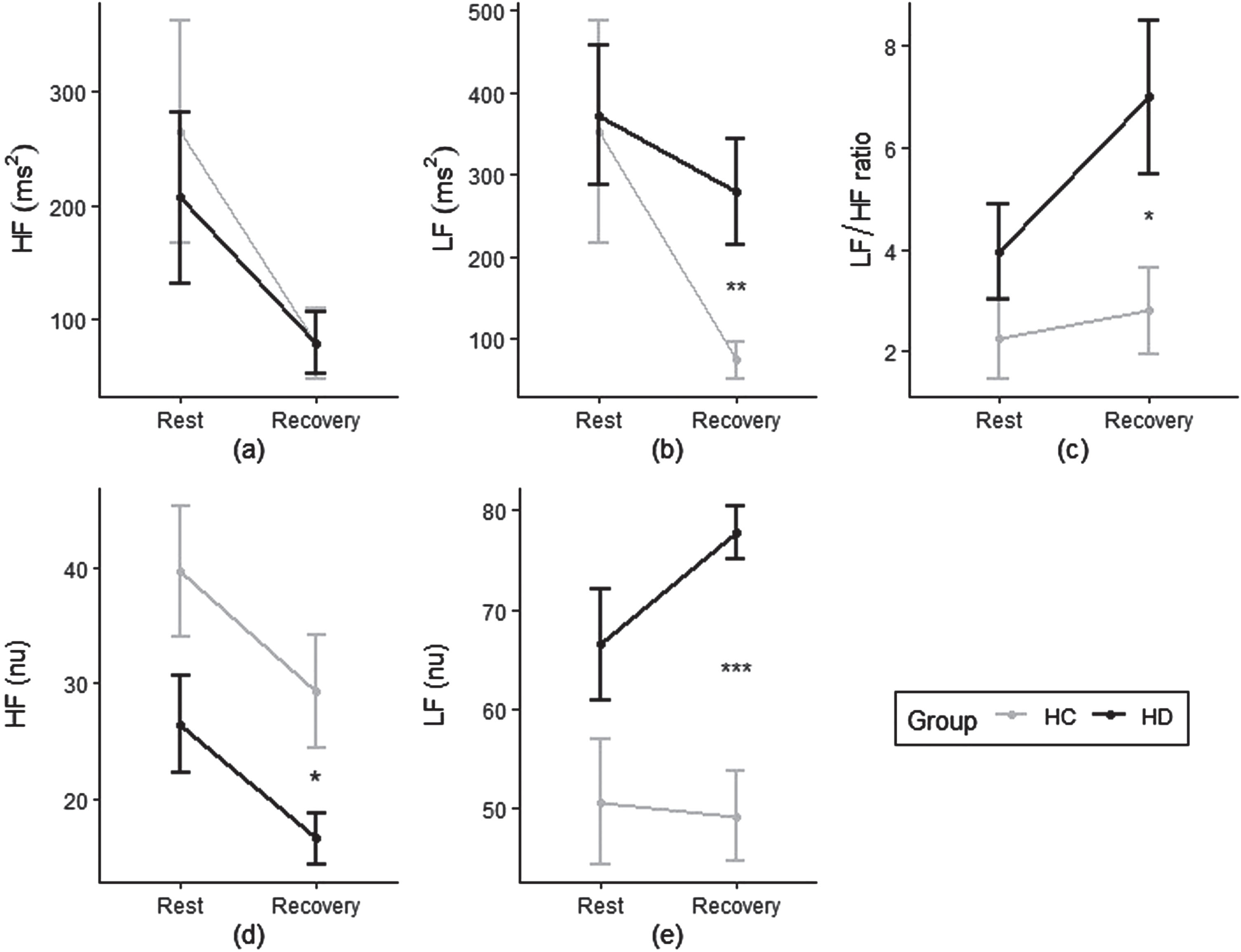
Heart rate variability (HRV) frequency domain measures a) High Frequency (HF); b) Low Frequency (LF); c) LF/HF ratio; d) HF normalized units (nu); and e) LF (nu) for HD and control groups at rest and recovery phase. HRV, heart rate variability; HD, participants with Huntington‘s disease; HC, healthy controls; HF, high frequency; LF, low frequency; nu, normalized units; HR, heart rate. *p < 0.05, **p < 0.01, ***p < 0.001 between groups.
DISCUSSION
In this study, we tested the HRV in early-to-mid stage HD (TFC stage 2 or lower) participants and controls during a maximal incremental exercise test to assess their cardiovagal modulation ability. Participants with HD had significantly lower HR reserve and HR peak compared with controls. These results indicate that people with early-to-mid stage HD have reduced aerobic exercise capacity, supporting previous studies that people with HD might have decreased aerobic metabolic ability and exercise capacity [19, 21]. We also found reduced time domain HRV measures in HD participants at rest, including RMSSD and SDSD, indicating decreased parasympathetic modulation [35, 39]. These results support previous observations that people with HD may have an imbalanced ANS [4] with a shift towards decreased vagal modulation [11, 13].
We observed that both groups showed a significant reduction in HRV time domain parameters at 30 s post-exercise phase and recovery phase, compared with rest phase, which supports previous findings that exercise could elicit a reduction in HRV time domain measures [36, 41]. Past studies also reported an intensity dose-response in which higher exercise intensity is associated with lower HRV time domain measures [16, 41]. Interestingly, although we observed that control participants experienced significantly higher exercise intensity than participants with HD (indicated by their significantly higher HR peak), controls did not show significantly lower HRV time domain measures at 30 s post-exercise or recovery phase than HD group. The reason might be that people with HD have reduced responsiveness to exercise, indicated by the significant interaction effect of HRV time domain parameters (i.e., RMSSD and SDSD decreased less in people with HD compared with controls in response to exercise), which indicates increased sympathetic modulation during acute post-exercise recovery [35, 42]. This lack of HRV difference with the presence of exercise intensity difference between groups suggests that people with HD may have a reduced dose-response relationship between HRV and exercise intensity compared to controls.
