Abstract
Huntington’s disease (HD) is a fatal genetic neurodegenerative disorder. It has mainly been considered a movement disorder with cognitive symptoms and these features have been associated with pathology of the striatum and cerebral cortex. Importantly, individuals with the mutant huntingtin gene suffer from a spectrum of non-motor features often decades before the motor disorder manifests. These symptoms and signs include a range of psychiatric symptoms, sleep problems and metabolic changes with weight loss particularly in later stages. A higher body mass index at diagnosis is associated with slower disease progression. The common psychiatric symptom of apathy progresses with the disease. The fact that non-motor features are present early in the disease and that they show an association to disease progression suggest that unravelling the underlying neurobiological mechanisms may uncover novel targets for early disease intervention and better symptomatic treatment. The hypothalamus and the limbic system are important brain regions that regulate emotion, social cognition, sleep and metabolism. A number of studies using neuroimaging, postmortem human tissue and genetic manipulation in animal models of the disease has collectively shown that the hypothalamus and the limbic system are affected in HD. These findings include the loss of neuropeptide-expressing neurons such as orexin (hypocretin), oxytocin, vasopressin, somatostatin and VIP, and increased levels of SIRT1 in distinct nuclei of the hypothalamus. This review provides a summary of the results obtained so far and highlights the potential importance of these changes for the understanding of non-motor features in HD.
NON-MOTOR FEATURES OF HUNTINGTON’S DISEASE
Huntington’s disease (HD) is more than a movement disorder. It is always caused by an expanded CAG repeat in the huntingtin (
The regulation of emotion, sleep and metabolism is governed by the hypothalamus and the limbic system. The hypothalamus is made up of interconnected nuclei that receive inputs both from the periphery, e.g., thyroid hormones, leptin, ghrelin and insulin, and the central nervous system (CNS). Besides regulating the endocrine axes of the body, its many different neuropeptide-expressing neuronal populations project within the region and to other areas of the brain to regulate emotion, sleep and metabolism. The hypothalamus is part of the larger limbic system that includes the hippocampus, gyrus cinguli, prefrontal cortex, insula, septal nuclei, amygdala, ventral striatum, ventral tegmental area and raphe nucleus [22, 23]. In light of the presence of non-motor features in HD, this system has gained increasing interest for investigations of changes in clinical material and experimental models of HD (previously reviewed in [24–27]). These authors reviewed the state of knowledge of this area in HD in 2012 in this journal [28] and provides here an updated overview of the major findings made in the hypothalamus and the limbic system in HD. The review is based on a literature search on the PubMed database up to 2019 with the search terms ‘Huntington disease’ ‘huntingtin’, ‘hypothalamus’, ‘limbic system’, ‘orexin’, ‘hypocretin’, ‘oxytocin’, and ‘vasopressin’.
HYPOTHALAMIC CHANGES IN CLINICAL HD DETECTED BY NEUROIMAGING STUDIES
A combination of neuroimaging studies and postmortem analyses of human hypothalamic tissue have identified alterations in the hypothalamic region in HD (Fig. 1, Table 1). Studies of the hypothalamic region in general are challenged by practical factors such as the scarcity of such tissue in brain banks and by intrinsic factors such as the difficulties to define the whole structure or specific nuclei within the hypothalamus due to the lack of clear anatomical borders. Nevertheless, structural analyses using voxel-based morphometry and mathematical modelling based on grey matter signals in the hypothalamic region have detected significant differences between premanifest HD and age- and sex-matched controls in several studies [18, 29–31]. Studies using positron emission tomography (PET) have found increased microglia activation and reductions in dopamine D2 receptors also in premanifest HD [32, 33]. Attempts to estimate the hypothalamic volume have not revealed any significant differences between HD and controls [34]. Nevertheless, these studies indicate that the hypothalamic region is affected early in clinical HD.
