Abstract
Background:
Huntington’s Disease Society of America Centers of Excellence (HDSA COEs) are primary hubs for Huntington’s disease (HD) research opportunities and accessing new treatments. Data on the extent to which HDSA COEs are accessible to individuals with HD, particularly those older or disabled, are lacking.
Objective:
To describe persons with HD in the U.S. Medicare program and characterize this population by proximity to an HDSA COE.
Methods:
We conducted a cross-sectional study of Medicare beneficiaries ages ≥65 with HD in 2017. We analyzed data on benefit entitlement, demographics, and comorbidities. QGis software and Google Maps Interface were employed to estimate the distance from each patient to the nearest HDSA COE, and the proportion of individuals residing within 100 miles of these COEs at the state level.
Results:
Among 9,056 Medicare beneficiaries with HD, 54.5% were female, 83.0% were white; 48.5% were ≥65 years, but 64.9% originally qualified for Medicare due to disability. Common comorbidities were dementia (32.4%) and depression (35.9%), and these were more common in HD vs. non-HD patients. Overall, 5,144 (57.1%) lived within 100 miles of a COE. Race/ethnicity, sex, age, and poverty markers were not associated with below-average proximity to HDSA COEs. The proportion of patients living within 100 miles of a center varied from < 10% (16 states) to > 90% (7 states). Most underserved states were in the Mountain and West Central divisions.
Conclusion:
Older Medicare beneficiaries with HD are frequently disabled and have a distinct comorbidity profile. Geographical, rather than sociodemographic factors, define the HD population with limited access to HDSA COEs.
Keywords
INTRODUCTION
Huntington’s disease (HD) is a fatal neurodegenerative genetic disorder caused by a cytosine-adenine-guanine (CAG) repeat expansion on the huntingtin gene [1, 2]. The prevalence of HD among U.S. Medicare beneficiaries was estimated to be around 13.1 per 100,000 persons in 2017 [3]. The CAG trinucleotide repeat leads to irregularities in protein function and ultimately to apoptosis of neurons in the brain [1, 4]. Individuals with HD usually present with a combination of psychiatric, cognitive, and motor impairments, including depression, dementia and chorea, which progressively worsen over 10–20 years after the initial onset of symptoms until death [1, 6].
Although research efforts offer great promise, currently there is no cure for HD [7]. The gold standard for treatment is comprehensive multidisciplinary care to manage drug responsive symptoms, maximize function and maintain the highest possible quality of life until death [7–11], Huntington’s Disease Society of America (HDSA) Centers of Excellence (COEs) are one source of onsite, same day multidisciplinary care, and are the entry point for nearly all HD research performed in the U.S. [11–14]. However, there are only 41 of these specialty centers across the U.S. in 2017, and they are not equally distributed from a geospatial perspective [14]. Research from other areas of medicine suggests that increasing distance from appropriate care can negatively affect health care outcomes [15–18]. Moreover, specialty-center based clinical studies often systematically exclude individuals who have social or demographic risk factors such as older age, minority race, and poverty, and clinical factors such as dementia or advanced disease. Descriptive data on HD populations in the U.S. that are inclusive of all care sites are lacking [2, 20]. To support efforts to provide equitable multidisciplinary care to all persons with HD, and to provide epidemiological data on adults from understudied groups living with HD, we used Medicare data to 1) describe the population of persons living with HD in the U.S. who are receiving insurance from the public Medicare program, and 2) to identify and characterize the proportion of HD patients that are proximal to an HDSA COE.
MATERIALS AND METHODS
Ethical compliance statement and data approval
This study utilized Centers for Medicare and Medicaid Services Research Identifiable Files, made available to Dr. Willis under a data use agreement for epidemiology and health services research on neurodegenerative disease. The Institutional Review Board at the University of Pennsylvania granted approval for this study with waiver for individual consent. The authors confirm that patient consent was not required for this work and that we have read the Journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Datasets
This cross-sectional study utilized Research Identifiable Files from the Centers for Medicare and Medicaid Services from the year 2017. We utilized the Carrier File (which contains encounter level data on physician and other medical provider visits, laboratory testing), and the Medicare Beneficiary Summary Base File [MBSF] (which contain individual level data on race/ethnicity, age, sex, beneficiary mailing zip code, date of death and Medicaid eligibility), and the Chronic Condition Warehouse Files (which contain individual level data over 69 chronic health conditions identified using algorithms applied to all available Medicare data). The Medicare program supplies inpatient and outpatient health and prescription insurance to individuals ages 65 and above. Individuals under the age of 65 can qualify for Medicare by virtue of being designated as permanently disabled from a chronic medical condition or having end stage renal disease (ESRD) [21, 22]. Individuals with conditions like HD often qualify for Medicare before age 65 due to the severity of the disease, and HD is known to be present in older adults. We included all individuals enrolled in the Medicare program ages 18 and older, which includes individuals who received benefits initially due to older age and individuals who originally qualified due to disability.
