Abstract
Background:
No pharmacological treatment has been demonstrated to provide a functional benefit for persons with Huntington’s disease (HD). Pridopidine is a sigma-1-receptor agonist shown to have beneficial effects in preclinical models of HD.
Objective:
To further explore the effect of pridopidine on Total Functional Capacity (TFC) in the recent double-blind, placebo-controlled PRIDE-HD study.
Methods:
We performed
Results:
The pridopidine 45 mg bid dosage demonstrated a beneficial effect on TFC for the entire population at week 52 of 0.87 (nominal
Conclusion:
Pridopidine 45 mg bid results in a nominally significant reduction in TFC decline at 52 weeks compared to placebo, particularly in patients with early-stage HD.
INTRODUCTION
Huntington’s disease (HD) is a progressive, fatal neurodegenerative condition characterized by behavioral, cognitive, and movement dysfunction [1]. These features contribute to gradual clinical worsening and subsequent functional decline. Current treatment approaches address choreiform movements and behavioral symptoms with limited success, but none to date are capable of modifying the evolving functional deficits that are inexorably observed with disease progression. New treatments that maintain or delay functional disability are of paramount interest and represent the major unmet medical need in the treatment of HD.
An emerging pathway with robust preclinical data suggestive of potential to provide benefit in HD is the Sigma-1 Receptor (S1R), a transmembrane chaperone protein located in mitochondria-associated membrane domains of the endoplasmic reticulum (ER) [2]. S1R regulates protein folding and degradation, with critical roles in calcium signaling, mitochondrial function, neuronal survival, synaptic plasticity, and activation of trophic factors [3, 4]. Pridopidine, a small molecule in clinical development for HD, was originally postulated to influence only motor symptoms via low-affinity dopamine D2 receptor antagonism [5]. More recent
Pridopidine has been investigated as a treatment for HD in three randomized, double-blind, placebo-controlled clinical trials: HART, MermaiHD, and PRIDE-HD [13–15]. These studies were initially designed to focus on symptomatic motor effects based on the hypothesis of dopamine modulation as pridopidine’s chief mechanism of action. Indeed, based on suggestion of motor improvement in HART and MermaiHD, the PRIDE-HD study tested four doses of pridopidine for their effects on the Total Motor Score of the Unified Huntington’s Disease Rating Scale (UHDRS-TMS). The Total Functional Capacity (TFC) score was also a pre-specified outcome measure [15, 16]. The TFC is a broad assay of functional status, consisting of five domains reflecting major lifestyle elements (capacity for work, finances, domestic chores, activities of daily living, home and caregiver status). It has established interrater reliability and validity, and served as the primary outcome measure for numerous HD trials [17–21]. Total scores range from 0 to 13, with higher scores indicating a greater capacity for independent function. TFC scores allow staging into early (HD1, TFC 11–13), early-mid (HD2, TFC 7–10), and more advanced disease states (HD3, TFC 4–6 and HD4, TFC 0–3). The TFC has attracted particular attention for HD studies based on willingness of the FDA and European regulatory agencies to accept this measure as a primary endpoint for clinical trials.
Based on an evolving understanding of pridopidine’s potent agonism at the S1R and regulatory agreement that TFC can serve as a single primary endpoint in pivotal trials, PRIDE-HD was extended from its original length of 26 weeks to 52 weeks [15]. This report provides
MATERIALS AND METHODS
The statistical analysis for PRIDE-HD is described in the primary manuscript [15]. In the present analysis, we examined the 45 mg BID dosage for all participants and in early HD participants with baseline TFC scores of 7–13 (HD1 and HD2). HD1 are defined as participants with baseline TFC 11–13, and HD2 are defined as participants with baseline TFC7–10. Dosages of pridopidine other than 45 mg BID showed progressive loss of benefit as dosage and drug exposure increased. This finding is consistent with the known bell-shaped effects of S1R agonists. Therefore, the 45 mg BID dosage was selected for more detailed analysis. A mixed model of repeated measures (MMRM) was used to evaluate data from the full analysis set at 26 and 52 weeks for these groups. Sensitivity analyses were performed using the multiple imputation method assuming Missing Not At Random (MNAR) and the ‘worst-case scenario’, where placebo values were imputed for missing data from all patients who discontinued therapy. TFC was a pre-specified endpoint in PRIDE. We performed
In PRIDE-HD, a total of 323 patients (from all treatment arms combined) completed 26 weeks of treatment. Out of these 323 patients, 262 (81%) continued to the second treatment period up to 52 weeks. The 61 participants (out of 323) who did not continue were enrolled into either Open-HART or Open-PRIDE. These participants had completed 26 weeks of treatment before the IRB approval for the extension of the trial to 52 weeks was granted.