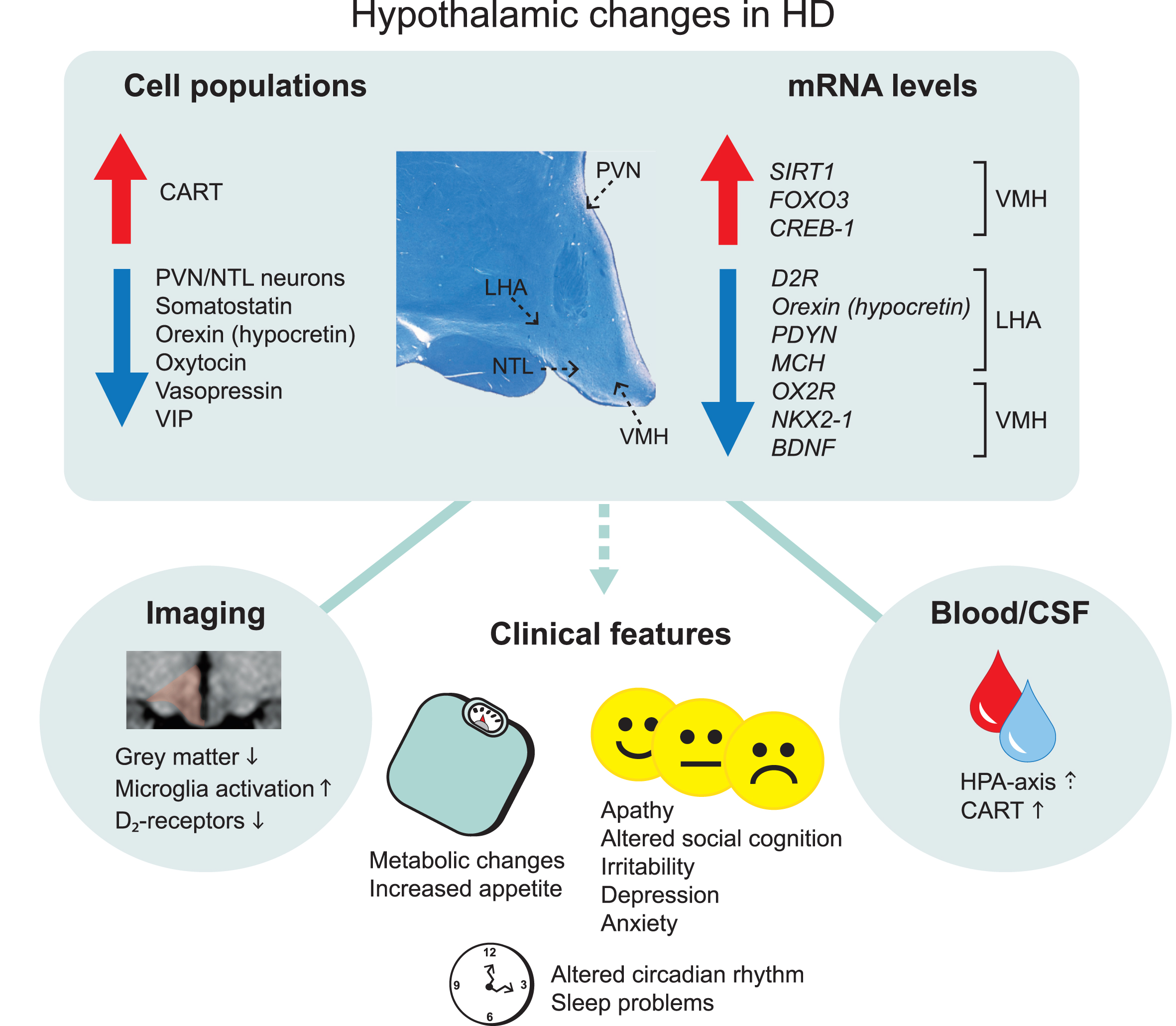
Overview of main hypothalamic changes in Huntington’s disease. Altered immunoreactivity and gene expression levels of cell populations detected in specific hypothalamic nuclei. Changes that result in upregulation and downregulation are indicated by the arrows. The location of specific hypothalamic nuclei is indicated by the dashed arrows on the cross-sectional human hypothalamic section stained for Cresyl violet and Luxol fast blue. Hypothalamic changes are also detected in the blood and cerebrospinal fluid as well as from different imaging paradigms. These changes are thought to be a key contributor to the clinical features in Huntington’s disease. CART, cocaine and amphetamine regulated transcript; CREB-1, cyclic AMP-responsive element-binding protein 1; D2R, dopamine D2 receptor; FOXO3, Forkhead box O3; HPA, hypothalamic–pituitary–adrenal; LHA, lateral hypothalamic area; MCH, melanin-concentrating hormone; NTL, nucleus tuberalis lateralis; NKX2-1, NK2 homeobox 1; OX2R, Orexin 2 receptor; PVN, paraventricular nucleus; PDYN, prodynorphin, SIRT1, sirtuin 1; VIP, vasoactive intestinal peptide; VMH, ventromedial hypothalamus.