Study population
We identified Medicare beneficiaries with HD as those with a health encounter with the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) code of G10 in the year 2017. We extracted sociodemographic data for the resulting HD cohort from the MBSF base file. Age in years was measured as a continuous variable, and then categorized (18–44, 45–54, 55–64, 65–74, 75–84, 85+). Race/ethnicity was categorized as follows: White, Black, Unknown + Other, Hispanic, Asian, and Native North American. We extracted data on original reason for Medicare benefits: being age 65 and older, having a qualifying disability or ESRD. We identified those individuals also receiving Medicaid insurance, known as “dual eligibles.” Medicaid insurance is provided to Medicare beneficiaries meeting low-income criteria, and dual eligibility is viewed as a marker of poverty in Medicare claims-based health services research [23]. We extracted Chronic Condition Warehouse File data on the diagnosis or treatment of common general medical, neurological, or disabling conditions [24]. Beneficiary mailing zip code is the most discrete geospatial variable available in Medicare Research Identifiable Files. Beneficiaries with a valid, identifiable zip code in the U.S., Puerto Rico, Guam, or the U.S. Virgin Islands were selected for the spatial analyses described below.
Proximity to HDSA COEs
Although high quality specialty care for HD is delivered at academic centers and specialty clinics across the country, Medicare claims data does not allow for automated identification of outpatient specialty centers, and there is no available source that maintains up-to-date data on all neurology centers providing a specific level of HD care in the U.S. Therefore, we focused on HDSA COEs, as these have met some external measures for specialty care and are primary centers for HD research. Addresses of all HDSA COEs in 2017 were retrieved from the HDSA website [14]. The zip codes of the HD patients and HD centers were converted to their latitude and longitude coordinates. Latitude/longitude data were imported into QGis 3.10, a geographic information system application, and distance matrix vector analysis [25] was performed to identify the HDSA COE most proximal to each person with HD. Next, the Google Maps Distance Matrix Application Programming Interface (API) [26] was used to calculate the distance, in miles and kilometers, between the closest HDSA COE and each HD patient. For beneficiaries living on an island or island territory (e.g., Hawaii, Puerto Rico, U.S. Virgin Island), if there was no HDSA COE on the island, we calculated the distance from the island airport to the airport of the nearest HDSA COE. We categorized the distance to nearest COE as ‘100 miles or less’, ‘101–250 miles’, ‘251–500 miles’, and ‘> 500 miles’, based on prior literature that posits a trip of 100 miles or less each way could be made safely in a single day [16, 27–29].
Statistical analyses
Study variables were analyzed using descriptive statistics appropriate for the type and distribution of each variable. Our descriptive analyses of comorbid disease burden include a comparison to the general Medicare population, and therefore was restricted to individuals ages 65 and older. We created a map depicting the variation in proportions of individuals living within a 100-mile drive of a COE in each state. Statistical analyses were performed using IBM PASW v26 and illustrated figures were created using Python and Microsoft Excel. Statistical significance was set at p < 0.05.
RESULTS
Study sample sociodemographic characteristics
Table 1 displays the demographic characteristics of our study sample. We identified 9,056 Medicare beneficiaries as having a diagnosis of HD in 2017. Just over half (51.4%) of Medicare beneficiaries with HD were under the age of 65, indicating they were currently qualified for Medicare benefits by virtue of disability. Moreover, among those ages 65 and above, 64.9% first qualified for Medicare due to disability, not age. Women slightly outnumbered men (54.5% versus 45.5%, p < 0.01). In this U.S. sample, most HD patients were White (83.0%), followed by Black (10.4%), Hispanic (2.9%), Other + Unknown (2.4%), Asian (0.9%), and Native North American (0.5%).