RESULTS
The PRIDE-HD study was initially designed to assess the effect of pridopidine on TMS at 26 weeks [15]. Due to recognition of the S1R as pridopidine’s main target after initiation of the study, suggesting a therapeutic potential beyond motor function, the ongoing trial was extended from 26 weeks to 52 weeks. Approximately 19% of participants reached the 26-week endpoint prior to their institution obtaining IRB approval for study extension (61 out of 323 participants). These participants went directly into the open-label extension study. Of the 323 participants who completed 26 weeks of treatment, 262 (81%) entered the second study period and continued treatment for 52 weeks.
Baseline demographic characteristics were similar between early HD participants (HD1 + HD2, TFC 7–13) who completed 26 weeks of treatment and those who completed 52 weeks of treatment (Table 1). There were no notable demographic differences between early HD participants who completed the full 52 weeks of treatment and those who dropped out (52-week non-completers) (Table 1). The dropout rates between the early HD (TFC 7–13) placebo and 45 mg BID groups were comparable (Table 2). 89% (55/62) of the placebo early HD participants completed the first 26 weeks of the study, vs. 81% (48/59) patients in the 45 mg BID group. 76% (42/55) of early HD placebo participants and 77% (37/48) of 45 mg BID participants who completed 26 weeks started the second treatment period, while 97% (41/42) of the early HD placebo group and 100% (37/37) of the 45 mg BD group who initiated the second study period completed 52 weeks (Table 2).