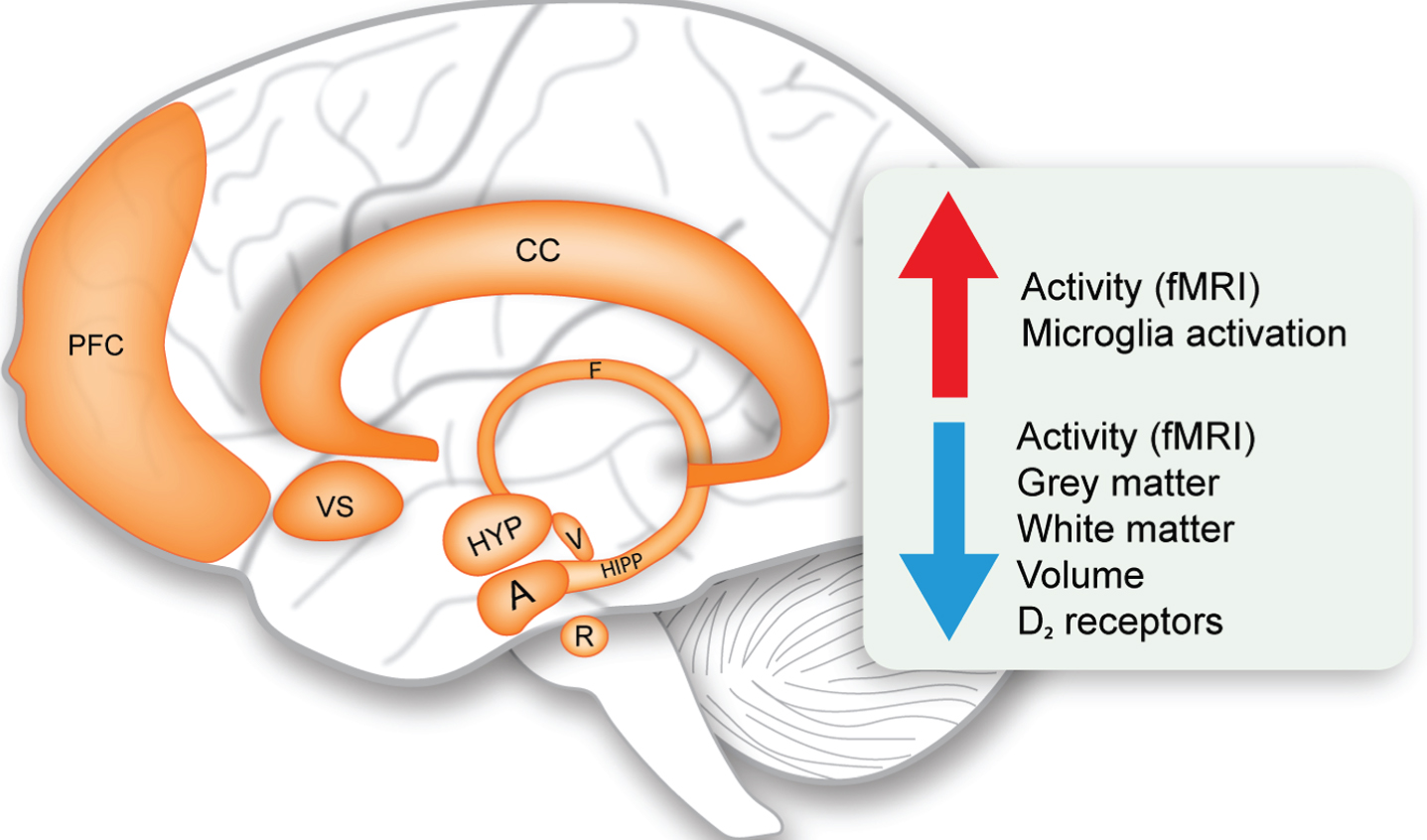
Limbic system changes in Huntington’s disease. Schematic representation of the structures of the limbic system and overview of the main upregulated and downregulated changes (indicated by the arrows). A, amygdala; CC, cingulate cortex; F, fornix; fMRI, functional magnetic resonance imaging; HIPP, hippocampus; HYP, hypothalamus; PFC, prefrontal cortex; R, raphe nucleus; V, ventral tegmental area; VS, ventral striatum.