Baseline characteristics of 9,056 Medicare beneficiaries with a claim for Huntington’s disease care, 2017
†Chi-square goodness-of-fit test –Bolded values indicate statistical significance. ESRD, end-stage renal disease; HD, Huntington’s disease; IQR, interquartile range; NA, not applicable; SD, standard deviation.
Comorbid disease patterns in geriatric HD
In our HD sample limited to adults ages 65 and older, dementia was the most prevalent neurological comorbid condition, diagnosed in 32.4%, followed by stroke/transient ischemic attack (16.4%). Among psychiatric diagnoses, depression and anxiety were most common (35.9% and 24.5%, respectively). Chronic cardiovascular disorders—atrial fibrillation, ischemic heart disease, congestive heart failure—were also common (14.6% –42.0%); alcohol and opioid abuse, colorectal and lung cancers were relatively rare (3.5% or less). HD patients rarely had non-cognitive neurological diagnoses (such as epilepsy, migraine, traumatic brain injury).
Figure 1 shows the prevalence of common medical conditions in Medicare beneficiaries with and without an HD diagnosis. Prevalence ratios (PRs) estimated using these data were greatest for psychosis (PR 2.98, 95% confidence interval [CI] 2.69–3.30), traumatic brain injury (PR 2.52, 95% CI 2.01–3.17), dementia (PR 2.18, 95% CI 2.07–2.29), and epilepsy (PR 2.04, 95% CI 1.75–2.41). Cardiovascular conditions (congestive heart failure, ischemic heart disease, atrial fibrillation) were less prevalent in HD; the PRs ranged from 0.67 (95% CI 0.62–0.73) for atrial fibrillation to 0.92 (95% CI 0.87–0.97) for heart failure. Diabetes, chronic obstructive pulmonary disease (COPD), and chronic kidney disease were similarly common between the two groups.
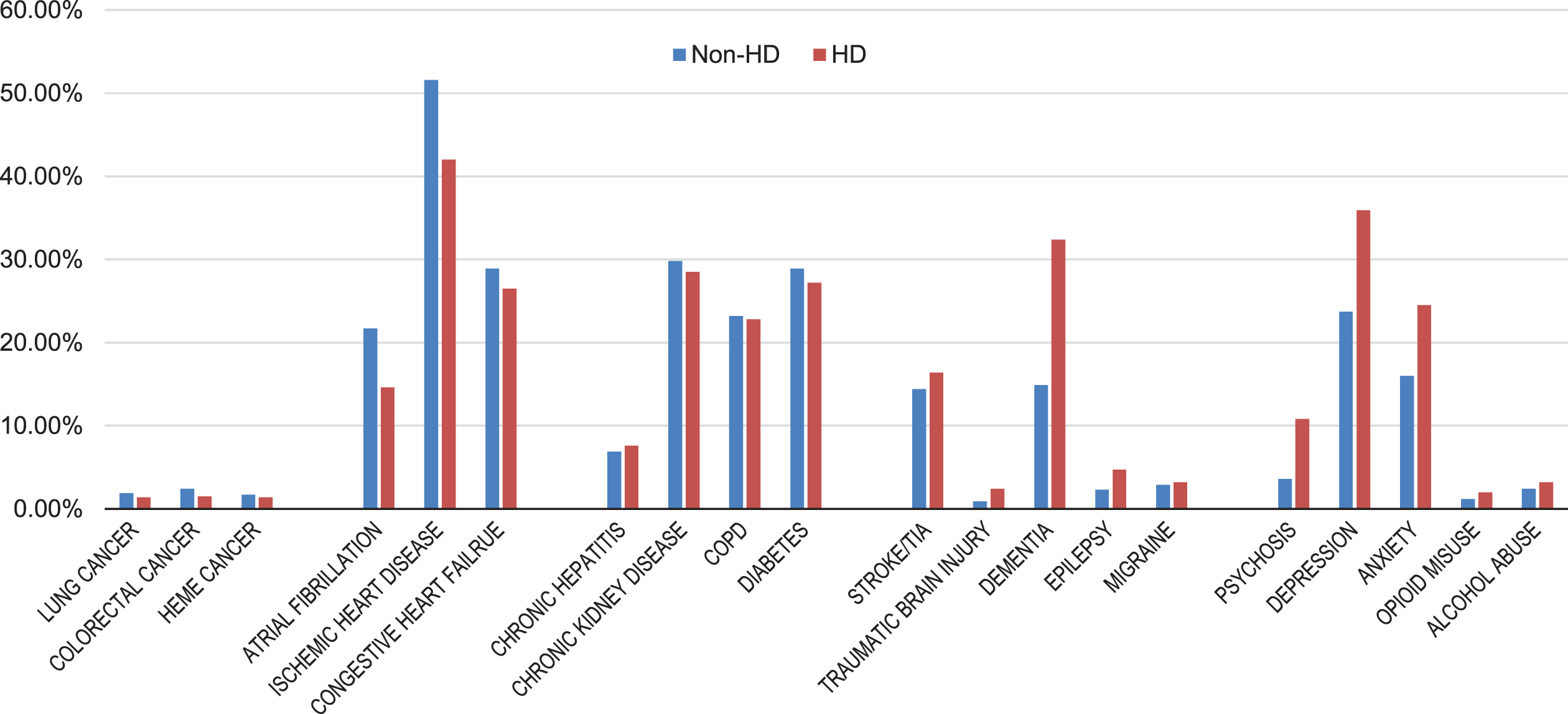
Prevalence of Common Chronic Conditions Among U.S. Medicare Beneficiaries with and without Huntington’s disease, 2017. Non-HD, without Huntington’s disease; HD, with Huntington’s disease; COPD, chronic obstructive pulmonary disease; HEME, hematologic; TIA, transient ischemic attack.