Demographic characteristics of Early HD participants (baseline TFC 7-13) completing 26 weeks and 52 weeks and those who did not complete 52 weeks
Disposition of Early HD Participants in Placebo and 45 mg BID Pridopidine Groups
Source: PRIDE-HD data.
Total Functional Capacity scores for all recipients on the 45 mg bid dosage (N = 75) at 26 and 52 weeks compared to placebo (N = 81) are displayed in Fig. 1. At 26 weeks, a trend towards improvement in change from baseline vs. placebo was seen in the pridopidine group (–0.49, SE 0.16 vs. –0.15, SE 0.17; difference between groups 0.34, nominal
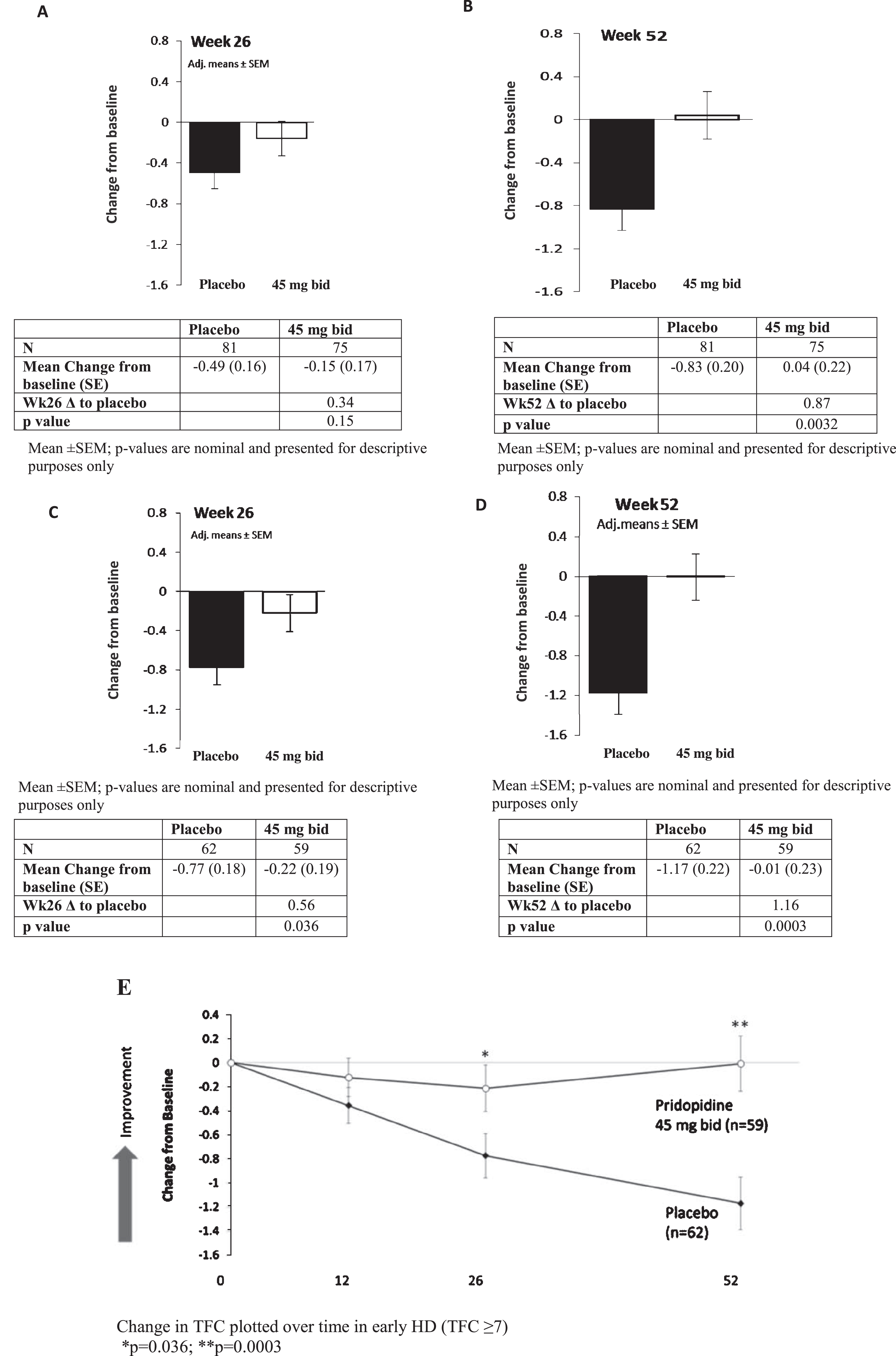
TFC change from baseline vs. placebo for pridopidine 45 mg bid at Weeks 26 and 52 in All Participants (A,B) and Early HD patients (C,D). Mean±SEM;
TFC change from baseline compared to placebo in the 45 mg BID group at Week 52 by disease stage
We performed multiple imputation analysis assuming Missing Not At Random (MNAR) and using the “worst case scenario” (Fig. 2). This method assumes that all missing data in the active treatment group follow the trajectory of the placebo group. Using MNAR for the entire population, the 45 mg bid pridopidine dose was superior to placebo at Week 52 (difference = 0.58; nominal
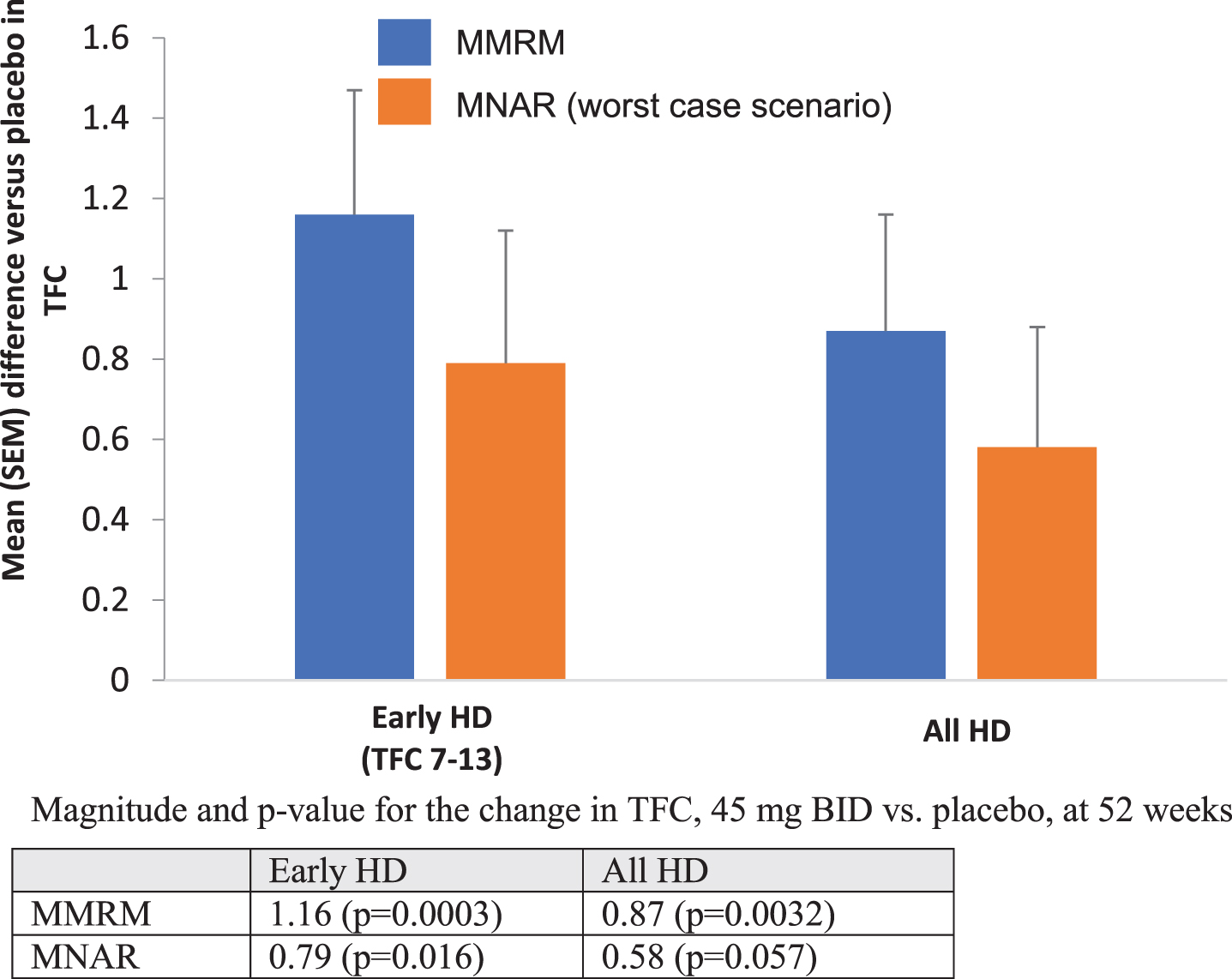
Mean TFC change from baseline vs. placebo at Week 52 for all participants and early HD cohorts (TFC 7–13): comparison of MMRM to MNAR. Magnitude and
Change from baseline to Week 52 in TFC domains for early HD subgroups (baseline TFC 7–13)
LS Mean, least square mean; Included all randomized patients with baseline TFC > = 7, who received at least one dose of study drug and had at least one post-baseline efficacy assesment;