Summary of main findings in the hypothalamus in clinical HD
AgRP, agouti-related protein; BDNF, brain-derived neurotrophic factor; CART, cocaine and amphetamine regulated transcript; CREB-1, cyclic AMP-responsive element-binding protein 1; CRH, corticotrophin releasing hormone; DTI, diffusion tensor imaging; FOXO3, forkhead box O3; MCH, melanin-concentrating hormone; NPY, neuropeptide Y; PET, positron emission tomography; SIRT1, sirtuin 1; TRH, thyrotropin-releasing hormone; VBM, voxel based morphometry; VIP, vasoactive intestinal peptide.
NEUROPATHOLOGICAL CHANGES IN THE HYPOTHALAMUS IN CLINICAL HD
Postmortem analyses of hypothalami from individuals with HD have revealed a number of changes in different nuclei of this region (Fig. 1, Table 1). The hypothalamus constitutes several nuclei such as the paraventricular nucleus (PVN), supraoptic nucleus (SON), infundibular nucleus, ventromedial nucleus of the hypothalamus (VMH), suprachiasmatic nucleus (SCN), nucleus tuberalis lateralis (NTL), lateral hypothalamic area (LHA) and mammillary bodies. The frequency of huntingtin inclusions as assessed using the EM48 antibody has been estimated to be between 0.5–2.8% in the different nuclei [35]. Although stereological estimation for the total number of neurons and cells in the whole hypothalamus in sections stained for the Nissl stain cresyl violet did not reveal any significant differences between HD and control cases, a specific loss of neurons has been detected in the PVN by 23% and in the NTL by 32% in HD tissue [35, 36]. Early analyses have also indicated a reduced number of somatostatin neurons in the NTL [37–39]. The function of this nucleus is still not well understood. Another study found a reduced number of neurons expressing oxytocin by 45% and vasopressin by 24% in HD cases compared to controls [35]. The number of oxytocin and vasopressin immunopositive neurons were reduced in a case of premanifest HD with Vonsattel grade 0, suggesting that loss of oxytocin and vasopressin may occur early in HD [40]. Oxytocin has been implicated in the ability to recognize facial expression of emotion, which has been found to be reduced already in premanifest HD [41, 42]. Acute intranasal administration of oxytocin to HD gene carriers normalized their altered brain activation pattern in response to recognizing facial expression of emotional disgust, indicating a role for oxytocin in altered emotion recognition in HD [43]. As for vasopressin, it is not known whether vasopressin levels or their activity are altered in HD. The vasopressin system has been linked to aggression in animals where aggressive behavior can be inhibited by vasopressin V1a receptor antagonists [44, 45]. There is an ongoing Phase 2 clinical trial with Azevan Pharmaceuticals using a vasopressin receptor antagonist (SRX246) against irritability in manifest HD (https://clinicaltrials.gov/ct2/show/NCT02507284).