We found that both groups showed a reduction in HRV frequency domain measures, including HF and LF power, HF (nu), and HF/LF ratio at recovery phase, compared with rest phase. The findings support previous observations that HRV frequency domain measures decreased in response to exercise [41, 44]. We observed that participants with HD had elevated LF power, HF (nu), LF (nu), and LF/HF ratio at recovery phase, indicating increased sympathetic activity during post-exercise recovery [35, 45]. These differences were not found at rest. Thus, these results reveal a lack of parasympathetic modulation in people with HD during post-exercise recovery, but not at rest before exercise. Moreover, this interaction effect supports the idea that using exercise as a stressor to put the body in a sympathetic dominant condition while monitoring HRV responses might be a valuable approach to investigate autonomic stress reactivity [16–18] since the deficit was only revealed during post-exercise recovery.
Our findings suggest that people with early-to-mid stage HD have reduced parasympathetic modulation and increased sympathetic activity not only at rest, but also during acute post-exercise recovery. This indicates that in people with HD, cardiac autonomic reactivity during post-exercise recovery may be impaired. Previous studies also showed that people with HD had reduced gluconeogenesis, metabolic, and cardiorespiratory capacity that affects recovery from exercise [19, 21]. Together, these results suggest that current exercise guidelines [46] may not apply to people with HD and a modified exercise prescription with special consideration over the recovery period may be required for people with HD, such as exercising in a lower percentage of HR reserve [19].
Our findings related to sympathovagal dysautonomia are also noteworthy. It has been suggested that sympathovagal dysautonomia with a sympathetic predominance could lead to fatal cardiac arrhythmias and cardiac failures in healthy populations and transgenic mice [5, 47]. Since sympathovagal dysautonomia with a sympathetic predominance has been reported in our results and several other studies [2, 13–15], it could potentially explain why one study found that cardiac failure was implicated in 30% of people with HD but in less than 2% of age-matched controls [5]. However, no study has focused on the potential cardiac consequences of sympathovagal dysautonomia in humans with HD [5]. Thus, further investigations of cardiac dysautonomia in people with HD are required.
Understanding the underlying mechanisms responsible for sympathovagal dysautonomia in HD is crucial for developing preventive and therapeutic strategies that can reduce the associated morbidity and mortality. It has been reported that ANS dysfunction in people with HD is associated with the damage caused by the progressive nerve cell apoptosis in CAN that performs high-level autonomic control [5]. Imaging studies showed strong associations between disease progression and the degeneration of the main components of CAN, including the insular cortex and hypothalamus [48]. Although neurological symptoms and pathology have been the major research topics, the pathology of HD is widespread, as the mutant Huntingtin gene is expressed throughout the body [49]. Evidence suggests that abnormal changes in peripheral tissues are not all secondary to brain dysfunction [50]. For example, transgenic mice studies showed that cardiac abnormalities might be caused by the expression of the mutant Huntingtin gene in cardiomyocytes [51]. However, the direct evidence linking brain or peripheral pathology in HD to cardiac autonomic dysfunction is limited [5, 50]. Thus, more studies that help establish causal links between brain and/or peripheral pathology in HD to autonomic dysfunction are needed to identify the underlying mechanisms causing sympathovagal dysautonomia in HD, which may help develop preventive and therapeutic approaches to reduce morbidity and mortality caused by sympathovagal dysautonomia in HD.
There are a few limitations in our study. Restrictions due to the COVID-19 pandemic affected recruitment; thus we ended up with a smaller sample size than expected. Moreover, we set a relatively low threshold for data exclusion, due to the large amount of artifacts generated during maximal exercise. A number of the participants with HD were taking medication, and we acknowledge these may have impacted the autonomic nervous system response. However, exploring these interactions was deemed to be beyond the scope of this study.
Conclusion
To our knowledge, this is the first study examining HRV during maximal exercise in people with HD. We found reduced aerobic exercise capacity and sympathovagal dysautonomia both at rest and during post-exercise recovery in people with HD, suggesting modified exercise prescriptions may be required for people with HD. Further investigations focusing on cardiac dysautonomia and underlying mechanisms of sympathovagal dysautonomia in people with HD are needed.
Footnotes
ACKNOWLEDGMENTS
We would like to express our sincere gratitude to the participants and their family members for their invaluable contributions and generous donation of time to this study. We would also like to acknowledge the PACE-HD study for providing us with data on individuals with HD, including Dr. Monica Busse and Dr. Cheney Drew of Cardiff University. Additionally, we extend our gratitude to the Neurorehabilitation Research Laboratory at Teachers College, Columbia University, for providing the setting for the study and to the Huntington’s Disease Center of Excellence, including Dr. Karen Marder, Paula Wasserman, Dr. Ashwini Rao, and Deborah Thorne for assistance with recruitment and study management. We also would like to acknowledge the invaluable contributions of our colleagues at the Neurorehabilitation Research Laboratory, including Gregory Youdan, Nia Irene Toomer-Mensah, and Julie Fineman, without whom this study would not have been possible.
FUNDING
This work was supported in part by the Jacques and Gloria Gossweiler Foundation.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