Responder analyses for participants in placebo and 45 mg bid pridopidine groups. N(%) of participants with
Change in TMS and Global Functional assessments in Early HD Responders (
Source: PRIDE-HD CSR
The composite UHDRS (cUHDRS) is a recent measure of interest, as its scoring system combines existing measurement scales to measure patient performance and function: UHDRS-TMS, UHDRS-TFC, SDMT (Symbol Digit Modality Test) and SWR (Stroop Word Reading Test). cUHDRS shows increased sensitivity over individual measures that is most obvious with increased duration [22]. PRIDE-HD did not measure SWR, but using available data the cUHDRS was calculated based on UHDRS-TMS, UHDRS-TFC and SDMT. Pridopidine shows a benefit in cUHDRS in early HD patients at 52 weeks (treatment effect of 0.6 points, nominal
cUHDRS at Week 52 in Early Stage HD Patients from PRIDE-HD
CI, confidence interval; Note: In PRIDE-HD cUHDRS score is derived from UHDRS-TFC, UHDRS-TMS and SDMT (without SWR).
DISCUSSION
This work is further analysis of TFC performance in the PRIDE-HD study, a randomized, placebo-controlled clinical trial of pridopidine in HD. PRIDE-HD was initially designed to assess the safety and efficacy of pridopidine on motor function at 26 weeks. After the trial started, emerging preclinical data indicated the primary target of pridopidine is the S1R, suggesting therapeutic potential beyond motor function. The ongoing trial was then extended from 26 weeks to 52 weeks to allow for more comprehensive assessment of outcomes that may require longer periods of time to show detectable therapeutic effects, including TFC.