Several studies have investigated the LHA in HD. The LHA controls several functions that are disturbed in HD and pathology in this area could therefore be a contributing if not causative factor for part of the clinical manifestation of the disease. Experimental lesions of the LHA established the “lateral hypothalamic syndrome” characterized by hypophagia, adipsia, hypoactivity and weight loss [46–48]. Later studies established an important role for the LHA in the regulation of sleep, energy balance, reward and motivated behaviors [49]. In HD, there is a 38% reduction in the number of orexin (hypocretin) immunopositive-neurons in HD and mRNA levels are also diminished compared to control cases [35, 50–52]. Orexin is a neuropeptide that is only expressed in the LHA and it plays an important role in the regulation of sleep, emotion and metabolism [53, 54]. Melanin-concentrating hormone (MCH) is another emotion-regulating neuropeptide that is expressed in neighboring neurons within the LHA [55]. A recent study showed significant reduction of MCH mRNA levels in HD cases compared to controls although the number of MCH-immunopositive neurons is not affected [52]. Analyses of mRNA levels of other factors in the LHA in HD cases indicated that the dopamine D2-receptor (D2R) levels are also reduced [52]. mRNA levels of D2R are not reduced in other hypothalamic areas suggesting that the signal of D2R reductions detected using PET may arise from the LHA specifically.
Sleep problems and altered circadian rhythm are common in patients with HD [13–18, 56]. The SCN is the key brain region for regulating sleep and circadian rhythm. Postmortem analyses of the SCN from patients with HD have shown that numbers of vasoactive intestinal peptide (VIP)- and vasopressin-immunoreactive neurons were reduced by 85% and 33% respectively in HD cases compared to controls [57]. This occurred in the presence of normal mRNA levels of these genes as well as unaffected number of neurons expressing melatonin receptors in this area [57]. Hence, there is specific pathology in the SCN in HD that may contribute to alterations in sleep and circadian rhythm.
HYPOTHALAMIC GENE EXPRESSION CHANGES IN CLINICAL HD
Alterations in specific hypothalamic genes have also been reported in HD (Fig. 1, Table 1). A recent study showed reduced expression of mRNA levels of brain-derived neurotrophic factor (
The metabolic regulators and energy sensors sirtuins (SIRT) have been implicated in HD. Neuroprotective effects of SIRT1 inhibitors have been shown in drosophila, mammalian cells and mouse models of HD [69]. One initial clinical trial to evaluate safety has been conducted using the SIRT1 inhibitor, selisistat, in HD patients [70]. SIRT1 acts in the hypothalamus to control metabolism and longevity partly through the regulation of maturation of hypothalamic peptide hormones [71–74]. A recent study showed an increase in mRNA levels and immunoreactivity of SIRT1 in both the LHA and VMH apart from other affected brain regions in HD such as the striatum and cerebral cortex [52]. No changes were found in the less affected cerebellum. Analyses of mRNA levels of downstream targets of SIRT1 such as FOXO3 indicated increased levels in the LHA and VMH in HD. FOXO3 has also been shown to be upregulated in the striatum in HD [75, 76]. This data indicate that the SIRT1 system is affected in sensitive brain regions in HD and that the hypothalamic effects of these factors need to be considered in future studies targeting this system in HD.
OTHER LIMBIC SYSTEM CHANGES IN CLINICAL HD
Although it is beyond the scope of this review to discuss in detail the alterations of the limbic system in HD, it is important to consider the interactions with other limbic structures which form widely distributed networks for a proper perspective. While the definition of the limbic system is not always clear, the anatomical structures generally accepted today and included in this review comprises of the hippocampus, amygdala, ventral striatum, nucleus accumbens, raphe nucleus, ventral tegmental area, cingulate cortex and prefrontal cortex [22, 23]. Table 2 summarizes the main alterations detected to date in the limbic system in clinical HD and in rodent models. Imaging studies using voxel based morphometry (VBM) in magnetic resonance images (MRI) have indicated increased atrophy and grey matter loss in these limbic structures already in premanifest HD [77–86]. Whereas histopathological studies on the limbic structures in HD are scarce, there is evidence for cell loss in the dorsal raphe nucleus, anterior cingulate cortex and prefrontal cortex in symptomatic HD cases with Vonsattel grade 1–4 [83, 88]. Functional MRI (fMRI) studies have reinforced our understanding of limbic system dysfunction in HD. Reduced functional connectivity, network integrity and activity was found in the HD hippocampus, amygdala, ventral striatum, cingulate cortex and prefrontal cortex using different fMRI paradigms assessing verbal working memory, emotional processing, interference/conflict resolution and attention/alertness [89–99]. Such alterations in activity patterns produce multidimensional maps that to some extent reflect internal states of brain processing in response to a task. These changes are already present in the premanifest stage suggesting that early changes in the limbic structures might contribute in part to disease pathogenesis. As cognition and emotional functions are derived from multiple circuit connections involving cortical and subcortical regions of the limbic system, it seems indisputable that limbic system dysfunction can be ascribed to non-motor features of HD. Indeed, volumetric abnormalities in the amygdala have been found to be related to the clinical profile of HD with high level of anxiety and cognitive symptoms occurring before the onset of motor symptoms [77]. This prevailing view of limbic system involvement in HD is also supported by findings from rodent HD models with alterations in volume, reduction in BDNF and serotonin levels reported in several rat and mouse models (see Table 2 for details). The full extent of limbic system pathology in HD is not well understood and more systematic neuropathological postmortem analyses of these structures are warranted.