HDSA COE proximity
In our sample, 9,003/9,056 (99.4%) individuals had a zip code which met criterion for proximity analysis. Overall, 57.1% of Medicare beneficiaries with HD resided within one day’s drive (0–100 miles) of an HDSA COE; 31.7% and 9.8% lived at 101–250 and 251–500 miles, respectively. As displayed in Table 2, proximity to a COE on this scale did not vary by age, sex, or dual eligible status. Compared to Whites and Blacks, a higher proportion of Asian (80.3%) and Hispanic (63.7%), Other/Unknown (70.2%), but a lower proportion of Native North American (31.1%) beneficiaries were in the most proximal group. As shown in Supplementary Table 1 and Fig. 2, individuals in the Mountain and Southwestern regions of the U.S., Alaska and Hawaii had the highest proportions of HD patients who lived greater than 100 miles from a COE. U.S. territories were not mapped.
Proximity to Huntington’s disease Centers of Excellence in Medicare beneficiaries with Huntington’s disease, 2017 (n = 9,003)
*Cells with number < 11 are suppressed due to Centers for Medicare & Medicaid Services policy.
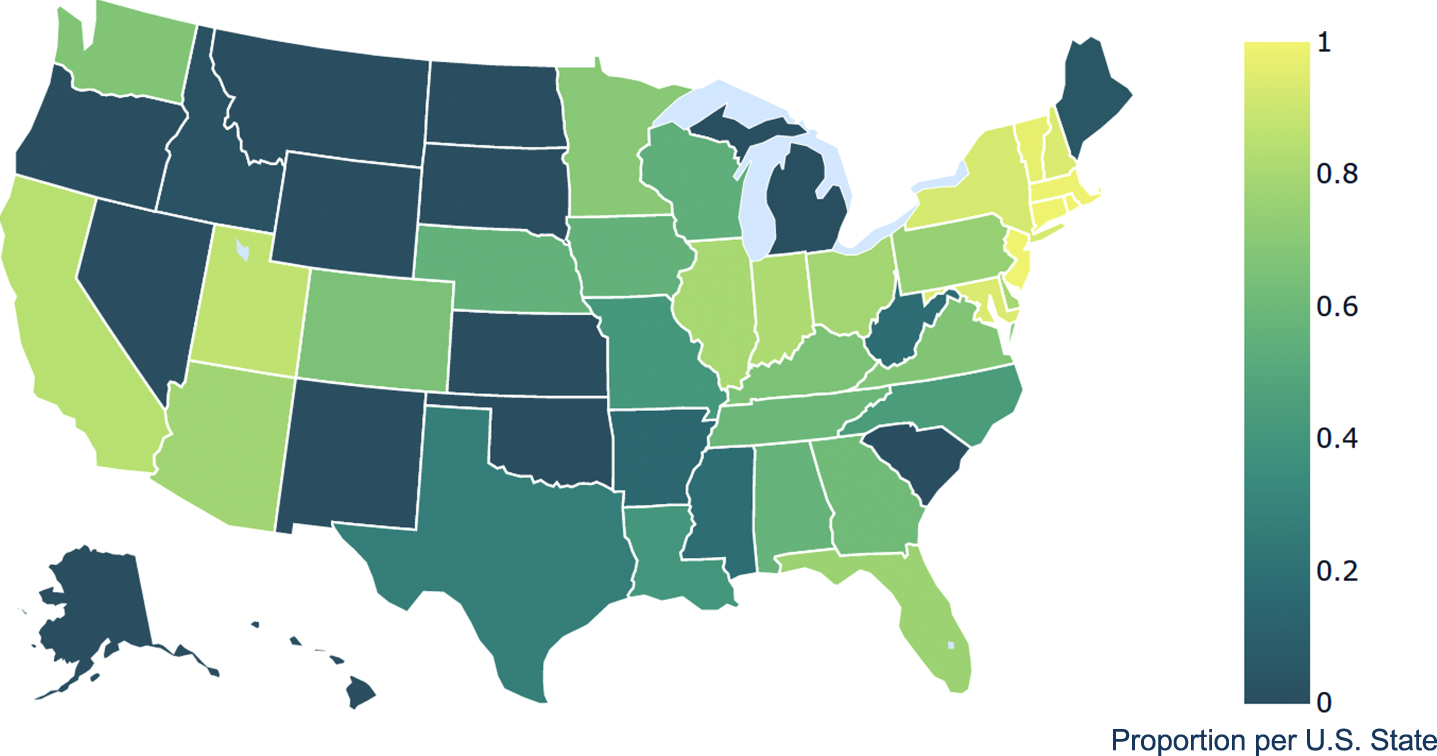
Geographical Variations of Medicare Beneficiaries with Huntington’s disease Living in Close Proximity (≤100 miles) to a Huntington’s disease Center of Excellence, 2017.
DISCUSSION
In this study, we characterized the U.S. Medicare population of individuals with HD from sociodemographic and clinical perspectives, and then examined the proximity of individuals in this population sample to specialty care. Our primary findings are 1) a large number of older and disabled adults with HD can be identified in Medicare datasets, 2) geriatric HD patients have a high comorbidity burden, and 3) there are geographical disparities in access to HDSA COE care.