Our analysis focuses on early-stage participants (HD1/HD2, TFC baseline 7–13) and the 45 mg bid dosage. Participants treated with 45 mg pridopidine BID had less TFC decline than placebo at week 52, demonstrating an almost 1-point difference (0.87, nominal
Due to the timing of independent review board (IRB) approval for extension of the study to 52 weeks, ∼19% of participants who completed 26 weeks of treatment did not continue treatment for 52 weeks (61 out of 323 participants). Demographic characteristics were similar between the placebo and 45 mg BID groups among early HD participants who completed 26 weeks, 52 weeks, or participants who discontinued over the duration of the study. Dropout rates between placebo and 45 BID were also comparable, suggesting these data are well-matched and suitable for TFC comparison at 52 weeks. To further validate the observed effect of pridopidine on functional decline at Week 52 with more conservative methods to address these missing data, multiple imputation was performed assuming MNAR and worst-case scenario, for which all missing data in the treatment group is assumed to follow the trajectory of the placebo group. Using this sensitivity analysis, pridopidine 45 mg BID showed an effect of 0.58 at week 52 (nominal
It is noteworthy that participants receiving pridopidine 45 mg bid displayed virtually no decline in mean TFC over the course of 1 year, an effect particularly visible for patients with milder disease (TFC 7–13). This is very different than observations from natural history studies and placebo groups in previous clinical trials, where the rate of TFC decline for active treatments are also consistently similar to placebo. Early HD patients (TFC 7–13) naturally decline at a mean rate of 0.97 points/year, while TFC 3–6 (HD3) and HD4: 0–2 decline at 0.38 and 0.06 points/year, respectively, likely reflecting a floor effect in more advanced disease [23]. The greater magnitude of benefit noted in early HD patients treated with pridopidine 45 mg BID likely reflects sufficient numbers of residual neurons and functional reserve to respond to an intervention, compared to late stage participants (HD3 and HD4) for whom advanced disease processes may lessen the possibility of protection or functional rescue.
The observed treatment effects on TFC change from baseline appear to be dosage-specific. Differences from baseline vs. placebo are substantially greater in the 45 mg bid dose group compared to higher dosages [15]. This dose provides essentially complete binding of S1R throughout the human brain on imaging studies [24]. S1R agonists, including pridopidine, are known to modulate numerous important survival pathways (calcium homeostasis, attenuation of oxidative stress, mitochondrial function, lessening of reactive astrogliosis and microglial-induced injury) and are characterized by a bell-shaped dose-response curve in multiple preclinical models [25–37]. Treatment with pridopidine both increases BDNF secretion in B104 neuroblastoma cells (unpublished data) and restores impaired synaptic plasticity in HD cortical neurons with a bell-shaped dose-response curve [38]. In the 6-OHDA Parkinson’s disease mouse model, low-dose but not high-dose pridopidine increases neuroprotection of dopaminergic neurons and restores behavioral abnormalities [39]. Evidence of a bell-shaped curve for S1R agonists is also observed in clinical trials. In a 14-day open-label trial in 30 patients with major depression assessing two doses of igmesine were evaluated, a S1R agonist. The lower dose (25 mg) showed the most efficacious response (83%) compared with the higher 50 mg dose (50%) [40]. Further confirmation for these data was obtained in a 6-week, large scale, double-blind, placebo-controlled, Phase 2 trial of 350 patients, where the strongest anti-depressive effect was seen with 25 mg/day (
Improvement in functional capacity—a measure which synthesizes motor, cognitive, and behavioral ability into relevant daily activities—is perhaps the most pressing unmet therapeutic need in HD. On September 22, 2015, the FDA held a public meeting to hear perspectives from people living with HD about disease symptoms, the impact of HD on their daily life, and their experiences with currently available therapies [42]. Participants strongly emphasized that disease burden left them or their loved ones unable to perform many, if not all, meaningful daily activities (working, driving, self-care, upkeep of household, etc.). The UHDRS-TFC captures these concerns, as it reflects elements of function with meaningful impact on patients’ lives. Thus, a therapy with the ability to beneficially modify TFC decline would be of significant therapeutic value. It may be the case that multiple different mechanisms will be required to optimize slowing of functional decline (e.g., huntingtin-lowering, growth-factor enhancing, anti-inflammatory, antioxidant, etc.). Given this uncertainty, it is critical that compounds with potential to lessen functional decline continue to be sought and tested in appropriately designed clinical trials. Longer pridopidine studies than those already conducted (12–26 weeks for HART, MermaiHD, and PRIDE before extension) may be required to see cumulative beneficial effects on functional outcomes that change slowly, like the TFC. The analyses described in this report support further investigation with a prospective, long term, placebo-controlled trial testing pridopidine 45 mg BID in early-stage HD using UHDRS-TFC as the primary outcome measure.
CONFLICT OF INTEREST
AM and KK have previously received grant support from Teva. MG and MH are previous employees of Teva. AM, KK, CWO are consultants for Prilenia therapeutics.
Footnotes
ACKNOWLEDGMENTS
The authors are grateful for the commitment of the PRIDE-HD study participants and their caregivers, without whom this work would not be possible. The original PRIDE-HD study was supported by Teva Pharmaceuticals.