Summary of main findings in the limbic system in clinical HD and rodent HD models
BDNF, brain-derived neurotrophic factor; PSA-NCAM, polysialylated neural cell adhesion molecule; TRH, thyrotropin-releasing hormone; VBM, voxel-based morphometry; fMRI, functional magnetic resonance imaging; DWI, diffusion weighted imaging.
EFFECTS ON CIRCULATING FACTORS RELATED TO THE HYPOTHALAMUS IN CLINICAL HD
As the hypothalamus is targeted by and regulates circulating (neuro-)endocrine factors in the blood and cerebrospinal fluid (CSF), it became interesting to investigate whether measurements of such circulating factors would be altered in HD as an indication of hypothalamic dysfunction and potentially disease progression. Several studies have been performed using both CSF and blood to measure neuroendocrine factors with often contradicting or negative results [16, 100–104]. The major results are summarized in Table 3. Out of the published results, the increase of cocaine and amphetamine regulated transcript (CART) levels in the CSF is interesting as it may reflect the increased number of CART-immunopositive neurons in the hypothalamus in HD [35, 105]. Also, a subtle change in the hypothalamic-pituitary-adrenal (HPA) axis may be present [106–109]. With the development of more sensitive assays and a better standardization of collected material, it is possible that future studies will reveal other hypothalamic-related factors with relevance for the non-motor features of HD.
Summary of main neuropeptide levels in the circulation in both clinical HD and rodent HD models
ACTH, adrenocorticotropic hormone; AgRP, agouti-related protein; CART, cocaine and amphetamine regulated transcript; FSH, follicle-stimulating hormone; GHRF, growth hormone-releasing factor; IGF-1, insulin-like growth factor 1; LH, luteinizing hormone; NPY, neuropeptide Y; TSH, thyroid-stimulating hormone; GIP, gastric inhibitory polypeptide; PP, pancreatic polypeptide; PYY, peptide YY.
LESSONS LEARNT FROM EXPERIMENTAL STUDIES IN HD
There are several animal models expressing different variants of the human mutant huntingtin gene. Several of these transgenic mice display hypothalamic pathology (Table 4). The R6/2 mouse model shows the highest degree of hypothalamic pathology with reduced number of neurons expressing orexin, oxytocin, vasopressin and VIP [50, 110–113]. Experimental studies provide a complementary approach to the analyses of clinical material as they provide the possibility to study and establish causative relationships. As the hypothalamus receives major input both from the periphery and the rest of the CNS, the question arises as to what extent are the changes detected in this region a direct consequence to the expression of mutant huntingtin in hypothalamic cells. Experiments using recombinant adeno-associated viral (rAAV) vector mediated delivery of different fragments of mutant huntingtin specifically to the hypothalamus have been conducted to address this point. These studies have shown a direct and early effect on the neuronal populations expressing orexin, oxytocin, vasopressin, MCH and BDNF in the hypothalamus, suggesting that the findings made in human postmortem hypothalamic tissue may be directly related to the effects of mutant huntingtin in this area [52, 115]. Other studies have been performed in order to investigate whether the inactivation of mutant huntingtin in the floxed BACHD mouse model using delivery of Cre-recombinase by rAAV vectors or different breeding strategies. rAAV-vector mediated delivery of Cre-recombinase into the hypothalamus has shown a causative link between hypothalamic dysfunction and non-motor phenotypes such as metabolic dysfunction and depressive-like behavior [114, 116]. However, experiments to inactivate mutant huntingtin in specific nuclei or cell populations in the hypothalamus using breeding of the BACHD mice with mice expressing Cre-recombinase under the VMH-specific promoter SF-1, under the PVN-specific promoter Sim-1, or the leptin receptor promoter, did not show any beneficial effects on the psychiatric or metabolic phenotype [117–119]. It would be useful to investigate the involvement of specific hypothalamic circuitries in the development of the early HD phenotype. Further studies using cell specific expression or inactivation of mutant huntingtin in combination with modulating the activity of specific circuitries using chemo- or opto-genetic techniques with analyses of behavioral outputs are likely to give further insight into the causal links between huntingtin-mediated hypothalamic dysfunction and non-motor features of HD.