There is limited focus on the health, care, and outcomes of older adults with chronic neurological conditions, which we term neuroaging research, thus our finding of a large number of geriatric HD patients may be surprising. Improved management of HD symptoms, preventive care, and comorbid disease care in general support greater survivorship for individuals with HD in any age group [30–36]. And, HD can manifest or become clinically evident at any age [2, 37]. Although beyond the primary purpose of this study, the HD prevalence in our data is 8.3 per 100,000 Medicare beneficiaries ages 65 and older, which is within the accepted prevalence rate of 5–10 people per 100,000 persons [3, 39]. Taken together, these findings provide initial conceptual support for further health services research in geriatric HD populations using these data.
In this predominantly older sample, neuropsychiatric, cardiac, and metabolic disorders were the most commonly diagnosed comorbidities. Psychiatric disease and dementia are known manifestations of HD [4, 40–43], with prevalence rates from specialty clinic studies ranging from 5–69% for dementia [44–47], 33–76% for mood disorders [48–51]. Our finding of a high prevalence of cardiac and metabolic diseases may reflect aging, HD, or both. Cardiovascular events are disproportionately high in the HD population [52, 53], and neurocardiovascular pathology has been demonstrated in both pre-manifest and early-stage HD [54–57]. Geriatric HD patients may therefore be an ideal group for the study of the long-term effects of HD on the cardiovascular system, or, among patients with HD manifest at older ages, study of the interplay between HD and normal cardiovascular aging. Moreover, recent studies demonstrated a correlation between pre-existing hypertension [58] or diabetes [59] and an earlier age of symptom onset in HD patients. Although causality has not been shown, it is important to determine if hypertension and diabetes care correlate with longer survivorship. In both paradigms, the influence of other mediating factors such as antipsychotic induced metabolic disorder, anticholinergic or serotonergic drug related dysautonomia, access to and utilization of excellent primary preventative care, exercise programs, nutritional attainment and food security may emerge as important mediating factors.