Summary of main hypothalamic findings in HD rodent models
HPA, hypothalamic–pituitary–adrenal; CART, cocaine and amphetamine regulated transcript; CRH, corticotrophin releasing hormone; GnRH, gonadotropin-releasing hormone; HAP1, huntingtin-associated protein 1; MCH, melanin-concentrating hormone; NPY, neuropeptide Y; POMC, proopiomelanocortin; TSH, thyroid stimulating hormone, VIP, vasoactive intestinal peptide.
CLINICAL TRIALS IN HD: FROM A HYPOTHALAMIC PATHOLOGY PERSPECTIVE
According to the clinical trials database (https://clinicaltrials.gov/), a total of 124 HD clinical trials have been completed to date worldwide with an additional 46 currently active or in the process of recruiting. These trials have largely focused on restoring motor or cognitive function and very few studies have focused on the non-motor disturbances. Gene therapy approaches to silence the disease-causing mutant HTT protein are currently at the forefront of treatment strategies for HD with the successful completion of the Phase 1/2a IONIS-HTTRx clinical trial (Ionis Pharmaceuticals) using intrathecal administration of antisense oligonucleotides (ASOs) to reduce both wild-type and mutant HTT [120]. A Phase 3 trial in adult patients with manifest HD (RG6042, Roche Pharmaceuticals) began in April 2019. The hypothalamus is located in a prime position anatomically, surrounding the third ventricle. Although it is likely that intrathecally-delivered ASOs can also target the hypothalamus, preclinical studies using different intrathecally-delivered HTT-lowering agents have not focused on whether there is target engagement in the hypothalamus. With the results of the ongoing HTT-lowering clinical trials, it will be imperative to determine whether intrathecally delivered ASOs reach the hypothalamus and if so, the extent to which this improves the non-motor symptoms and signs. In preparation for future preclinical and clinical trials with HTT-lowering agents, it is worth considering that hypothalamic pathology exists alongside or perhaps even before striatal and cortical pathology. It will be important to further investigate what brain regions need to be targeted to impact upon non-motor features of HD and to pinpoint a treatment timeframe during the life of an affected individual where it is still possible to prevent or reverse symptoms and eventually halt disease progression.
CONCLUSION
The hypothalamus and other areas of the limbic system are part of the brain regions affected in HD. The changes detected mainly in the hypothalamus include effects on factors that regulate motivated behavior, emotions, social cognition, sleep and/or metabolism with reduced levels of orexin, oxytocin, vasopressin, VIP and somatostatin and increased levels of SIRT1 in specific hypothalamic nuclei. Experimental studies in mice have begun to establish causative relationships between expression of mutant huntingtin in the hypothalamus and its effects on neuropeptide expression as well as in the development of depressive-like behavior and metabolic disturbances. Hence, pathology in the hypothalamus may have relevance for the development of non-motor features in HD. Further studies are still needed to investigate the overall extent of these changes in the limbic system and the potential of modulating affected targets for symptomatic effects and/or disease modification in HD.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
RYC is supported by the Swedish Society for Medical Research and NEURO Sweden. SG is supported by the Swedish Brain Foundation.