Twenty-three states and three U.S. territories had a COE that was not easily reachable by at least half of their HD populations. This finding suggests that HDSA COE use among older HD patients is likely impacted by geographical disparities in access to such care, and that solutions to disparities in care will need to overcome geographical barriers as a first step. From a clinical perspective, HDSA COEs provide coordinated specialty care in neurology, psychiatry, psychology, palliative medicine, social work, physical, occupational and speech therapies, and genetic counseling. These clinical care specialists may be accessed locally, and some of the services listed are not needed frequently. Therefore, a negative clinical impact of the geographical disparities cannot be assumed. However, our finding of similar-to-higher proportions of historically underrepresented minorities or marginalized populations in proximity to HDSA COEs should be considered seriously, as it suggests that factors other than distance influence specialty center referrals and the demographic composition of HD trial participant samples. Efforts to better understand these patterns must not be limited to patient-impugning hypotheses such as language barriers and historical mistrust of the medical and scientific communities. Research focused on understanding how the current academic center-based neurology specialty care models and processes (e.g., referral and recruitment practices) contribute to unequal use of services, even in uniformly insured populations, is more likely to lead to scalable, transportable, and effective solutions.
Study strengths
Strengths of this study include its ability to focus on the Medicare population, including geriatric HD, which is underrepresented in the literature. Our data identifies several areas for further inquiry: health service use and disparities, cardiovascular disease development and evolution, drug-disease interactions in the aging HD population. Our study also provides data that is useful for planning future sites for COEs, suggesting that telemedicine or video-based solutions may be an appropriate first choice for HD patients living in some areas of the U.S. [60].
Study limitations
The validity of HD codes in administrative health care databases is not yet known [61, 62], but genetic diseases, especially fatal ones, in general have good validity or acceptable accuracy [63–66]. Our cross-sectional design cannot account for individuals moving closer or away from COEs nor provide evidence that proximity to a COE associates with health care use, quality or outcomes; these relationships will be the focus of future longitudinal analyses. Although we chose previously published metrics for what is considered an acceptable driving distance for a single day, HD patients as a group may tolerate longer or shorter travel distances. Our use of Medicare claims allows a previously unpublished census of older adults with HD, at the expense of providing information on individuals with HD under the age of 65 who do not have Medicare coverage. Furthermore, Medicare claims do not include life history data, so we cannot make determinations about the duration of disease or time since diagnosis with this dataset. Finally, we examined the most recent year of data in our repository, 2017. Since then, more COEs have been established and the number of medical centers (COE designated or not) able to offer multidisciplinary HD care and research access may have changed in a manner that decreases, increases, or has no effect on the disparities we described here.
Progress in the care and management of chronic neurological conditions demands research at intersection of neurology and aging. In this study, we found that U.S. Medicare beneficiaries with HD are frequently disabled and have complex medical histories. There are also geographical barriers to HDSA COE access for some. Therefore, future research opportunities, particularly those which seek to understand HD in aging cohorts, may wish to consider including teleresearch recruitment options and focusing on improving telemedicine feasibility and outreach.
Footnotes
ACKNOWLEDGMENTS
This project was funded by the discretionary funds to Dr. Willis through the University of Pennsylvania Department of Neurology Translational Center for Excellence for Neuroepidemiology and Neurological Outcomes Research.
CONFLICT OF INTEREST
Dr. Pham Nguyen receives support from the National Institute of Health [Grant #R01 NS099129] and Acadia, both unrelated to this work. Dr. Gonzalez-Alegre has received consulting fees unrelated to this work from Spark Therapeutics, Eisai Therapeutics and NeuExcell, and research funding from the National Institute of Health. Dr. Willis receives financial support from National Institutes of Health [Grant #R01 NS099129], Acadia, and the University of Pennsylvania. Ms. Bravo declared no competing interests for this work.
